Abstract
Functional popliteal artery entrapment syndrome (FPAES) presents a challenge for clinicians in the field today. The relative rarity of the disease coupled with its affliction primarily in young, athletic individuals makes it a difficult condition to diagnose. Treatment modalities have been limited to surgical intervention, with more novel methods being developed over the last several years. We present a case report and review the diagnostic and management approaches for FPAES, shedding light on proposed future interventions.
Keywords
Case presentation
A 21-year-old female with a history of competitive dancing presented to the vascular surgery clinic with a 5-year history of increasing left leg claudication, paresthesias, and weakness exacerbated by dancing and pirouettes on her left leg. On presentation, the patient had palpable lower extremity pulses in the neutral position. However, upon active plantarflexion, the dorsalis pedis and posterior tibialis pulses and signals disappeared on the left side. Monophasic Doppler waveforms were found in the left tibioperoneal trunk and left popliteal artery during plantarflexion and dorsiflexion maneuvers (Figure 1). Given these results, a diagnostic angiogram was performed, which showed near occlusion of her popliteal artery with delayed runoff during active plantarflexion, confirming our diagnosis of popliteal artery entrapment syndrome (PAES) (Figure 2). Magnetic resonance imaging (MRI) analysis of the knee was inconclusive and no muscular abnormality could be identified. The patient elected to continue with conservative management and follow-up if symptoms progressed.
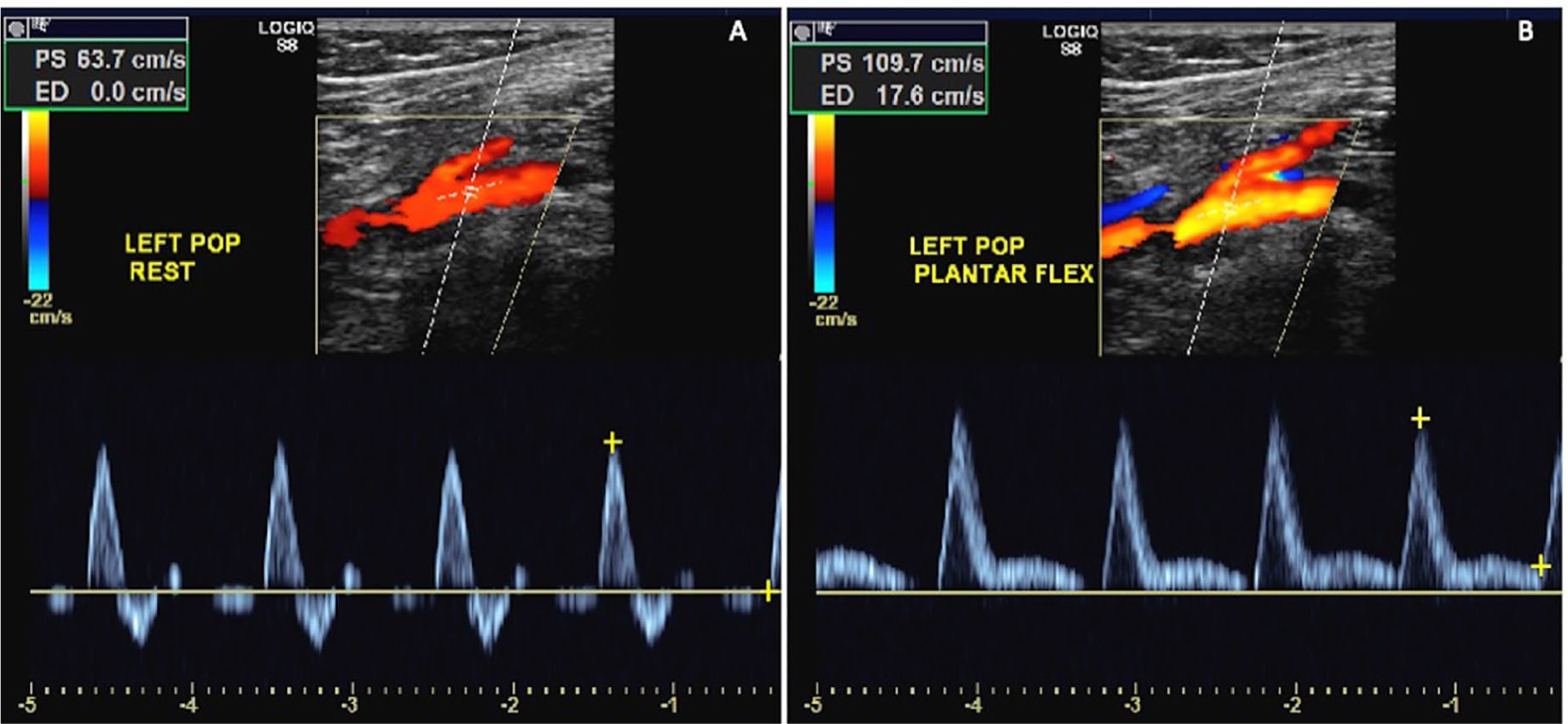
