Abstract
Background:
This retrospective comparative cohort study evaluated the clinical outcome of angiosome-guided endovascular arterial reconstructions in chronic limb-threatening ischemia (CLTI) due to multilevel peripheral artery disease (PAD).
Methods:
Patients treated in an endovascular fashion for CLTI with tissue loss due to multilevel PAD were analyzed. Limbs were classified as having undergone either angiosome-guided (direct) revascularization (DR) or nonangiosomic (indirect) revascularization (IR). DR was defined as uninterrupted in-line flow to the affected angiosome, revascularization through the pedal arch was also considered direct. Groups were adjusted with propensity score (PS) matching and compared for amputation-free survival (AFS), freedom from major adverse limb events (MALE), and healing rate at 12 months.
Results:
A total of 174 patients (81 men, mean age 70.0 ± 10.4 y) were included. PS matching produced two groups of 55 patients each: DR (24 men, mean age 71.7 ± 10.7 y) and IR (26 men, mean age 72.0 ± 9.4 y). The matched groups had no significant differences in baseline variables. At 12 months there were no significant differences in AFS (73.2% vs 71.6%; p = 0.841), freedom from MALE (71.7% vs 66.1%; p = 0.617), and healing rate (72.7% vs 72.0%; p = 1.000) between DR and IR, respectively.
Conclusion:
This study failed to support the use of angiosome concept in CLTI due to multilevel disease.
Introduction
Lower limb revascularization in chronic limb-threatening ischemia (CLTI) with tissue loss is a demanding procedure, which sometimes may not achieve its ultimate goals – limb salvage and wound healing – even when the patency is preserved. 1 One of the ways to explain this has been the angiosome concept, originally described by Taylor et al. 2 It divides the foot between different angiosomes – blocks of tissue, each receiving blood from a separate arterial branch. 3 Direct revascularization (DR) implies that flow is restored to the affected foot angiosome either via in-line flow or via collateral perfusion through the pedal arch. It has been reported to improve the long-term outcome compared to indirect revascularization (IR), when the affected angiosome is perfused indirectly through collaterals.4–9 However, evidence regarding the effect of DR has been conflicting, with some studies showing benefit for both limb salvage and wound healing,4,5,10 yet others reporting only improved wound healing5,11–14 or even no difference at all. 15 The role of pedal arch and major collaterals was also reported.4,8,15–17 In recent years, the angiosome concept has been much debated, its possible benefit reconsidered and called into question.1,18 Importantly, almost all studies of angiosome-oriented endovascular treatment included only infrapopliteal lesions. Only a few of them7,13 dealt with a mixed cohort of mostly infrapopliteal disease with a minor proportion of multilevel occlusions and no analysis of possible contribution from the inflow disease. At the same time, CLTI is known to be associated with occlusive lesions involving several arterial segments and these multilevel changes are diagnosed in the vast majority of patients with rest pain or tissue loss.19–22 Therefore, the role of angiosome-guided endovascular therapy in multilevel PAD is still unclear. As well, this type of lesion distribution brings up some new factors, which can be far more predictive of the long-term outcome than angiosome perfusion status (e.g., the presence of iliac disease, deep femoral artery occlusion, and length and type of femoropopliteal lesion).23–25 Therefore, the aim of this study was to evaluate the angiosome concept in a pure cohort of patients with CLTI and multilevel PAD.
Methods
Study design
This single-center retrospective cohort study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Ethics Board (Ethics Committee’s approval protocol #0049). Informed consent was provided by all study patients. We retrospectively analyzed the hospital database to find patients with ischemic tissue loss due to PAD who underwent endovascular interventions. Exclusion criteria were (i) claudication or rest pain without tissue loss (Rutherford 3–4); (ii) restenosis in an infrainguinal bypass; (iii) purely infrapopliteal disease; (iv) unsuccessful revascularization, where no improvement in patency was achieved; (v) patients in whom no crural arteries were patent at the end of the procedure; and (vi) presence of occlusive lesions in superficial femoral artery (SFA) or popliteal artery at the end of the procedure (e.g., foot revascularization via deep femoral artery [DFA] or SFA collaterals).
Patient cohorts
The remaining patients were classified as either DR or IR – in accordance with the angiosome principle. The 7-day rates of major amputation were compared between the groups. The early amputees were excluded from further calculations based on the assumption that early major amputation occurs due to delayed rather than indirect revascularization, and in order to refine the survival analysis. After exclusion of 7-day major amputations, the DR and IR groups were compared by baseline demographic, clinical, and angiographic variables. To equalize the study groups, propensity score (PS) matching was performed with appropriate predictors. The resulting PS-matched cohorts were again compared using the same baseline demographic, clinical, and angiographic variables, as well as procedural characteristics to check for significant differences. The role of angiosome-guided revascularization was assessed by comparing amputation-free survival (AFS) and cumulative freedom from major adverse limb events (MALE), as well as the 12-month healing rate between the two study groups both prior to and after the PS matching. Owing to the retrospective nature of the study, healing rate was assessed only in patients for whom the 12-month data were available. Possible contribution from the number of peroneal artery terminal branches and the completeness of pedal arch in the IR cohort to 12-month AFS and freedom from MALE was measured in the matched population. The role of other pertinent covariates, namely the number of crural runoff arteries and the extent of tissue loss, was tested using a multivariable proportional hazards model.
