Abstract
Cardio-Oncology has blossomed as a new field in cardiovascular medicine, in large part due to new therapies, which may have cardiovascular sequelae. Despite this, anthracyclines still serve as cornerstone therapy for most pediatric cancers, several solid tumors and hematological malignancies. Cardiotoxicity is the main limiting concern with anthracyclines, and this is particularly an issue in patients in extremes of age (both young and old patients). Pediatric hearts are susceptible for cardiotoxicity, while in older patients, concomitant risk factors may contribute to lower threshold for cardiotoxic effects. With increasing patient survival, a significant increase in elderly cancer patients and long-term cardiotoxicity effects of anthracyclines, a better mechanistic understanding of age-dependent processes—that define cardiotoxicity—is needed. This review sheds light on how age affects underlying molecular pathways of anthracycline-associated cardiotoxicity and aims to provide preventive strategies that can be used in clinical practice.
Introduction
The population of cancer survivors is growing due to earlier detection and better treatment options. 1 Today, overall 5-year survival in pediatric and adolescent cancer patients exceeds 80%. 1 As of January 2019, an estimated 16.9 million Americans are considered cancer survivors, representing approximately 5% of the total United States (US) population. 2 More than 65,000 children (0-14 years), 47,000 adolescent (15-19 years) and 10.8 million elderly (>65 years) are considered cancer survivors. 3 Cancer currently affects more than 1 in 3 people during their lifetime 1 and is, together with cardiovascular (CV) disease, the leading cause of death in developed countries. 4 Paradoxically, increases in cancer survivors go hand in hand with increases in cardiac patients.
Anthracyclines emerged as an effective anti-cancer treatment more than 50 years ago and still form the cornerstone therapy for pediatric cancers, sarcoma, hematological malignancies and a subset of breast cancers. 5,6 Unfortunately, anthracycline use is limited by unintended cardiotoxicity, 7 which presents as a disease spectrum from asymptomatic systolic dysfunction to clinical heart failure (HF). 8 Acutely, anthracyclines can also cause arrhythmia and a myocarditis-like syndrome. 9,10 CV complications due to anthracycline treatment are the main non-cancerous cause of morbidity and mortality in childhood cancer survivors, 5,11,12 persisting up to 45 years after treatment. 12,13 In fact, pediatric cancer survivors are 15 times more likely to develop congestive HF 14 and 7 times more likely to die from cardiac causes 15 as compared with their peers that do not have a history of cancer.
Anthracyclines continue to serve as the main treatment regimen for common pediatric cancers, with more than 50% of the pediatric cancer patients treated with these chemotherapeutics. 16 -18 In addition, elderly patients experience higher rates of cardiotoxicity and HF compared with their younger counterparts when treated with anthracyclines. 19 However, in contrast to the abundancy of clinical trials in pediatric cancer patients, data are lacking on the elderly population. This, in combination with cardiotoxicity risk, results in under treatment and premature treatment termination in elderly cancer patients. 20 -24 Efforts to understand the molecular mechanisms of chemosensitivity in patients in extremes of age will contribute to 1) reduction of cardiotoxicity and eventually 2) optimal therapy for all cancer patients.
Risk Factors for Cardiotoxicity
Multiple risk factors predispose to anthracycline-induced cardio-toxicity, of which cumulative anthracycline dose is the most critical one. Subclinical cardiac dysfunction 25,26 and histopathologic changes (i.e. microfibrillar derangement, vacuolation) 27 can already be seen in patients with so-called “low dose” or “normal dose” administrations (100 and 240 mg/m2, respectively), underscoring the fact that no “safe dose” for anthracyclines exists. Other risk factors include pre-existing CV disease, concomitant chemotherapy or treatment with trastuzumab, mediastinal radiation, female sex, 28 -30 increased cardiac biomarker levels during and after administration 31 and genetics. 32 In addition to the above-mentioned risk factors, age plays an important role in anthracycline-induced cardiotoxicity, with pediatric and elderly patients being most at risk (Figure 1). 30 Why extremes of age are most susceptible to cardiotoxicity is not entirely clear. However, the biology may be fascinating and insights into mechanisms may lead to preventive and treatment strategies for patients.
