Abstract
Critical limb ischemia represents the advanced stage of peripheral artery disease, a health problem with increasing prevalence. Critical limb ischemia is associated with significant mortality, limb loss, pain, and diminished health-related quality of life. Public awareness and early diagnosis are necessary for an effective treatment with early risk factor modification, smoking cessation, and exercise therapy. Herein, we present an overview of the epidemiology as well as the clinical stages of the disease, and estimate that there are 6.5 million patients with critical limb ischemia in the US, Europe, and Japan based on global population-based studies. At least 75% of these patients, accounting for approximately 4.8 million patients, are amenable to endovascular therapy.
Keywords
Introduction
In its most severe form, lower extremity peripheral artery disease (PAD) manifests as chronic limb-threatening ischemia (CLTI), also known as critical limb ischemia (CLI), defined by limb-threatening rest pain and ulceration. 1 Although open and endovascular revascularization techniques are both commonly used to prevent limb loss, there exists significant variation in treatment algorithms, and limited data are available to guide the choice of an appropriate revascularization strategy. 2 Further understanding of CLI epidemiology, natural history, and risk stratification is needed in order to evaluate the success of current therapies and provide evidence-based standard of care practices for treatment of these patients. The current review focuses on the epidemiology, evolving classification, and recent trends in management strategies in CLI, in addition to evaluating notable population-based databases used to facilitate evidence-based improvements in CLI management and care.
Methodology
Literature searches were performed in PubMed, Medline, and Google Scholar examining publications originally in or translated into English from January 1, 1985 to July 1, 2019. The search terms used were: ‘critical limb ischemia’, ‘peripheral artery disease’, ‘epidemiology’, ‘classification’, ‘vascular registries’, and ‘endovascular therapy’. Both landmark and recently published articles were evaluated.
PAD epidemiology and risk factors
PAD affects approximately 202 million people globally. 3 From 2000 to 2010, the number of patients with PAD increased by 28.7% in low- to middle-income countries (LMIC) and by 13.1% in high-income countries (HIC). 3 PAD prevalence is particularly high in Southern and sub-Saharan Africa, North America, as well as Western and Northern Europe. 4 Prevalence rates of PAD increase with age in both sexes, with HIC and LMIC affected in a similar manner. In a retrospective cohort analysis of United States employers’ health plans and Medicare and Medicaid programs from 2003 to 2008, among adults aged > 40 years, the annual incidence and prevalence of PAD were 2.69% and 12.02%, respectively. 5
Of risk factors associated with symptomatic PAD, the influence of smoking and diabetes are greatest, followed by age, hypertension, and hypercholesterolemia.3,6 Smoking is the number one preventable risk factor for the development and progression of PAD. 7 Additionally, smoking cessation carries a class 1 level of recommendation in guidelines of PAD management, and has been shown to substantially reduce PAD progression to CLI and subsequent amputation.8,9
Hyperglycemia, hypertension, abdominal obesity, and low high-density lipoprotein (HDL) cholesterol levels are significant contributors to both metabolic syndrome (MetS) and PAD. Unsurprisingly, a total of 40–50% of the population with PAD suffer from MetS.10,11 Data from three National Health and Nutrition Examination Surveys (NHANES) from 1999 to 2004 showed that the prevalence of PAD in individuals with MetS is 7.0%, compared to 3.3% in healthy subjects. 11 As such, individuals with MetS carry twice the risk of developing PAD. As a result of their close relationship, the presence of either PAD or MetS should prompt screening for both conditions in order to minimize risk factors early on. 12 Hematological factors may play a role in the increased risk of atherosclerotic disease in individuals with MetS. In a cohort of 4,622 community-dwelling older adults, C-reactive protein (CRP) and fibrinogen, markers associated with the pro-inflammatory state of MetS, were also independently associated with increased incidence of PAD. 13 Results from another population-based study of 1,592 people confirmed that diabetic individuals and those with impaired glucose tolerance have a higher prevalence of PAD, as well as elevated levels of fibrinogen, von Willebrand factor (VWF), tissue plasminogen activator, and fibrin D-dimer, as compared to healthy subjects. 14
Because PAD and cardiovascular disease are associated with similar risk factors, PAD is a marker of systemic atherosclerotic disease, and results in atherosclerosis in multiple vascular beds. Based on the REduction of Atherothrombosis for Continued Health (REACH) Registry data, the Trans-Atlantic Inter-Society Consensus (TASC) II Document reports that 4.7% of patients with symptomatic PAD had concomitant coronary artery disease (CAD), 1.2% had concomitant cerebrovascular disease (CVD), and 1.6% had both. Moreover, approximately 65% of patients with PAD had clinical evidence of other vascular disease. 6 In a similar Slovenian study, 13% of patients with PAD had concomitant CAD, 9.2% had CVD, and 3.1% had both; 15 while in a Korean study, 4.6% had concomitant CAD, 1.3% had CVD, and 1.5% had both. 16 Pooled data from 11 studies across six countries found that the age-, sex-, risk factor-, and CVD-adjusted relative ratios were 1.45 for CAD and 1.35 for stroke in people with PAD compared to those without. 17
Only a few studies have evaluated the genetics of PAD. 18 In a recent genome-wide association study of PAD in the Million Veteran Program, 19 PAD loci reaching genome-wide significance were identified. 19 Eleven of the 19 loci were associated with disease in coronary, cerebral and peripheral vascular beds. The notable loci included low-density lipoprotein (LDL) cholesterol, the lipoprotein lipase pathway, and circulating lipoprotein(a). 19 Four of the variants were specific for PAD, including Factor V Leiden (F5 p.R506Q), highlighting the role of thrombosis in the peripheral vascular disease. 19 In addition, these results confirm mechanistic similarities among coronary, cerebral, and peripheral atherosclerosis. Identification of genetic variants associated with PAD may help unravel the biological basis of PAD and improve understanding of both the causes of similarity as well as the variation in degree of atherosclerosis from one arterial bed to another.