Preoperative assessment
All patients underwent basic clinical evaluation by vascular specialist and appropriate laboratory tests. The amount of tissue loss and wound healing was assessed on admission and at discharge, respectively. Imaging methods included duplex ultrasound (DUS) and digital subtraction angiography (DSA) in all patients. DUS was used to confirm the presence of occlusive lesions. Ankle pressure, toe pressure, and ankle–brachial index (ABI) were measured where available and tissue loss was graded according to the Wound, Ischemia, and foot Infection (WIfI) classification system. 26
Endovascular procedure
Endovascular procedures were performed by experienced interventionalists according to the standard of care approach.
The strategy of infrapopliteal angioplasty was to open as many runoff arteries as possible, preferably including the one that supplied the affected angiosome to achieve DR. The number of patent crural arteries at completion angiography and the presence of functioning terminal branches of peroneal artery (calcaneal branch and perforating branch) were assessed during the intervention. All patients underwent duplex ultrasound the day after the intervention to assess early patency.
Dual antiplatelet therapy (clopidogrel 75 mg/d + ASA 100 mg/d) was administered for 6 months postoperatively, with acetylsalicylic acid 100 mg/d continued indefinitely thereafter.
Wound care
Wound care included antimicrobial therapy, serial debridement and, occasionally, negative pressure wound therapy and skin flaps.
Follow-up and reinterventions
Data on 12-month AFS, freedom from MALE, and healing rate were obtained from telephone contacts. Indications for reintervention at follow-up were signs of CLTI relapse, including rest pain and/or ischemic tissue loss, with confirmed restenosis or re-occlusion of limb arteries.
Endpoints and definitions
Ischemic tissue loss was defined as the clinical syndrome of rest pain with tissue loss and confirmed by DUS and DSA showing occlusive lesions located in iliac, femoral, and/or popliteal segments (purely infrainguinal disease was an exclusion criterion). Necrotic changes were classified according to the WIfI grading system for amputation risk.
Foot angiosomes were defined in accordance with Attinger et al. 3 : three angiosomes fed by posterior tibial artery (PTA) branches – instep and hallux (medial plantar artery), lateral midfoot and forefoot (lateral plantar artery), medial and plantar aspects of the heel (calcaneal branch); two angiosomes receiving blood from the peroneal artery – lateral aspect of the heel (calcaneal branch) and anterolateral portion of the ankle + lateral forefoot; one angiosome of the dorsalis pedis artery (DPA) – dorsum of the foot. Direct revascularization was defined as restoring direct in-line blood flow to the affected angiosome. Importantly, revascularization via complete pedal arch (e.g., dorsum of the foot from lateral plantar artery or toes from the DPA) was also considered direct. If the necrotic changes involved several angiosomes, revascularization was defined as direct only if uninterrupted flow was restored to each of the affected angiosomes. All other variants of revascularization were classified as indirect. Pedal arch quality was defined according to Troisi et al. 15 Complete pedal arch was defined when both the DPA and at least one of the plantar arteries were patent and joined to each other; incomplete pedal arch was defined when the DPA or one of the plantar arteries were patent but not joined to each other; and absent pedal arch was defined when neither the DPA nor at least one of the plantar arteries were patent.
Technical success of the procedure was defined as < 50% residual stenosis at any point of the treated arterial segment. MALE was defined as the combination endpoint of target lesion revascularizarion (TLR) (any revascularization of the same arterial segment, either open or endovascular) and major amputation (limb amputation above the ankle). AFS was defined as the combination endpoint of overall survival and limb salvage (freedom from major amputation). Wound healing was defined as complete closure of the skin defect with no need for further wound dressings.
Statistical analysis
Data are presented as counts with percentages for nominal and ordinal variables; means and SDs for normally distributed interval variables; medians and ranges for interval variables with skewed distribution.
Categorical variables were compared with Fisher’s exact test. Continuous variables were compared with independent samples t-test or Mann–Whitney U-test. Statistical significance was defined as p < 0.05.
PS was calculated for each case with all pertinent variables used as predictors: demographic (age, sex), clinical (relevant comorbidities, WIfI stage), and angiographic (presence and type of occlusive lesions in each of the limb segments; SFA lesion length; pedal arch patency). After calculating the PS, one-to-one optimal matching was performed.
