Abstract
Guidelines for management of normotensive patients with acute pulmonary embolism (PE) emphasize further risk stratification on the basis of right ventricular (RV) size and biomarkers of RV injury or strain; however, the prognostic importance of these factors on long-term mortality is not known. We performed a retrospective cohort study of subjects diagnosed with acute PE from 2010 to 2015 at a tertiary care academic medical center. The severity of initial PE presentation was categorized into three groups: massive, submassive, and low-risk PE. The primary endpoint of all-cause mortality was ascertained using the Centers for Disease Control National Death Index (CDC NDI). A total of 183 subjects were studied and their median follow-up was 4.1 years. The median age was 65 years. The 30-day mortality rate was 7.7% and the overall mortality rate through the end of follow-up was 40.4%. The overall mortality rates for massive, submassive, and low-risk PE were 71.4%, 44.5%, and 28.1%, respectively (p < 0.001). Landmark analysis using a 30-day cutpoint demonstrated that subjects presenting with submassive PE compared with low-risk PE had increased mortality during both the short- and the long-term periods. The most frequent causes of death were malignancy, cardiac disease, respiratory disease, and PE. Independent predictors of all-cause mortality were cancer at baseline, age, white blood cell count, diabetes mellitus, liver disease, female sex, and initial presentation with massive PE. In conclusion, the diagnosis of acute PE was associated with substantial long-term mortality. The severity of initial PE presentation was associated with both short- and long-term mortality.
Keywords
Introduction
Acute pulmonary embolism (PE) is a common condition associated with significant morbidity and mortality. 1 Clinical guidelines recommend risk stratification of acute PE according to hemodynamic status and presence of right ventricular (RV) enlargement or strain2–4 into three categories: massive/high-risk, submassive/intermediate-risk, and low-risk PE.
While extensive data are available on short-term mortality after acute PE,5,6 fewer studies have assessed long-term mortality after an acute PE and there are limited data associating initial presentation severity with long-term outcomes. Furthermore, the long-term mortality of subjects with submassive PE has not been compared with those with low-risk PE.
We sought to determine the long-term mortality among subjects hospitalized for an acute PE and to establish whether the severity of the initial PE presentation is associated with long-term mortality. We also sought to determine the causes and baseline predictors of long-term mortality in this cohort.
Methods
Study population
We utilized hospital record data and International Classification of Diseases, Ninth Revision codes (ICD-9-CM codes 415.1x) to identify patients diagnosed with acute PE between 2010 and 2015 at the University of Toledo Medical Center. All identified cases were confirmed to have the diagnosis of acute PE on the basis of a computed tomography pulmonary angiogram (CTPA) or a moderate or high probability ventilation-perfusion (V/Q) scan that was available for review within our electronic medical record. We excluded subjects who were missing a diagnostic study within our system, such as those transferred from another facility who did not have a diagnostic study uploaded into our system. We also excluded subjects who were missing variables needed for classification of acute PE severity. Baseline demographic, clinical, laboratory, and imaging variables were abstracted from the electronic medical record and de-identified. Institutional Review Board approval was obtained and the informed consent requirement was waived.
Classification of acute PE severity
Baseline PE severity was categorized as follows: massive/high-risk PE, submassive/intermediate-risk PE, and low-risk PE. Massive PE was defined as systolic blood pressure (SBP) ⩽ 90 mmHg or requirement for vasopressor therapy or presentation with cardiac arrest. Submassive PE was defined as SBP > 90 mmHg AND at least one of the following risk markers: right ventricular to left ventricular (RV:LV) diameter ratio > 0.9 OR troponin I > 0.40 ng/mL OR brain natriuretic peptide (BNP) level > 90 pg/mL. 2 Low-risk PE was defined as SBP > 90 mmHg and RV:LV ratio ⩽ 0.9, troponin I ⩽ 0.40 ng/mL and BNP ⩽ 90 pg/mL. The RV:LV diameter ratio was measured on echocardiogram and CTPA. If there was discrepancy between the two imaging modalities for classification, then the higher RV:LV diameter ratio was utilized for classification. For both imaging modalities, a four-chamber view was utilized and the RV and LV diameters were measured 1 cm from the tricuspid and mitral valve in a subannular plane, as previously described. 7
Endpoints
The primary outcome was all-cause mortality. Mortality data were obtained from the Centers for Disease Control (CDC) National Death Index (NDI). Short-term mortality was defined as death within 30 days of presentation and long-term mortality was defined as death from day 31 through end of follow-up. Causes of death were adjudicated by a panel of investigators (RG, MR, and ZA) who reviewed abstracted death certificate data from the NDI and classified each death into a primary category (Cardiac Disease, PE, Malignancy, Respiratory Disease, Infection/Sepsis, Cerebrovascular Disease, Dementia, Vascular Disease, and Other causes).
