Abstract
Pulmonary embolism (PE) is a major cause of mortality and morbidity. It is known that the risk of death varies by provoking factors; however, it is unknown if the risk of death persists beyond the initial diagnosis among patients with cancer-associated and non-cancer provoked patients. In this study, we aimed to investigate the effect of cancer on overall, short- and long-term mortality in a cohort of consecutive incident PE patients. Using administrative databases, we identified all incident cases of PE between 2004 and 2012 in Alberta, Canada. Cases were stratified by provoking factors (i.e. unprovoked, provoked, and cancer-associated). A multivariate Cox survival model was used to estimate the hazard ratios of short- and long-term death. We identified 8641 patients with PE, among which 42.2% were unprovoked, 37.9% were provoked and 19.9% were cancer-associated. The 1-year and 5-year survival probabilities were 60% (95% CI: 57–64%) and 39% (95% CI: 36–43%) in patients with cancer-associated PE, 93% (95% CI: 92–94%) and 80% (95% CI: 78–81%) in provoked PE, and 94% (95% CI: 93–95%) and 85% (95% CI: 83–87%) in unprovoked PE, respectively. Compared to patients with unprovoked events, both short-term and long-term survival in patients with cancer-associated PE have a higher observed risk of all-cause mortality in all age groups (p<0.001). In contrast, patients with provoked events had a similar short- and long-term all-cause mortality. While PE has a significant mortality in all risk groups, patients with cancer have a higher risk of short-term mortality compared to patients with unprovoked PE.
Introduction
Acute pulmonary embolism (PE) is a common and potentially fatal disease.1,2 Owing to population aging and improvement in diagnostic techniques, PE has been increasingly diagnosed more often in the last few years 3 and is now considered a major cause of mortality, morbidity, and hospitalization, with 10–30% of all patients suffering mortality within 30 days.1,4 Despite advances in the diagnosis and management of PE, short- and long-term mortality rates remain high. 5 In the acute phase, mortality seems to be directly related to the characteristics of the thromboembolic event (i.e. clot burden) and to the rate of its short-term recurrence. Distinctively, the long-term prognosis of PE may be more influenced by the presence of underlying comorbidities or provoking factors.6,7
Several studies have shown that mortality from PE is greatest during the short-term period but even patients who survive beyond this period have an increased risk of death compared to the general population.3,8 It is unknown if the risk of death persists in the short- and long-term beyond the initial diagnosis among cancer provoked and non-cancer provoked patients. In this study, we aimed to investigate the effect of cancer on overall, short- and long-term mortality in a cohort of consecutive incident PE patients diagnosed from 2004 to 2012.
Materials and methods
Study design and data sources
The University of Alberta Health Research Ethics Board (Pro00044383) approved this study. In this retrospective study, six uniquely de-identified administrative health databases in Alberta, Canada, were linked to identify adults (age ≥18 years) diagnosed with incident PE. The databases include: (1) the ambulatory care database, which covers emergency department and outpatient clinic visits (April 1, 2002 to March 31, 2014); (2) the hospital inpatient discharge database (April 1, 2002 to March 31, 2014); (3) the physician claims database (April 1, 2002 to March 31, 2014); (4) the Alberta Blue Cross database for prescription information on patients ≥65 years old (April 1, 2002 to March 31, 2014); (5) the population registry database (April 1, 2002 to March 31, 2014); and (6) Vital Statistics (i.e. death registry from January 1, 2002 to December 31, 2012).