PAD mortality
In 2016, the overall any-mention age-adjusted death rate for PAD was 14.8 per 100,000. Any-mention death rates in males were 18.2 for non-Hispanic white individuals, 22.8 for non-Hispanic black individuals, 7.6 for non-Hispanic Asians or Pacific Islanders, 17.5 for non-Hispanic American Indians or Alaska Natives, and 14.1 for Hispanic males. In females, rates were 12.5 for non-Hispanic white individuals, 15.7 for non-Hispanic black individuals, 5.9 for non-Hispanic Asians or Pacific Islanders, 14.6 for non-Hispanic American Indians or Alaska Natives, and 10.0 for Hispanic females.20,21
Using segmental blood pressure and Doppler ultrasound, Criqui et al. investigated the association of large-vessel PAD with rates of mortality from all cardiovascular diseases and from coronary heart disease. In this regional study, a 15-fold increase in rates of mortality due to cardiovascular disease was found among subjects with large-vessel PAD. 22 Among 400 patients with PAD, aortoiliac disease was associated with an increased risk of mortality or cardiovascular events, compared with infrainguinal disease. 23
A meta-analysis of 48,294 patients from 16 cohorts demonstrated a reverse-J-shaped association between ankle–brachial index (ABI) and mortality, where participants with an ABI of 1.11–1.40 were at lowest risk. Males with a ABI ⩽ 0.9 carried a threefold increase in risk of all-cause mortality compared with those with a normal ABI, and a similar risk was observed in females. 24 A similar reverse-J-shaped association was observed between ABI and cardiovascular mortality. 24
PAD detection and screening
Until 1999, there were no national efforts in the US to detect PAD in community-based office practice. Subsequently, the PAD Awareness, Risk, and Treatment: New Resources for Survival (PARTNERS) program was developed to assess the feasibility of detecting PAD using ABI in office-based practices, and to evaluate utilization of risk-reduction strategies in community practice. 25 The program showed that despite having a prevalence of 29%, PAD was underdiagnosed and undertreated in primary care practice, in terms of risk factor modification and use of antiplatelet therapies, as compared with other cardiovascular diseases. Furthermore, an ABI value of < 0.9, the primary diagnostic criteria used in the aforementioned study and in most real-life clinical scenarios, has only moderate sensitivity (80%) for detecting PAD, further underestimating the true prevalence of PAD. 3
Guideline recommendations for screening with ABI vary. The US Preventive Services Task Force suggests there are not sufficient data to justify screening for PAD with the ABI in asymptomatic patients. 26 A systematic review of eight guidelines from various countries on screening for PAD found five guidelines that advocated for PAD screening in middle-aged individuals at elevated risk for CVD, and three found the evidence insufficient to recommend screening. 27 Nevertheless, recent studies suggest there is a link between low ABI and increased risk for myocardial infarction, stroke, and overall mortality, potentially justifying the screening of asymptomatic individuals. 28
CLI definition and epidemiology
CLI represents the end stage of PAD, and portends poor limb outcome without intervention. In addition to common signs of PAD, including diminished pulses, decreased capillary refill, pallor on elevation, and atrophic changes, patients with CLI often suffer from rest pain, tissue loss, and hemodynamic evidence of arterial insufficiency. 29
The annual incidence and prevalence of CLI in the US are 0.35% and 1.33%, respectively. 5 Up to 10% of patients with PAD may have CLI, 6 and 5–10% of patients with asymptomatic PAD or intermittent claudication will progress to CLI over a 5-year period. 2 The prevalence of both PAD and CLI is predicted to increase with the aging of the American population and global increase in MetS, diabetes, and smoking. 6 Independent factors associated with progression of PAD to CLI include age, smoking, diabetes, 30 and end-stage renal disease (ESRD). 31
CLI outcomes and mortality
Data from the Nationwide Inpatient Sample demonstrates that hospital admission rates for CLI remained constant from 2003 to 2011. 32 CLI is associated with an impaired quality of life and a 50–60% 5-year mortality rate.6,33 Its economic impact on the US is estimated to be greater than US$4 billion in annual health care costs. 33 In a meta-analysis of six studies, the pooled data on the treatment of 2,144 lower extremities with CLI showed that the rate of any revascularization procedure was 70.4%, of primary amputation 8.4%, and of conservative treatment 20.3%. 34 The pooled 1-year limb salvage rate after conservative treatment of CLI was 57.4%, while the survival and amputation-free survival rates were 75.4% and 51.4%, respectively. 34 Regardless of disease severity or procedure performed, in-hospital mortality was higher in women than men, even after adjustment for age and baseline comorbidities: 2.3% versus 1.6% after percutaneous transluminal angioplasty or stenting and 2.7% versus 2.2% after open surgery. 35
CLI classification
Fontaine, Rutherford, and the Peripheral Academic Research Consortium (PARC)
The first formal definition of CLI proposed by Fontaine et al. in 1954 included the presence of rest pain or tissue loss, without specific hemodynamic measurements. 36 In 1986, the Society of Vascular Surgery/International Society of Cardiovascular Surgery (SVS/ISCVS) guidelines were published, which included the well-known Rutherford classification, 37 which included objective hemodynamic criteria such as pulse volume recording (PVR), ankle pressure, and toe pressure. Of note, this classification system for chronic ischemic disease is different from the well-known Rutherford acute limb ischemia staging system.
Unfortunately, strict hemodynamic criteria are often excluded in clinical and research practice, and because the definitions used to define PAD vary across clinical trials, there exists a significant challenge to data accrual and uniformity of findings. Recognizing this problem, PARC, together with the US Food and Drug Administration and the Japanese Pharmaceuticals and Medical Devices Agency, developed a series of consensus definitions, including the clinical presentation, anatomic depiction, interventional outcomes, surrogate imaging and physiological follow-up, as well as clinical outcomes of patients with PAD. 38 The PARC group provided hemodynamic support for the definition of CLI, using ankle and toe pressures, transcutaneous oxygen, and skin perfusion measurements in patients with tissue loss or ischemic rest pain (Table 1). However, the PARC definition has not gained popularity in clinical and research practices.
BEST-CLI, Best Endovascular Versus Best Surgical Therapy for Patients With Critical Limb Ischemia; PARC, Peripheral Academic Research Consortium; PVR, pressure volume recording; TcPO2, transcutaneous oxygen perfusion.