Cumulative probabilities of achieving an endpoint were estimated by Kaplan–Meier method and compared using log-rank test. The possible role of pertinent covariates was evaluated in a univariate proportional hazards model. The chi-squared test was used to assess statistical significance, which was defined as p < 0.05.
Statistical analyses were done using IBM SPSS, Version 26.0 (IBM Corp., Armonk, NY, USA).
Results
The search identified 249 patients. After exclusion criteria had been applied, a total of 183 patients were left for further analysis. They were classified as DR (n = 123) and IR (n = 60). The 7-day major amputation rate was four (3.3%) versus five (8.3%) in DR and IR cohorts, respectively (p = 0.156). After exclusion of early major amputations, the groups were compared by their demographic, clinical, and angiographic variables (Table 1). Significant differences were found in the history of myocardial infarction (MI), history of stroke, presence of occlusive lesions in the external iliac artery (EIA), deep femoral artery, median SFA lesion length, presence of occlusive lesions in the anterior tibial and peroneal arteries, as well as completeness of the pedal arch. After PS-matching there were no significant differences between the matched DR and IR cohorts. The ischemic tissue loss commonly involved the forefoot angiosomes (posterior tibial and dorsalis pedis arteries). Most patients in the PS-matched cohorts (n = 87; 79.1%) had SFA disease combined with crural lesions. The remaining had PA lesions combined with either inflow (i.e., iliac) or runoff disease, or both.
Baseline demographic, clinical, and angiographic characteristics of direct versus indirect revascularization cohorts before and after propensity score matching.
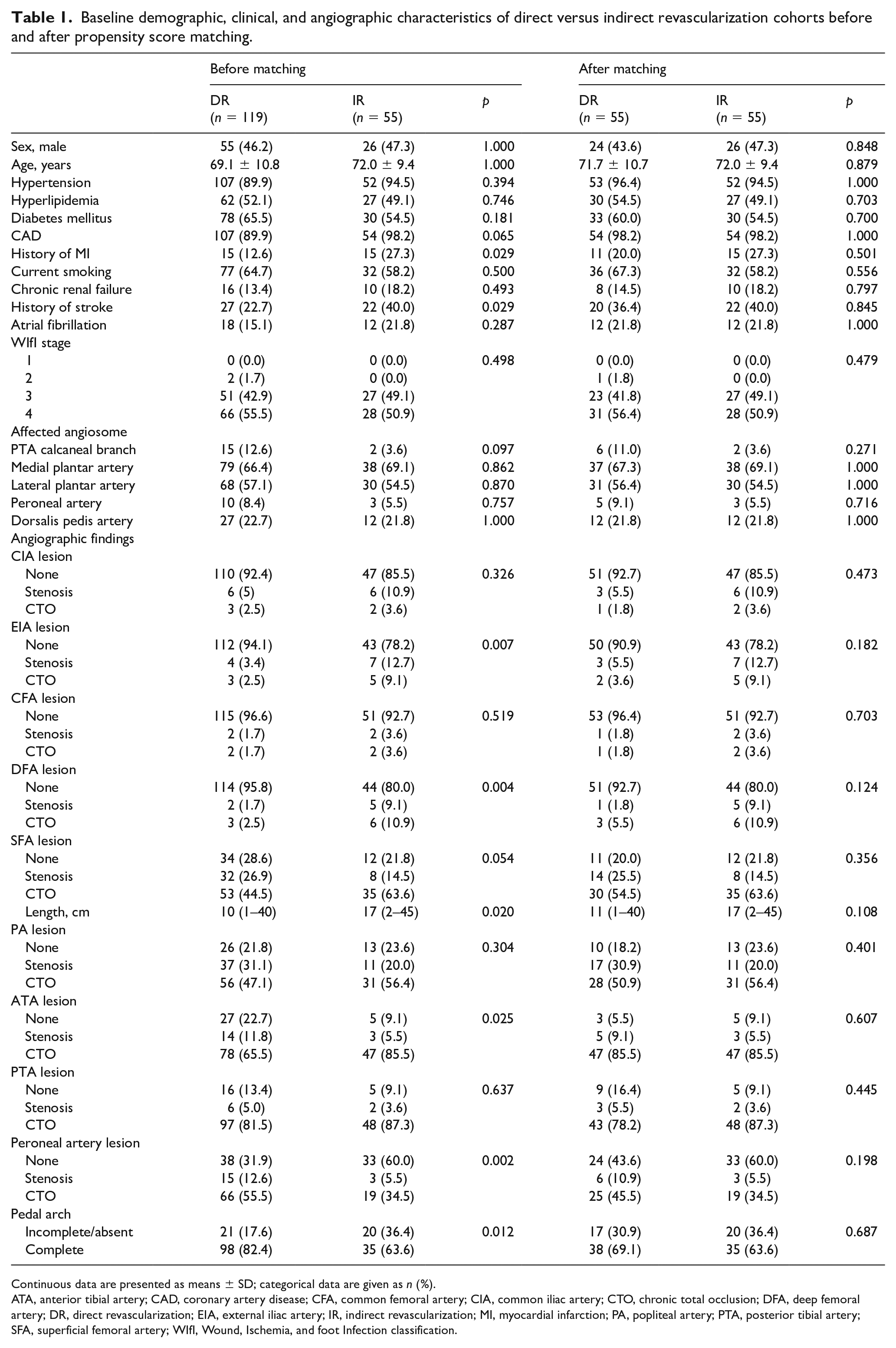
Continuous data are presented as means ± SD; categorical data are given as n (%).