Statistical analysis
Demographic, clinical, laboratory, and imaging variables appear as continuous or categorical data types. Continuous data were tested for goodness-of-fit to the normal distribution using the Shapiro–Wilk test. If not normally distributed, the log-transformation of the variable was assessed for normality. Continuous data are presented as mean ± SD of the untransformed or log-transformed version or, if neither were normally distributed, as median with IQR. Categorical data are presented as frequencies with percentages. Comparisons of continuous data were evaluated using analysis of variance (ANOVA) or the Kruskal–Wallis rank-sum test for three-way comparisons, and two-way comparisons were evaluated using two-sample t-tests or the Wilcoxon rank-sum test. For categorical variables, contingency table analysis with the chi-squared test was used, or, if the frequency of counts for some factors was low (⩽ 5), Fisher’s exact test was used to compare groups. Unadjusted survival estimates are presented using the Kaplan–Meier method. Log-rank testing was employed to compare the mortality rates between groups. Landmark analysis was used as a means to explore the short-term and long-term mortality rates in separate analyses using a 30-day cut-off point. Only patients alive at the landmark cut-off point were included in the separate long-term survival analysis. Additionally, for the landmark analysis, long-term survival was only compared for submassive and low-risk PE since there were too few remaining patients in the massive PE subgroup. Cox proportional hazards regression analysis was used to identify independent predictors of mortality. Using step-wise selection with Akaike information criteria (AIC) and clinically relevant variables, the following factors were entered into the Cox proportional hazards model: risk stratification of PE (massive, submassive, low risk), age, sex, body mass index (BMI), myocardial infarction (MI), congestive heart failure (CHF), peripheral vascular disease (PVD), dementia, liver disease, diabetes mellitus, chronic kidney disease (CKD) stages III–V, cancer, tobacco use, white blood cell count (WBC), hemoglobin, creatinine, major bleeding, and transfusion. Model diagnostics were performed using the Cox–Snell residual plot to check for goodness-of-fit. The extended Cox model was used to test for interaction among model predictors and time. The p-values for all covariate–time interactions were greater than 0.05, confirming the null hypothesis assumption of proportional hazards, and indicating that the fitted Cox regression hazard models are adequate. All statistical analyses were performed using SAS version 9.3 (SAS Institute, Cary, NC, USA) and R version 3.00 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Baseline characteristics
