Abstract
Background:
The scope of clinical guidelines for the management of chronic limb-threatening ischemia (CLTI) is limited to atherosclerotic disease of the lower-extremity arteries. This study aimed to reveal the clinical features and prognoses of CLTI due to inflammatory nonatherosclerotic etiologies undergoing revascularization.
Methods:
DECOPON (DatabasE of ChrOnic limb-threatening ischemia Presenting ischemic ulcer and gangrene due to nON-atherosclerotic disease) is a multicenter retrospective study that evaluated 465 limbs of 414 patients with CLTI undergoing revascularization for ischemic tissue loss with inflammatory nonatherosclerotic etiologies between 2010 and 2020. Clinical features and prognoses were compared with 930 limbs of 809 patients with atherosclerotic CLTI during the same period.
Results:
The frequency of conventional atherosclerotic risk factors was significantly lower in the inflammatory nonatherosclerotic group. The inflammatory nonatherosclerotic group had more severe WIfI (wound, ischemia, and foot infection) W and fI grades, and more frequent infrapopliteal and inframalleolar artery diseases with a rarer presence of arterial calcification (all p < 0.05). Adjunctive therapy was more frequently performed in the inflammatory nonatherosclerotic group (p < 0.05). The inflammatory nonatherosclerotic group had a lower incidence rate of wound healing (0.64-fold, p < 0.001) and a higher incidence rate of major reintervention (2.30-fold, p = 0.006), whereas the risk ratio of all-cause mortality was 0.83-fold (p = 0.067). The association of inflammatory nonatherosclerotic disease with the delayed wound healing and the susceptibility to major reintervention remained significant even after adjustment for covariates (both p < 0.001).
Conclusions:
Patient, limb, and lesion characteristics of inflammatory nonatherosclerotic CLTI were considerably different from those of atherosclerotic CLTI. Inflammatory nonatherosclerotic CLTI was less likely to heal than atherosclerotic CLTI.
Background
Chronic limb-threatening ischemia (CLTI), especially presenting with ischemic tissue loss, is the most advanced phenotype of lower-extremity artery disease (LEAD), and is associated with an extremely high risk of amputation, mortality, and impaired quality of life.1,2 CLTI has become a global healthcare issue, and its management has attracted increasing attention. 3 Accordingly, clinical guidelines on management of CLTI have been published in recent years, providing systematic and practical strategies.1,2,4,5 However, the scope of these guidelines is limited to atherosclerotic disease of the lower-extremity arteries; they do not address inflammatory nonatherosclerotic etiologies. Indeed, CLTI due to inflammatory nonatherosclerotic etiologies is less common and firstly considered for conservative therapy but is often consequently indicated for revascularization in clinical practice. The aim of this study was to reveal the clinical features and prognoses of CLTI due to inflammatory nonatherosclerotic etiologies undergoing revascularization in comparison with CLTI due to atherosclerotic disease.
Methods
Study design and participants
The DatabasE of ChrOnic limb-threatening ischemia Presenting ischemic ulcer and gangrene due to nON-atherosclerotic disease (DECOPON) study is a retrospective multicenter study conducted in 55 cardiovascular centers across Japan. The study included limbs of CLTI patients presenting with ischemic tissue loss due to inflammatory nonatherosclerotic etiologies (cases) and atherosclerotic disease (controls), who underwent revascularization between January 2010 and December 2020. The data of limbs were extracted from the medical records of each participating center at a ratio of 1:2 (case: controls, respectively): 465 limbs from 414 case patients, and 930 limbs from 809 control patients. For each ‘case’ limb, we selected two ‘control’ limbs that underwent revascularization on the closest date to the date of the case.
This study was performed in accordance with the Declaration of Helsinki and was approved by the ethics committee of all participating hospitals. As this was an observational study and it did not include human biological specimens, the need for written informed consent was waived in accordance with the ethical guidelines for medical and health research involving human subjects in Japan. However, relevant information regarding the study was open to the public, and opportunities for refusal were provided.
Evaluation of baseline characteristics and revascularization
In this study, inflammatory nonatherosclerotic disease included: (1) Buerger’s disease, (2) scleroderma, (3) systemic lupus erythematosus (SLE), (4) rheumatoid arthritis (RA), (5) polyarteritis nodosa, (6) eosinophilic granulomatosis with polyangiitis, (7) granulomatosis with polyangiitis, and (8) vasculo-Behçet’s disease. The presence of inflammatory nonatherosclerotic disease and baseline characteristics of the patient, lower limb, and anatomical severity were retrospectively reviewed using clinical medical records. The condition of ischemic tissue loss and wound infection was retrospectively evaluated using photographs that were taken before any treatment including surgical debridement and revascularization. Wounds in the index limbs were evaluated based on the Wound, Ischemia, and foot Infection (WIfI) classification system. 2 The presence and severity of wound infection was also assessed according to the WIfI classification system, into which the Infectious Diseases Society of America (IDSA) and the International Working Group on the Diabetic Foot (IWGDF) perfusion, extent/size, depth/tissue loss, infection, sensation (PEDIS) classifications of diabetic foot infection 6 was adapted. The severity of ischemia and hemodynamic changes in the index limb before and after revascularization were evaluated using the ankle–brachial index, ankle pressure, and skin perfusion pressure. The anatomical location and severity of arterial lesions were routinely evaluated using duplex ultrasound as a noninvasive test. If arterial disease with hemodynamic significance was detected by duplex ultrasound, the presence of significant arterial lesions was diagnosed by digital subtraction angiography before revascularization. The presence of arterial calcification was evaluated by fluoroscopy without contrast in two directions. The severity of aortoiliac disease was assessed using the TransAtlantic Inter-Society Consensus (TASC) II classification, and the severity of femoropopliteal, infrapopliteal, and inframalleolar artery lesions was graded using the global limb anatomic staging system (GLASS). The treatment strategy was determined by a team of vascular specialists, including vascular surgeons, interventional cardiologists, and radiologists, in each hospital in clinical practice. Each hospital can select both surgical and endovascular treatments and each revascularization strategy was based on a generally accepted protocol for the treatment of CLTI in clinical practice.1,2 During surgical therapy, an autogenous vein graft is generally used for bypass surgery, whereas prosthetic bypass is limited in cases without a usable autogenous vein. During endovascular therapy (EVT), the primary stenting strategy was generally applied to aortoiliac lesions, whereas the provisional stenting strategy was usually applied to femoropopliteal lesions. The device used in each arterial lesion was selected at the physician’s discretion, determined by anatomical severity. An atherectomy device was not used because it was not approved in Japan during the study period. Angioplasty with a noncoated balloon was performed to treat the infrapopliteal and inframalleolar artery lesions. Attending physicians examined clinical symptoms, including wound condition, hemodynamic status evaluated by measurement of the ankle–brachial index and skin perfusion pressure, and vessel patency mainly assessed by duplex ultrasound in clinical settings. The follow-up protocol largely relied on the manner of participating hospitals, but was generally scheduled every 2–4 weeks until complete wound healing and thereafter every 3 months for as long as possible.
