Abstract
Submassive pulmonary embolism (PE) remains a vexing entity, and the appropriate use of thrombolytic therapy for this subgroup continues to be actively debated. Catheter-directed thrombolysis has shown efficacy for submassive PE and is gaining momentum because of theoretically improved safety. This review poses and responds to four questions that explore the complex issues surrounding optimal therapy of submassive PE.
Introduction
In 2008, the US Surgeon General and National Heart, Lung, and Blood Institute (NHLBI) announced a national Call to Action that declared pulmonary embolism (PE) a major US public health threat. The Call to Action noted that PE afflicts 300,000–600,000 and kills 100,000–180,000 Americans annually, and causes profound disability in many survivors. 1 PE patients presenting to medical attention fall into three categories.2,3 At the most severe end of the spectrum, patients with ‘massive’ or ‘high-risk’ PE 4 are hypotensive and at imminent risk of death (25–65% mortality), so aggressive therapies such as thrombolysis are considered standard treatment. At the other end, patients with ‘low-risk’ PE are stable, have normal heart function, and have an excellent prognosis with use of anticoagulation alone (<1% mortality), so aggressive therapy is not needed.4–6 In contrast, the optimal treatment strategy for the middle group of patients – ‘submassive’ or ‘intermediate-risk’ PE, 4 which represents >25% of PE cases7–9 – is not clear. Submassive PE patients are initially stable with a normal blood pressure. However, they show evidence of right ventricular (RV) dysfunction and/or ischemia, have a higher risk of clinical deterioration (e.g. require intubation, cardiopulmonary resuscitation (CPR), or vasopressor support), and have a mortality rate of 2–3%.10,11 While advanced therapies such as thrombolysis have shown benefit in submassive PE, they also increase risk. Equipoise prevails, with some clinicians frequently employing thrombolysis and others adopting a more conservative ‘watch and wait’ approach.
The following review addresses four key questions: (1) Should submassive PE patients be treated with systemic thrombolysis? (2) Is catheter-directed thrombolysis a better choice than systemic thrombolysis for submassive PE? (3) Can thrombolytic therapy improve long-term outcomes? (4) What is the optimal design for a definitive future thrombolytic trial for submassive PE?
Question 1: Should submassive PE patients be treated with systemic thrombolysis?
In 1993, Goldhaber et al. randomized 46 patients presenting with acute PE to receive alteplase plus heparin and another 55 to receive heparin alone. 8 The investigators demonstrated a significant improvement in RV function and pulmonary perfusion in patients treated with alteplase plus heparin compared with heparin alone. Moreover, 17% of patients treated with heparin alone had worsening of RV function compared with only 2% of those treated with heparin plus alteplase. Five heparin-only subjects had recurrent PE compared with zero alteplase subjects.
These results were supported by the large prospective registries conducted in Europe and the United States (MAPPET and ICOPER), which showed that RV dysfunction, even in patients without hypotension, was associated with a higher 30-day mortality, and that the use of thrombolytic drugs in these patients was associated with a decreased mortality rate.2,3,12
In 2000, Grifoni et al. examined a prospective cohort of 209 PE patients and found 65 with RV dysfunction without hypotension. 9 Ten per cent of these patients deteriorated during their hospitalization and 5% died, compared with zero PE-related deaths in patients without RV dysfunction.