(A) Normal, triphasic waveform seen on color duplex images with Doppler velocity waveforms of the left popliteal artery in neutral positioning. (B) Abnormal, monophasic waveform seen on color duplex images with Doppler velocity waveforms of the left popliteal artery during plantarflexion.
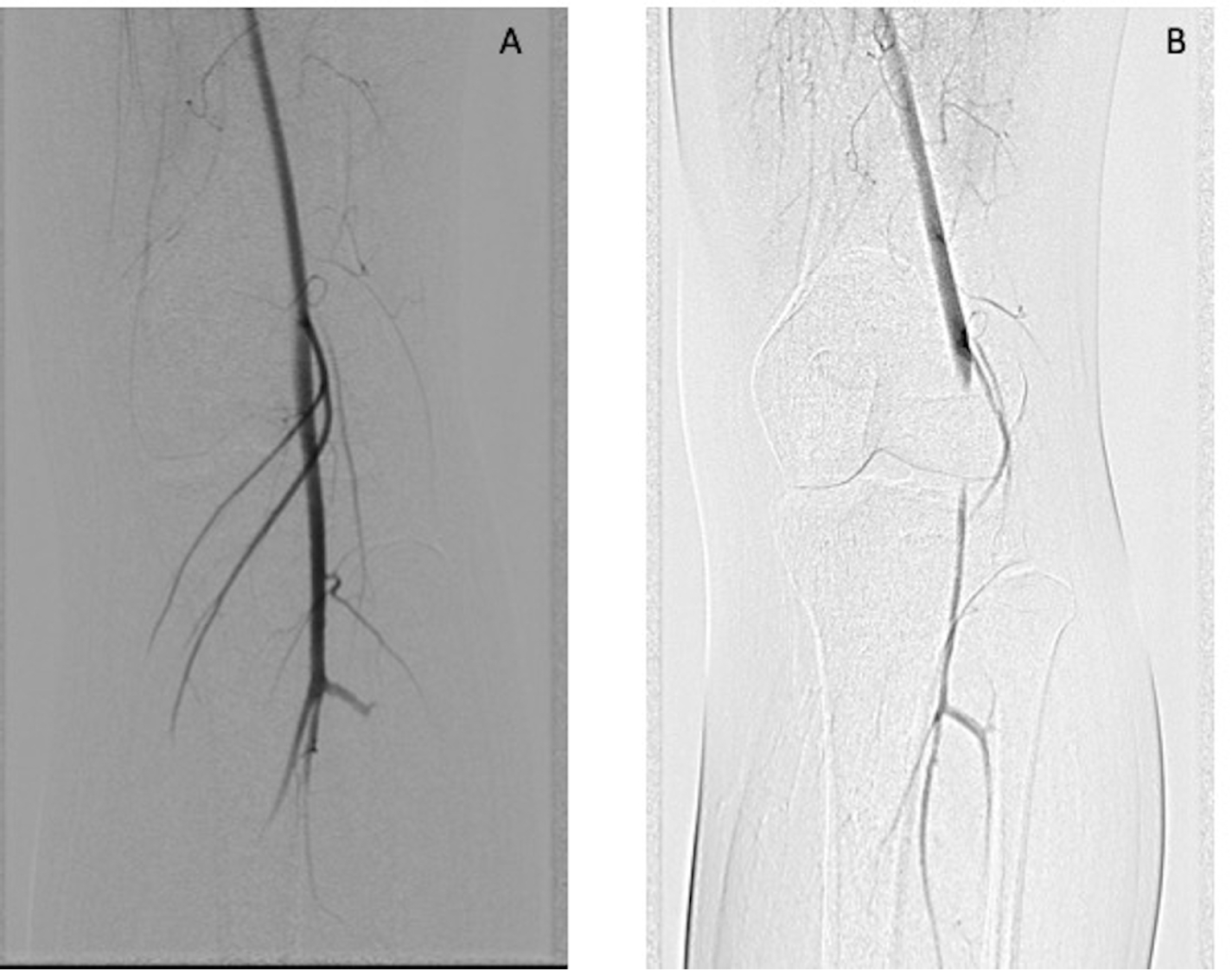
(A) Angiogram showing normal flow within the left popliteal artery in neutral positioning. (B) Near occlusion of the left popliteal artery with plantarflexion.
Introduction
PAES has been a known condition in medicine since 1879 when it was first described by Stuart as a variation in the course of the popliteal artery. 1 There has been growing literature on the topic ever since, most prominently in the past two to three decades.2–6 The bulk of the literature has highlighted the anatomic variant. Functional PAES (FPAES) was described initially by Rignault et al. in 1985. 7 While an anomalous association between the popliteal artery and the myofascial structures in the popliteal fossa characterizes anatomic PAES, FPAES usually presents with symptoms of lower extremity claudication and paresthesias without any identifiable anatomic variations in the popliteal fossa. Affecting primarily young, athletic individuals, the condition is often confused with other diseases that originate in the lower extremity, making diagnosis challenging. Given the dynamic nature of the disease, it is necessary to modify imaging protocols to prevent misdiagnosis. Surgical management has remained the primary treatment so far, with some patients requiring more extensive vascular reconstructions given the extent of disease progression secondary to chronic compression.