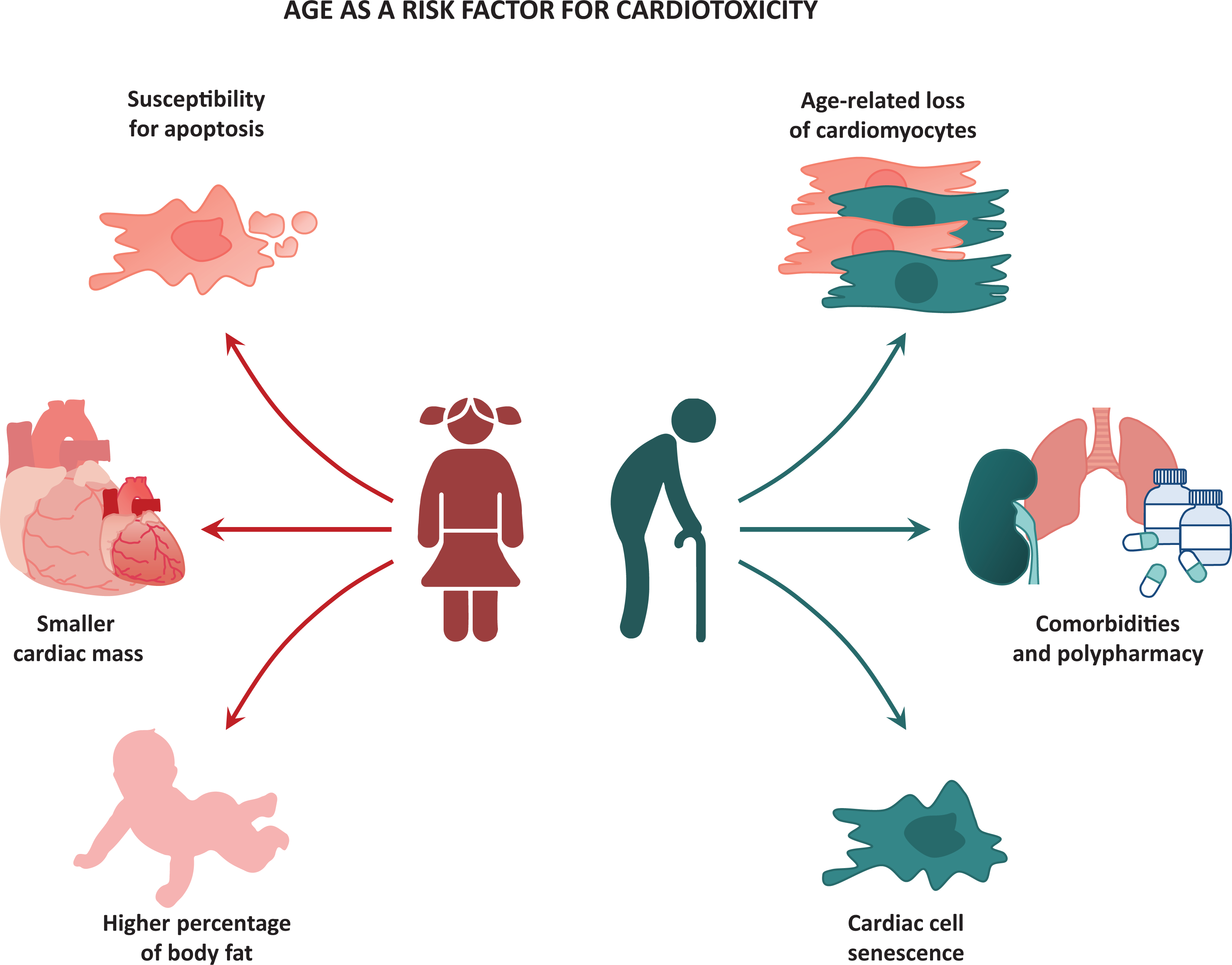
Age as a risk factor for anthracycline-associated cardiotoxicity.