Best Endovascular Versus Best Surgical Therapy for Patients With Critical Limb Ischemia (BEST-CLI) trial
BEST-CLI is an ongoing randomized controlled clinical trial aimed at providing Level 1 evidence for the best treatment strategies and standard of care for PAD. In this trial, CLI is defined as arterial insufficiency with gangrene, a nonhealing ischemic ulcer, or rest pain, and is corroborated by at least one hemodynamic criteria including ankle pressure, toe pressure, transcutaneous oxygen pressure or transmetatarsal PVR. 39 Despite its use of PARC consensus criteria to classify CLI, this landmark study uses different thresholds (Table 1), prohibiting generalization across studies.
Wound, Ischemia, and foot Infection (WIfI) classification
All commonly used classifications of CLI rely on arterial perfusion but do not account for clinical factors that may affect outcomes, such as wound severity and infection. Moreover, these clinical schemata fail to identify patients at risk of amputation, and do not determine the best therapy for individual patients. Inconsistencies in the definition and heterogeneity of clinical presentation of CLI, as well as advances in revascularization strategies, have rendered many of the current classification systems inadequate. Given these limitations, the SVS recently developed a novel staging scheme for lower-extremity threatened limbs, known as WIfI (Wound, Ischemia, and foot Infection). 40 This staging system is based on the extent and depth of wounds, the degree of ischemia, and the presence and extent of infection (Table 2). 40 A severity grade of 0 to 3 (none, mild, moderate, severe) is assigned to wound, ischemia, and foot infection. On the basis of these three scores, patients are further assigned to four threatened limb clinical stages, from clinical stage 1 (very low risk) to stage 4 (high risk), corresponding to estimated risk of amputation. The underlying premise of WIfI is that the risk of amputation increases with increasing disease burden, and the predicted benefit of revascularization can be derived from the WIfI score and clinical stage. Emerging studies of the WIfI system suggest that this classification has practical and clinical validity in predicting outcomes after open and endovascular revascularization in various patient groups (e.g. diabetic, hemodialysis dependent, etc.).41–51 Additionally, both the WIfI composite and WIfI mean scores are found to be predictive of major amputation, as well as additional endpoints such as survival and restenosis. 41 The recently published ‘Global vascular guidelines on the management of chronic limb-threatening ischemia’ recommended using the WIfI grading system to standardize patient diagnosis, management, and reporting. 1 Nevertheless, further efforts are warranted to validate the WIfI classification system with multicenter data and to incorporate the SVS WIfI classification into clinical decision-making algorithms, in conjunction with a comorbidity index and anatomic classification. 44
WIfI grading system 40 .
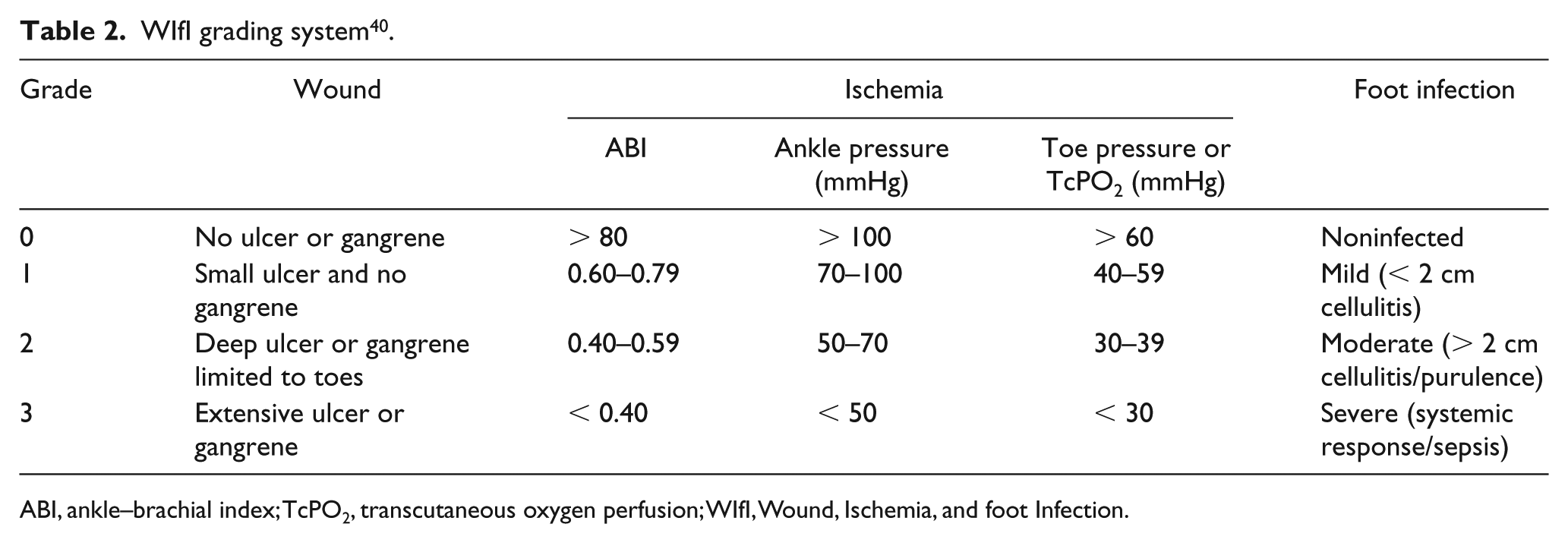
ABI, ankle–brachial index; TcPO2, transcutaneous oxygen perfusion; WIfI, Wound, Ischemia, and foot Infection.
Epidemiology of PAD in vascular registries
Studying the epidemiology and outcomes of intervention for lower extremity arterial occlusive disease remains a major challenge. Regional and national registries have emerged as a method to predict the best therapy for CLI based on patient demographic and anatomic factors. Each of these registries provides valuable clinical information despite limited generalizability as well as the inherent drawbacks associated with retrospective collection of data. Below, we review the major findings from these registries.
Vascular Quality Initiative (VQI)
The VQI is a US clinical registry that provides anatomic and perioperative details which allow for analysis of variation in surgical practice. In a recent study of regional variation in treatment of PAD with respect to patient selection, treatment, and process measures, 16,145 bypass and 35,338 endovascular procedures were analyzed. 52 Patients with CLI accounted for 56% of those treated with bypass, and 46% of those treated with peripheral vascular interventions (PVI). For both bypass and PVI, there was significant variation in the proportion of octogenarians (bypass: 9.0–22%; PVI: 14–27%) and women (bypass: 27–41%; PVI: 38–47%) being treated for CLI. 52 There was also large variation in the proportion of non-white patients being treated across both techniques. Common PAD comorbidities, such as diabetes mellitus, coronary artery disease, congestive heart failure, and chronic obstructive pulmonary disease, also showed significant variation within each treatment modality. There was significant clinical practice variation in areas with established guidelines and therefore potentially unwarranted variation, as well as in areas with clinical ambiguity. 52 Data derived from the VQI national registry highlight the need for quality improvement efforts to reduce unwarranted variation and identify best practice guidelines where none exist.