Diagnostic modalities
Most commonly identified clinically by neuromuscular symptoms in the posterior calf, FPAES can often be confused with other conditions that manifest in the vicinity, including chronic recurrent exertional compression syndrome (CRECS), medial tibial stress syndrome, tibial stress fracture, and soleal sling syndrome, among others. 8 Overall, it is a condition that occurs commonly in younger patients, resulting in claudicating symptoms that resolve with cessation of exercise. No anatomic abnormalities or aberrant associations can be found between the posterior compartment muscles and the vascular structures. The distinguished findings in patients with FPAES include hypertrophy of the gastrocnemius, plantaris, or soleus muscles, resulting in impingement on the vascular structures secondary to exercise. Turnipseed et al. looked at functional PAES and CRECS, both of which present with a similar set of symptoms. 9 As per the study, which looked at 854 patients over the course of 20 years, only 20 out of 614 patients were treated for popliteal entrapment, with 13 attributed to FPAES, attesting to the relative rarity of the condition. Hislop et al. shed more light on this, describing FPAES as a condition prone to occur bilaterally and distinct from CRECS given a much quicker resolution of post-exertional pain. 10 Several different vascular imaging modalities have been utilized to aid in the diagnosis of FPAES, including first-line tools such as ankle–brachial indices (ABIs) and duplex ultrasonography (US), to more intricate imaging methods such as magnetic resonance imaging (MRI/MRA) (Figure 3) and conventional angiography.