We studied 183 subjects hospitalized with acute PE from 2010 to 2015. Baseline demographics, clinical characteristics, vital signs, and laboratory values are shown in Table 1. The median age was 65 years. Comorbid conditions were frequent, including CHF, prior MI, chronic obstructive pulmonary disease (COPD), diabetes mellitus, and tobacco use. The population was categorized as massive, submassive, and low-risk PE: 3.8% of subjects met the definition of massive PE, 65.0% submassive PE, and 31.1% low-risk PE. In general, higher acute PE severity was associated with older age, higher Charlson Comorbidity Index, higher white blood cell count (WBC), higher troponin and BNP levels, and longer length of stay (Table 1). When comparing subjects with submassive versus low-risk PE, subjects with submassive PE had older age at presentation and a higher burden of comorbid conditions including more frequent rates of CHF, PVD, and prior cerebrovascular accident (CVA). The majority of the study population was treated with conventional anticoagulation therapy alone. Systemic thrombolysis was administered to three patients (1.6%). Five patients (2.7%) underwent catheter-directed therapy. CTPA was the diagnostic study for PE in most (81%) subjects and a V/Q scan was used for the remainder (19%). When compared with low-risk PE, the RV:LV ratio was increased among subjects with massive and submassive PE by both echocardiogram and CTPA (Table 2).
Baseline demographic and laboratory values.
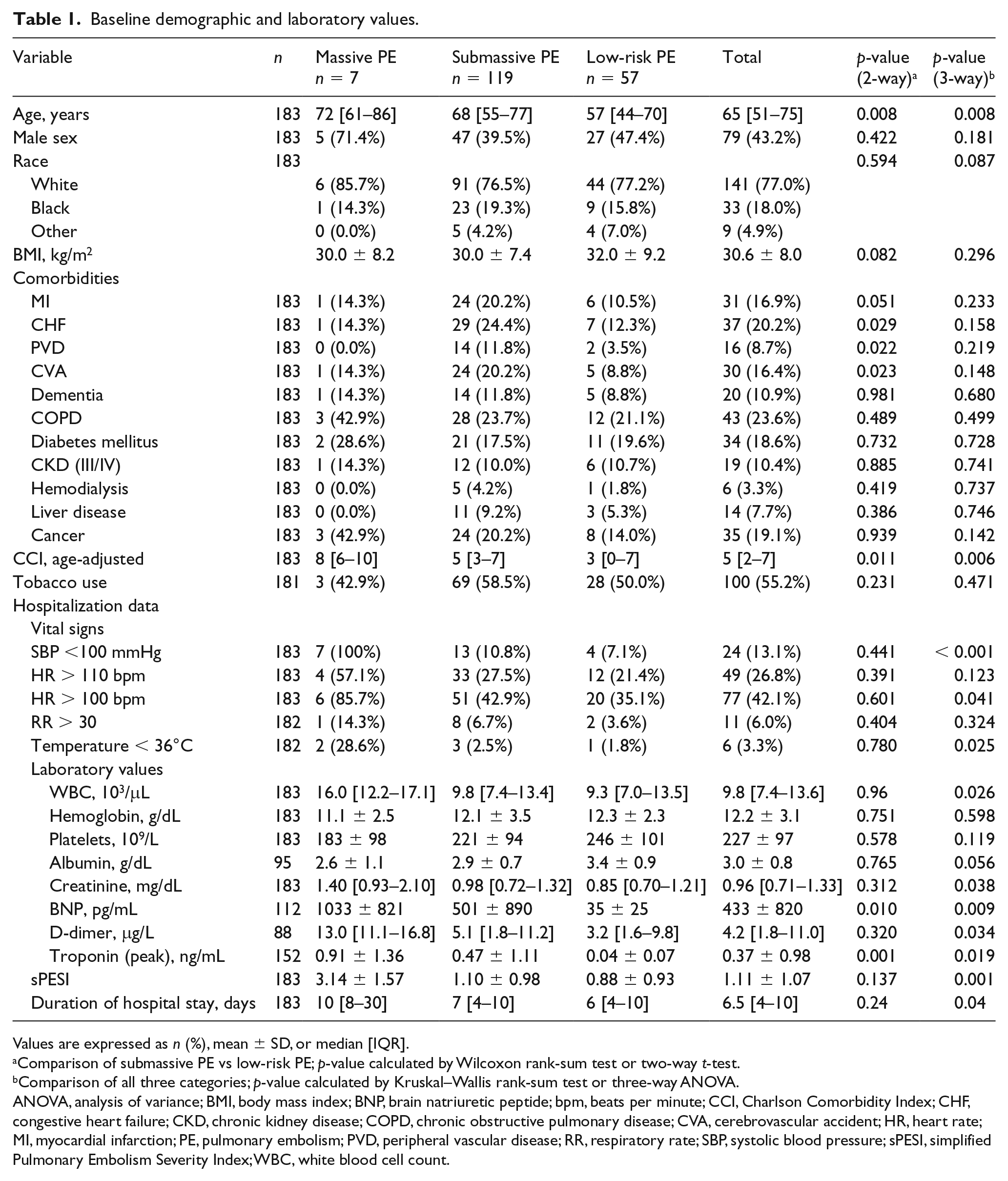
Values are expressed as n (%), mean ± SD, or median [IQR].
Comparison of submassive PE vs low-risk PE; p-value calculated by Wilcoxon rank-sum test or two-way t-test.
Comparison of all three categories; p-value calculated by Kruskal–Wallis rank-sum test or three-way ANOVA.
ANOVA, analysis of variance; BMI, body mass index; BNP, brain natriuretic peptide; bpm, beats per minute; CCI, Charlson Comorbidity Index; CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; CVA, cerebrovascular accident; HR, heart rate; MI, myocardial infarction; PE, pulmonary embolism; PVD, peripheral vascular disease; RR, respiratory rate; SBP, systolic blood pressure; sPESI, simplified Pulmonary Embolism Severity Index; WBC, white blood cell count.
Imaging findings from subjects with massive, submassive, and low-risk PE.
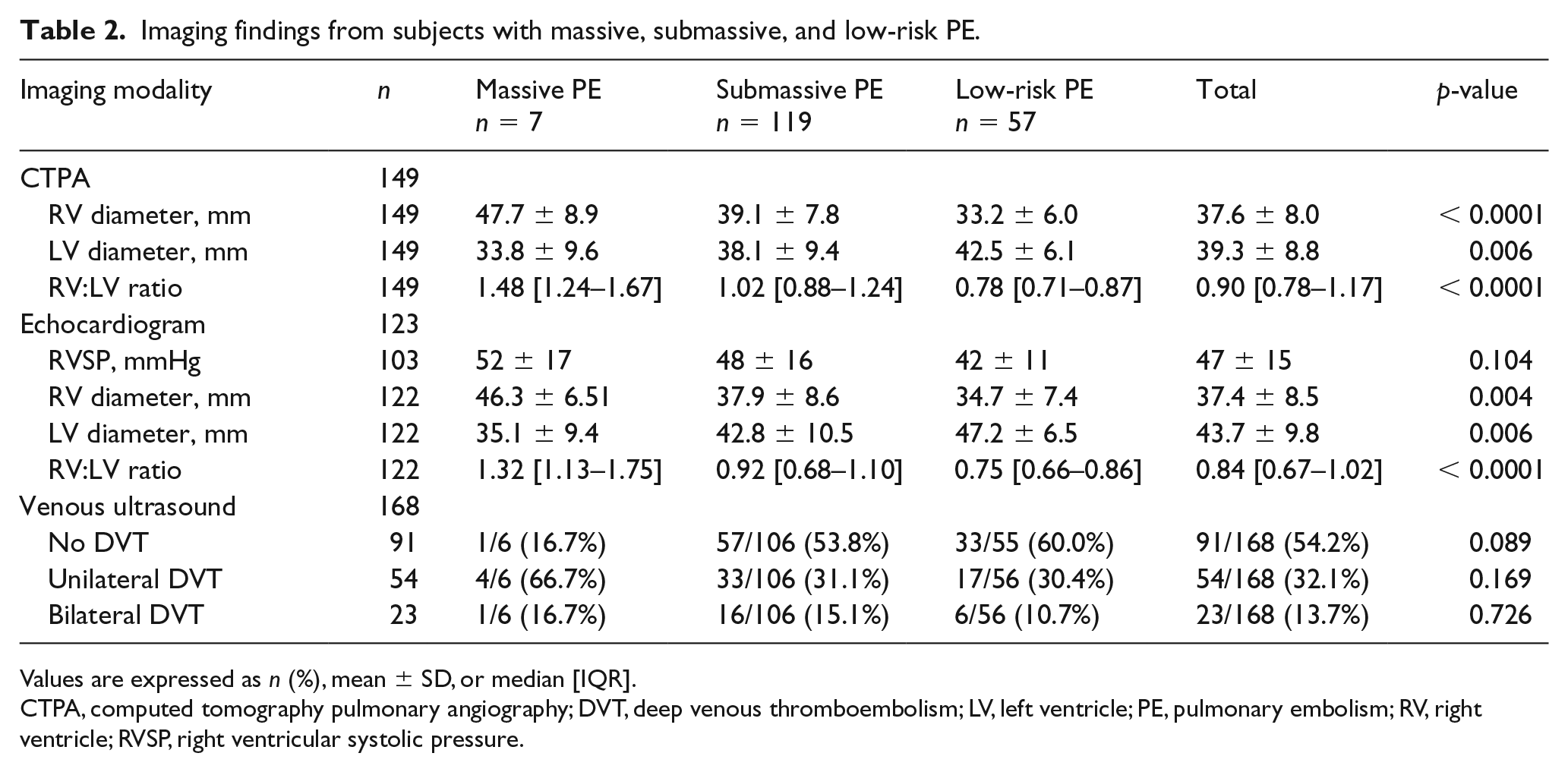
Values are expressed as n (%), mean ± SD, or median [IQR].