Blue Cross Blue Shield of Michigan Cardiovascular Consortium (BMC2 PVI)
BMC2 PVI is a prospective, multicenter, observational quality improvement registry funded by Blue Cross Blue Shield of Michigan. 53 In a study of 44,650 PVIs performed in 43 hospitals from 2006 to 2013, a significant temporal trend was observed for PVI indications in treating CLI. 54 In 2006, 24.1% of the 1,677 PVIs were performed for CLI while 47.5% of the 9,185 PVIs were performed for CLI in 2013. The largest increase in PVI performed was for femoropopliteal disease, with no significant trend in aortoiliac or below-the-knee pathology.
Swedish National Quality Registry for Vascular Surgery (Swedvasc)
Swedvasc was launched in 1987 and reached full coverage of all Swedish vascular surgery centers in 1994. 55 Approximately 3,000 PAD-related procedures are registered in the Swedvasc annually. In a recent analysis of data from 2008 to 2013, 16,889 patients were treated for PAD, of whom 63% had CLI. 56 Age-adjusted diabetes mellitus and chronic renal insufficiency rates for patients with CLI on admission for revascularization were 47.2% and 6.7%, respectively.
The incidence of amputation in patients with CLI during the first 6 months following revascularization was 12.0%. Thereafter, the incidence declined to approximately 2% per year. The cumulative combined incidence of death or amputation 3 years after revascularization was 48.8% in CLI patients. Among patients with CLI, compared to patients with intermittent claudication, the prevalence of diabetes, ischemic stroke, heart failure, and atrial fibrillation was approximately double and renal failure was nearly triple, even after adjustment for age. 56
Korean Vascular Intervention Society Endovascular Therapy in Lower Limb Artery Diseases (K-VIS ELLA)
The K-VIS ELLA Registry is a multicenter observational study with retrospective and prospective cohorts of patients with lower extremity PAD undergoing PVI from January of 2006 to July of 2015. In an analysis of this cohort, 1,036 (33.7%) of 3,073 patients with CLI were treated with PVI in 31 Korean hospitals. 57 The diabetes mellitus rate, ESRD rate, and body mass index (BMI) for patients with CLI were 77.7%, 21% and 23.4 ± 4.0, respectively. Type 2 diabetes occurs at a lower mean BMI in East Asian patients, compared with those of European descent. 58 Consistent with the rapid rise in the prevalence of MetS and type 2 diabetes in Asian countries, Korean patients with PAD exhibited higher rates of coexisting diabetes and renal failure compared to other developed countries. 59 Rutherford categories 4, 5, and 6 comprised 22.2%, 48.5%, and 28.9% of CLI cases, respectively. The femoropopliteal artery (41.2%) was the most common target vessel for endovascular treatment, followed by the aortoiliac (35.6%) and infrapopliteal arteries (23.2%). TASC II type C/D aortoiliac (48.0%) or femoropopliteal lesions (60.2%) were frequent targets of PVI.
CLI-specific registries
First-Line Treatments in Patients With Critical Limb Ischemia (CRITISCH)
The CRITISCH registry is a prospective registry analyzing the effectiveness of treatment strategies in 1200 patients with CLI who were treated in 27 vascular centers in Germany from January 2013 to September 2014. 60 In this cohort, 24%, 54%, and 22% of the patients had Rutherford categories 4, 5, and 6 lesions, respectively. PVI was used in 54% and open bypass in 24% of patients. Patients with CLI receiving statin therapy had an increased rate of amputation-free survival (AFS) and lower rate of death, and major adverse cardiovascular and cerebral events. An important finding of this analysis was the beneficial role of statins in improving the AFS rate in challenging CLI subgroups, namely patients with diabetes (47%) and ESRD (43%). 61
One-year AFS for PVI and open bypass was 75% and 72%, respectively, with the endovascular-first approach achieving a noninferior AFS rate compared with open bypass. 61 Treatment strategy had no impact on time until death, major amputation, reintervention, and/or above-ankle amputation. Regardless of the revascularization method, chronic kidney disease, age, lack of statin therapy, Rutherford category 6, and previous vascular intervention, were identified as risk factors for amputation.61,62
Bypass and Endovascular Therapy Against Critical Limb Ischemia from Hyogo (BEACH)
BEACH is a two-center (Kansai Rosai Hospital and Shinsuma General Hospital in Japan) registry, which includes 459 consecutive patients with CLI who underwent revascularization between January 2007 and December 2011. 63 In this cohort, 68% of patients had diabetes mellitus, 47% were hemodialysis dependent, 18% had rest pain, and 82% had tissue loss. Age > 75 years, nonambulatory status, hemodialysis dependency, and ejection fraction < 50% were independent predictors of 2-year mortality. The BEACH score derived from these predictors allows for risk stratification of patients with CLI undergoing revascularization. 63
Poor-Risk Patients With and Without Revascularization Therapy for Critical Limb Ischemia (PRIORITY)
PRIORITY is a multicenter prospective observational study of 662 patients who presented with CLI from January 2014 to April 2015 and had disability in activities of daily living (ADL) and/or impairment of cognitive function. 64 In this cohort, the average BMI was 21.1, rate of diabetes 63.7%, and hemodialysis dependency 44.7%. The 1-year overall survival rate was not significantly different between the revascularization (55.9%) and nonrevascularization groups (51%) in high-risk patients with CLI.
