Abstract
Background
Critical limb ischemia (CLI), the most advanced form of peripheral artery disease (PAD), is associated with a high risk of adverse cardiovascular and limb events, including mortality and major amputation.1,2 Treatment goals for CLI are focused on cardiovascular risk management, quality of life improvement, and limb preservation, with revascularization being an important strategy for improving blood flow and potentially preserving limb viability. 3 Procedural strategies to improve blood flow include endovascular peripheral vascular intervention (PVI) and lower-extremity bypass grafting. While the comparative effectiveness of PVI and bypass grafting is still being evaluated, the endovascular approach offers the potential benefit of fewer upfront procedural risks and has become the most frequently used treatment.4 –7 Rates of major amputation in the context of CLI-related admissions overall have decreased and the risk of undergoing an amputation 30 days after endovascular PVI is variable, with a single-center study documenting a rate of 6%, a regional study documenting a 2% rate, and a review of trials for endovascular procedures in CLI documenting a rate of 1.9%.8 –10
Key performance benchmarks for the quality of CLI treatment include avoiding major adverse cardiac events and major adverse limb events, including major amputations within 30 days of treatment.10,11 Although no formal agreed-upon quality benchmarks exist for 30-day amputation complication risks following PVI, target goals for testing new therapeutics in CLI clinical trials have suggested a goal of amputation rates below 3% within 30 days of catheter-based interventions. 10 Extending CLI treatment quality goals for avoiding amputation requires deeper insights into current performance, both in frequency and variability across sites, in real-world clinical practice.
To address this need as a foundation for developing quality measures for CLI care and supporting the use of electronic health record (EHR) data to more efficiently capture quality metrics, we sought to describe 30-day major amputation rates, and their variability, following PVI across care centers in the United States. To do this, we used the Cerner Health Facts EHR data from 2010 to 2018 from over 700 participating sites across the United States to (1) document 30-day amputation rates for CLI following PVI discharge, (2) study variability of the rates across centers, and (3) explore the degree to which patient-level and site-level factors were explaining this variability.
Methods
Study design and patient selection
The de-identified Health Facts (Cerner Corporation, Kansas City, MO, USA) database is a research data warehouse created from EHR data. Health Facts data represent over 69 million unique patients from inpatient, outpatient, and emergent visits, and provide procedure-level data that include medical history, medications, and procedure history.
For this study, we applied the following inclusion criteria: (1) CLI documented during the index procedure using administrative codes from the International Classification of Diseases, 9th and 10th revisions (ICD-9/10) (online Supplementary Table 1); (2) an inpatient or outpatient PVI procedure documented through Current Procedural Terminology, version 4 (CPT-4) codes (online Supplementary Table 1); and (3) procedures that occurred between 2010 and 2018.7,12 Procedures were excluded if (1) the patient was less than 18 years old; (2) they had documented acute limb ischemia; and (3) the procedure was performed at a site with fewer than 50 PVIs during the study period.
The University of Missouri-Kansas City and Yale University institutional review boards determined research performed with the de-identified Health Facts database qualifies as not human subjects research (Protocols 14-567 and 2000031627, respectively).
Primary outcome
Thirty-day major amputations were defined as lower-extremity amputations at the level of the ankle or above that occurred within 30 days following a PVI discharge. Nontraumatic, lower-limb major amputations were identified using ICD-9/10 codes and CPT-4 codes (online Supplementary Table 1). The nontraumatic amputation outcome specifically did not include ICD-9/10 codes for traumatic amputation (online Supplementary Table 1). Conversions between ICD-9 and ICD-10 codes were based on previously published methods. 13 Patients with amputations that occurred during the index admission associated with the PVI procedure were not included as it was not possible to discern whether these amputations were periprocedural complications or planned amputations.
Patient characteristics
Demographic and socioeconomic variables included age, sex, race, and marital status. Race is documented in Health Facts as White, Black, Asian, Hispanic, Biracial, and Unknown. Marital status was classified as married versus not married.
Encounter visit type was classified as inpatient, outpatient, emergent, and other/unknown encounter type.
Medical history was derived from patient encounters using ICD-9, ICD-10, and CPT-4 codes. History variables include previous limb amputation, transient ischemic attack, sepsis, renal disease, osteomyelitis, malnutrition, malignant cancer, hypertension, heart failure, diabetes mellitus, depression, chronic kidney disease, and coronary artery disease. Clinical variables were defined using Elixhauser comorbidity definitions and review of previously published administrative code definitions.12,14,15
Minor amputation, surgical revascularization, and repeat PVI outcomes within 30 days following the PVI procedure discharge were identified using ICD-9 and ICD-10 codes (online Supplementary Table 1).