CTPA, computed tomography pulmonary angiography; DVT, deep venous thromboembolism; LV, left ventricle; PE, pulmonary embolism; RV, right ventricle; RVSP, right ventricular systolic pressure.
Short-term, long-term, and overall mortality rates
Overall mortality over a median follow-up of 4.1 [IQR 1.1– 4.9] years was 40.4% (Figure 1A). Unadjusted survival rates differed significantly according to whether patients presented with massive, submassive, or low-risk PE, where the respective mortality rates were 71.4%, 44.5%, and 28.1% (p < 0.001) (Figure 1B). In a landmark analysis which divided the follow-up period into short- (⩽ 30 days) and long-term (from 31 days until study end) time periods, mortality rates differed significantly among PE severity categories (Figure 2). Short-term mortality in the massive, submassive, and low-risk subgroups was 57.1%, 6.7%, and 3.5%, respectively (log-rank p < 0.001). Long-term mortality in the submassive and low-risk PE groups was 40.5% and 25.5%, respectively (log-rank p = 0.032) (Figure 2). There were only three subjects remaining in the massive PE group beyond 30 days, and their survival was therefore not compared to those in the submassive or low-risk PE groups. Lastly, we further subdivided the submassive group into intermediate/low versus intermediate/high-risk groups and we found similar risk for long-term all-cause mortality (online Supplementary Figure 1). 2
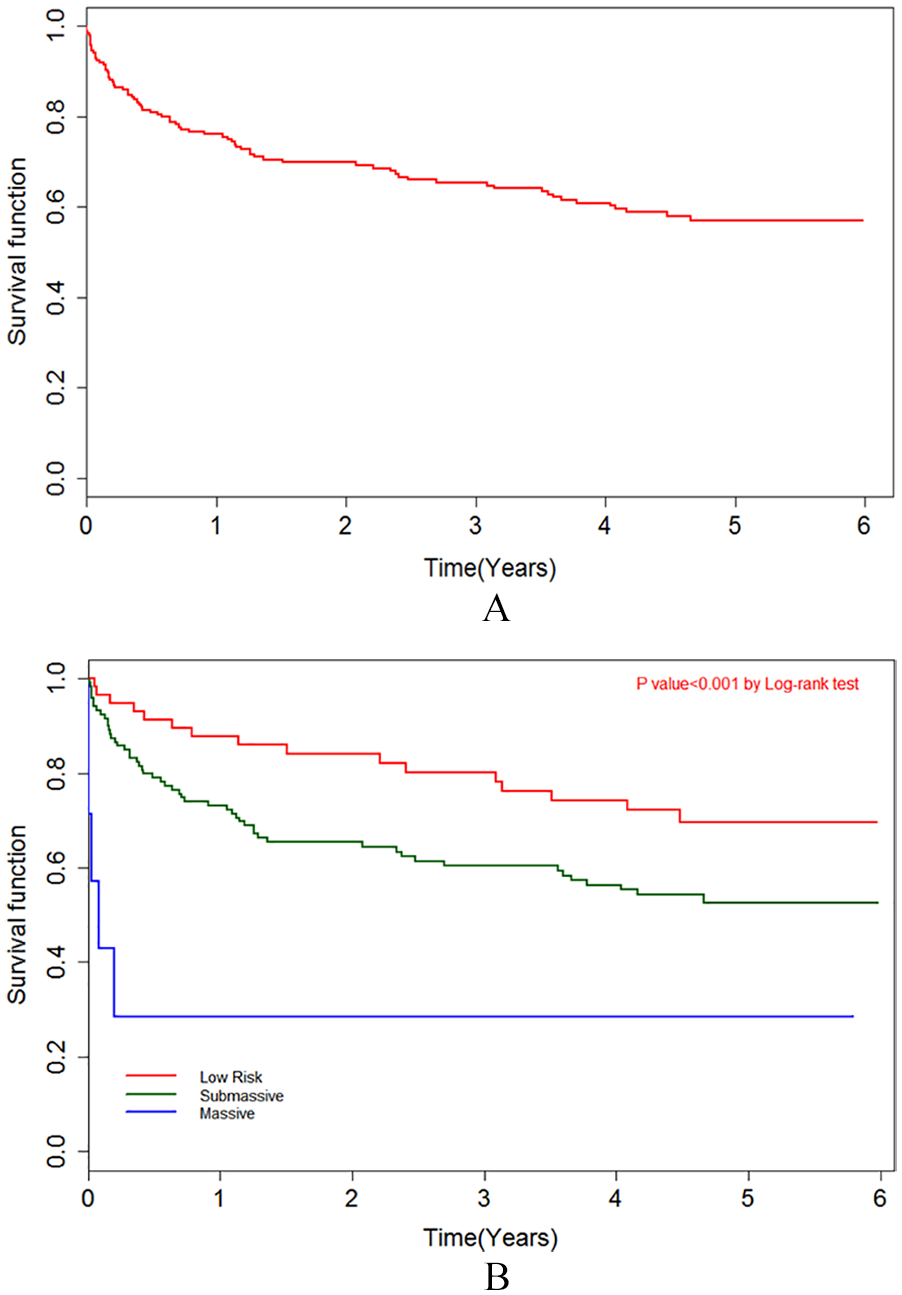
(A) Kaplan–Meier curve depicting all-cause mortality. (B) Kaplan–Meier curve depicting all-cause mortality for massive, submassive, and low-risk PE.