Young Age as a Risk Factor for Cardiotoxicity
In children, anthracycline therapy can cause a decrease in left ventricular mass and wall thickness, increases in afterload and reduction of ventricular mass, sometimes called Grinch syndrome. 33 This can eventually result in asymptomatic LV dysfunction or congestive HF, 16 affecting 57% and 16% of the children, respectively, with the youngest children (<4 years) being most at risk. 29
Differences in sensitivity for anthracyclines between children and non-elderly adults might partly be due to the lipophilic nature of anthracyclines. Rodvold et al. showed that the clearance of anthracyclines is reduced with increased fat percentage. 34 The higher percentage of body fat in (younger) children 35 might result in higher anthracycline levels in blood and non-adipose tissues. 29 Additionally, younger children have a smaller cardiac mass, thus the impact of anthracyclines on myocardial growth inhibition might be greater. 29
Advanced Age as a Risk Factor for Cardiotoxicity
In the last years, the elderly population (≥65 years of age) has increased exponentially, making oncologists face the unpredictability of chemotherapeutic effects in a large cohort of the cancer population. Studies have shown that elderly are more at risk for cardiotoxicity in comparison with their younger counterparts, even at lower cumulative doses of anthracyclines. 19 This risk is even increased with 1.6 to 6.8-fold according to the American Society of Clinical Oncology (ASCO) Guidelines. 36
One of the reasons the old heart is particularly vulnerable to cardiotoxicity by chemotherapy is probably the age-related loss of cardiomyocytes. This subsequently leads to the decrease of myocardial volume, which is correlated to increased CV events. 37,38 It has also been shown that pharmacokinetics of anthracyclines alter during aging, resulting in significant increased doxorubicin concentrations, particularly evident in the heart. 39,40
Aging in general is often accompanied by the development of comorbidities, including hypertension, high cholesterol 41,42 and diabetes mellitus, 43 which directly translate in a higher number of prescribed medications. 44,45 Some retrospective studies suggest that age, comorbidities and functional performance account for a reduced tolerance of chemotherapy and lead to worse outcomes, even if the cancer responds equally to treatment. 46,47 Although drug-related problems are also common in the general elderly population, older cancer patients show more severe toxicities compared to age-matched individuals. 48 In elderly cancer patients, the risk factors of cardiotoxicity can be recapitulated as a “snowball effect”, as described by Reddy et al.: the diagnosis of cancer in combination with age-related factors forms the snowball, the cancer treatment and toxicity effects on peripheral tissues enlarge the snowball and set it in motion, to cause CV morbidity and mortality, eventually. 38
The fear for cardiotoxicity in elderly patients leads to other issues: under treatment and under representation in clinical trials. Although it has been shown that optimal therapy effectively decreases symptoms and improves quality of life and overall survival, 20 -22,49 older patients are often treated based on age and the number of comorbidities alone. 50 As a result, they often do not receive guideline-recommended treatment. In addition, elderly are substantially under represented in clinical trials, both in the cancer 51 and CV 52 field. Although the majority (63%) of US cancer patients was 65 years or older, elderly patients only accounted for approximately 25% in trials between 1993 and 1996. 51 Elderly patients were considered less eligible for cancer trials and physicians indicated age alone as a reason to exclude patients from trials. 53,54 Understanding the molecular mechanisms of cardiotoxicity in elderly will not only reduce cardiotoxicity itself, but will also contribute to optimal treatment in this patient population.
Molecular Mechanisms of Cardiotoxicity
Multiple mechanisms have been proposed for chemotherapy-induced cardiotoxicity, most of which suggest a strong correlation between cardiotoxicity and cardiac cell death, in particular cardiomyocyte death. 55 Apoptosis, 56 autophagy 57 and necrosis 58 are probably the best known cell death mechanisms, but necroptosis, 59 pyroptosis 60 and ferroptosis 61 have also been described to contribute to anthracycline-induced cardiac cell death. Although quite a number of cardiotoxicity-pathways have been studied extensively, the interplay between all pathway proteins is still not fully clear.