Inpatient medications reported included antiplatelets, antithrombotic therapies, antihypertensives, and lipid-lowering documented at the time of encounter and were identified using National Drug Codes (NDC) as specified in online Supplementary Table 1. Medications are reported only for inpatient procedures.
Site characteristics
Site characteristics included teaching status (teaching vs nonteaching), which is a site-reported status dependent on presence of an accredited medical residency program, categorized bed size (1–5, 6–99, 100–199, 200–299, 300–499, and 500+ beds), United States Census regions (Midwest, Northeast, South, and West), and rurality (urban vs rural).
Statistical analysis
The main analysis was performed with the procedure as the unit of observation. For descriptive purposes, the characteristics of patients undergoing PVIs were summarized as means and SDs for continuous variables and proportions for categorical variables for the overall study population.
Median risk ratios (MRR) were calculated to describe variation in the 30-day amputation outcome across clinical sites, with patient-level factors, site-level factors, and Census region entered the model in subsequent steps. The Poisson MRR measure quantifies outcome variability using a Poisson prediction method, rather than a typical logistic regression method, to identify variability in binary outcome among sites when the outcome of interest is relatively rare.16,17 A MRR describes the average likelihood a patient would experience a 30-day major amputation event at one random site versus any other site, after adjusting for patient- and site-level covariates. A MRR of one indicates no variation in outcomes by site, and a risk ratio greater than one indicates variation at the site level. For example, a MRR of 1.10 indicates that two statistically similar patients would have 10% difference in the odds of undergoing amputation within 30 days of procedure if treated at one random site versus another. The MRR model accommodates for variability in sample size and down-weights sites with smaller sample sizes that may otherwise be outliers in raw 30-day major amputation rates.
Patient-level covariates for our 30-day amputation predictive models were selected from the pool of available variables if they were associated with 30-day amputation outcomes at the 0.10 level of significance, through univariate testing using chi-squared, Mann–Whitney-U, and Student’s t-tests, as appropriate. 9 Further adjustments were sequentially made, adding site urban versus rural status, teaching status, bed size classification, and, in the last step, an additional adjustment for United States Census region was made. Risk ratios for the correlates of amputation were derived from the modified, binary outcome Poisson regression, with a random site effect and including robust standard errors to account for variance misspecification.
As a sensitivity analyses, the analyses were repeated using the patient (rather than the procedure) as the unit of observation, using only the index procedure, to test whether repeat procedures were driving outcomes. All statistical tests were two-tailed and considered significant at p < 0.05. All analyses were performed using the RStudio integrated development environment (version 1.4.1717; PBC, Boston, MA, USA) using R (version 4.1.0; R Foundation for Statistical Computing, Vienna, Austria). 18
Results
A flowchart describing the cohort selection process is shown in Figure 1. A total of 20,204 unique procedures from 179 sites in 16,173 patients undergoing PVI for CLI were identified. The mean age of patients undergoing these procedures was 69.0 ± 12.6 years, 58.0% were male, 64.4% were White, 25.3% were Black, 0.9% were Asian, 0.7% were Hispanic, 0.0% were Biracial, and 8.5% had Unknown race (Table 1). Most patients had controlled diabetes mellitus (61.3%), controlled hypertension (76.6%), and a third presented with chronic kidney disease (34.8%). Procedures had the most representation of data from sites in the South United States Census region (48.4%), at teaching facilities (73.4%), and at urban sites (80.4%).