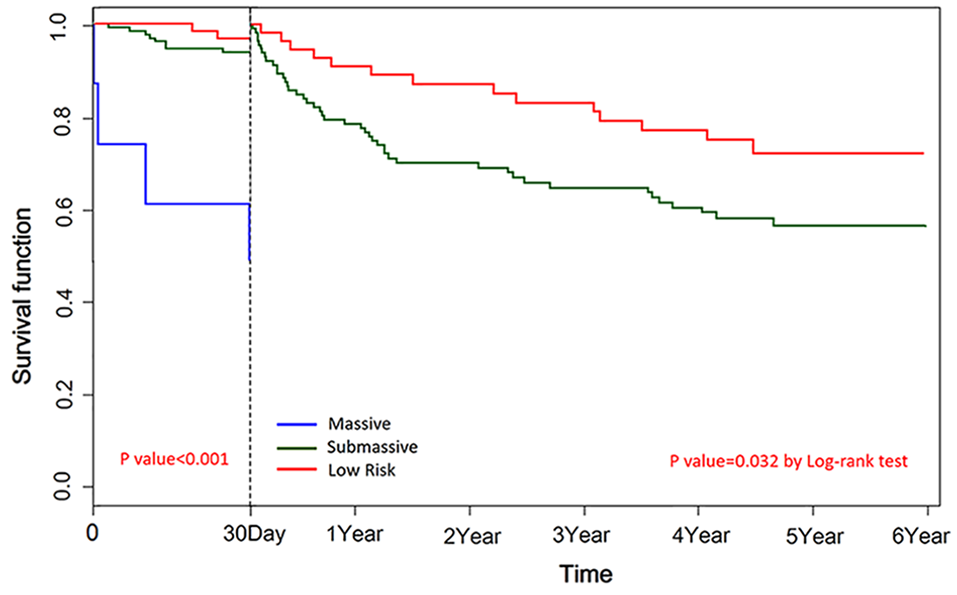
Landmark analysis dividing mortality into short-term mortality (within 30 days) and long-term mortality (31 days through end of study period).
Causes of death
The most frequent causes of death were malignancy (29.7%), cardiac disease (14.8%), respiratory disease (13.5%), and PE (10.8%) (Figure 3). Cause of death was also tabulated separately for short- and long-term time periods. PE accounted for more short-term deaths and malignancy for more long-term deaths (online Supplementary Table 1). Since subjects with known malignancy at baseline have a different prognosis than those without known malignancy, we next excluded subjects with known malignancy. In this analysis, we found a similar relationship between acute PE severity and long-term mortality among subjects with no known malignancy at the time of their PE presentation (online Supplementary Figures 2A and 2B).