ATA, anterior tibial artery; CAD, coronary artery disease; CFA, common femoral artery; CIA, common iliac artery; CTO, chronic total occlusion; DFA, deep femoral artery; DR, direct revascularization; EIA, external iliac artery; IR, indirect revascularization; MI, myocardial infarction; PA, popliteal artery; PTA, posterior tibial artery; SFA, superficial femoral artery; WIfI, Wound, Ischemia, and foot Infection classification.
During the endovascular procedure (Table 2) the most commonly treated segments were SFA and PA followed by below-the-knee (BTK) interventions. The DR group had a significantly higher rate of multiple crural revascularization (more than one patent runoff artery at completion angiography) – 85.4% compared to only 40.0% among IR patients (p < 0.001). Successful angioplasty of the anterior tibial artery (ATA) and PTA (feeding the most commonly affected angiosomes) was predictably more prevalent among DR patients.
Details of endovascular procedure in propensity score-matched direct versus indirect revascularization cohorts.
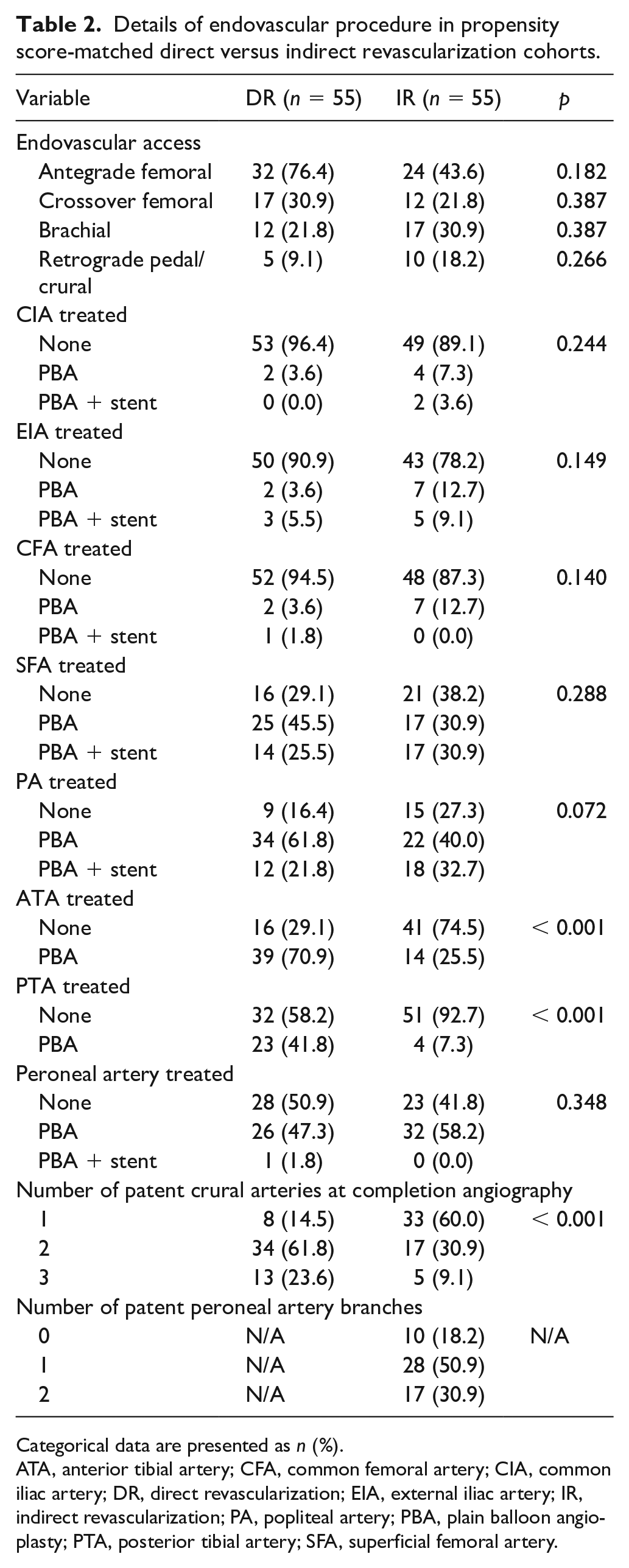
Categorical data are presented as n (%).