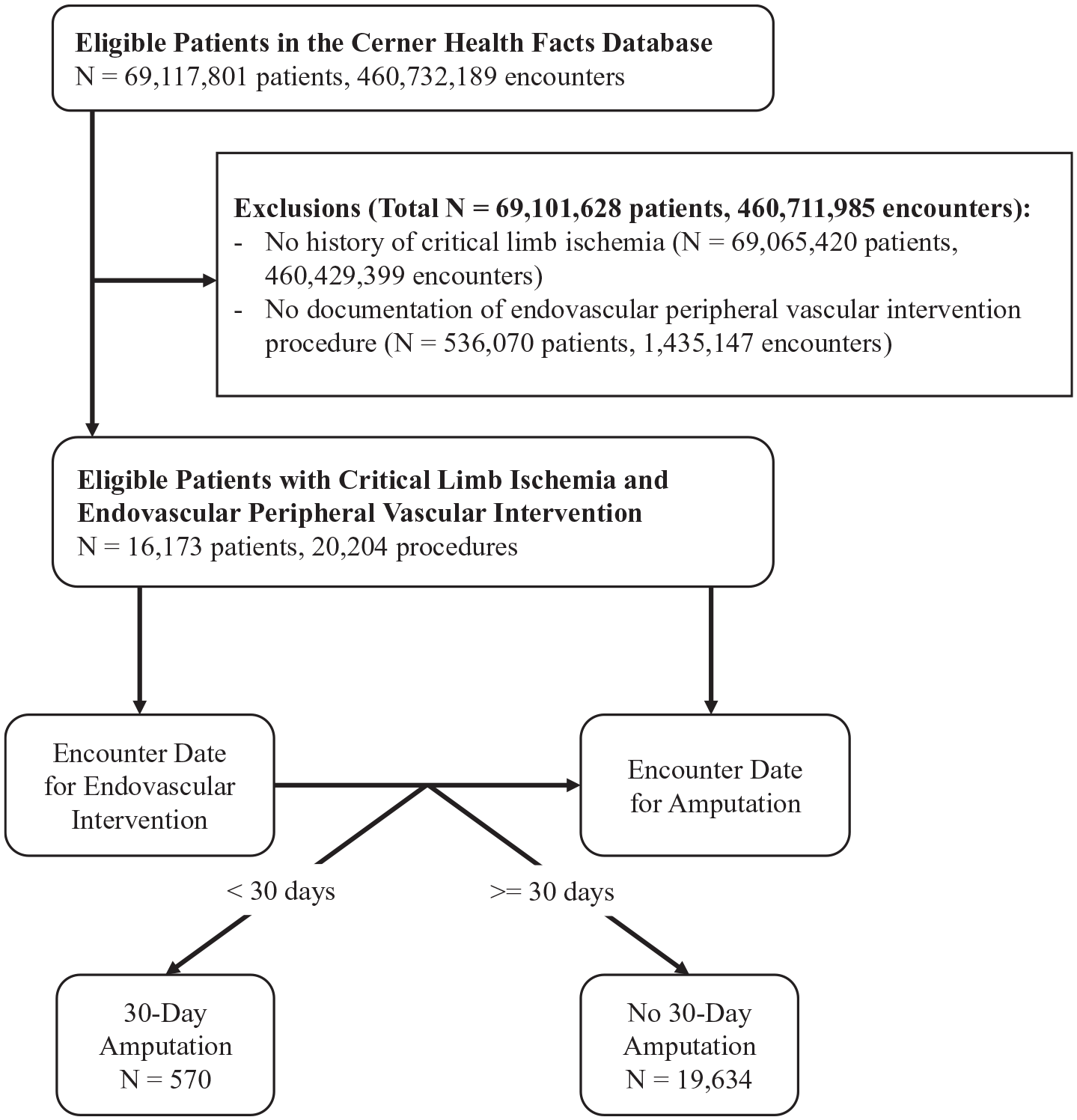
Cohort selection process.
Patient characteristics for peripheral vascular endovascular interventions in critical limb ischemia (at procedural level).

Values are presented as n (%) unless otherwise noted.
Only for patient encounters documented as ‘inpatient’ (denominator n = 6,130).
Across all 179 sites included in the analysis, the mean rate of nontraumatic major amputation within 30 days following PVI discharge was 2.8% (Table 2). Within 30-days of procedure discharge, minor amputation occurred after 10.5% of procedures and surgical revascularization after 5.2%. For the 16,173 patients undergoing PVI, 8.8% underwent repeat PVI procedures within 30 days of the initial procedure discharge. The median amputation rate was 2.5% (IQR 1.3–3.6%) across all sites, ranging from 0.0% to 10.0% (Figure 2). The MRR for site variability in the unadjusted model was 1.40 (95% CI 1.35–1.46; Table 3). Patient characteristics that were retained for model adjustment were age greater than or less than 65 years, sex, race, marital status, coronary artery disease, depressive disorder, diabetes, heart failure, previous lower-limb amputation, malignant cancer, hypertension, chronic kidney disease, malnutrition, osteomyelitis, sepsis, and previous transient ischemic attack. Race was categorized into White, Black, and Other (Asian, Hispanic, Biracial, and Unknown) categories for the modeling only due to the smaller proportions of the Asian, Hispanic, Biracial, and Unknown groups.
Crude rates for 30-day outcomes following peripheral vascular endovascular intervention for critical limb ischemia.