Definition
The definition of comorbidities are described in the online Supplemental Material. Wound healing was defined as complete epithelialization of the wound for the first time and healing time was defined as the time between initial revascularization and complete epithelialization. Major amputation was defined as any limb loss extending above ankle level. Any reintervention indicated major and minor interventions after the index procedure. Major reinterventions included a new bypass graft, jump/interposition graft revision, or the use of thrombectomy or thrombolysis in stents upon loss of primary-assisted patency. Minor reinterventions were defined as endovascular procedures (angioplasty or stenting) without thrombectomy or thrombolysis, and minor surgical revisions (patch angioplasty). Reintervention was clinically driven and was indicated in cases with angiographic evidence of significant stenosis at the treated segment and documentation of recurrent clinical symptoms. 7
Outcome measures
The outcome measure of efficacy was wound healing. The adverse outcome measures included any reintervention, major reintervention, major amputation (failure of limb salvage), all-cause mortality (failure of overall survival), cardiovascular death, and noncardiovascular death.
Statistical analyses
Data are presented as the mean ± SD for continuous variables or as the median (IQR) and percentage for categorical variables, unless otherwise indicated. A p-value less than 0.05 was considered statistically significant, and 95% CIs were reported when appropriate. We treated the case and corresponding controls as pairs. The association of the etiology (inflammatory nonatherosclerotic vs atherosclerotic disease) with the incidence of the first events was investigated using the Poisson mixed-effect models. Details are provided in the online Supplementary Material.
Results
Of the 465 limbs of 414 patients with inflammatory nonatherosclerotic disease, 206 limbs of 174 patients (206/174) were affected by rheumatoid arthritis, 132/122 by scleroderma, 49/49 by Buerger’s disease, 42/38 by systemic lupus erythematosus, and 36/31 by other inflammatory nonatherosclerotic disease. The baseline characteristics, lower limb, lesion, and treatment strategies of the study population are summarized in Tables 1, 2, and 3, and the online Supplementary Table, respectively. In brief, compared with the atherosclerotic group, the inflammatory nonatherosclerotic group had lower prevalence of diabetes mellitus (56.3% vs 82.7%), chronic renal failure (25.6% vs 61.1%), and chronic heart failure (18.8% vs 25.6%) (all p < 0.05) (Table 1). As Table 2 shows, the WIfI W and fI grades were more severe in the inflammatory nonatherosclerotic group (p = 0.009 and 0.019, respectively), whereas the WIfI I grade and clinical stage were not significantly different (p = 0.27 and 0.099, respectively). The inflammatory nonatherosclerotic group had more severe infrapopliteal and inframalleolar artery disease (p = 0.039 and p < 0.001, respectively), whereas femoropopliteal arterial disease was less severe (p = 0.040). The severity of aortoiliac artery disease was not significantly different between the two groups (p = 0.57). The inflammatory nonatherosclerotic group had fewer arterial calcifications in all arterial regions (all p < 0.05). As shown in Table 3, endovascular and surgical revascularization strategies were similarly performed between groups (endovascular, 88.6% vs 89.5%; surgical, 13.3% vs 12.4%), but adjunctive therapy including lumbar sympathetic ganglion block and cell therapy was more frequently performed in the inflammatory nonatherosclerotic group (5.4% vs 0.6% and 1.3% vs 0.1%, respectively). Hemodynamic improvement after revascularization was less marked in the inflammatory nonatherosclerotic group than in the atherosclerotic group (increase in skin perfusion pressure by ⩾ 20 mmHg, 64.1% vs 74.4%).
Baseline patient characteristics.