CLI-specific registries dedicated to endovascular interventions
Japan Below-the-Knee Artery Treatment (J-BEAT)
J-BEAT is multicenter, retrospective, nonrandomized registry which includes 900 consecutive patients seen in the cardiology departments of six Japanese hospitals between April 2004 and October 2010. In this cohort, 406 patients presented with CLI (17% rest pain and 83% tissue loss), of whom 69% were diabetic and 60% hemodialysis dependent. In examining the efficacy of balloon angioplasty in treating isolated infrapopliteal artery disease in patients with CLI, hemodialysis dependent patients had a higher target extremity revascularization rate compared to the general population. Nevertheless, the long-term AFS rate (77.1% at 5 year) after balloon angioplasty for isolated infrapopliteal lesions in patients on hemodialysis is comparable to published PVI data. 65 Nonambulatory status, the presence of gangrene, and higher CRP levels were strong predictors of poor prognosis, amputation, and death after PVI in patients with CLI on hemodialysis.
A Prospective, Multi-Center, Three-Year Follow-Up Study on Endovascular Treatment for Infra-Inguinal Vessel in Patients with Critical Limb Ischemia (OLIVE)
OLIVE is a prospective multicenter registry study that enrolled 314 consecutive patients who underwent infrainguinal PVI for CLI (12% Rutherford 4, 73% Rutherford 5, and 15% Rutherford 6) from December 2009 to July 2011 in Japan. 66 In this cohort, the average BMI was 22, rate of diabetes 71%, and ESRD 61%. Three-year AFS, freedom from major amputation, and wound-free survival rates were 55.2%, 87.9%, and 49.6%, respectively. The authors concluded that the 3-year clinical results of PVI were reasonable in patients with CLI and infrainguinal lesions, despite a high reintervention rate and moderate ulcer recurrence rate. 66
Randomized Amphirion Drug-eluting Balloon (DEB) vs Standard Balloon Angioplasty for the Treatment of Below the Knee Critical Limb Ischemia (IN.PACT DEEP)
IN.PACT DEEP is a prospective, multicenter, patient-blinded randomized controlled trial of Amphirion DEB (Medtronic, Santa Rosa, CA, USA) versus balloon angioplasty (PTA) for the treatment of infrapopliteal arterial disease in patients presenting with CLI. 67 In 13 European centers, 358 patients with CLI (17% Rutherford 4, 80% Rutherford 5, and 3% Rutherford 6) undergoing PVI were randomized (2:1) to DEB or PTA from 2009 to 2012. In this cohort, the average BMI was 27, diabetes rate was 60%, and ESRD rate was 10%. While both treatment modalities showed similar rates of clinically driven target lesion revascularization (DEB: 9.2% vs PTA: 13.1%), there was a trend towards an increase in the 1-year major amputation rate in the DEB group (DEB: 8.8% vs PTA: 3.6%; p = 0.080). 67
Limitations of population-based registries
The creation of large clinical databases and national registries allows for capture of vital information that affects health care delivery and patient outcomes. Nevertheless, all databases are limited to in-hospital events and suffer from patient selection and geographical biases. The true incidence rates of risk factors, complications, and mortality may be underestimated in community centers and overestimated in tertiary care centers. For instance, the rate of diabetes and renal failure substantially varied in PAD- and CLI-specific databases (Table 3). Additionally, each of these registries has its own limitations that are listed in Tables 4 and 5.
Prevalence of diabetes and renal failure in major vascular clinical registries.
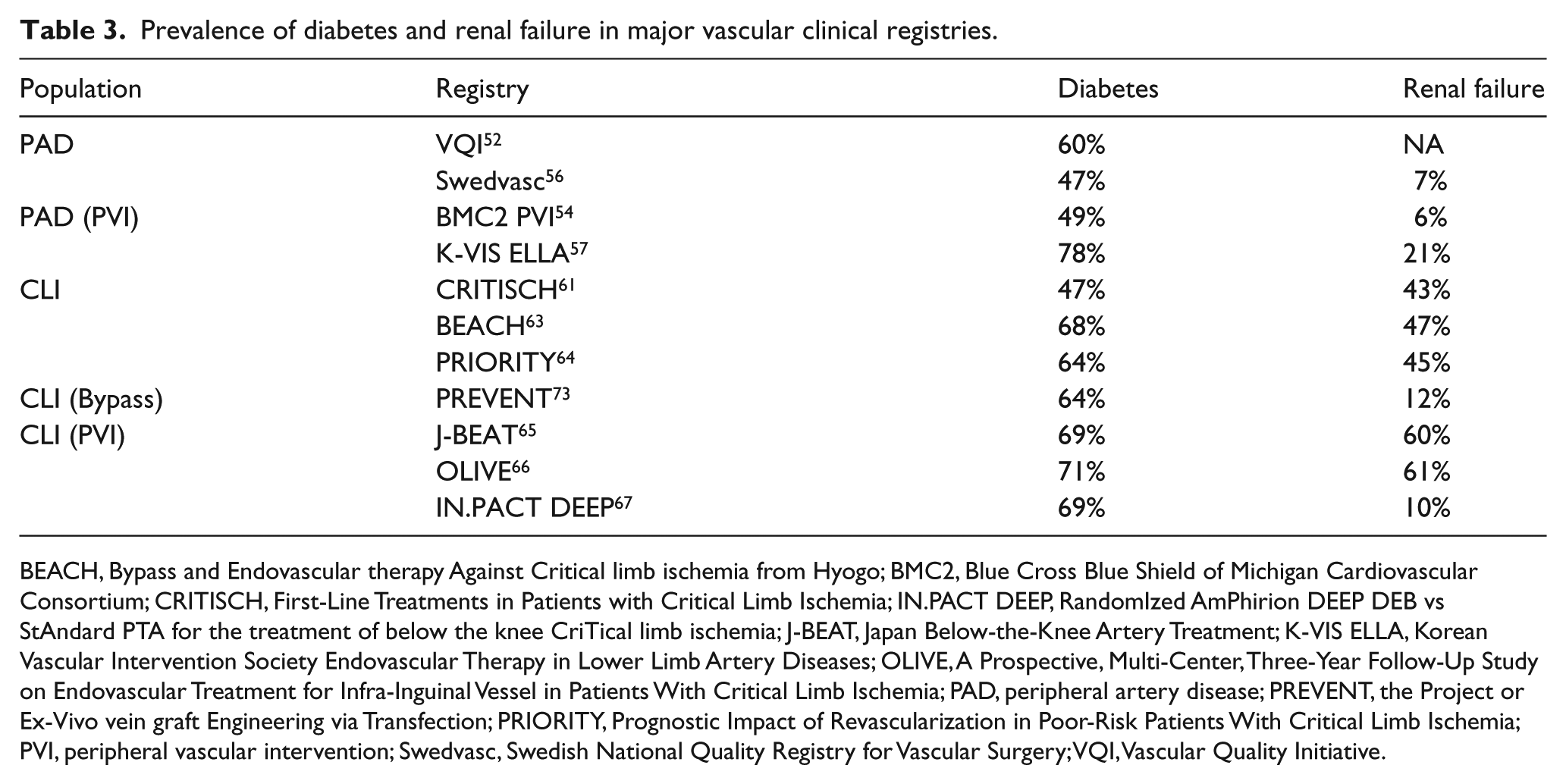
BEACH, Bypass and Endovascular therapy Against Critical limb ischemia from Hyogo; BMC2, Blue Cross Blue Shield of Michigan Cardiovascular Consortium; CRITISCH, First-Line Treatments in Patients with Critical Limb Ischemia; IN.PACT DEEP, RandomIzed AmPhirion DEEP DEB vs StAndard PTA for the treatment of below the knee CriTical limb ischemia; J-BEAT, Japan Below-the-Knee Artery Treatment; K-VIS ELLA, Korean Vascular Intervention Society Endovascular Therapy in Lower Limb Artery Diseases; OLIVE, A Prospective, Multi-Center, Three-Year Follow-Up Study on Endovascular Treatment for Infra-Inguinal Vessel in Patients With Critical Limb Ischemia; PAD, peripheral artery disease; PREVENT, the Project or Ex-Vivo vein graft Engineering via Transfection; PRIORITY, Prognostic Impact of Revascularization in Poor-Risk Patients With Critical Limb Ischemia; PVI, peripheral vascular intervention; Swedvasc, Swedish National Quality Registry for Vascular Surgery; VQI, Vascular Quality Initiative.