Data elements and variables definition
The combined databases provide information on patient demographics, comorbidities, outcomes of admission (e.g. length of hospitalization, cost), and the International Statistical Classification of Diseases and Related Health Problems, Tenth Revision, Canada (ICD-10-CA) diagnosis and procedure codes. The databases also provide information about the data of diagnosis, procedure, location of service, and provider specialty. Additionally, the Alberta Vital Statistics records information on all-cause mortality that occur within Alberta were used. To select incident PE cases, we used a validated algorithm comprised of the presence of a PE ICD-10 diagnosis with concurrent imaging codes without a PE code for up to 2 antecedent years (washout period). The sensitivity and specificity for identifying patients with PE using this algorithm in our data were 74.83% (95% confidence interval (CI): 67.01–81.62) and 91.86% (95% CI: 89.29–93.98), respectively. 9
Risk factors and comorbidities
Comorbidities were defined as those present at the time of the incident emergency department, hospital, or outpatient visit at which the diagnosis of PE was made, and were identified based on ICD codes entered for that index visit and all other contacts with the health care system in the 1 year prior to the index PE visit (using the hospitalization, ambulatory care, and physician claims databases).10,11 Risk factors were defined as: (1) the presence of cancer (within 1 year preceding the index PE diagnosis visit); (2) whether the patient had undergone major surgery, major trauma, or hip fracture within the last 3 months preceding the diagnosis of PE; or (3) had recent (within 3 months from delivery) or ongoing pregnancy. A hospitalization of more than 3 days for indications other than those stated above in the last 3 months preceding venous thromboembolism (VTE) occurrence was also identified. The VTE event was considered unprovoked if risk factors were absent. If one or more risk factors were identified, then the venous thromboembolic event was defined as either non-cancer-associated or cancer-associated, if cancer was identified in the year preceding the VTE event. The identification of cancer was based on any hospital discharge diagnosis code for cancer during or up to 12 months preceding the VTE indexed events. Non-melanomatous skin cancers were excluded. The primary end point was all-cause mortality. Death was determined through the Alberta Vital Statistics data, which does not contain information on cause of death. Short-term death was defined as 3 months or less, whereas long-term is defined as more than 3 months.
Statistical analysis
Using pertinent ICD codes, patients were stratified into three groups according to the presence or absence of risk factors to unprovoked (absence of any risk factor), provoked, or cancer-associated PE events. Variables were summarized as means and standard deviations or frequencies and percentages, accordingly. Adjusted Kaplan–Meier curves were constructed to describe PE survival stratified by the provoking risk factors. Log-rank tests were used to assess differences in survival between the three groups. Post hoc tests comparing the curves by pairs were performed when the log-rank test was statistically significant.
Multivariate Cox proportional hazard regression models were used to estimate hazard ratios (HRs) for mortality after PE diagnosis for people with non-cancer provoked events and cancer-associated events compared to individuals with unprovoked events as the reference group while adjusting for the following covariates at baseline: age group (≤50, 51–70, and >70 years and over), sex (female, male), year of diagnosis, and presence of comorbidities. The time dependent variable in the models was 3 months since PE diagnosis. Three months are considered the minimum duration required to complete the active treatment of an acute episode of PE.12,13 Another Cox proportional hazards model was used to estimate the effect of predictors on short- and long-term mortality, adjusting for age groups as above, sex, year of diagnosis and provoking factor. Censoring for the analysis occurred upon the date of death or March 31, 2012, whichever came first. After the univariate variables testing, confounding identification, all first-order interaction terms between age, sex and risk factors were tested one at a time. Significant interaction terms (p < 0.05) were incorporated in the HR estimates. Log-likelihood ratio tests were used to evaluate whether adjusted Cox regression models that included interaction terms provided a significantly better fit than the main-effects Cox regression models.
All mortality outcomes were reported with 95% confidence intervals (CIs) around the estimates. A two-sided p-value < 0.05 was considered statistically significant. Statistical analyses were performed with Stata Statistical Software, Release 12 (College Station, TX: StataCorp LP, 2011).
Results
A total of 8641 consecutive adults were newly diagnosed with PE between April 1, 2004 and March 31, 2012 (34,854 person-years) with a median follow-up of 8 years, of which 42.2% were unprovoked, 37.9% were provoked and 19.9% were cancer-associated. The median age is 59 years (IQR 43–74 years) and 57.6% were women. Baseline characteristics differed substantially depending on provoking factors (Table 1). Patients with no identified provoking factors (i.e. unprovoked PE group) were younger and had fewer comorbidities than those with provoked and cancer-associated events.
Baseline characteristics of patients with unprovoked, non-cancer provoked and cancer-associated incident pulmonary embolism (PE) in Alberta, Canada.