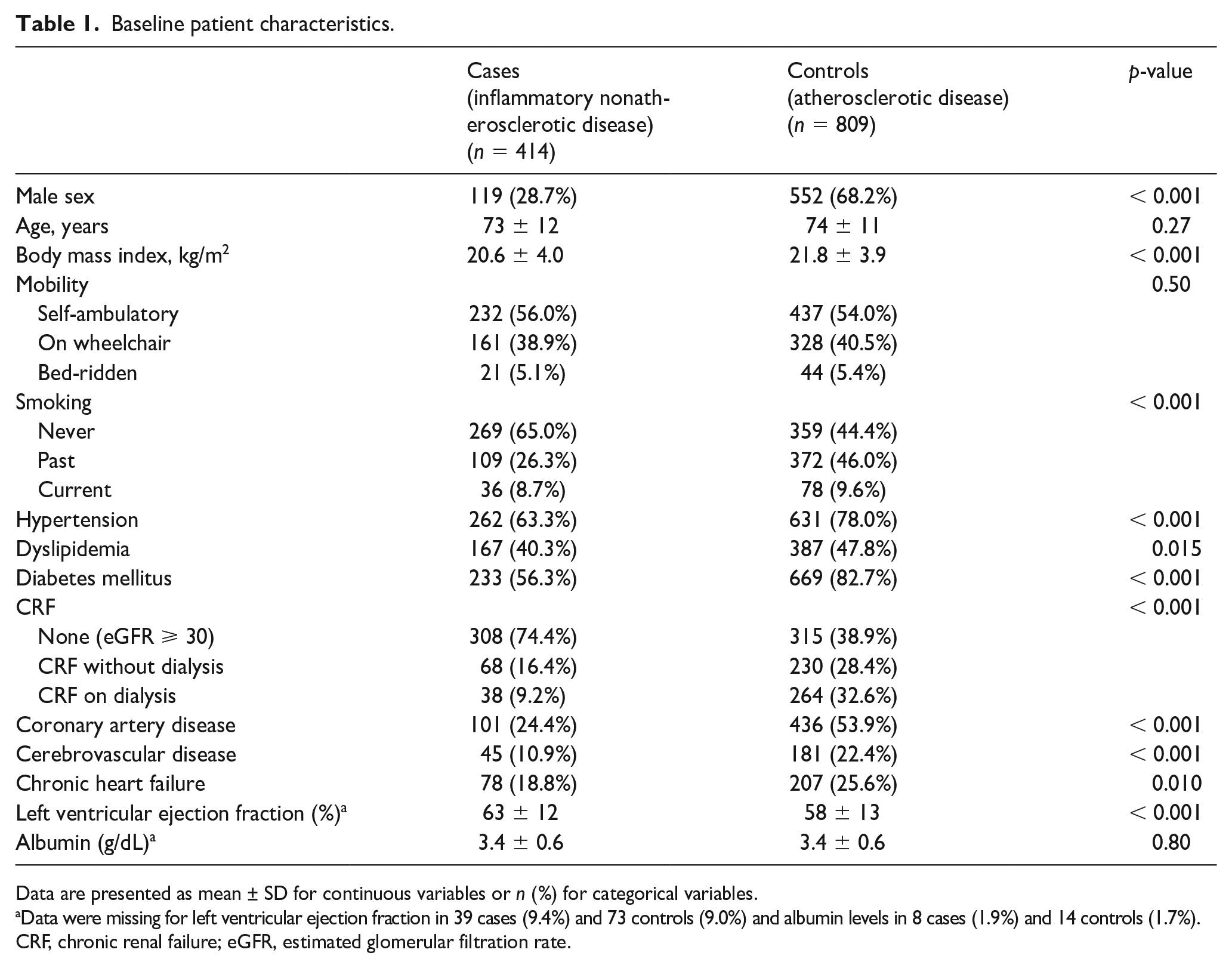
Data are presented as mean ± SD for continuous variables or n (%) for categorical variables.
Data were missing for left ventricular ejection fraction in 39 cases (9.4%) and 73 controls (9.0%) and albumin levels in 8 cases (1.9%) and 14 controls (1.7%).
CRF, chronic renal failure; eGFR, estimated glomerular filtration rate.
Baseline limb characteristics.
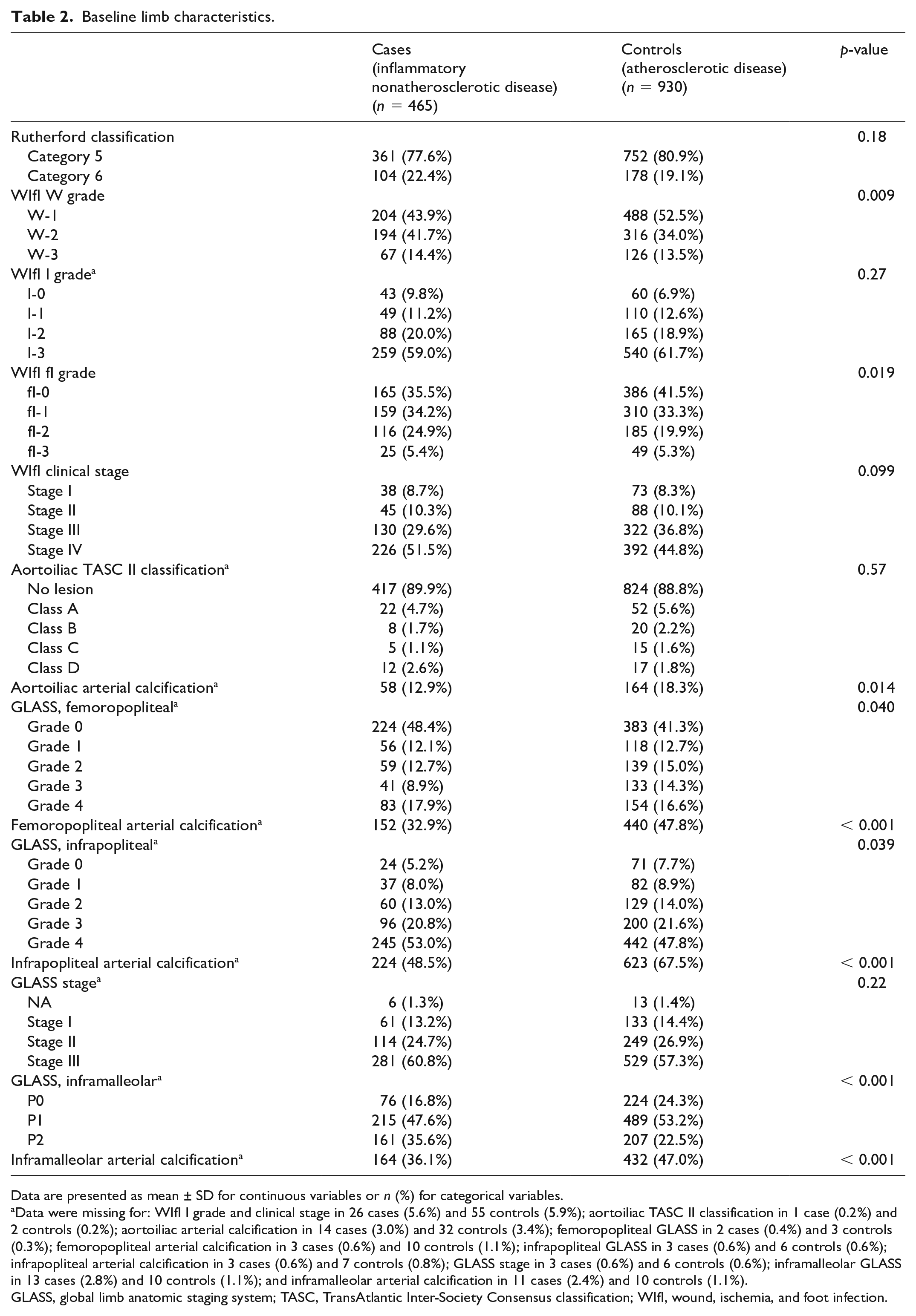
Data are presented as mean ± SD for continuous variables or n (%) for categorical variables.
Data were missing for: WIfI I grade and clinical stage in 26 cases (5.6%) and 55 controls (5.9%); aortoiliac TASC II classification in 1 case (0.2%) and 2 controls (0.2%); aortoiliac arterial calcification in 14 cases (3.0%) and 32 controls (3.4%); femoropopliteal GLASS in 2 