The accumulating evidence prompted the first randomized trial of systemic thrombolysis in normotensive patients with RV dysfunction. Konstantinides et al. randomized 256 acute PE patients with these characteristics to either receive alteplase plus heparin or heparin alone. 10 The composite endpoint of death or clinical deterioration was met significantly more often in the heparin only group. However, this result was driven by the need for rescue thrombolytics in the heparin only group; mortality was no different between the groups. Overall mortality in the study was significantly lower than the registry data (hypothesized to be due to more intense patient monitoring in a randomized controlled trial (RCT)), and it was postulated that a much larger sample size would be required to demonstrate a mortality benefit. Accordingly, the investigators initiated a follow-up trial with four times the number of subjects, using tenecteplase this time instead of alteplase, taking advantage of the former drug’s ability to be administered as a rapid bolus. Another important difference was the enrichment of the subject population with sicker submassive PE patients by requiring both RV dysfunction and an elevated troponin. It was thought that the combination of sicker patients and a larger study would result in a mortality difference. However, like the 2003 study, the PEITHO trial showed no mortality benefit. 11 The rate of clinical deterioration in subjects treated with tenecteplase plus heparin was lower than that of subjects treated with heparin alone, but major and intracranial bleeding was significantly higher in the tenecteplase group. With systemic thrombolysis, a large dose of fibrinolytic drug (e.g. recombinant tissue plasminogen activator (rt-PA) 100 mg) is injected into a peripheral vein in order for an adequate concentration to be delivered to the pulmonary arteries. The large non-directed dose circulates systemically resulting in increased minor, major, and intracranial bleeding compared with anticoagulation alone. This appears to be a class effect, with similar major and intracranial bleeding rates for both tenecteplase and alteplase.3,10,11
Three meta-analyses have been published in the past year examining the effect of thrombolytics on short-term outcomes; the most widely cited demonstrated a slight but significantly decreased mortality rate (1.39% vs 2.92%) in patients with submassive PE treated with thrombolytics compared with those treated with heparin alone. 13
It is unclear which other factors should be considered when determining whether to administer thrombolytics to a submassive PE patient. For example, should central thrombus or extensive thrombus burden prompt more concern and more aggressive therapy? Central thrombus (saddle embolus or thrombus in one of the main pulmonary arteries) appears to correlate with RV dysfunction, 14 and predicts a higher rate of 30-day mortality or clinical deterioration.15–17 On the other hand, large thrombus burden (as measured by percentage or semi-quantitative indices such as Mastora or Qanadli) does not appear to correlate well with adverse outcomes.18–20 While the criteria for stratification and therefore treatment escalation at present are based upon hemodynamics and RV function, central location of thrombus may be another factor that prompts intervention because thrombus location plays a role in patient selection for catheter-directed therapy and surgical embolectomy.
Another issue is the use of ‘low’ or ‘half-dose’ systemic thrombolysis to reduce the rate of bleeding. The most widely cited manuscript is the MOPETT trial, which found low bleeding rates and good clinical outcomes in patients receiving a maximum of 50 mg of alteplase rather than the standard dose of 100 mg over 2 hours. 21 The study had several methodological limitations including non-standard definitions of intermediate-risk PE and an unclear randomization strategy. Additionally, the reported bleeding rate in both arms was much lower than prior literature. A systematic review suggested that lower dosing strategies had lower major bleeding rates but preserved the benefits of lower 30-day mortality and recurrent PE rates. 22
Thus, the rationale for using systemic thrombolytics for submassive PE thus far has been to prevent short-term adverse outcomes including mortality and clinical deterioration. Proving the former has been challenging in individual studies, while proving the latter has been accomplished several times. Avoiding clinical deterioration may be a worthy reason to judiciously employ systemic thrombolysis; however, the risk of bleeding has tempered enthusiasm and prompted a search for alternatives.
Ideally, patients who are in the high-risk submassive group should be evaluated by a multi-disciplinary team that includes an interventionalist, a cardiothoracic surgeon, and an intensivist and/or emergency physician. Systemic thrombolysis should be considered for patients without contraindications who are at low risk of bleeding, with the biggest risks being advanced age (>65 years) and cancer, and patients who have worrisome signs and symptoms indicating impending clinical deterioration. While none of the options for escalation (systemic thrombolysis, catheter-directed thrombolysis (CDT), or surgical embolectomy) has proven superior, systemic thrombolysis may be particularly useful in a patient who is at low risk of major bleeding or who is unwilling to undergo catheter-directed therapy or surgery.
Question 2: Is catheter-directed thrombolysis a better choice than systemic thrombolysis for submassive PE?
In contrast to systemic thrombolysis, CDT delivers the fibrinolytic drug directly into the thrombus through a multi-sidehole catheter that is placed into the thrombus under imaging guidance (Figure 1). A variation on the standard multi-sidehole catheter is an ultrasound-facilitated catheter (EKOS Corp., Bothell, WA, USA; a subsidiary of BTG plc), which was approved by the Food and Drug Administration in 2014 for the treatment of acute PE with a 24 mg dose of alteplase. The theoretical advantages of this mode of drug delivery are: (1) equivalent or better clot removal because of a higher drug concentration within the clot; and (2) less bleeding, since a much lower overall dose of the fibrinolytic drug (e.g. 15–30 mg of rt-PA) is required and is concentrated in the thrombus.