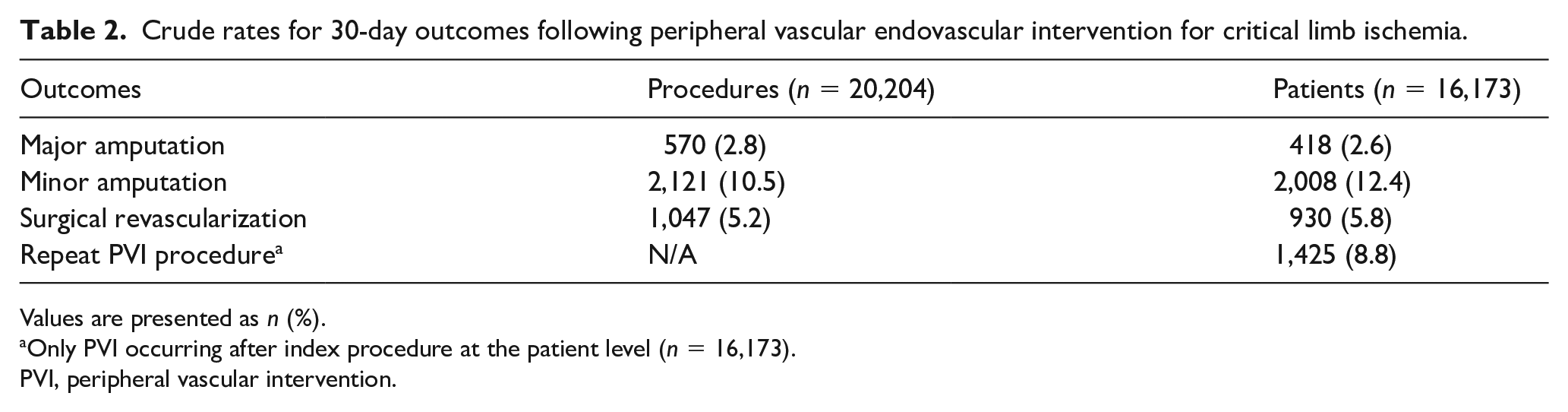
Values are presented as n (%).
Only PVI occurring after index procedure at the patient level (n = 16,173).
PVI, peripheral vascular intervention.
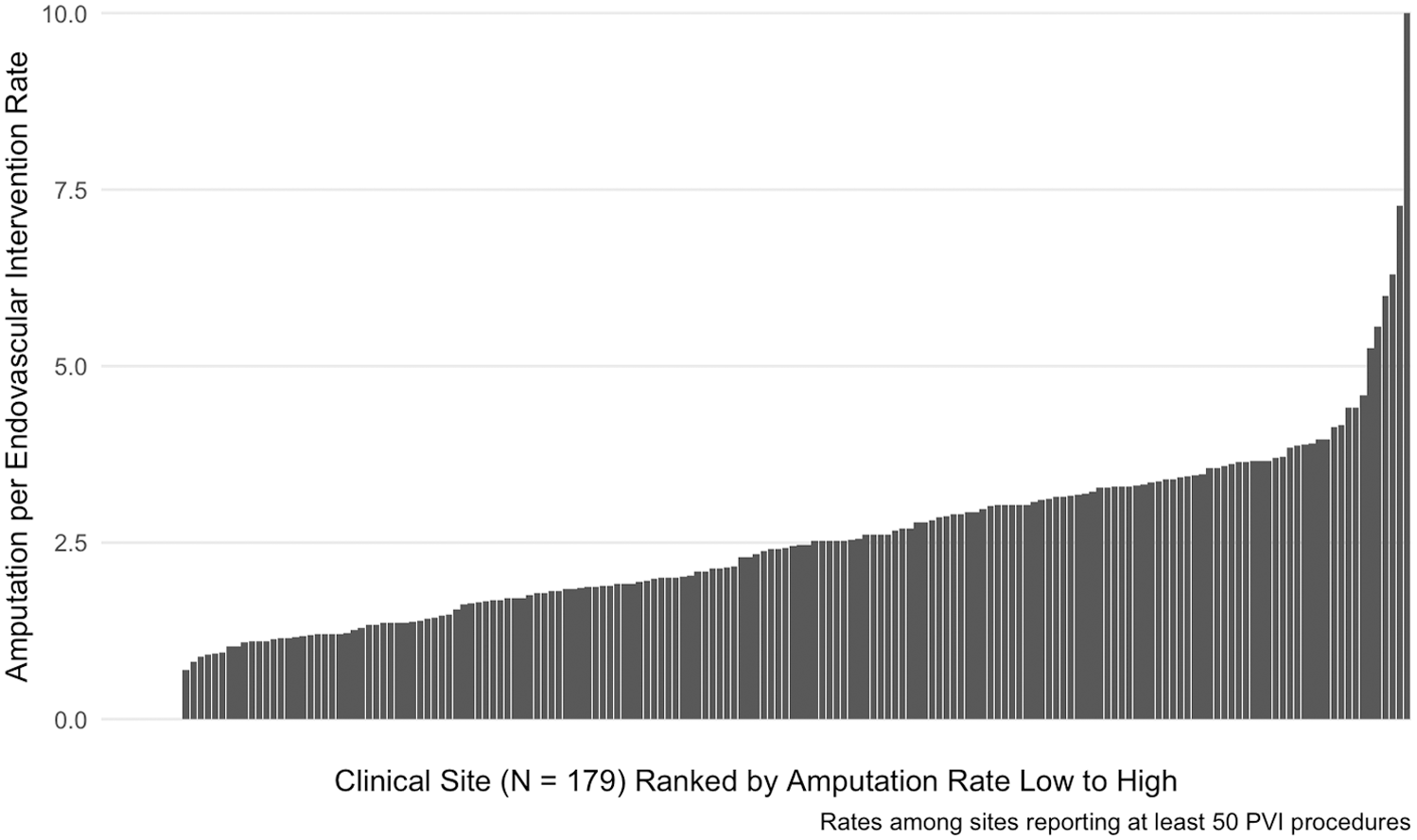
30-day major amputation rate (%) relative to the number of PVI procedures by site in patients undergoing endovascular peripheral vascular intervention for critical limb ischemia.
Risk of experiencing 30-day major amputation following peripheral vascular intervention for critical limb ischemia by site.

Number of sites = 179.
Patient characteristics include dichotomous age greater than or less than 65 years, biological sex, race, marital status, documented coronary artery disease, depressive disorder, diabetes, heart failure, previous lower-limb amputation, malignant cancer, hypertension, kidney disease, malnutrition, osteomyelitis, sepsis, and previous transient ischemic attack.
Site characteristics include teaching vs nonteaching site, urban vs rural status, and bed size classification.
After adjusting the model for patient-level factors at the time of procedure, the median relative difference in the odds of major amputation at 30 days between sites was 30% (MRR 1.30, 95% CI 1.26–1.35). Further adjustment for site-level characteristics (teaching vs nonteaching site, urban vs rural status, and bed size classification) explained much of the variability (MRR 1.14, 95% CI 1.12–1.16), whereas adjusting for Census region resulted in a small additional attenuation (MRR 1.11, 95% CI 1.09–1.13). Malnutrition (risk ratio (RR) 1.81, 95% CI 1.38–2.38), previous lower-limb amputation (RR 1.97, 95% CI 1.60–2.42), diabetes (RR 1.42, 95% CI 1.13–1.78), and Black race (RR 1.42, 95% CI 1.15–1.74) were all independently associated with an increased risk of 30-day major amputation in the final adjusted model (Figure 3). Although much of the MRR was explained by the sequential adjustments of the patient-level and site-level variables, none of the site-level variables emerged as independent predictors of 30-day amputation in a model fully adjusted for patient-level factors (Figure 4).