cases (0.4%) and 3 controls (0.3%); femoropopliteal arterial calcification in 3 cases (0.6%) and 10 controls (1.1%); infrapopliteal GLASS in 3 cases (0.6%) and 6 controls (0.6%); infrapopliteal arterial calcification in 3 cases (0.6%) and 7 controls (0.8%); GLASS stage in 3 cases (0.6%) and 6 controls (0.6%); inframalleolar GLASS in 13 cases (2.8%) and 10 controls (1.1%); and inframalleolar arterial calcification in 11 cases (2.4%) and 10 controls (1.1%).
GLASS, global limb anatomic staging system; TASC, TransAtlantic Inter-Society Consensus classification; WIfI, wound, ischemia, and foot infection.
Treatment strategies and postrevascularization hemodynamic change.
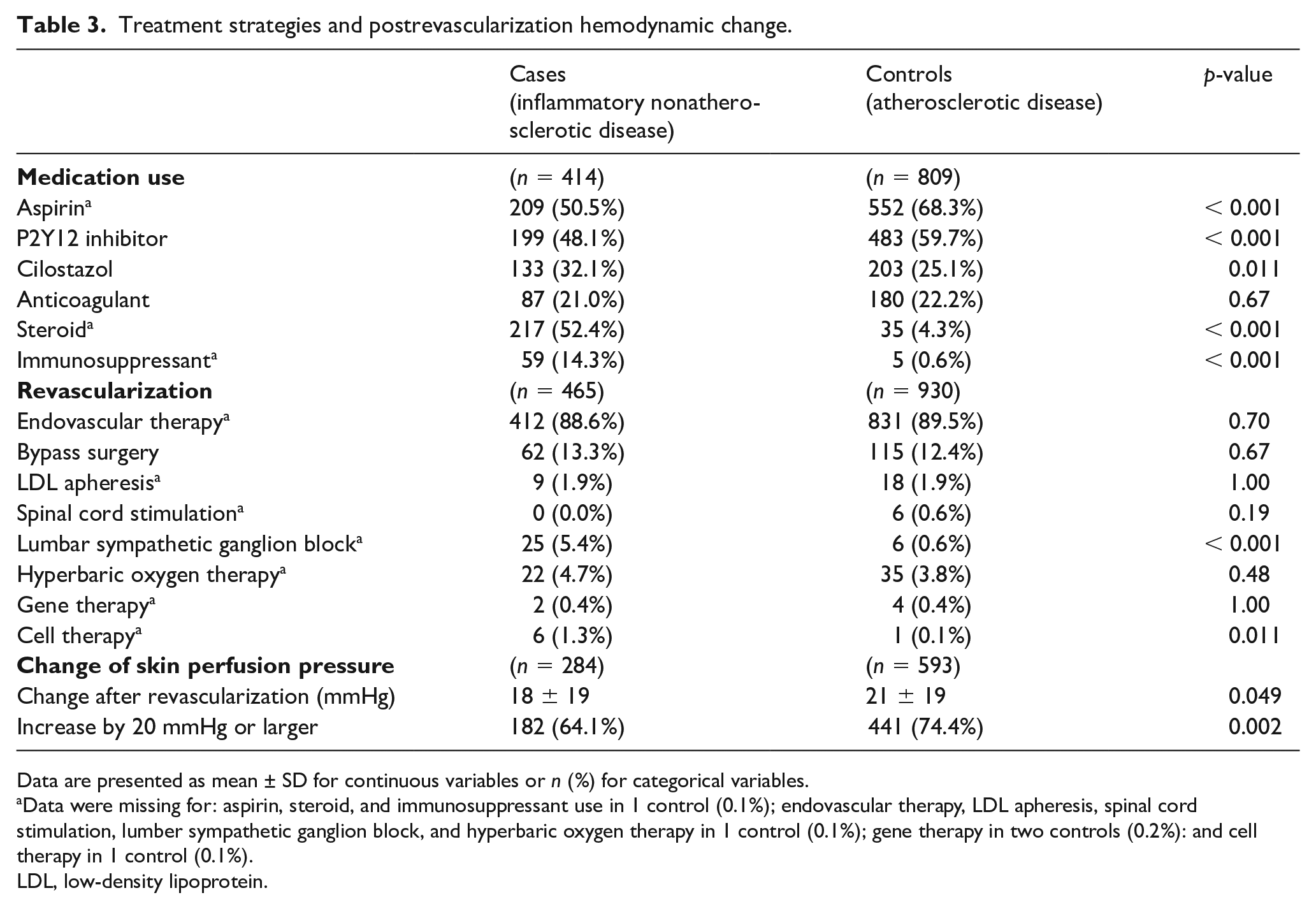
Data are presented as mean ± SD for continuous variables or n (%) for categorical variables.