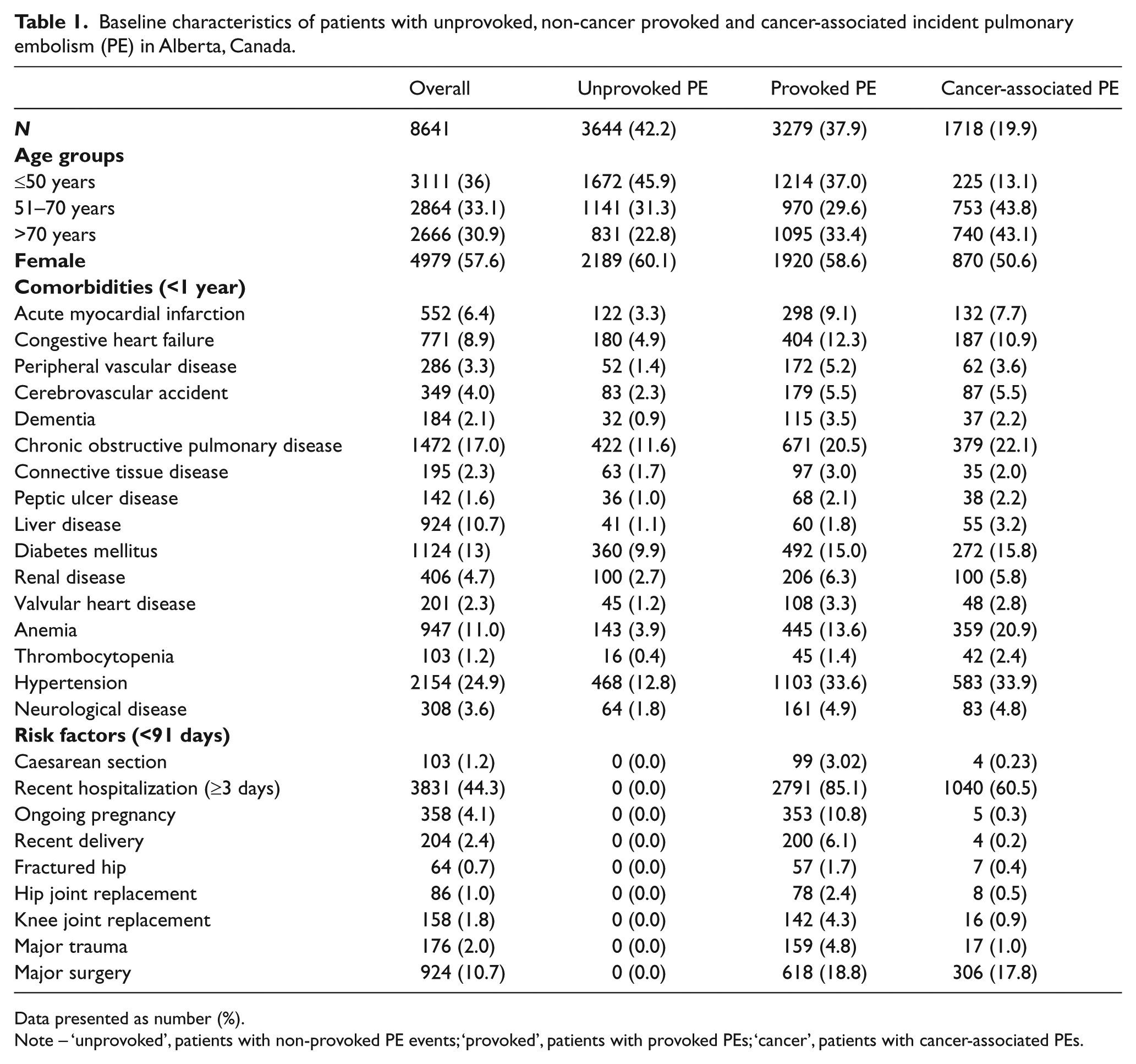
Data presented as number (%).
Note – ‘unprovoked’, patients with non-provoked PE events; ‘provoked’, patients with provoked PEs; ‘cancer’, patients with cancer-associated PEs.