The reason why the heart (and not other organs) is particularly susceptible for anthracycline toxicity is not completely understood. However, it has been postulated that the abundancy of mitochondria present in cardiomyocytes—and the attractive power of anthracyclines to these mitochondria—contribute greatly to this. 62 -64 Additionally, cardiac tissue only displays a low level of anti-oxidant enzymes and one of the main anti-oxidants, glutathione, is decreased in the presence of anthracyclines, 65 resulting in an impaired defense against anthracycline-associated reactive oxygen species (ROS). 66 Anthracyclines do not only target DNA or mitochondria, but also directly damage important functional and structural cardiac proteins, including titin and dystrophin. This causes systolic and diastolic dysfunction and potentially increases the risk of dilated cardiomyopathy. 67
Molecular Mechanisms of Cardiotoxicity in Young Age
A recent paper provides an intriguing mechanism for increased risk of anthracycline-induced cardiotoxicity in young tissue compared to adult tissue. 68 In this paper, the investigators utilized BH3 profiling, a novel assay which directly measures the functional state of the mitochondrial apoptotic pathway in cells of several tissues. Young cells and adult cells of mice were exposed to titrated doses of BIM and BID, activator BH3-only proteins. This causes activation of BAX and/or BAK, mitochondrial permeabilization and subsequently triggers apoptosis. 69,70 Using BH3 profiling, the threshold of tissue undergoing apoptosis can be measured. Tissue can be classified as “primed” for apoptosis if mitochondria permeabilize readily upon BIM and BID titration or “unprimed” if a large reserve of anti-apoptotic proteins results in less sensitivity to apoptosis and apoptosis “refractory” if apoptosis can be blocked because insufficient amounts of cell death machinery is present. It is known that this degree of “priming” is correlated with sensitivity to chemotherapy in multiple cancers. 68,71
Interestingly, adult heart, kidney and brain tissue showed to be apoptosis “refractory”—cells of these tissues were almost completely insensitive to BIM and BID and lacked pro- and anti-apoptotic mitochondrial proteins. 68 Unlike adult tissues, tissues of newborn mice were found to be “primed” for apoptosis. Consistent with these findings, adult mice did not show signs of cardiac damage after doxorubicin treatment, while loss of both Bax and Bak1 was needed to reverse anthracycline-induced cardiotoxicity in young mice. This decrease in apoptotic potential in post-mitotic cardiomyocytes has been suggested previously as a main contributor to chemosensitivity. 72 Mass spectrometry proteome data showed equivalent protein expression in mice and human, suggesting comparable apoptosis regulation in both species. 68,73
An important shared feature between young, embryonic cells and cancer cells is the high degree of proliferation. 74 Therefore, enhanced priming might come at the expense of capacity to proliferate, making young cells and cancer cells relatively susceptible for apoptosis. 68 The oncogene c-Myc, a transcription factor implicated in proliferation and differentiation, is a potential modulator in the process of “cell priming” for apoptosis. c-Myc is known to directly regulate BAX expression 75,76 and shows higher expression in young heart, kidney and brain tissues compared with non-elderly adult tissues. Although not determined in heart tissue, it was confirmed that c-Myc can directly bind to pro-apoptotic genes Bax, Bim and Bid, indicating that c-Myc drives an apoptotically primed state (Figure 2). 68

Simplified representation of proposed mechanisms of anthracycline-associated cardiotoxicity in young age. In the presence of doxorubicin, the oncogene c-Myc directly binds to pro-apoptotic genes Bax, Bim and Bid, activating the pro-apoptotic effector proteins BAX and/or BAK. This results in mitochondrial outer membrane permeabilization and subsequent release of cytochrome c from the mitochondrion. In the cytosol, cytochrome c forms complexes with APAF1 and caspase-9 to form an apoptosome and induce apoptosis. APAF1, apoptotic protease activating factor 1; BAK, BCL-2 antagonist/killer 1; BAX, BCL-2-like protein 4; BIM, BCL-2-like protein 11; BID, BH3 interacting-domain death agonist; DOX, doxorubicin; MOMP, mitochondrial outer membrane permeabilization.