ATA, anterior tibial artery; CFA, common femoral artery; CIA, common iliac artery; DR, direct revascularization; EIA, external iliac artery; IR, indirect revascularization; PA, popliteal artery; PBA, plain balloon angioplasty; PTA, posterior tibial artery; SFA, superficial femoral artery.
Survival analysis (Figure 1) showed that in both the unmatched and the matched population the 12-month AFS and freedom from MALE were very similar between DR and IR cohorts: AFS (81.9% vs 71.6%; p = 0.220) and freedom from MALE (66.1% vs 73.1%; p = 0.374) for the unmatched; AFS (73.2% vs 71.6%; p = 0.841) and freedom from MALE (71.7% vs 66.7%; p = 0.617) for the matched.
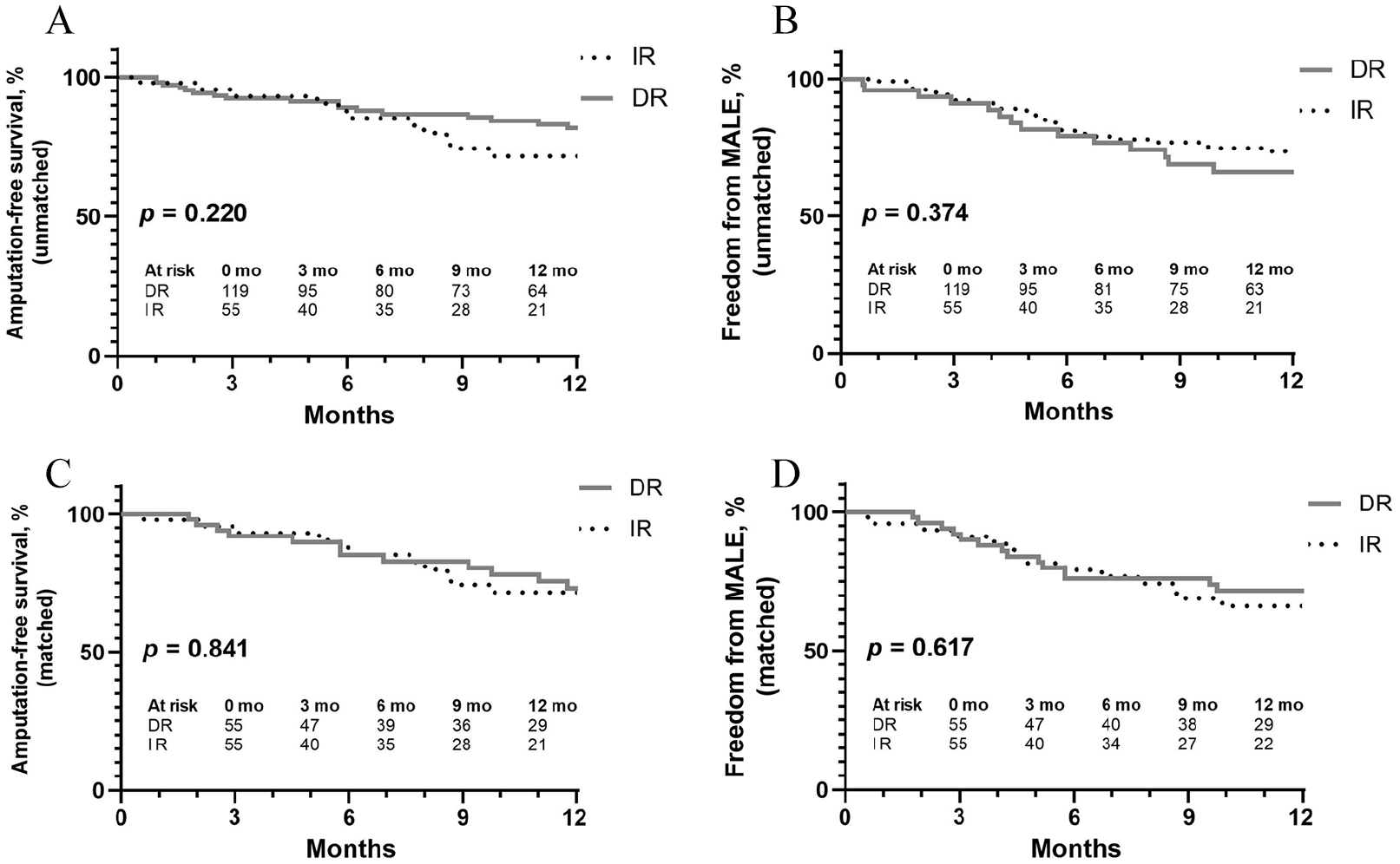
Kaplan–Meier assessment of 1-year outcomes of direct versus indirect revascularization.