Patients with cancer-associated PE had a 1-year survival of 60% (95% CI: 57–64%) and 5-year survival of 39% (95% CI: 36–43%). Further, patients with provoked PE events secondary to major risk factors other than cancer had a 1-year survival of 93% (95% CI: 92–94%) and a 5-year survival of 80% (95% CI: 78–81%), and patients with unprovoked PE had a 1-year survival of 94% (95% CI: 93–95%) and a 5-year survival of 85% (95% CI: 83–87%) (Figure 1). Patients with both unprovoked and provoked PE events have a median survival time of 8 years compared to 2.18 years in patients with cancer-associated events.
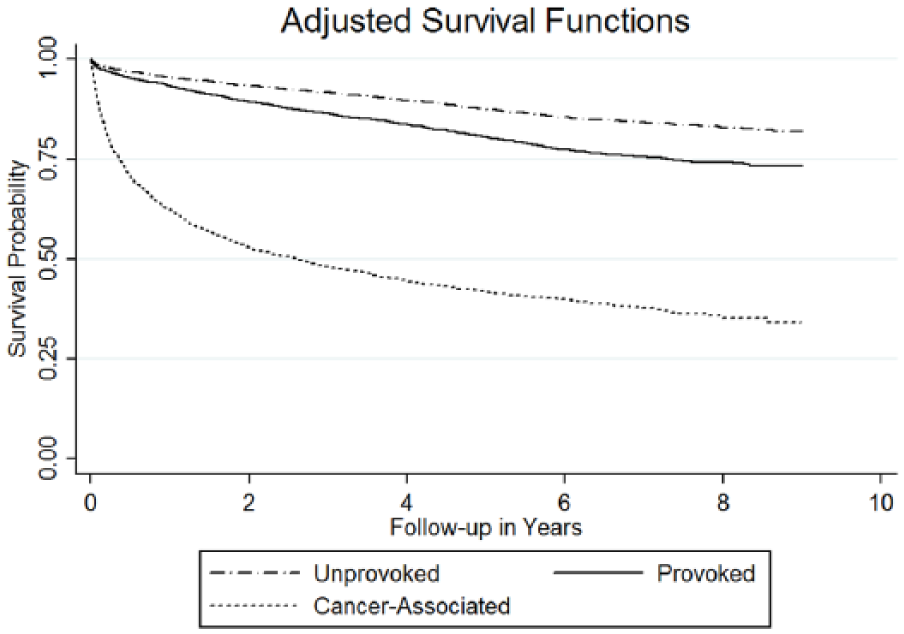
Overall survival probability after pulmonary embolism stratified by risk factors.
The predictive analysis of short- and long-term mortality in patients with PE was performed using the variables listed in Table 2, plus the year of diagnosis. In women, both short- and long-term follow-up in patients with cancer-associated PE have a significantly higher observed risk of all-cause mortality (p-value < 0.001) compared to patients with unprovoked PE (Table 3). This difference was evident across all age groups. In addition, there was no significant difference in the HR of death during the short- or long-term follow-up in patients with provoked PE in comparison to unprovoked PE. Similarly, men with cancer-associated PE events had a significant higher risk of death in the short-term phase compared to patients with unprovoked PE and in all age categories. On the other hand, in comparison to men with unprovoked PE, analysis of men with provoked PE showed no difference in all cause mortality in the short- and long-term follow-up except in patients aged 51–70 years. As shown in Table 3, for male patients in the 51–70 years old age group, the HR of all-cause mortality during the first 3 months was 1.22 (95% CI: 0.47–3.18) for non-cancer provoked PE patients compared to a HR of 1.68 (95% CI: 1.11–2.55) during the long-term follow-up.
Multivariate predictors of mortality in patients with pulmonary embolism (PE) before and after 3 months of diagnosis.