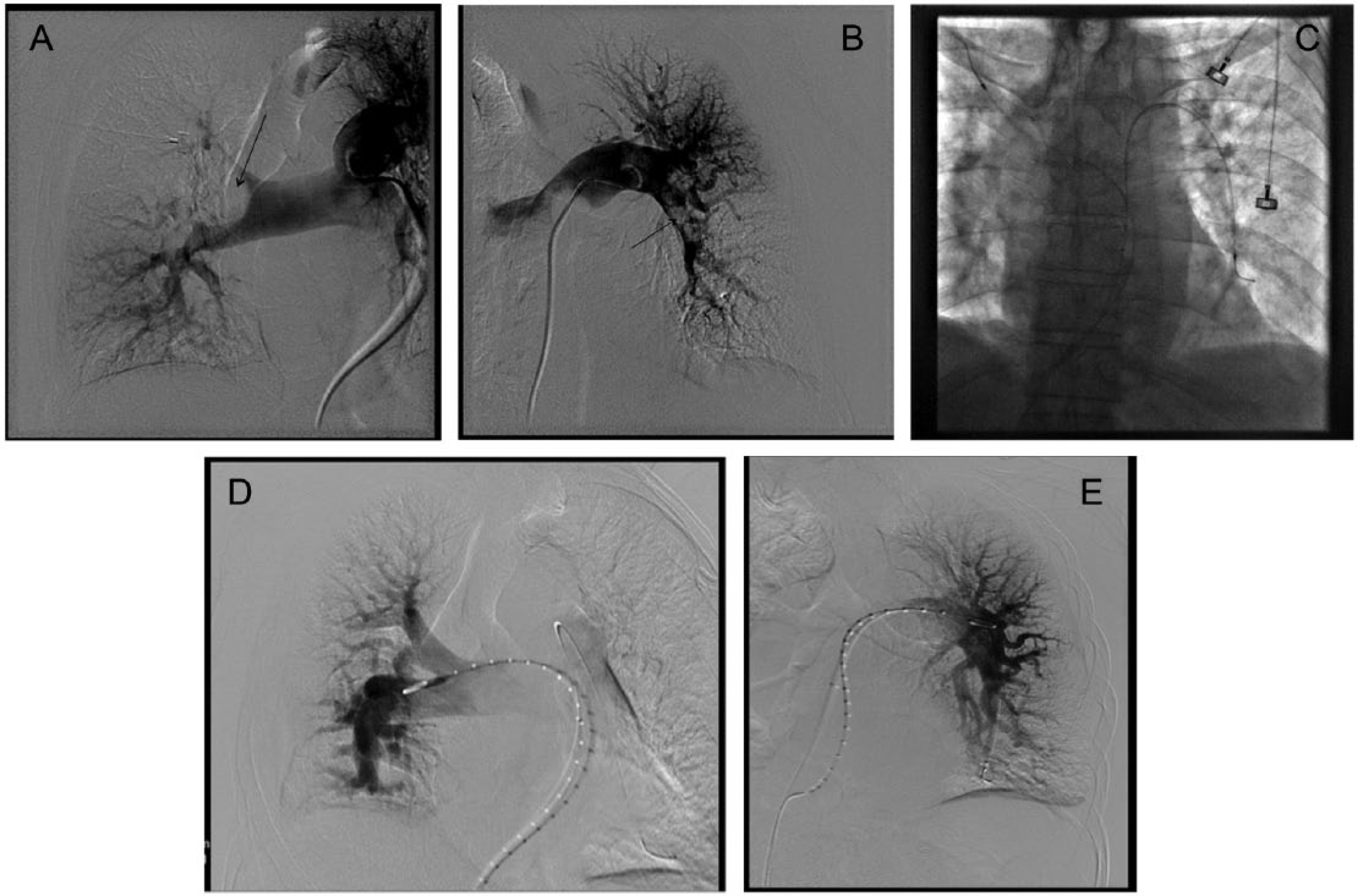
Representative case of catheter-directed therapy. Panel A is a right pulmonary angiogram demonstrating a large excavating thrombus (arrow) in the main right pulmonary artery. Panel B is the left pulmonary angiogram showing thrombus (arrow) in the left lower lobe pulmonary artery. Panel C demonstrates the placement of bilateral multi-sidehole standard infusion catheters. Panels D and E are the right and left respective angiograms 24 hours later after the administration of rt-PA at 1 mg/h × 24 hours, showing marked thrombus reduction and improved perfusion.