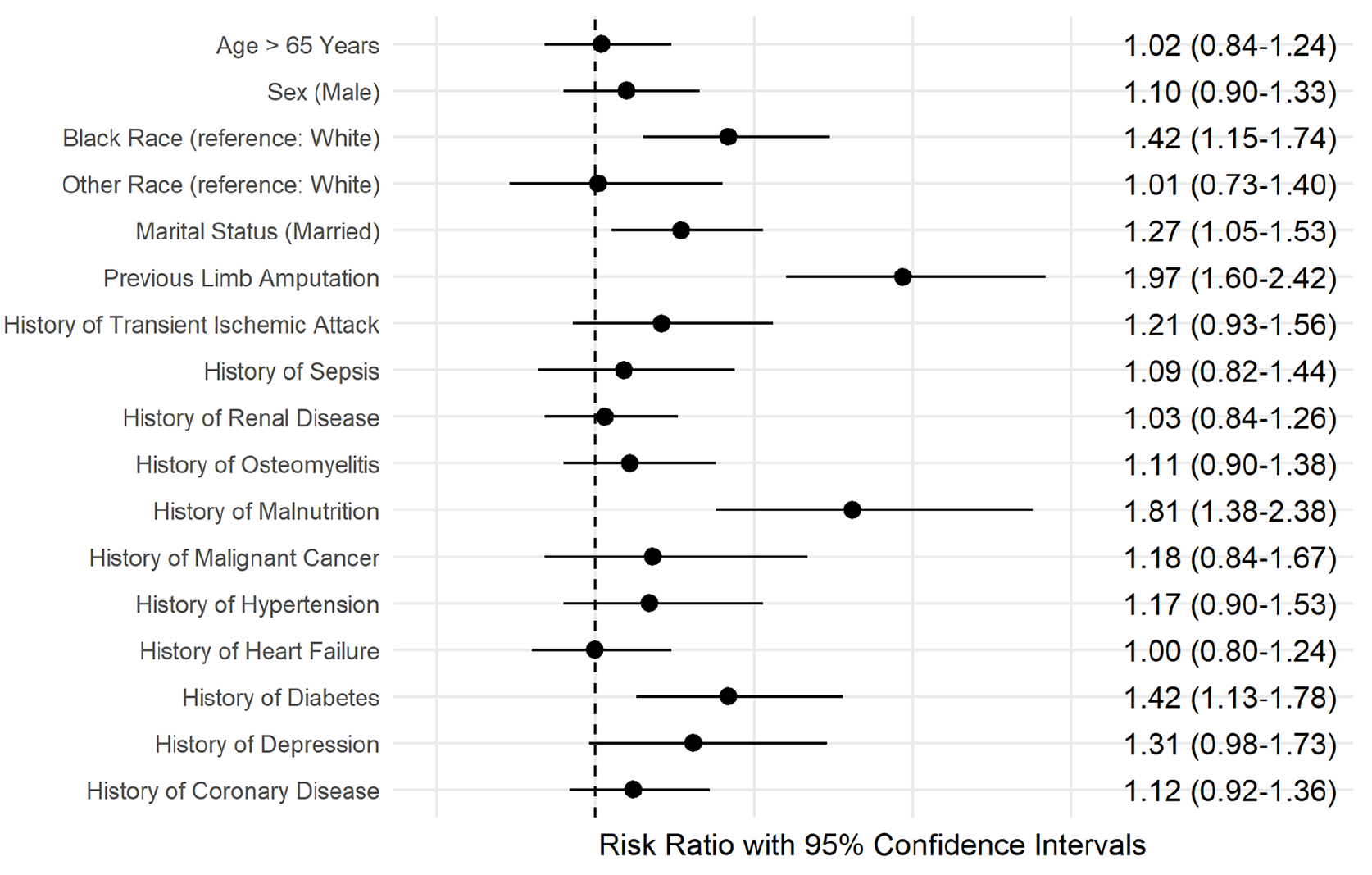
Patient-level factors associated with 30-day major amputation in patients undergoing endovascular peripheral vascular intervention for critical limb ischemia.
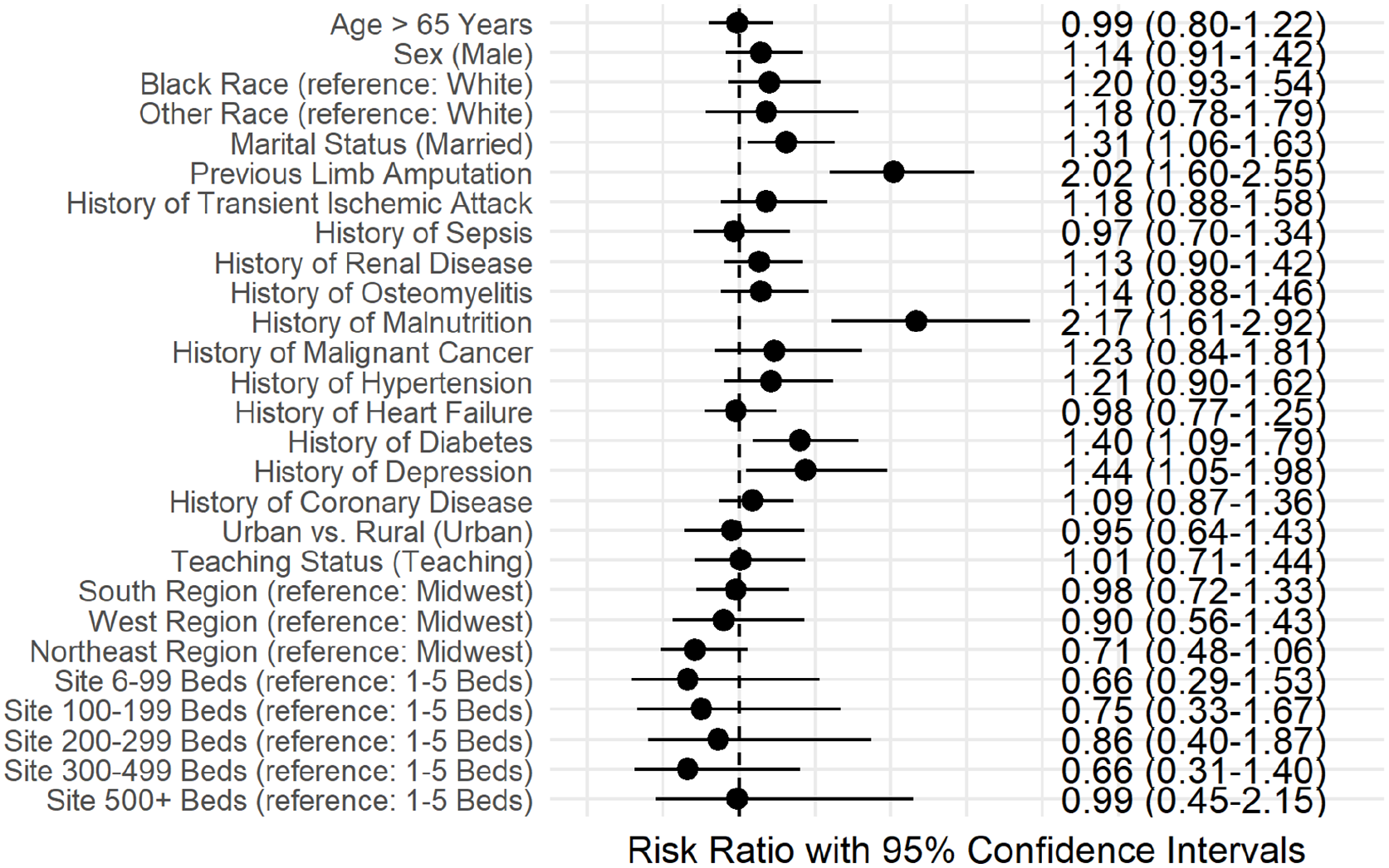
Patient- and site-level factors associated with 30-day major amputation in patients undergoing endovascular peripheral vascular intervention for critical limb ischemia.
Restricting the cohort to only index PVI procedures resulted in 16,173 index PVI procedures performed at 178 sites. The 30-day major amputation rate was similar, with 2.6% of PVI procedures having subsequent 30-day amputations (Table 2). The median amputation rate was 2.2% (IQR 1.0–3.4%) and the amputation rate ranged from 0.0% to 8.2%. Predictors of 30-day amputation aligned with the main analysis and included malnutrition (RR 1.81, 95% CI 1.38–2.38), previous lower-limb amputation (RR 1.97, 95% CI 1.60–2.42), diabetes (RR 1.42, 95% CI 1.13–1.78), and Black race (RR 1.42, 95% CI 1.15–1.74) (online Supplementary Figure 1). The unadjusted MRR was 1.38 (95% CI 1.33–1.43). After patient-level factor adjustment it was 1.29 (95% CI 1.25–1.33), it was 1.12 (95% CI 1.10–1.14) after site-level factor adjustment, and it was 1.11 (95% CI 1.09–1.13) after adjusting for Census region (online Supplementary Table 2).
Discussion
Using the national Health Facts dataset, we documented current rates and variability of 30-day amputations, which is vitally important to evaluate CLI quality of care. We found that 30-day amputations occurred in 2.8% of included procedures, with rates ranging from 0.0% to 10.0% across sites. The rate of 30-day amputation was variable across sites, which was explained by both patient- and site-level factors. Patient-level factors including being a person of color, nutritional status, and a history of amputation and diabetes were associated with an increased odds of 30-day amputation.
Our work provides national, real-world estimates for rates of 30-day major amputations, which to date have only been reported in regional registry data such as the Vascular Study Group of New England or in the context of clinical trials.9,10 The Society for Vascular Surgery Vascular Quality Initiative Peripheral Vascular Intervention Registry has quickly become one of the nation’s primary resources for the evaluation of endovascular care evaluation. 19 However, insights from the Health Facts database provide expanded evidence for the quality of care patients with CLI experience at sites beyond those participating in registries using EHR data, and demonstrates feasibility to apply this across other EHR data warehouses.