Data were missing for: aspirin, steroid, and immunosuppressant use in 1 control (0.1%); endovascular therapy, LDL apheresis, spinal cord stimulation, lumber sympathetic ganglion block, and hyperbaric oxygen therapy in 1 control (0.1%); gene therapy in two controls (0.2%): and cell therapy in 1 control (0.1%).
LDL, low-density lipoprotein.
The inflammatory nonatherosclerotic group had a lower incidence rate of wound healing than the atherosclerotic group (incidence rate, 75.1 vs 117.1 per 100 limb-years; p < 0.001). The 1-year cumulative incidence rate of wound healing was calculated to be 59.1% (95% CI, 53.7% to 63.8%) versus 75.2% (71.9% to 78.2%) (Figure 1). The inflammatory nonatherosclerotic group had higher incidence rates of major reintervention (8.0 vs 5.8 per 100 limb-years; p = 0.006) and major amputation (21.5 vs 15.4 per 100 limb-years; p < 0.001), whereas they had a lower incidence rate of cardiovascular death (9.5 vs 20.2 per 100 person-years; p = 0.007). There was no significant intergroup difference in the incidence of any reintervention (52.5 vs 67.2; p = 0.64), all-cause mortality (41.5 vs 62.2 per 100 person-years; p = 0.067), and noncardiovascular death (25.3 vs 32.7 per 100 person-years; p = 0.85). The freedom rates from those adverse events are shown in Figure 2. Table 4 indicates the incidence risk ratios of inflammatory nonatherosclerotic disease relative to atherosclerotic disease for clinical outcomes. The association of inflammatory nonatherosclerotic disease with delayed wound healing was still significant after adjustment for covariates (adjusted incidence rate ratio, 0.61 [0.49 to 0.76]; p < 0.001), after adjustment for the propensity score (0.62 [0.50 to 0.77]; p < 0.001), and after inverse probability weighting (0.65 [0.58 to 0.73]; p < 0.001). Inflammatory nonatherosclerotic disease was also significantly associated with an increased risk of major reintervention after adjustment for the propensity score (4.57 [1.82 to 11.48]; p = 0.001) and inverse probability weighting (4.87 [2.94 to 8.06]; p < 0.001). Inflammatory nonatherosclerotic disease was significantly associated with an increased risk of major amputation and noncardiovascular death after adjustment for propensity score (1.71 [1.05 to 2.77] and 1.70 [1.15 to 2.51]; p = 0.031 and 0.008), whereas it was significantly associated with a lower risk of all-cause and cardiovascular death after inverse probability weighting (0.85 [0.72 to 1.00] and 0.67 [0.49 to 0.91]; p = 0.046 and 0.010) (Table 4).
Association of inflammatory nonatherosclerotic disease for clinical outcomes.