The 12-month healing rate in the unmatched DR and IR cohorts was 75.3% vs 52.7%, respectively (p = 0.476). After PS matching, the healing rate at 12 months was again found to be similar between DR and IR cohorts, as well as in patients with different WIfI stage, number of crural runoff vessels, and pedal arch condition (Table 3).
Wound healing at 12 months as a function of different risk factors.
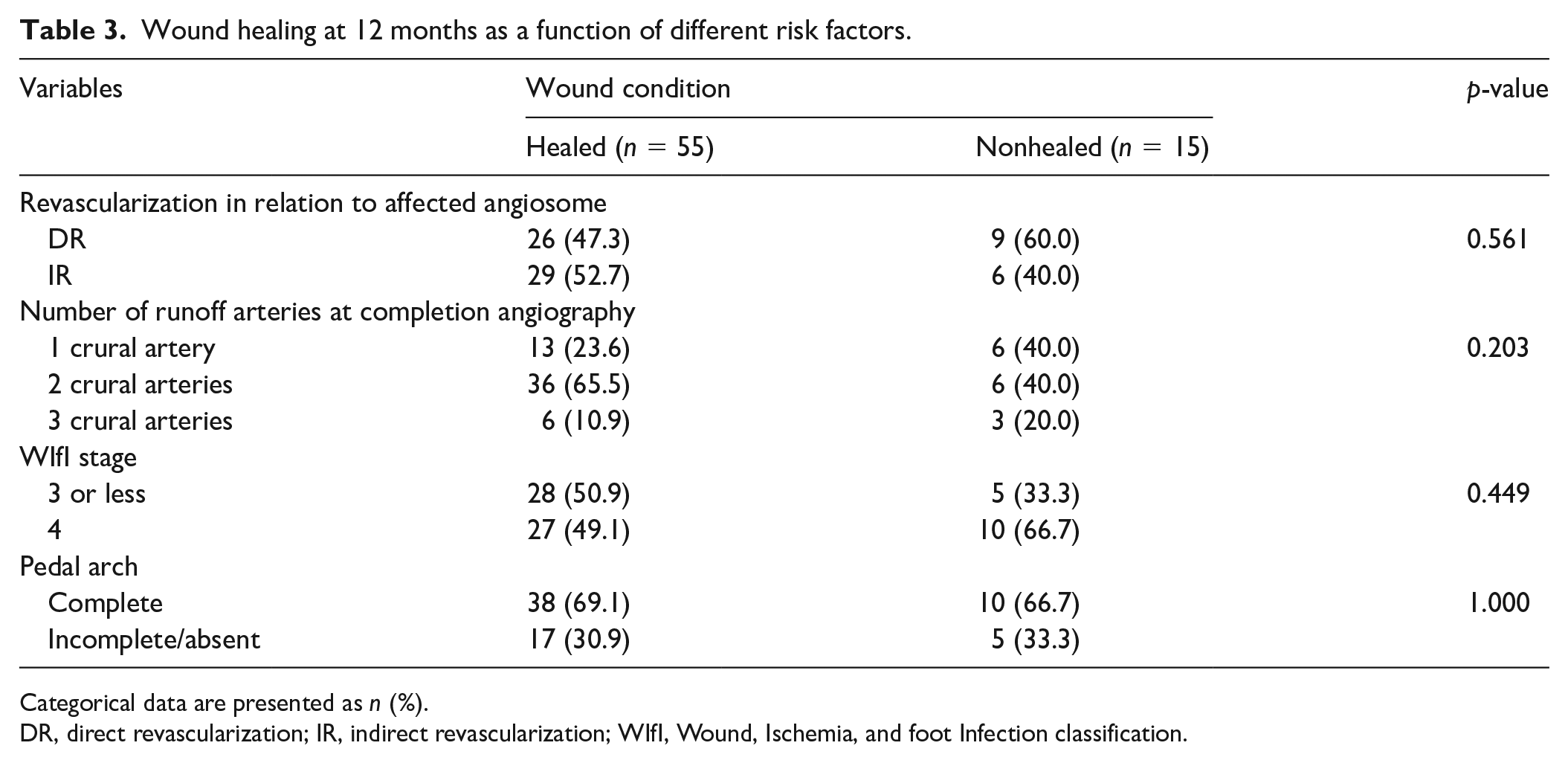
Categorical data are presented as n (%).
DR, direct revascularization; IR, indirect revascularization; WIfI, Wound, Ischemia, and foot Infection classification.