The variability of 30-day amputation rates across sites ranged from 0% to 10% in the poorest performing site. Our results indicate that site-level variables, including teaching status, rurality, and bed size classification, explained a substantial proportion of the 30-day outcome variability between sites; however, in models fully adjusting for patient-level factors, site-level variables and United States Census region did not emerge as independent predictors of individual 30-day amputation risk. Patient-level factors that remained predictive after site-level factor adjustment may indicate patients with differing risk profiles between the sites included in our study. Prior work has established that rates of major amputation in Medicare patients undergoing peripheral vascular procedures varies widely between sites and geographies and increased intensity of vascular care is associated with fewer lower-limb amputation rates. 20 The rate we report aligns with previously documented rates between 2% and 6% and falls below the suggested 3% objective performance metric for the evaluation of peripheral procedures, but does identify outliers as 41% of the sites had rates above this threshold.8 –10 Our collected site variables did not explain all of the variability, thus future work will need to further explore practices that are associated with lower amputation rates, which can inform quality efforts to lowering them at a national level.
Other than practices performed at the site level, potentially modifiable targets for lowering 30-day amputation rates were also identified at the patient level, including nutritional status and diabetes. For patients with PAD, nutritional goals have not been referenced as a key treatment guideline; the importance of nutrition in CLI care is increasingly being recognized.1,3 Being a person of color was associated with increased risk of 30-day amputation. Racial inequalities in vascular care have been well-documented in diagnosis, the quality of care received, and in outcomes for persons of Black race.3,21 –24 Our results suggest a higher risk for 30-day amputation in minorities. Beyond defining quality metrics of high-quality CLI care, further priority should be given to programs targeting earlier detection of PAD and CLI in health systems and communities to further prevent amputations. 25 If these outcomes can be predicted, further investigation into how these correlates might be modified is warranted. Data-driven approaches, such as the one we present here, will be necessary to move to a population health-based model of CLI prevention and care.
Study limitations
Our study is limited due to the observational nature of the data, which leaves potential for residual confounding. For example, in our data we were unable to verify whether 30-day amputation outcomes were unplanned outcomes versus staged interventions. Next, Health Facts only contains medication information for inpatient encounters, and could therefore only be documented for PVI procedures that took place in the inpatient setting. Complete information on the medical management of patients with CLI could have further enhanced our ability to risk stratify patients. No detailed information about anatomical lesions intervened upon was available, which could have further explained which types of lesions are most at risk to follow with an amputation. Furthermore, the use of EHR data sources includes risk of misclassification and underreporting of risk factors (e.g., malnutrition). Importantly, no specialty type information was accessible, which could have further contextualized the extent of multidisciplinary CLI care that patients received. Finally, our findings may only be generalizable to centers subscribing to the Cerner EHR system.
Conclusions
Our study of a national cohort of patients with CLI used real-world EHR data to identify variability in 30-day major amputation outcomes in patients undergoing endovascular PVI. Although criteria of care excellence exist for treatments such as percutaneous coronary intervention, they have yet to be developed for the treatment of CLI. 26 The 30-day amputation risk following PVI may be one metric that can be collected from EHR data sources. Outliers were identified, and substantial variability across sites suggest that there is an opportunity to better understand practices associated with better versus poorer performing sites and sites that see a higher at-risk population for amputations that would benefit from targeted CLI quality improvement programs.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X221098097 – Supplemental material for Variability in 30-day major amputation rates following endovascular peripheral vascular intervention for critical limb ischemia
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X221098097 for Variability in 30-day major amputation rates following endovascular peripheral vascular intervention for critical limb ischemia by Jeremy B Provance, John A Spertus, Philip G Jones, Mark A Hoffman, Matthew C Bunte, Todd R Vogel, Carlos Mena-Hurtado and Kim G Smolderen in Vascular Medicine
Footnotes
Correction (December 2022):
Article updated to replace Figure 2. The original figure was a sensitivity analysis on a subset (N = 80) of the 179 sites. The correct figure includes data for all 179 sites
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. John A Spertus reports a consulting relationship with Merck, Janssen, Novartis, MyoKardia, Bayer, and United Healthcare; grant funding from Abbott Vascular; ownership of the copyright to the Seattle Angina Questionnaire (SAQ), Kansas City Cardiomyopathy Questionnaire (KCCQ), and Peripheral Artery Questionnaire (PAQ); and service on the board of directors of Blue Cross Blue Shield of Kansas City. Carlos Mena-Hurtado reports consulting for Abbott, Cardinal Health, Medtronic, Optum Labs, and Cook. Kim G Smolderen receives research grants from J&J, Cardiva, and Abbott; and is a consultant for Optum Labs, Cook, and Abbott. The remaining authors have no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