List of PAD registries in the US, Europe, and Japan.
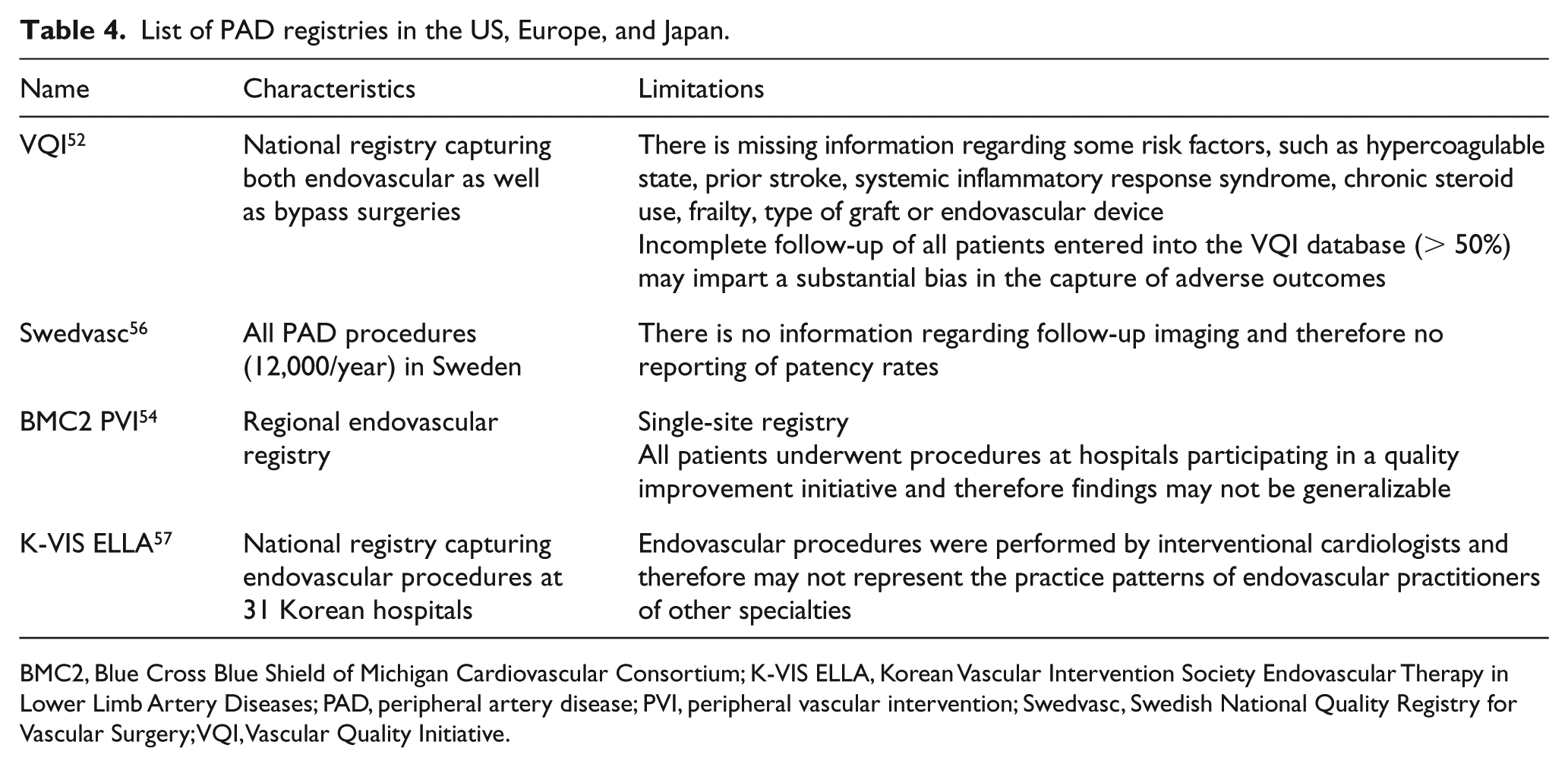
BMC2, Blue Cross Blue Shield of Michigan Cardiovascular Consortium; K-VIS ELLA, Korean Vascular Intervention Society Endovascular Therapy in Lower Limb Artery Diseases; PAD, peripheral artery disease; PVI, peripheral vascular intervention; Swedvasc, Swedish National Quality Registry for Vascular Surgery; VQI, Vascular Quality Initiative.
List of CLI registries in the US, Europe, and Japan.