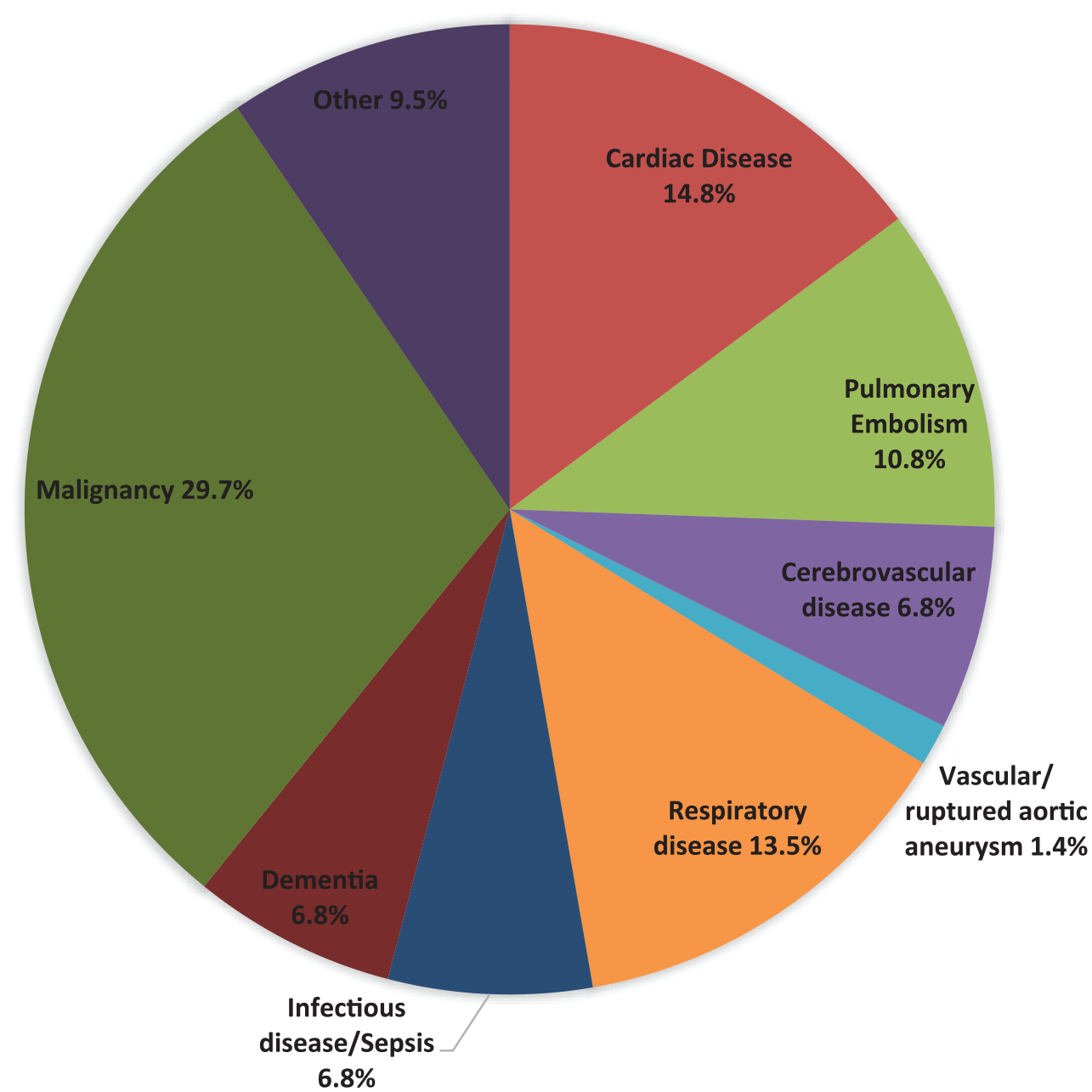
Causes of death during follow-up after acute PE.
Independent predictors of all-cause mortality
Predictors of all-cause mortality in an unadjusted analysis are shown in Figure 4A. Next, we performed a multivariable adjusted analysis. In a Cox proportional hazards model, all-cause mortality was significantly higher in the massive PE group than in the low-risk group (hazard ratio = 6.82; 95% CI 2.12–21.91; p = 0.01). Additional independent predictors of all-cause mortality include age, WBC, malignancy, diabetes mellitus, liver disease, and female sex (Figure 4B). Higher BMI was associated with a lower risk of mortality. The adjusted overall mortality in the submassive PE group (44%) was numerically higher than in the low-risk group (28%), but was not statistically significant in the multivariate model (hazard ratio = 1.43, p = 0.27) (Figure 4B). Troponin was not an independent predictor of long-term mortality in the multivariable model.
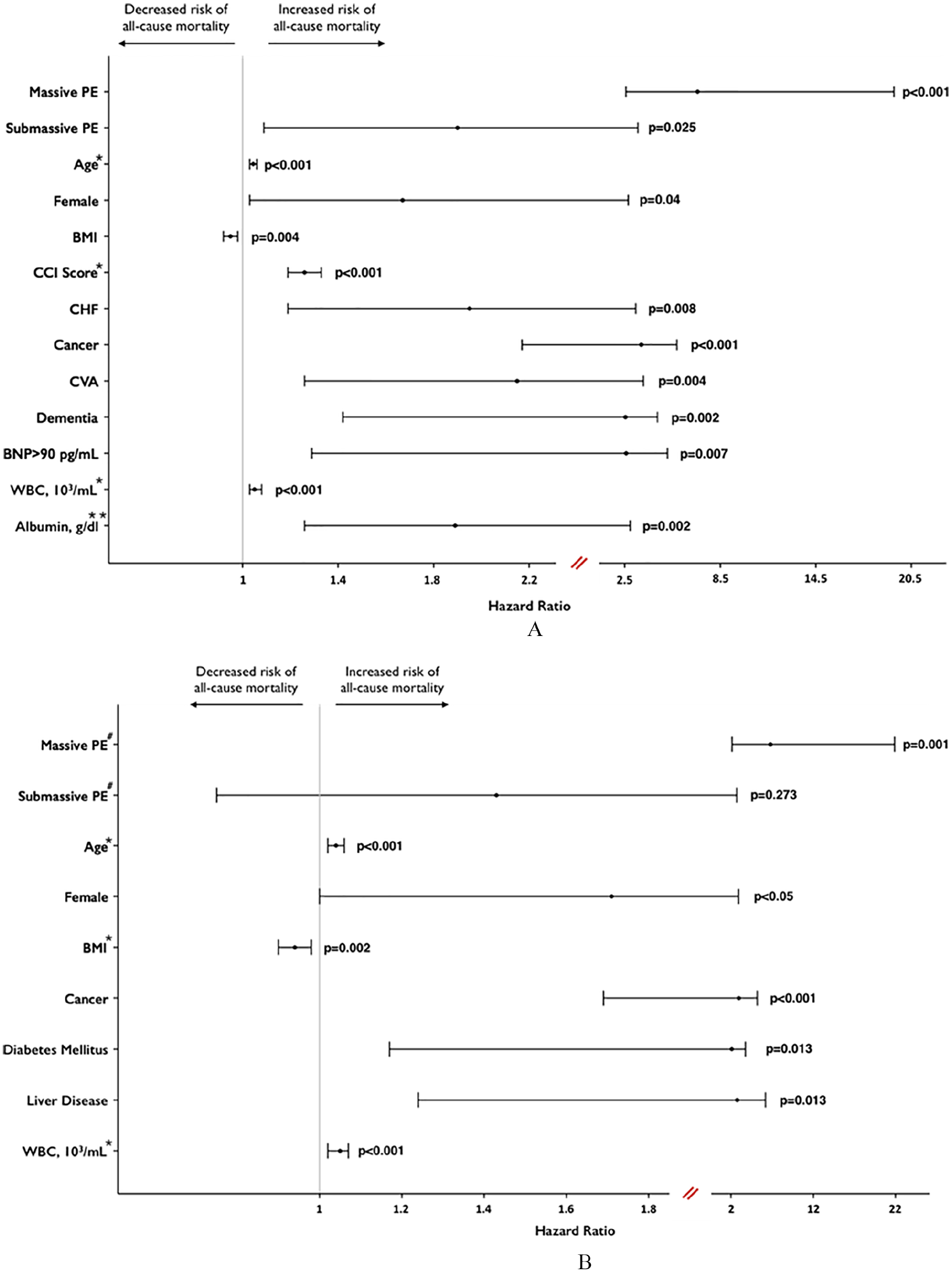
(A) Univariate predictors of all-cause mortality in long-term follow-up after acute PE. (B) Predictors of all-cause mortality in long-term follow-up after acute PE in a multivariate model.
Discussion
In this study of patients who presented with acute PE and were followed for a median of 4.1 years, we observed a relatively high overall mortality rate (40.4%) and determined that initial risk stratification of patients into low, intermediate, and high-risk categories was associated with both short- and long-term mortality.
Our findings are consistent with others, which have demonstrated substantial long-term mortality after a diagnosis of acute PE. In the control arm of the Prévention du Risque d’Embolie Pulmonaire par Interruption Cave (PREPIC) study (n = 200), which consisted of acute PE subjects treated with anticoagulation, the overall mortality was 51% at 8 years. 8 Similarly, a retrospective study of patients with acute PE (n = 1023) found an overall mortality rate of 35.5% during a mean follow-up of approximately 4 years. 9 Our findings are also similar to prior studies that have demonstrated short-term mortality is predicted by initial PE severity, especially among those with massive PE.6,10–18 However, little is known about the long-term mortality following acute submassive PE when compared with other PE risk groups. Recent work has focused on risk stratification of normotensive acute PE patients using imaging assessment of RV size12,13 and using biomarkers of RV injury (troponin) or RV strain (BNP), but again, these studies are largely focused on short-term risk stratification.14–18 The current study provides important new information regarding long-term mortality risk after an acute PE.