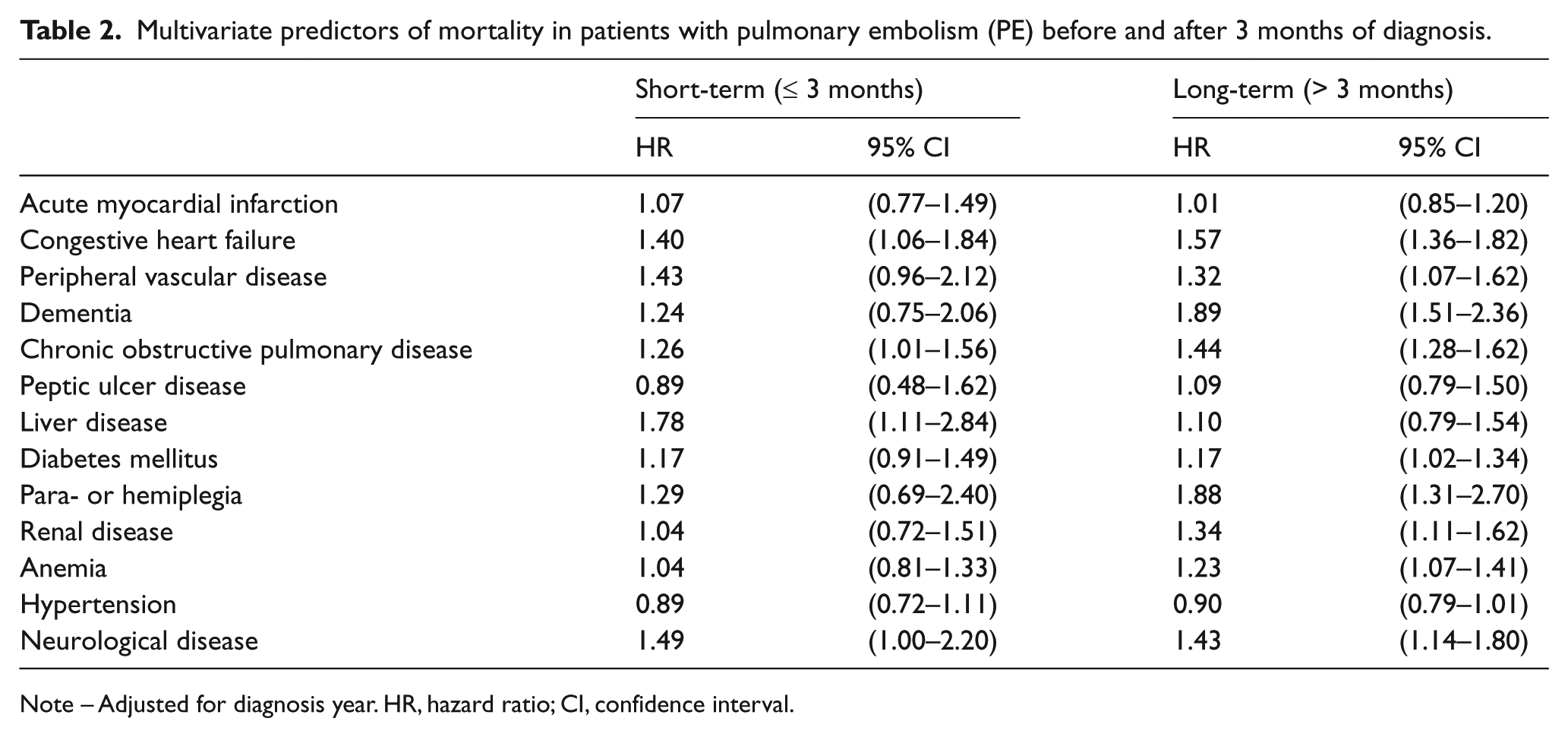
Note – Adjusted for diagnosis year. HR, hazard ratio; CI, confidence interval.
Multivariate Cox regression analysis of short- and long-term all-cause mortality hazard ratios in patients with unprovoked, provoked and cancer-associated pulmonary embolism (PE) in Alberta, Canada.