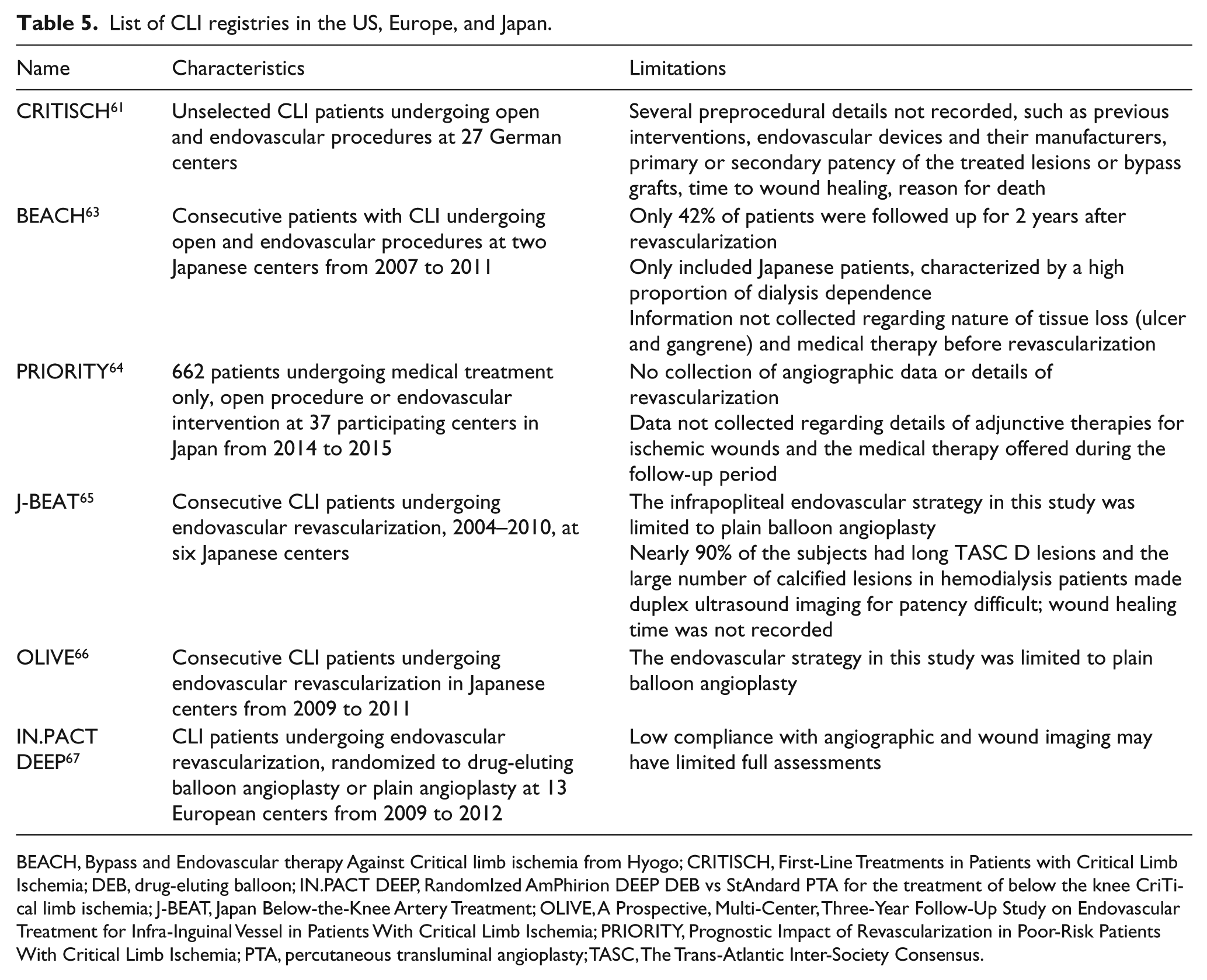
BEACH, Bypass and Endovascular therapy Against Critical limb ischemia from Hyogo; CRITISCH, First-Line Treatments in Patients with Critical Limb Ischemia; DEB, drug-eluting balloon; IN.PACT DEEP, RandomIzed AmPhirion DEEP DEB vs StAndard PTA for the treatment of below the knee CriTical limb ischemia; J-BEAT, Japan Below-the-Knee Artery Treatment; OLIVE, A Prospective, Multi-Center, Three-Year Follow-Up Study on Endovascular Treatment for Infra-Inguinal Vessel in Patients With Critical Limb Ischemia; PRIORITY, Prognostic Impact of Revascularization in Poor-Risk Patients With Critical Limb Ischemia; PTA, percutaneous transluminal angioplasty; TASC, The Trans-Atlantic Inter-Society Consensus.
Summary and three fundamental questions regarding CLI
There are currently at least 200 million people with PAD worldwide 3 and the estimated prevalence of PAD is 12% and rapidly increasing. 5 Progression of PAD to CLI is closely associated with age, smoking, diabetes, 30 and ESRD. 31 There are several evolving lower extremity threatened limb grading systems; the most recent guidelines recommend use of the WIfI classification system that grades wound extent, degree of ischemia, and severity of infection to guide clinical management in all patients with suspected CLI. 1
How many patients suffer from CLI?
It is not possible to accurately determine the prevalence of CLI globally, particularly in developing countries, secondary to a lack of population-based data. However, based on CLI prevalence of 800 per 100,000, as ascertained by Biancari, 34 the number of patients with CLI is approximately 2,595,676 in the USA, 2,551,917 in Europe, 1,019,876 in Japan, and 6,167,468 in these regions combined. Similarly, based on the World Health Organization PAD data and report of an 11.1% prevalence of CLI in this cohort, 4 the estimated number of patients with CLI in these three regions combined is approximately 6,485,884 (Table 6). Based on the distribution of Rutherford classifications reported in CRITISCH, Rutherford category 5 (55%) is estimated to have the highest number of CLI presentations (3,567,236) in developed countries.
Estimated number of patients with CLI in the US, Europe, and Japan.