Causes of death
When we analyzed the CDC NDI to adjudicate the causes of death, the most frequent causes of death were malignancy, cardiac events (including MI and CHF events), respiratory diseases (e.g. pneumonia, COPD), PE, and stroke. Cancer-related mortality was the largest single category for long-term mortality, with about 29.7% of deaths related to malignancy. The relation between malignancy and PE mortality is well established. 19 The rate of cancer-related death was higher in our cohort, when compared with other cohorts. 9 This may relate to the high incidence of known malignancy at baseline, in addition to other factors associated with malignancy such as the high rate of smoking in our cohort. However, when we excluded patients with known cancer at baseline, we still found a similar trend for the association between initial PE severity and long-term mortality rates among the three different categories of PE severity (online Supplementary Figure 2B). Recent studies have assessed whether anticoagulation can reduce the risk of venous thromboembolism (VTE) and VTE-related mortality in patients with cancer and should inform preventive strategies for VTE among patients with cancer.20,21
A significant portion of long-term mortality was related to cardiovascular events such as MI, heart failure, and stroke. Given the frequency of cardiovascular mortality in long-term follow-up after acute PE, there may be opportunities for enhanced primary and secondary prevention of cardiovascular disease after a diagnosis of acute PE. In fact, there are opportunities to target both recurrent VTE risk and cardiovascular risk. For example, aspirin is known to reduce the risk of both MI and recurrent VTE. 22 Similarly, statins are well known to reduce cardiovascular events, and may also reduce incident and recurrent VTE events.23,24 Low-dose rivaroxaban appears to be beneficial for secondary prevention among subjects with atherosclerotic vascular disease and may reduce rates of VTE as well. 25 A focus on transitioning from acute treatment goals to long-term treatment goals may identify opportunities for long-term risk reduction.