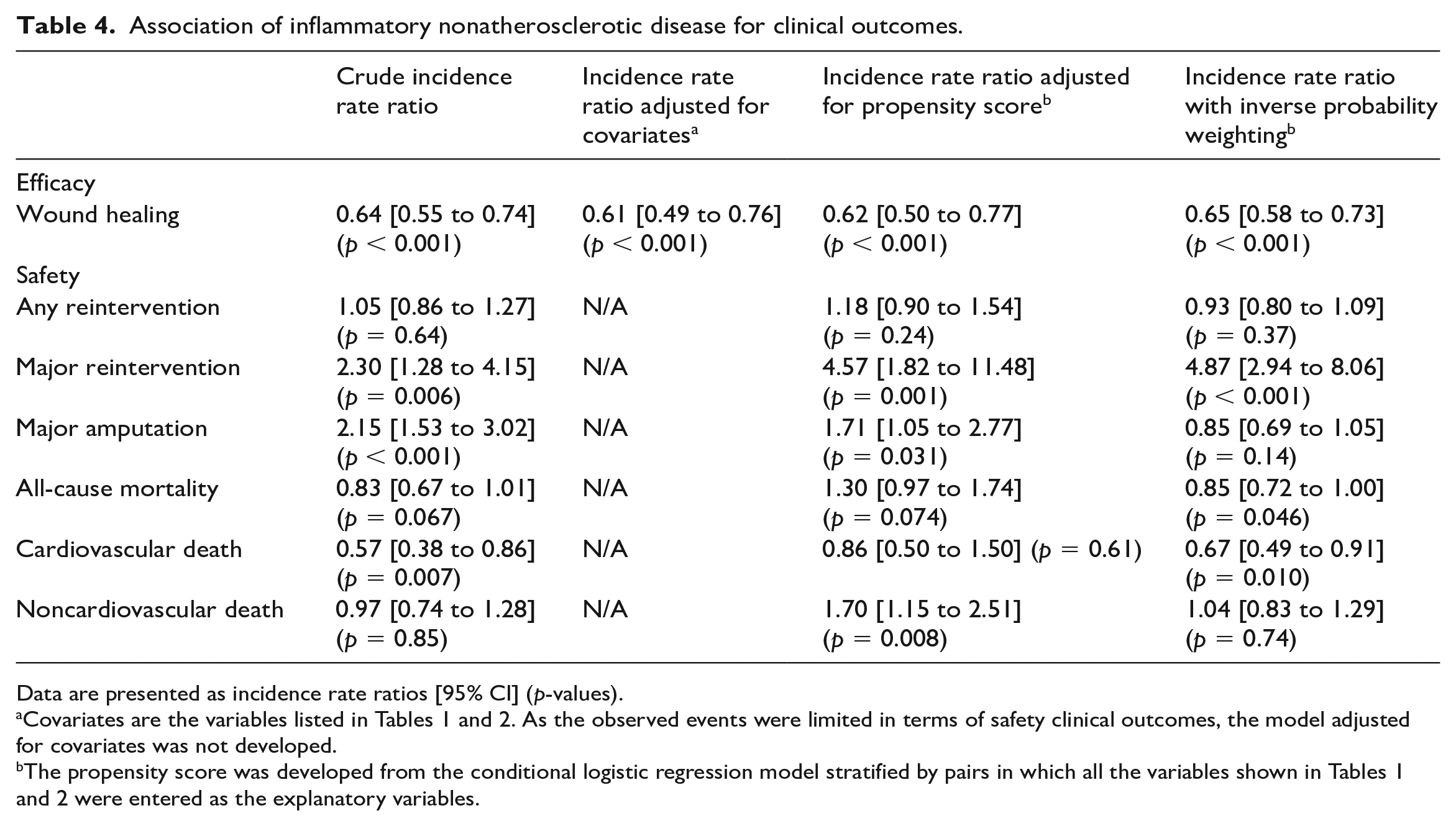
Data are presented as incidence rate ratios [95% CI] (p-values).
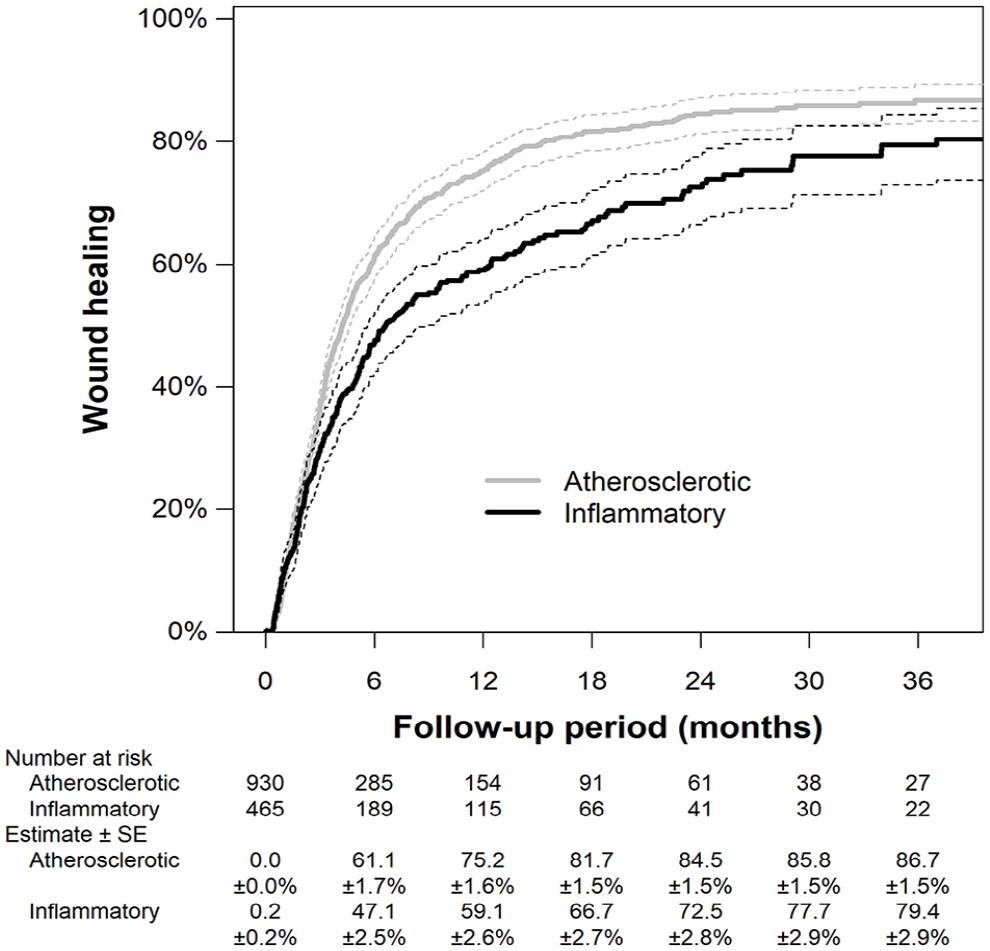
Cumulative incidence rate of wound healing.