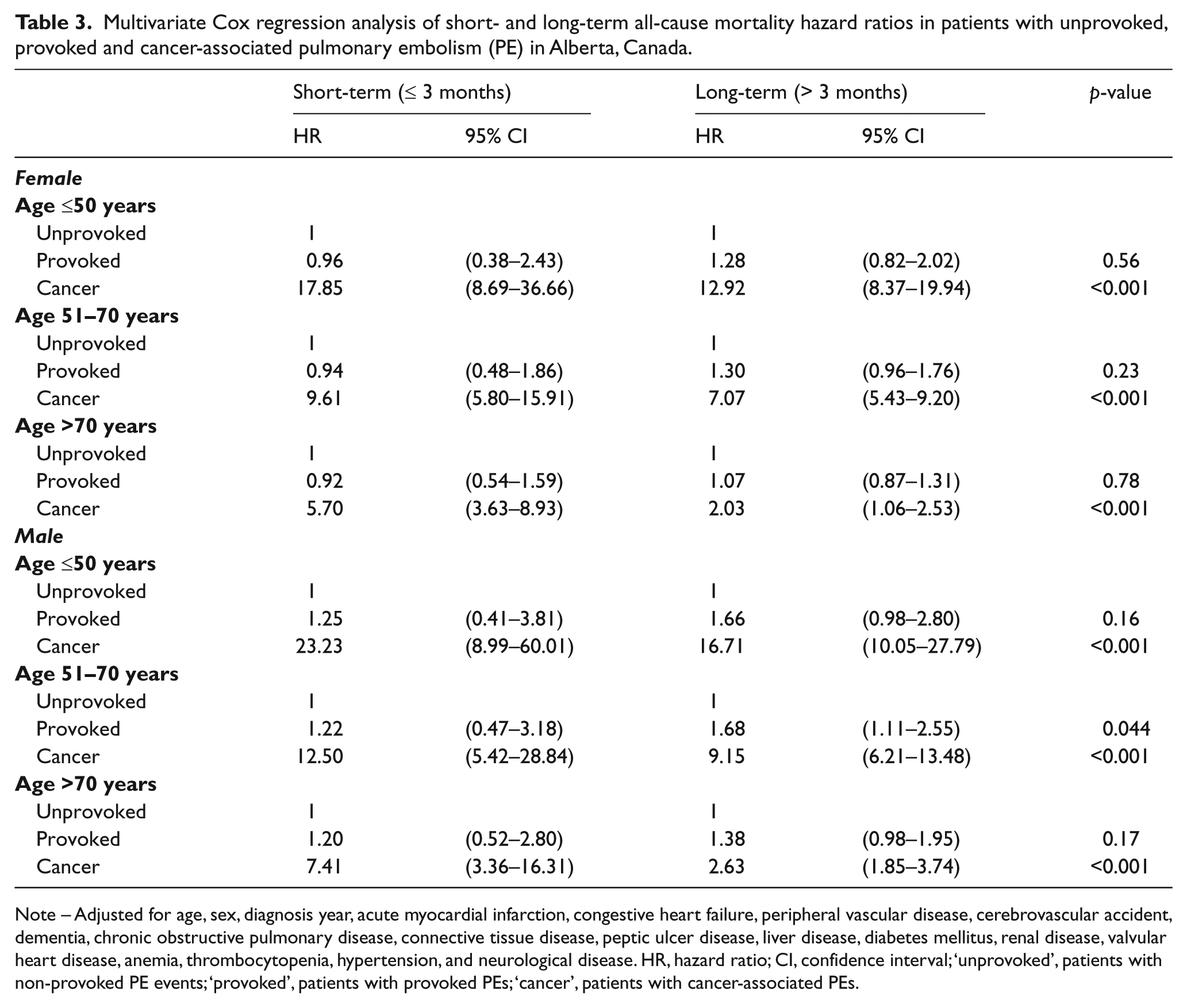
Note – Adjusted for age, sex, diagnosis year, acute myocardial infarction, congestive heart failure, peripheral vascular disease, cerebrovascular accident, dementia, chronic obstructive pulmonary disease, connective tissue disease, peptic ulcer disease, liver disease, diabetes mellitus, renal disease, valvular heart disease, anemia, thrombocytopenia, hypertension, and neurological disease. HR, hazard ratio; CI, confidence interval; ‘unprovoked’, patients with non-provoked PE events; ‘provoked’, patients with provoked PEs; ‘cancer’, patients with cancer-associated PEs.
Discussion
This study is the first to evaluate the short- and long-term survival in patients with first PE with and without cancer. We found that the probability of survival in patients with PE was the lowest in patients with cancer, with a median survival of 2 years. This excess mortality risk persists throughout the study period, which demonstrates a consistently increased mortality regardless of underlying comorbidities. Moreover, patients with cancer-associated PE have a higher risk of dying during the first 3 months of diagnosis compared to patients with otherwise unprovoked PE or PE provoked by risk factors other than cancer.