Molecular Mechanisms of Cardiotoxicity in Advanced Age
A recent paper emphasizes that cellular senescence—the complex phenomenon in which cells cease to divide—might alleviate chemosensitivity in non-cancerous tissues in elderly subjects. 77 It is known that senescent cells accumulate during aging and contribute to age-related dysfunction of several organ systems, including the heart. The underlying molecular pathways include accumulation of DNA, telomere shortening and increased expression of cell cycle inhibitors p16INK4a and p53. 78,79 Senolytics—drugs that selectively induce apoptosis of senescent cells—have shown to delay the onset of age-associated disorders in mice. 80,81 Therefore, cardiac senescence might contribute to anthracycline-induced cardiotoxicity, even years after last treatment. 82
It has been shown previously that several chemotherapeutic agents can induce senescence in peripheral tissues. These therapy-induced senescent (TIS) cells exhibit similar characteristic changes as can be seen in aged cells. In fact, neonatal rat cardiomyocytes that are exposed to doxorubicin show increased expression of senescence-associated β-galactosidase and decreased telomerase activity. 82 In a study by Demaria et al senescence was induced in mice and human fibroblasts in vitro and in several organs in mice in vivo by exposing them to doxorubicin. 77 TIS cells showed increased expression of pro-inflammatory senescence-associated secretory phenotype (SASP) genes in several tissues and higher blood plasma levels of pro-inflammatory cytokines, indicating local and systemic inflammation. Additionally, doxorubicin treatment resulted in decreased cardiac function, most likely caused by inflammation and/or cardiac senescence. Contrary to popular belief, the majority of cardiac senescent cells were found to be endothelial cells and cardiac fibroblasts, but not cardiomyocytes. Interestingly, clearing of these senescent cells in vivo, using a p16-3MR transgenic mouse model, prevented the development of chemotherapy-associated adverse reactions. These results were confirmed by human data, showing that expression of the cell cycle inhibitor p16INK4a in T-cells predicted for chemotherapy-induced fatigue in breast cancer patients. 77,83
Telomere shortening and dysfunction—processes that often accompany aging—may also contribute to increased susceptibility for chemotherapy-induced cardiotoxicity in elderly and may enhance cellular senescence. Sahin et al. proposed that telomere dysfunction activates the tumor suppressor gene p53, resulting in repression of PGC-1α and PGC-1β, main regulators of mitochondrial biogenesis. Tert-/- mice (telomerase deficient with severe telomere dysfunction) showed decreased expression levels of PGC-1α and PGC-1β and their downstream gene pathways in cardiac tissue. Additionally, a reduction in mitochondrial DNA content and density was seen, indicating a link between telomere function and mitochondrial biology. Subsequently, Tert-/- mice were treated with doxorubicin to study the effect of anthracyclines in telomere dysfunction. Anthracycline treatment resulted in a significant decreased cardiac function, as shown by fractional shortening (FS), which could be partially saved by knock-out of p53. The telomere-p53-PGC-mitochondria axis may therefore contribute significantly to cardiac failure after chemotherapy treatment (Figure 3). 84

Simplified representation of proposed mechanisms of anthracycline-associated cardiotoxicity in advanced age. In the presence of doxorubicin, the processes of cellular senescence and telomere dysfunction are induced and/or enhanced. Senescent cells show increased expression of pro-inflammatory senescence-associated secretory phenotype genes and higher levels of pro-inflammatory cytokines in plasma. Telomere dysfunction results in activation of the tumor suppressor gene p53, which represses PGC-1α and PGC-1β transcription, thereby inhibiting downstream protein targets and disturbing mitochondrial biogenesis. Mitochondrial dysfunction results in production of reactive oxygen species, reduction in oxygen consumption and reduction in ATP synthesis, thereby enhancing cellular senescence. CXCL10, C-X-C motif chemokine ligand 10; DOX, doxorubicin; ERRα, estrogen-related receptor alpha; IL-1α, interleukin-1 alpha; MMP3, matrix metalloproteinase-3; PGC-1α, peroxisome proliferator-activated receptor gamma coactivator 1-alpha; PGC-1β, peroxisome proliferator-activated receptor gamma coactivator 1-bèta; PPARα, peroxisome proliferator-activated receptor alpha; NRF-1, nuclear respiratory factor 1; ROS, reactive oxygen species; SASP, senescence-associated secretory phenotype; TFAM, mitochondrial transcription factor A.