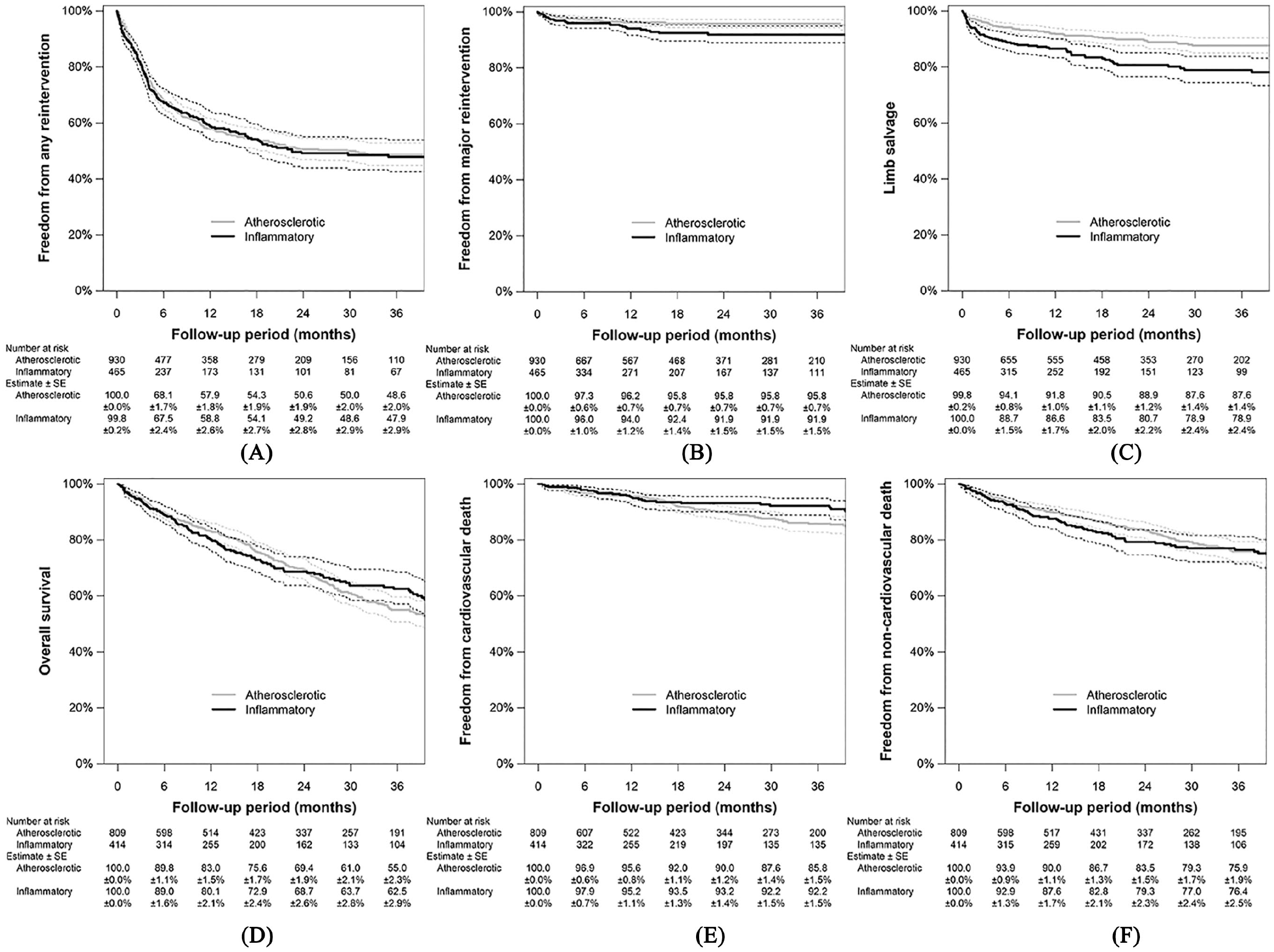
Safety outcome measures: freedom from any reintervention
Discussion
This multicenter and retrospective study revealed the clinical features and prognoses of CLTI with ischemic tissue loss due to inflammatory nonatherosclerotic disease versus atherosclerotic LEAD undergoing revascularization. The inflammatory nonatherosclerotic group had a lower prevalence of conventional atherosclerotic risk factors and relevant comorbidities, but more severe wounds, infections, and infrapopliteal and inframalleolar artery disease with a rarer presence of arterial calcification. The inflammatory nonatherosclerotic group had a lower rate of wound healing, even after adjusting for covariates.
LEAD based on inflammatory nonatherosclerotic etiologies, including vasculitis and collagen-tissue related disease, can be mainly characterized as inflammatory and thrombotic vascular changes, and is clearly distinguished from atherosclerotic etiology.8,9 Epidemiological studies have demonstrated that the prevalence and incidence of LEAD is higher in patients with nonatherosclerotic disease than in the general population. 9 When an ischemic wound due to inflammatory nonatherosclerotic disease is resistant to conservative therapy, lower-extremity revascularization has been empirically indicated, similarly to CLTI due to atherosclerotic disease.10–13 However, to date, data on CLTI due to inflammatory nonatherosclerotic disease are limited. To the best of our knowledge, this is the largest study to directly compare the clinical features and prognoses of CLTI patients undergoing revascularization between inflammatory nonatherosclerotic and atherosclerotic disease.
Patient characteristics
Male sex, smoking, hypertension, dyslipidemia, diabetes mellitus, and chronic renal failure are well-known risk factors for atherosclerosis, and coronary artery disease, cerebrovascular disease, and chronic heart failure are major comorbidities associated with atherosclerosis. It is not unexpected that the inflammatory nonatherosclerotic group had a lower prevalence of these features. A lower frequency of arterial calcification could be explained by a lower prevalence of diabetes mellitus and chronic renal failure, both of which are well-recognized as accelerators of vessel calcification. Mean serum albumin levels in the nonatherosclerotic group were 3.4 ± 0.6 g/dL, as low as those in the atherosclerotic disease group. Patients with CLTI due to inflammatory nonatherosclerotic etiologies would have as high a risk of malnutrition as those with CLTI due to atherosclerotic disease.