The reported mortality after VTE varies considerably in the existing literature14,15 according to advanced age, 16 cardiovascular disease, 15 underlying cancer, 17 recurrent VTE, or other medical conditions. 14 Similar to our findings, a Norwegian population-based study of 740 patients with a first-time VTE diagnosed between 1995 and 2001 compared mortality risk after 8 years of follow-up with that in a population comparison cohort. 18 Patients with underlying cancer had a 13-fold age- and sex-adjusted increased mortality risk compared with the comparison cohort. This risk remained 2.5-fold increased for cancer patients who survived 3 years. The mechanism behind the increased long-term mortality risk remains to be further investigated, but it likely reflects both the severity of underlying disease and a VTE-associated excess mortality rate.
Supported by findings from major VTE registries, provoked PE (cancer and non-cancer) was more common than unprovoked PE in our cohort.19,20 These findings suggest that optimizing thromboprophylaxis in these settings in all patient groups might substantially lower the incidence of thrombosis. 21 In contrast to patients with cancer-associated PE, the 1- and 5-year survival in patients with unprovoked events and events provoked by major risk factors other than cancer were somewhat similar. This is explained by the fact that in the control group of patients with an unprovoked VTE, the risk of recurrence has been consistently higher than provoked VTE after discontinuing anticoagulants, especially over the medium term (1–2 years), but the prognostic differences between patients with unprovoked and provoked PE disappear with time. 22 Despite the similarity, the proportion of mortality for both provoked and unprovoked PE at 5 years is about one in five patients, which emphasizes that acute PE is an important clinical problem with a poor prognosis for short-term and long-term survival.
A 20-year review of data from 1979 to 1998 found that the age-adjusted death rate for PE was 94 per million individuals. 23 Extrapolating to today’s Canadian population suggests that an estimated 3290 people die each year from this disease. But it is noted that PE is often undiagnosed, and thus the true death rate is almost certainly substantially higher. In fact, community-based epidemiological studies suggest that roughly one in five individuals die almost immediately from PE, while 40% die within 3 months.4,24 Additionally, the great improvement in myocardial infarction (MI) and stroke mortality trends25,26 suggests that PE may possibly be the leading cause of death in the near future and that much more effort should be dedicated to PE management in order to achieve successful trends in mortality, similar to those observed with MI and stroke.
With the advent of direct oral anticoagulants, a cornerstone strategy to lower VTE-related mortality is to primarily prevent thrombotic events in the first place, followed by optimizing treatment during the acute treatment period, with secondary prevention based on the risk of VTE recurrence.
The mortality rate was highest in patients with cancer, compared to patients who presented with unprovoked PE. In cancer patients, the risk of death was significantly higher during the acute phase (first 3 months), which may be explained by the clinical deterioration after PE, 27 and the presence of thrombosis may signify a more advanced cancer. In addition, we found that patients with events provoked by temporary risk factors had a similar risk of death in the acute phase and long-term. 22 However, this result should be interpreted with caution because, as in most registries, neither diagnostic work-ups nor therapy were controlled.
Strengths and limitations
The present study has several limitations. First, the AB-VTE registry is an observational registry that has inherent limitations in terms of its susceptibility to bias and confounding, restricting its ability to define causality. In addition, there is no external control of the data entered, and there is no external adjudication of the events. Second, treatment varied with local practice and patients were not treated with a standardized anticoagulant regimen. Analysis of different time periods can be used to scrutinize the effect of treatment change over time, but unfortunately without ascertaining the treatment pattern or the exact reason of death it is difficult to interpret. Cause of death cannot be ascertained from the Alberta Vital Statistics data; therefore we reported all-cause mortality.
The strength of the present study is its large size, involving a cohort of more than 8000 patients followed for nearly a decade, and thus should more closely reflect current populations in terms of age demographics and the prevalence of baseline comorbidities. In addition, a validation study allowed us to confirm the diagnosis of VTE and this minimizes a major misclassification bias in our study.
Conclusion
In summary, we found that PE is a common condition with a high mortality in all risk groups; however, patients with cancer have a persistent and substantial risk of short- and long-term mortality.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Ghazi Alotaibi is supported by a scholarship from King Saud University. MS McMurtry is supported by the Heart and Stroke Foundation of Canada.