Preventive Strategies
It is known that anthracycline-based regimens result in significantly higher subclinical cardiotoxicity and cardiac event rates compared to non-anthracycline treatment (OR 6.25 [95% CI 2.58-15.13]; OR 5.43 [95% CI 2.34-12.62], respectively). 85 Despite growing interest and expansion of new therapeutic strategies, it is still unimaginable that conventional anti-cancer drugs, including anthracyclines, can be completely replaced in the near future. 86 Therefore, determination of toxicity and development of prevention strategies are essential for optimal cancer treatment and for minimizing toxic side-effects, especially in the most vulnerable patient populations, the young and the old. 87 Beside reduction of the total cumulative anthracycline dose and continuous anthracycline infusion, other strategies for cardioprotection in anthracycline treatment regimens include the following: 1) choice of anthracycline agent, 2) adjustment to a liposomal anthracycline formulation and 3) administration of cardioprotective agents.
Choice of Anthracycline Agent
Most oncology treatment protocols assume that the hematologic toxicity of anthracyclines is equivalent to the cardiotoxicity that can occur as a devastating side-effect of the same compound. 88 However, it has been suggested that not all anthracycline agents entail the same risk and cause cardiotoxicity to the same degree. For example, studies imply that daunorubicin might be less cardiotoxic compared to other types of anthracycline agents. 26,89 A multicenter cohort study showed a higher risk for development of cardiomyopathy in doxorubicin-treated patients compared to daunorubicin-treated ones (HR 4.6 vs 2.8) 22 years after diagnosis. 88 In contrast, studies on the cardiotoxicity profile of epirubicin are inconclusive. While some studies suggest reduced cardiotoxicity by epirubicin in comparison with doxorubicin, 90,91 others mention isoequivalent toxicity profiles of the 2 anthracycline agents. 88 Beside these conventional therapies, efforts have been made to reduce cardiotoxicity by modifying the chemical structure of anthracyclines. Probably the most interesting agent is N-benzyladriamycin-14-valerate (AD 198). This chemically modified anthracycline has shown to be less cardiotoxic compared to doxorubicin, without losing its anti-tumor efficacy. 92
Liposomal Anthracycline Formulations
By encapsulating anthracyclines in liposomes—vesicles with a lipid bilayer—they are prevented from delivery to tissues with tight capillary junctions, including the heart. In this manner, cardiotoxicity may be reduced and the blood circulation time optimized. Currently, multiple pegylated liposomal anthracycline formulations are approved by the FDA or in clinical trials. 93 Randomized controlled trials (RCT) have shown that liposomal anthracyclines are less cardiotoxic compared to conventional formulations, 91,94 and have also shown to be beneficial in elderly patients. 93,95 -97 In a multicenter phase II trial in elderly (>60 years) with diffuse large B-cell lymphoma, patients received standard RCHOP therapy with pegylated liposomal doxorubicin instead of the “regular” formulation. This treatment regimen showed an acceptable level of cardiotoxicity (4% with high-grade cardiac events; 20% with subclinical decline in ejection fraction) and comparable oncologic efficacy to other studies using conventional doxorubicin protocols. 95 However, median follow-up time of the patients was 46 months and late cardiotoxic effects could therefore not be ruled out. 12,13 A randomized phase III trial in elderly patients (aged 60-75 years) with newly diagnosed secondary acute myeloid leukemia showed improved overall survival when treated with CPX-351 (liposomal encapsulation of cytarabine and daunorubicin) compared to standard-of-care (9.56 versus 5.95 months respectively, HR 0.69 [95% CI 0.52-0.90], P = 0.003). 93 In contrast, RCT data are lacking in the pediatric cancer population, resulting in no recommendation of liposomal doxorubicin in children. Although most studies with liposomal doxorubicin focused on the pegylated form, it is known that this formulation might display hand-foot syndrome, making the non-pegylated doxorubicin likely to be better tolerable. 95
Cardioprotective Agents
In the last decade, efforts have been made to reduce cardiotoxicity by development of cardioprotective agents as addition to anthracycline therapy. Neurohormonal antagonists, including angiotensin-converting enzyme inhibitors (ACEi), angiotensin II-receptor blockers (ARBs) and β-blockers, have shown to be highly effective in HF treatment. Therapy has shown to reverse anthracycline-induced cardiotoxicity in animal models 98 and to be beneficial as (primary) prevention in patients. 99,100 A study of Cardinale et al. in 2,625 patients showed substantial recovery of cardiac function with ACEi and β-blocker treatment, after anthracycline-associated decrease in left ventricular ejection fraction (LVEF). 101 Another study of the same research group suggested that patients can be stratified upon Troponin levels, to identify patients at risk for chemotherapy-induced cardiotoxicity. Prophylactic neurohormonal antagonist therapy in these high-risk patients should be initiated directly after anthracycline treatment. 31 However, the vast majority of studies included a relatively “healthy” adult patient population treated with lower cumulative anthracycline doses, suggesting that future trials should focus more on high-risk populations, including very young and very old patients.