Three prospective clinical studies have confirmed that CDT effectively lyses pulmonary arterial thrombi, improves pulmonary blood flow at 48 hours, and more rapidly restores RV function (at 24 hours and 3 months) compared with anticoagulation alone. The ULTIMA investigators randomized 59 patients to receive either ultrasound-assisted CDT plus heparin or heparin alone. CDT normalized the RV/LV ratio faster than heparin alone. 23 No major bleeding was observed in either arm. In SEATTLE II, Piazza et al. enrolled 150 patients with either submassive or massive PE into a single-arm, ultrasound-assisted CDT study. At 48 hours, there was a significant reduction in the RV/LV ratio and pulmonary artery (PA) pressure. 24 Sixteen moderate bleeds and one severe bleed were recorded. The PERFECT prospective global registry enrolled >100 massive and submassive PE patients, and showed a >80% clinical success rate with no major bleeds and a significant reduction in PA pressure. 25 No patients had intracranial bleeding in any of these studies.
It is unclear whether ultrasound-facilitated, catheter-directed therapy offers improved fibrinolysis and safety over standard multi-purpose catheters. The PERFECT registry found no difference in technical and clinical outcomes between the two catheter types. A randomized trial comparing ultrasound-assisted and standard catheters in acute iliofemoral deep vein thrombosis demonstrated no difference in technical or clinical success between the catheters. 26
These results are provocative, and demonstrate the efficacy of CDT, but they cannot be used to routinely recommend CDT over systemic thrombolysis or anticoagulation alone for submassive PE patients. Some remaining questions include: (1) Is the procedure truly safe? The 11% major bleeding rate demonstrated in SEATTLE II requires further exploration; and (2) Does CDT improve clinically relevant short- and long-term outcomes?
In light of this preliminary data, CDT should be weighed against systemic thrombolysis in patients with high-risk submassive PE being considered for thrombolytic therapy. CDT is particularly efficacious when (1) there is central thrombus into which a catheter can be embedded, and (2) a patient is stable enough to tolerate a prolonged thrombolytic infusion. Given its efficacy and theoretical lower bleeding risk, CDT may be preferred over systemic thrombolysis in the majority of high-risk submassive PE cases, although randomized, well-powered studies are needed to validate this practice.
Question 3: Can thrombolytic therapy improve long-term outcomes?
There is a growing body of literature chronicling the deleterious effects of acute PE on long-term quality of life and exercise tolerance. A study by Pengo et al. indicated that 4% of all patients with a first incidence of symptomatic PE went on to develop chronic thromboembolic pulmonary hypertension, a number higher than suspected prior to their investigation. 27 In addition, several other studies (described below) indicate that functional impairment occurs at an even higher rate.
Standard anticoagulant therapy does not actively dissolve thrombus, so residual thrombus remains in the pulmonary arteries of many patients, increasing pulmonary vascular resistance and the pressure load on the right ventricle.28,29 In a prospective study of 254 patients, 73 (29%) had residual perfusion defects on follow up at a median of 12 months. 30 Notably, this subgroup walked shorter distances on the 6-minute walk test and had higher dyspnea scores. In a separate prospective study, residual pulmonary vascular obstruction >35% correlated with a significantly higher risk of death at 6 months. 31 RV damage sustained at the time of the PE can reduce the heart’s ability to adapt to exercise.32–35 In one study of PE patients with high pulmonary pressures on admission, 57% treated with anticoagulation alone had residual pulmonary arterial hypertension after 28 months of follow up. 21 In a separate study of 144 anticoagulated submassive PE patients, 27% of patients were found to have higher pulmonary pressures at the 6-month follow up than at the initial presentation. 36
As alluded to above, these physiologic findings may have functional consequences for patients, referred to by some as ‘the post-PE syndrome’.37,38 In one prospective cohort of 109 previously healthy patients who developed acute submassive PE, 41% displayed poor exercise tolerance (assessed using the 6-minute walk test or New York Heart Association (NYHA) status) or persistent RV dysfunction (by echocardiography) 6 months later. 35 These objective findings were paralleled by patients’ self-reported functional impairment − 20% of them described being unable to shop, needing home oxygen, or having perceptions of poor health status. In Kline et al.’s study from above, 46% of those patients treated with heparin alone who had higher pulmonary pressures on follow up failed the 6-minute walk test or had an NYHA status >3. 36 In a long-term study of 120 patients with PE, the 6-minute walk distance was significantly lower than population norms and 16% had 6-minute walk distances less than 350 meters. 39 In comparison, only 8% of patients with no comorbidities had a 6-minute walk distance less than 350 meters.