Initial PE and recurrent PE-related mortality
In the short-term period, 4/14 (29%) of the mortalities were attributed to acute PE. All four of these deaths were in the massive PE group. Our prevalence of massive PE (~4%) is similar to that observed in other studies. Among 2392 patients with acute PE and a known initial blood pressure in the International Cooperative Pulmonary Embolism Registry (ICOPER), 108 (4.5%) had massive PE, using a definition that was similar to ours. 26
During follow-up, recurrent PE also caused about 7% of long-term deaths, those occurring after 30 days from initial PE presentation. Subjects with initial VTE will have average VTE recurrence rates of approximately 20–30% over 5 years.27,28 As demonstrated in the current study, some of these recurrent VTE events will be fatal. Recent studies have demonstrated good safety and efficacy with reduced dose, long-term anticoagulation for the prevention of recurrent VTE.29–31 It remains to be determined whether reduced dose, long-term anticoagulation can reduce long-term mortality rates in this population.
Comorbid conditions and predictors of mortality
We also found a greater burden of comorbid conditions and older age at presentation among subjects with submassive versus low-risk PE (Table 1). Although submassive PE was associated with increased mortality in the landmark analysis and the univariate analysis, submassive PE presentation was not an independent predictor of long-term mortality in the multivariate analysis. The older age at presentation and greater burden of comorbid conditions among subjects with submassive PE likely explain these findings. Interestingly, increased BMI was associated with reduced risk of all-cause mortality. These findings support the obesity paradox that has been previously demonstrated for subjects with VTE and for other conditions.32,33
Initial PE treatment and risk of long-term mortality
The majority of subjects in the current study were treated with anticoagulation. A PE Response Team (PERT) was initiated at our institution after the period of this study, and acute PE therapies have evolved significantly in recent years. Nonetheless, it remains to be determined if more aggressive acute PE therapies can impact long-term mortality risk. Prior studies have attempted to determine whether systemic thrombolysis can reduce short- or long-term mortality among subjects with submassive PE. The largest randomized controlled trial of intravenous thrombolysis versus anticoagulation alone for acute submassive PE, the PEITHO trial, has reported both short-term and long-term results. 5 The long-term mortality rates did not differ between subjects randomized to thrombolysis versus anticoagulation alone. 34 Our study demonstrates higher all-cause mortality rates compared with the PEITHO trial long-term follow-up. This is likely related to a higher burden of comorbid illness in our cohort, with the current cohort having higher rates of CHF, COPD, and other comorbid illnesses compared with the PEITHO trial population.
Overall, PE remains a significant cause of short-term mortality. Furthermore, as demonstrated in the current study, subjects diagnosed with an acute PE have a substantial long-term mortality risk. Further research is needed to improve both short-term and long-term outcomes following a diagnosis of acute PE.
Limitations
This is a single-center, retrospective, observational cohort study with all of the limitations inherent in this type of study. Given the retrospective study design, data were missing for some variables. The sample size for each variable is listed in Table 1 and Table 2. Ascertainment of cause of death based on death certificate information has known limitations and inaccuracies. 35 Nonetheless, the CDC NDI is a well-established method for ascertaining all-cause mortality and also allowed us to categorize the causes of death. There are varying definitions for PE risk stratification groups. Our cohort had a high number of comorbidities and a high frequency of submassive PE. We used an inclusive definition of submassive PE to better differentiate submassive PE versus low-risk PE and to best define the low risk referent population. Similarly, we defined massive PE as SBP ⩽ 90 mmHg or requirement for vasopressor therapy or presentation with cardiac arrest. There are additional and broader definitions for massive PE, and our frequency of massive PE may have differed with alternative definitions. Tricuspid annular plane systolic excursion (TAPSE) is an echocardiographic parameter of RV systolic function and has been associated with both short- and long-term mortality after an acute PE.36–38 Unfortunately, during the time of this study, measurement of TAPSE on echocardiograms was not standard at our institution, so we had too many missing variables to pursue TAPSE as a variable in the multivariable model assessing predictors of long-term mortality. BNP also had too many missing variables and could not be used in the multivariable model. The findings of this study may not be representative of different populations with different levels of acute and chronic illnesses. In addition, the small sample size limits the allowable degrees of freedom in our multivariable model and this analysis may be underpowered.
Conclusions
In this contemporary cohort of patients diagnosed with acute PE, the risk of long-term mortality was substantial. Initial PE severity was associated with both short- and long-term mortality. The causes of death were diverse and baseline predictors of mortality were identified. More research is needed to reduce the long-term risk of mortality after an acute PE.
Supplemental Material
10.1177_1358863X19886374_Supplementary_material – Supplemental material for Long-term mortality after massive, submassive, and low-risk pulmonary embolism
Supplemental material, 10.1177_1358863X19886374_Supplementary_material for Long-term mortality after massive, submassive, and low-risk pulmonary embolism by Rajesh Gupta, Zaid Ammari, Osama Dasa, Mohammed Ruzieh, Jordan J Burlen, Khaled M Shunnar, Hanh T Nguyen, Yanmei Xie, Pamela Brewster, Tian Chen, Herbert D Aronow and Christopher J Cooper in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Gupta and Dr Cooper are co-owners of Thermomorph, LLC, a company developing medical devices for VTE treatment. The remaining authors have nothing to disclose.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from the University of Toledo to the principal investigator (RG). The University of Toledo did not have any role in study design, data analysis, manuscript writing, or the decision regarding journal submission.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