Since ROS formation is considered one of the main drivers of drug-induced cardiotoxicity, natural anti-oxidants were considered potential cardioprotective drugs. Vitamin E and N-acetylcysteine failed to protect from cardiotoxicity in long-term clinical trials 17 and also experiments with ROS scavengers failed to rescue cardiomyocytes from cardiotoxicity. 102 Although some compounds with anti-oxidants activities (i.e. berberine and carvedilol) have shown to be beneficial, 99,103 the contribution of oxidative stress in drug-induced cardiotoxicity is increasingly questioned. Some even state there is “the need to move beyond the oxidative stress hypothesis.” 17
To date, dexrazoxane is the only cardioprotective agent approved by the Food and Drug Administration (FDA) and European Medicines Agency (EMA). Although the exact mechanisms remain elusive, dexrazoxane is thought to chelate iron and inhibit free radical formation in cardiac tissue. Because of its protective effect on acute cardiotoxicity, dexrazoxane is currently widely applied in clinical practise. 91,104 -106 Dexrazoxane leads to reduction of cardiac injury, as reflected by Troponin T levels 107 and echocardiographic parameters. 108 A multicenter randomized phase III study found a significant reduction in cardiac events (39% vs 13%) and congestive HF (11% vs 1%) with dexrazoxane in high-risk patients previously treated with anthracyclines. 104 Also in pediatric cancer patients, dexrazoxane has shown to be an effective cardioprotectant, 109,110 with the greatest effect seen in females. 108 However, in 2007 a study was published that claimed that dexrazoxane might increase the incidence of secondary malignancies in patients with Hodgkin’s disease (HD). 111 Despite this article was criticized for its statistical analyses, 112,113 physicians did not universally support the use of dexrazoxane in children. Since 2017, the contraindication for dexrazoxane treatment in children and adolescents was lifted by the EMA. In contrast to the abundancy of data on dexrazoxane in children, data on elderly patients is scarce, 114 although this high-risk population might benefit highly from this.
Conclusion
It should be emphasized that modern cancer medicine leads to increased survival rates, with the population of cancer survivors growing each year. Even with improved techniques and treatment options, cardiotoxicity still is a limiting factor in chemotherapy treatment. Patients in extremes of age are especially at risk, making age an important consideration in Cardio-Oncology. In this review we discussed that the underlying mechanisms causing anthracycline-associated cardiotoxicity, might differ between the young and the old. In children, the heart may be particularly vulnerable for chemotherapy-induced cardiotoxicity due to the enhanced “priming” of cardiac cells for apoptosis, while in elderly, chemosensitivity seems to be driven by cardiac senescence and telomere dysfunction. Anthracyclines might even target different cardiac cell types during aging. Although several drugs have shown to be beneficial in minimizing cardiotoxicity effects, studies are usually conducted in relatively “healthy” adults. The ability to extrapolate this evidence to children or elderly is questionable. Therefore, more research is needed to better understand age-related processes concerning cardiotoxicity to optimize treatment and develop effective prevention strategies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Moslehi has served a consultant for Bristol-Myers Squibb, Takeda, Deciphera, AstraZeneca, Nektar, Audentes Therapeutics, TripleGene, Boston Biomedical, ImmunoCore, Janssen, and Myovant. The other authors report no potential conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Meijers was supported by funding from the Niels Stensen Fellowship and the Netherlands Heart Institute. Ms. Screever was supported by funding of the Junior Scientific Masterclass.