These PE-induced functional impairments may contribute to reduced quality of life (QOL). In a prospective study of 392 patients surveyed on average 3.6 years after the diagnosis of acute PE, QOL scores were significantly reduced on seven of the nine subscales (especially the physical role limitation and physical functioning subscales) of the Short Form-36 (SF-36) generic QOL measure compared to age- and sex-matched population norms. 40 These lower scores appear to translate to an inability to work, walk up stairs, or perform household tasks. In the TOPCOAT trial of patients with submassive PE, 28% of patients had exercise intolerance or a low perception of wellness after 3 months. 42
Making sense of this heterogeneous long-term data is not simple, and predicting which individuals presenting with acute submassive PE are going to develop long-term symptoms is not possible with the current data, although one study suggests that a high pulmonary artery systolic pressure (>50 mmHg) at baseline and a failure to normalize after 1 month may be associated with persistent pulmonary hypertension after 1 year. 32
There are some data to suggest that up-front thrombolytic therapy can reduce the incidence of the post-PE syndrome. In a single-center RCT of 121 patients with acute PE, systemic thrombolysis was associated with a significant reduction (16% vs 57%, p <0.001) in the rates of pulmonary hypertension (defined as systolic PA pressure >40 mmHg by echocardiography) at the 2–3-year follow up. 21 In an observational study of 162 patients, patients treated with systemic thrombolysis were less likely to have an increase in their pulmonary artery pressures from baseline to 6 months compared with patients treated with heparin alone. 36 In a small, double-blind, placebo-controlled RCT (n = 72), RV function was improved at 6 months with use of systemic thrombolysis plus heparin compared with heparin alone. 41 In TOPCOAT, systemic thrombolysis recipients were more likely to have normal RV function, exercise capacity, and perception of physical wellness at 3 months compared with patients treated with anticoagulation alone. The thrombolysis recipients had higher Physical Component summary SF-36 scores at 3 months, providing support for the notion that thrombolytic therapy may improve long-term, patient-reported QOL in submassive PE patients. 42
Again, these data are provocative but not conclusive, and by no means is it possible to routinely recommend thrombolytic therapy with its attendant bleeding risks to prevent exercise intolerance. Physiologically, it is tempting to postulate that quickly normalizing RV function and pulmonary perfusion would limit RV inflammation and remodeling and ultimately improve long-term outcomes, but prospective randomized data are needed to test this theory. At the least, patients with abnormal echocardiograms or elevated pulmonary pressures at baseline should be monitored longitudinally with imaging, laboratory analysis, and functional testing to ensure that they do not develop persistent pulmonary hypertension and/or exercise intolerance. If they do, early referral to a pulmonary hypertension center can optimize outcomes.
Question 4: What is the optimal design for a definitive future thrombolytic trial for submassive PE?
First, clinicians and PE researchers need to arrive at a consensus regarding which submassive PE patients should be considered for thrombolytic therapy. The combination of RV dysfunction and biomarker elevation has been associated with poorer 30-day outcomes,43,44 and accordingly the latest European Society of Cardiology guidelines stratify the submassive group (aka the ‘intermediate-risk’ group) into high and low risk, with the former group having both RV dysfunction and biomarker elevation. 4 While this is a reasonable initial framework, understanding which patients are at risk for long-term morbidity may modify this stratification.
Second, the appropriate outcomes need to be selected. As recent randomized trials have shown, submassive patients, when aggressively identified and monitored, have relatively low in-hospital mortality. However, the incidence of short- and long-term morbidity may be higher, so targeting short- and long-term outcomes important to patients and providers, including clinical deterioration, recurrent VTE, exercise tolerance, and QOL, may be more practical and impactful.
Third, the study design needs to address current gaps in the literature (Figure 2). Repeating a short-term systemic or catheter-directed thrombolytic trial for submassive PE would be redundant. However, examining long-term outcomes for thrombolytic therapy, whether with systemic thrombolysis or catheter-directed thrombolysis, is a worthy undertaking. Comparison with anticoagulation alone is a must, so randomization is key. It ought to be noted that such a trial needs to be pragmatic and adequately powered, which may make a three-arm study difficult to conduct.