The number of patent crural arteries at completion angiography in the entire matched population, as well as the quality of pedal arch and the number of functioning peroneal branches in the IR cohort exerted no significant influence on 12-month cumulative AFS and freedom from MALE (Table 4).
Risk factors for 12-month amputation-free survival and freedom from MALE according to multivariable proportional hazards model.
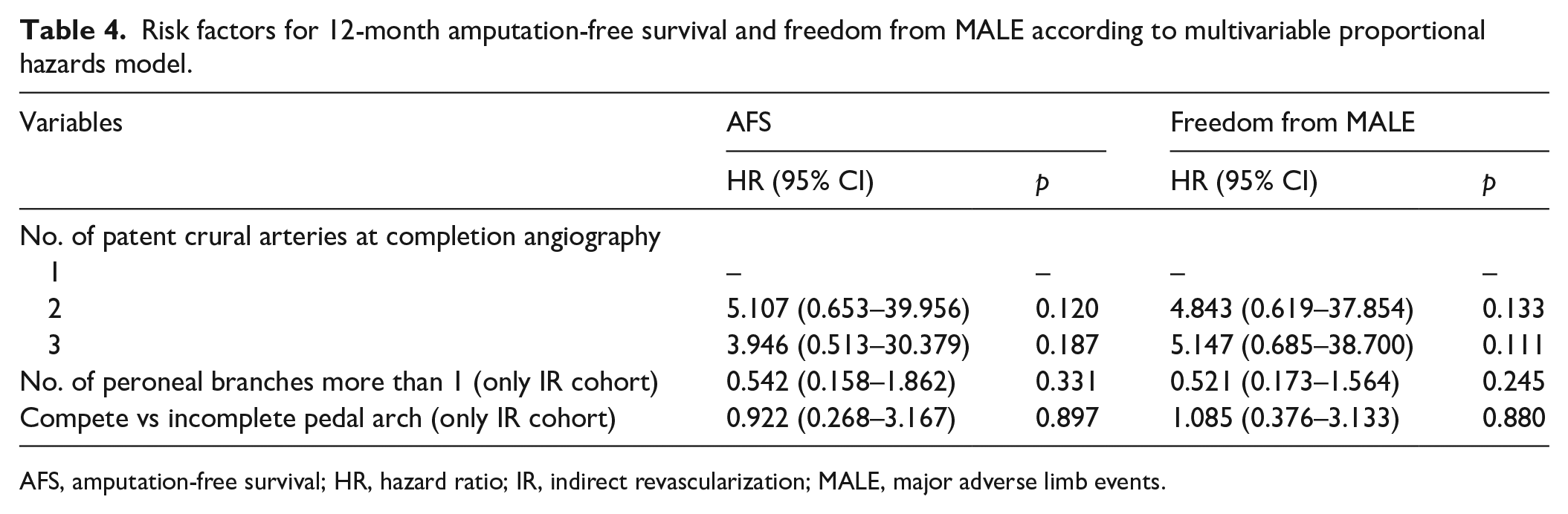
AFS, amputation-free survival; HR, hazard ratio; IR, indirect revascularization; MALE, major adverse limb events.
According to the multivariable proportional hazards model, there was no significant contribution from the number of runoff arteries > 1 (HR 1.792 [0.689–4.657], p = 0.232; HR 1.050 [0.433–2.550], p = 0.914), WIfI stage 4 versus 3 (HR 1.330 [0.585–3.022], p = 0.496; HR 0.865 [0.403–1.855], p = 0.709), and DR versus IR (HR 1.223 [0.475–3.147], p = 0.677; HR 0.841 [0.360–1.966], p = 0.690) on the 12-month AFS and cumulative freedom from MALE, respectively. However, the low number of patients with nonhealing wounds (n = 15) greatly limits the reliability of the analysis.
Discussion
Owing to the high proportion of long and totally occlusive lesions, 1 as well as severe calcinosis, 19 particularly in crural arteries, 27 multilevel infrainguinal reconstructions can become time-consuming, associated with high radiation exposure and contrast volume.28–30
This raises a question of whether the direct revascularization of foot angiosomes should be pursued in this highly challenging category of patients. Evidence to support this concept are very sparse.7,13,14 Our study focused exclusively on patients with multilevel disease and excluded those with purely infrapopliteal disease, who have been amply investigated before in studies of the angiosome concept,4–6,8–13,15–17 comprise a separate clinical entity of smaller vessel diameter, shorter affected segment, different patterns of collateral circulation and, therefore, were excluded.
In our study, the unmatched DR and IR cohorts appeared significantly different in the distribution of some key factors predicting the long-term outcome: prevalence of EIA lesions, length of SFA occlusions, rate of stroke/MI, and pedal arch quality. After applying the PS-matching procedure, we obtained two comparable groups.