Wound severity and prognoses
The severity of the wound and infection was greater in the inflammatory nonatherosclerotic group. The proinflammatory and prothrombotic nature of inflammatory nonatherosclerotic etiologies might impair in vivo protective responses against tissue destruction, resulting in more extensive wounds. 14 Structural changes in the connective tissue might also adversely affect appropriate immune responses, causing bacterial infection. Furthermore, the administration of steroids and immunosuppressants would compromise the immune system, exacerbating bacterial infection.15,16 Increased severity of infrapopliteal and inframalleolar artery disease in inflammatory nonatherosclerotic arteries suggests that inflammatory nonatherosclerotic etiologies might be more prone to diffuse distal arterial involvement.
Severe wounds, infections, and artery disease are major risk factors for delayed wound healing. 17 It is intuitively reasonable that the inflammatory nonatherosclerotic group had a lower rate of wound healing. However, the rate was still lower after adjustment for these covariates, indicating that other obstacles to wound healing would be present in inflammatory nonatherosclerotic disease. One possible obstacle is inferior vessel patency after revascularization. The inflammatory nonatherosclerotic group was less likely to achieve sufficient hemodynamic improvement after revascularization and had a higher risk of major reintervention and amputation. Inflammatory nonatherosclerotic artery disease might be more prone to recoil after endovascular therapy and graft failure, including anastomotic complications after bypass surgery. Insufficient hemodynamic improvement after revascularization may have been secondary to impaired perfusion beyond pedal arteries, especially if indicated by skin perfusion pressure. In general, inflammatory nonatherosclerotic etiologies, including vasculitis and collagen disease, are likely to have impairments in the digital arteries. 10 The higher frequency of toe ulcers and gangrene in the current study might imply such characteristics. Distal artery disease would not only impair tissue perfusion but also work as poor runoffs of recanalized vessels that potentially adversely affect vessel patency and consequently resulted in major amputation.
Another potential obstacle is structural changes in the connective tissue due to the underlying disease. Altered histological structures would interfere with the healing process. Furthermore, chronic administration of steroids and immunosuppressants would also adversely affect the respective steps of wound healing, including hemostasis, inflammation, proliferation, and remodeling.15,16 However, steroids protect against restenosis. 9 Future studies are warranted to reveal the impact of these drugs on clinical outcomes.
Mortality risk
The risk of all-cause mortality was not significantly different between the inflammatory nonatherosclerotic group and the atherosclerotic group, except after inverse probability weighting. Although the mechanisms remain unknown, it would be of clinical interest that the risk would be broadly similar despite the considerably different baseline characteristics. The fact that CLTI develops per se might determine their prognosis. On the other hand, the propensity score-adjusted model, but not the other models, indicated that the inflammatory nonatherosclerotic group had a higher risk of noncardiovascular death, though the crude comparison and that after inverse probability weighting suggested that they had a lower risk of cardiovascular death. The causes of death might differ between the two groups.
Study limitations
First, the data were retrospectively collected from medical records; the retrospective nature of the study could be associated with potential biases. In general, inflammatory nonatherosclerotic CLTI does not primarily undergo revascularization and the population that this study included would not be a representative of a general population with CLTI due to inflammatory nonatherosclerotic disease. Second, the diagnosis of inflammatory nonatherosclerotic disease was extracted from medical records and, therefore, on a clinical basis. Arterial lesions were not histopathologically examined; therefore, cases complicated by inflammatory nonatherosclerotic etiologies, atherosclerosis, and/or Raynaud’s could not be excluded. Third, detailed data regarding the duration, severity, and activity of inflammatory nonatherosclerotic disease, and treatment regimens including appropriated medical and wound therapy prior to revascularization were unavailable. Those aspects might influence prognosis. Fourth, the severity of tissue loss and artery disease was assessed at each participating center and not at a core laboratory, and therefore, the procedure was not standardized.
Conclusions
This multicenter, retrospective, observational study revealed the clinical features and prognoses of CLTI presenting with ischemic tissue loss due to inflammatory nonatherosclerotic disease and undergoing revascularization, in comparison with atherosclerotic disease. The inflammatory nonatherosclerotic group had a lower rate of wound healing, even after adjusting for covariates.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231164659 – Supplemental material for Clinical outcomes of chronic limb-threatening ischemia due to inflammatory nonatherosclerotic versus atherosclerotic etiologies undergoing revascularization
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231164659 for Clinical outcomes of chronic limb-threatening ischemia due to inflammatory nonatherosclerotic versus atherosclerotic etiologies undergoing revascularization by Osamu Iida, Mitsuyoshi Takahara, Akio Kodama, Terutoshi Yamaoka, Yoshimitsu Soga and Toshiaki Mano in Vascular Medicine
Footnotes
Acknowledgements
The authors would like to thank the medical staff and clinical research coordinators at the participating centers.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The DECOPON study was supported by the Research Association for Lower Limb Artery Revascularization (LIBERAL) sponsored by the following companies (in alphabetical order): Boston Scientific Japan K.K., Kaneka Medix Corp., OrbusNeich Foundation, and Terumo Corp. The funding companies had no role in the design of the study, selection of the enrolled patients, treatment strategy, revascularization procedures or equipment, or collection, analysis, or interpretation of the data.
Data availability statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