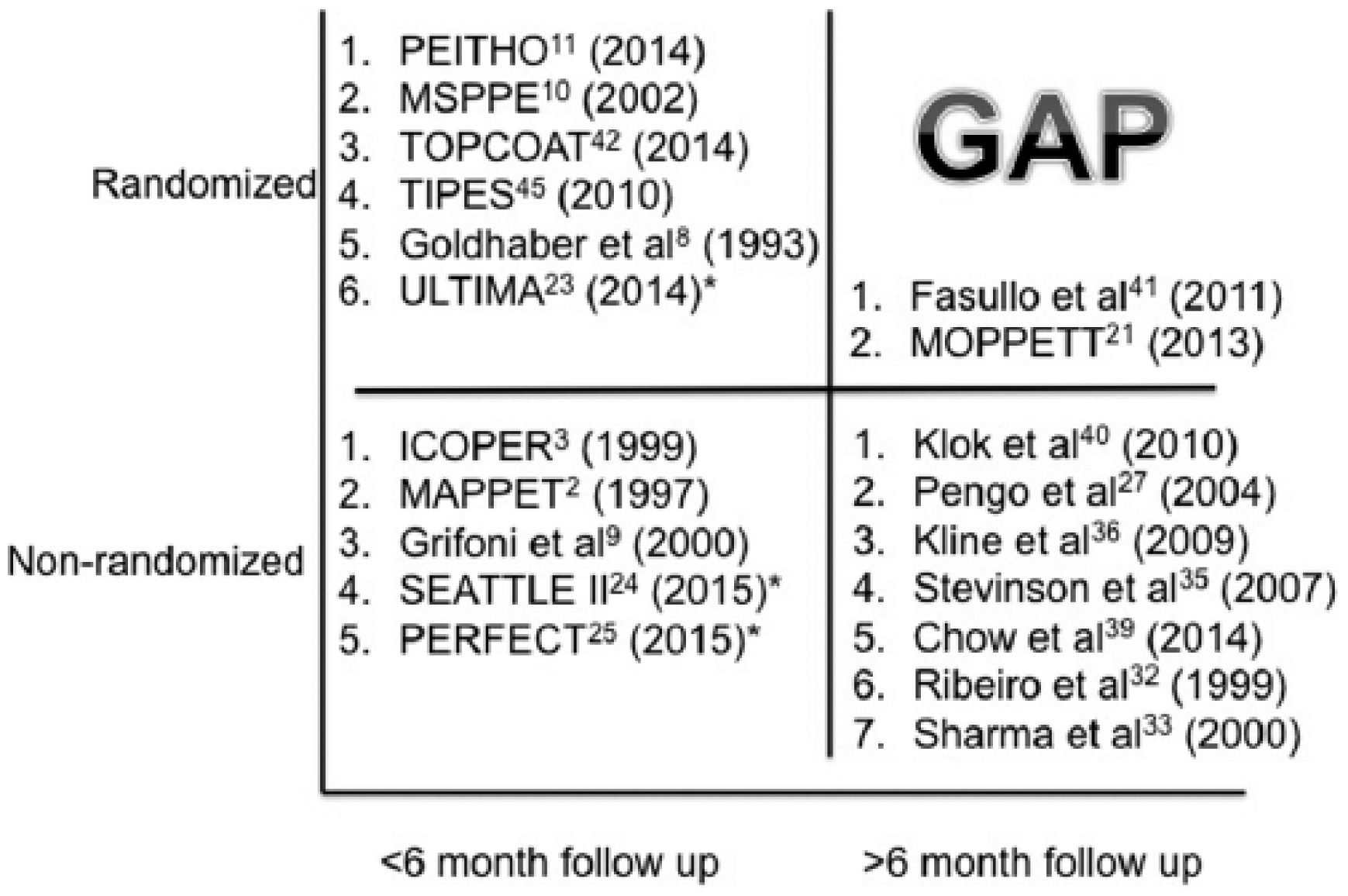
Data gap analysis demonstrating a paucity of well-powered randomized trials with long-term outcomes. Notably, no catheter-directed thrombolysis (CDT) studies have examined outcomes after 6 months. Starred trials utilize CDT. Trials are numbered in order of decreasing number of subjects.
If the above principles are followed and a rigorous, well-conceived RCT is conducted, the confusion surrounding this subset of PE patients will be replaced with certainty, and guideline documents will be more definitive in their recommendations for acute submassive PE.
Footnotes
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