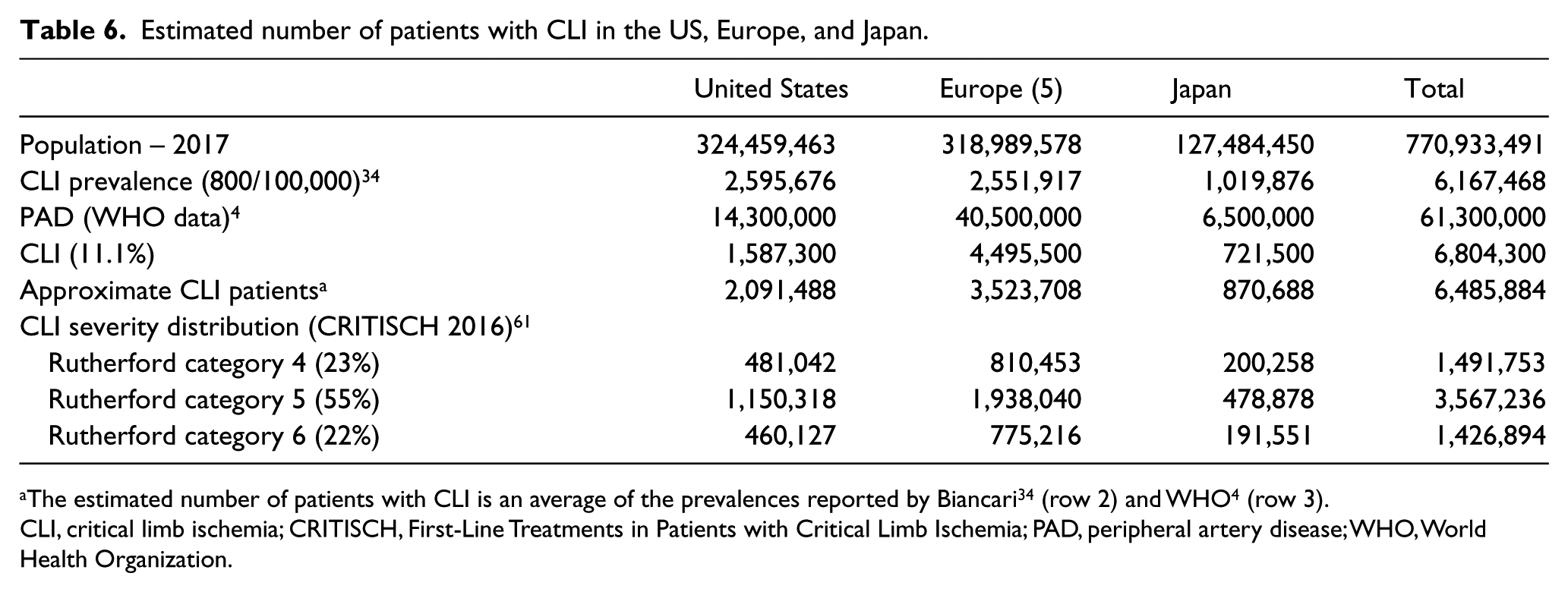
The estimated number of patients with CLI is an average of the prevalences reported by Biancari 34 (row 2) and WHO 4 (row 3).
CLI, critical limb ischemia; CRITISCH, First-Line Treatments in Patients with Critical Limb Ischemia; PAD, peripheral artery disease; WHO, World Health Organization.
How many patients have extensive ischemia amenable to therapy versus requiring major amputation?
With pooled CLI prevalence of 800 per 100,000, more than 90% of CLI is amenable to therapy (70% to revascularization and 20% to conservative management) and less than 10% to primary amputation. 34 Therefore, the estimated number of patients amenable to therapy is 5.2–5.8 million. However, the annual major amputation rate has been persistently declining. 32 Ulceration (Rutherford category 5/6) is a significant predictor of amputation and death in diabetic patients, and is associated with a 21% amputation rate. 68
Despite a less than 10% amputation rate in patients with CLI, significant variation exists in the extent of vascular care provided in the year prior to major amputation. 70 Of the 20,464 patients with CLI in the Medicare database who underwent major amputations from 2003 to 2006, 54% of patients had no vascular procedures, including diagnostic angiography, 1 year prior to amputation. 69
How many patients with CLI are treated with endovascular therapy?
Since advances in endovascular technology have expanded the treatment options available for PAD, endovascular interventions are now performed much more commonly than bypass surgery. 70 In a study of the Nationwide Inpatient Sample from 2003 to 2011, there was a significant increase in endovascular treatment of CLI (from 5.1% to 11.0%), which was accompanied by lower rates of in-hospital mortality and major amputation, as well as shorter length of stay. 32 Conversely, there was a significant decrease in surgical revascularization rates (from 13.9% to 8.8%). Of note, the rate of CLI admission remained constant during this time period.
In an analysis of the VQI procedural database from 2010 to 2013, PVI was performed more frequently (61%) compared to bypass and was primarily offered to older patients and those with more comorbidities (e.g. diabetes and hemodialysis dependency). 71 Patients undergoing lower extremity bypass (31%) were more likely to have a history of previous interventions. Patients treated with PVI had lower perioperative mortality, although this benefit did not apply to patients with fewer comorbidities. In a similar study of the VQI database from 2009 to 2014, 61% of bypass procedures in symptomatic patients with PAD were performed for CLI and 73% of the PVI for symptomatic PAD were performed in patients with CLI. Overall, 54.5% of patients with CLI were treated with PVI and 45.5% with bypass surgery. 72 Based on these large cohort studies in patients who undergo treatment for CLI, at least 55% are treated with PVI. Therefore, at least 4.8 million patients with CLI are amenable to endovascular therapy in the US, Europe, and Japan.
Conclusion
CLI is associated with an excessively high risk for cardiovascular events and mortality as well as poor prognosis with respect to limb preservation. With its increasing incidence, CLI poses a substantial burden on patients and health care providers. Prevention strategies and best medical therapy, including diet, exercise, smoking cessation, preventive foot care, and antihypertensive, antithrombotic, lipid-lowering, and glycemic control agents, should be offered to patients with CLI. Initiatives should be taken to increase the use of a globally accepted CLI staging system that includes clinical, anatomical, hemodynamic, and prognostic criteria (i.e. WIfI). Comparative effectiveness studies need to select populations that reflect the general CLI demographics encountered in real-life clinical settings. Large global collaborative efforts can improve understanding of prevalence, outcomes, and treatment costs of CLI to promote evidence-based treatment of these patients.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