Patients in most studies of endovascular angiosome-guided revascularization (except Acin et al. 13 ) had at least one patent crural artery at completion angiography. In those with IR this artery was most commonly the peroneal artery and the ischemic tissue loss usually involved the forefoot (i.e., the PTA/ATA angiosomes). Hence, the question why the IR outcomes were similar or inferior to those of DR is largely a question of: how good were the collaterals connecting the peroneal artery to the forefoot angiosomes?
Zheng et al. 4 and Varela et al. 8 found that IR without apparent collaterals to the affected angiosomes had significantly worse limb salvage and ulcer healing compared to IR with collaterals. The number of peroneal artery terminal branches (2 vs 1) was also reported to improve the AFS in ischemic tissue loss; however, the study included only open reconstructions. 17 The pedal arch may play an important part in collateral perfusion of the forefoot, with some publications showing improved AFS15–17 and wound healing15,16 in patients with complete compared to incomplete or absent arch.
In our cohort of patients with multilevel PAD, we failed to reveal any effect of the number of peroneal branches and pedal arch quality on the 12-month AFS and freedom from MALE in the IR group.
One of the ways to explain this inconsistency between different trials could be the selection bias, which occurs because of the time interval between the onset of symptoms and the first assessment by a vascular specialist. The longer the interval, the higher might be the likelihood that patients with a poor collateral network will proceed to primary major amputation due to more rapid progression of gangrene and eventually be knocked out from the study sample. The per cent of primary amputations due to CLTI in everyday practice ranges between 20% and 40%31,32 and the time from wound onset to assessment in studies of angiosome concept can be as long as 2.7 months. 33 Thus, the angiosome concept might work earlier in the course of the disease and be futile in late-comers. A subgroup or correlation analysis would help confirm or reject this hypothesis in future studies.
The number of patent crural arteries at completion angiography has been suggested to affect the long-term outcome.13,34 Kobayashi et al. 34 found that two patent tibial arteries were associated with better wound healing and higher freedom from TLR compared to a single patent artery. According to Acin et al., 13 the 12-month healing rate and limb salvage were significantly improved in patients with more than one runoff artery compared to zero runoff; however, no difference was found between patients with one runoff artery and those with two or three arteries. Importantly, both studies had substantial differences between single and multiple revascularization cohorts, either in the rate of DR34 or sex, smoking, and concurrent treatment of the femoropopliteal sector, 13 which could have biased the comparison. Likewise, in our study, we found significant difference in the number of patent runoff arteries between DR and IR groups. Therefore, both variables were included into a multivariate proportional hazards model together with another possible confounder – the extent of tissue loss.17,33 None of them appeared to affect the AFS or freedom from MALE. It is possible that these factors behave differently in multilevel disease.
We assessed wound healing at a single time point in a limited sample of patients. Therefore, Kaplan–Meier survival analysis and Cox regression could not be used to provide more sensitive and comprehensive evaluation. On the other hand, in some of the relevant studies where the healing was measured as a cumulative likelihood, no significant effect of any of the pertinent variables was found as well.15,35 Apart from the above-mentioned possibility of late-look bias, this variability can be explained by the confounding effect of wound care protocol. According to Mii et al., 36 more aggressive debridement after revascularization for CLTI was able to decrease the mean healing time almost twice (from 82 to 48 days), overtaking the reported modest gain in wound closure speed after angiosomic revascularization versus revascularization via collaterals (e.g., 6.8 vs 7.9 months according to Alexandrescu et al. 11 ). Thus, a minor difference in wound healing might be obscured by variations in wound care practices across outpatient hospitals, where patients are followed after being discharged from the tertiary care center. Therefore, vascular teams that are unable to arrange frequent and standardized wound care until complete epithelialization are less likely to detect a possible perfusion-related effect on healing rate.
Overall, according to our study, angiosome-oriented revascularization in multilevel disease does not need to be pursued at any cost provided that collateral perfusion from a single crural artery is achieved. However, the results can’t be directly extended to patients with tissue loss located within heel angiosomes or involving multiple angiosomes – a cohort that still requires additional analysis.
Study limitations
Limitations of the study include (i) single-center and retrospective design, (ii) some endpoints, particularly wound healing, assessed by telephone contact, (iii) variabilities in the outpatient wound care protocol, and (iv) healing rate measured at a single time point, not as cumulative likelihood.
Conclusion
This study did not find evidence to support angiosome-guided revascularization in patients with ischemic tissue loss due to multilevel PAD. The number of terminal branches arising from peroneal artery, the crural runoff, and the pedal arch quality did not affect the long-term results. Larger studies with standardized wound care protocols are required to obtain more robust data on possible perfusion-related factors that predict the outcomes of endovascular treatment in CLTI with occlusive lesions involving several arterial segments.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