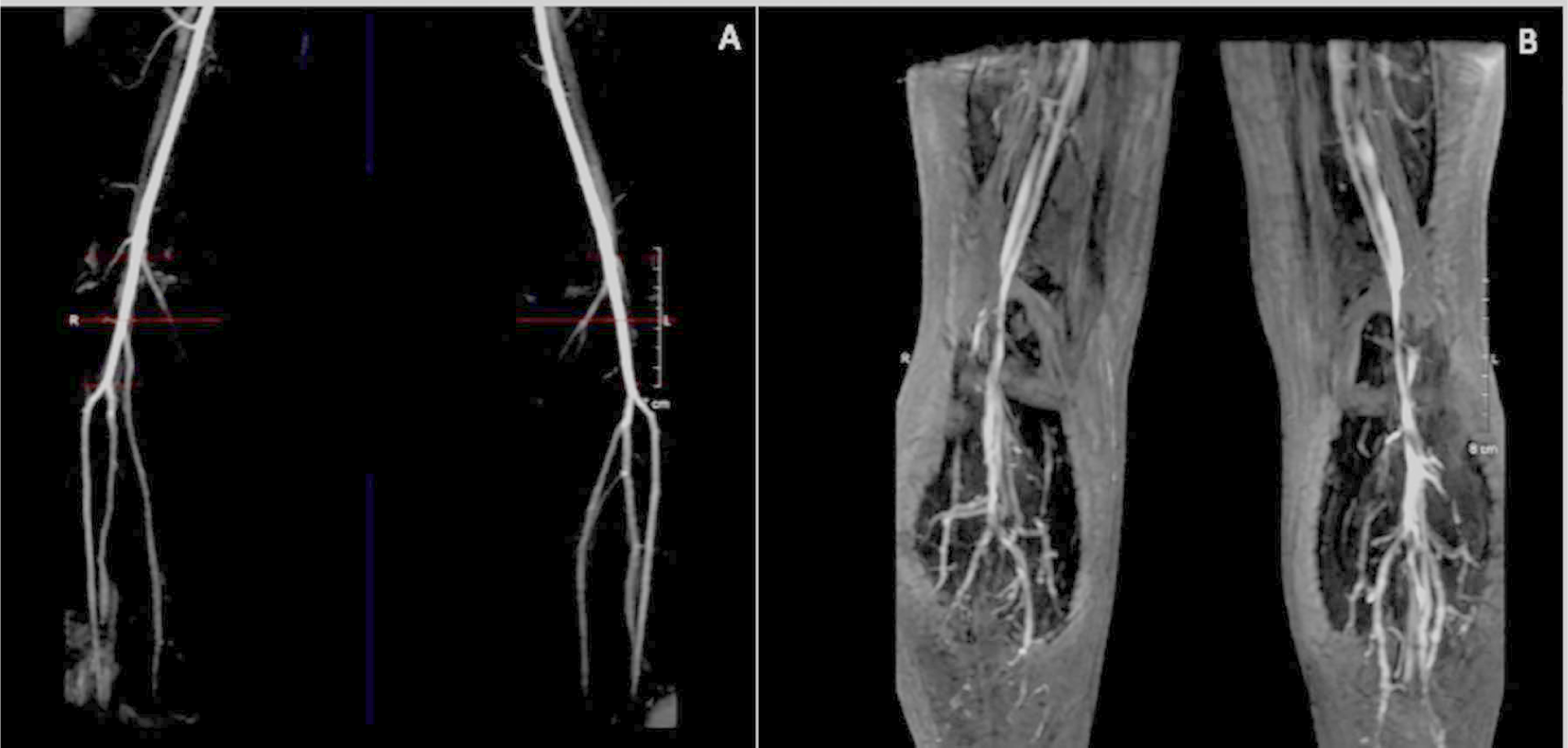
(A) MRA demonstrating normal flow within the popliteal artery. (B) Narrowing of the mid popliteal artery due to compression by the medial head of the gastrocnemius muscle due to hypertrophy of the bilateral medial heads of the gastrocnemius muscles in the setting of FPAES.
Brown et al. demonstrated the use of provocative ABIs and popliteal artery diameter changes on duplex US in a study consisting of 90 limbs (29 symptomatic and 61 asymptomatic), finding a greater reduction in ABIs and popliteal artery diameter on duplex US in the symptomatic group on 1 minute of running compared to the asymptomatic group. 11 Lane et al. performed provocative ABIs (6 km/h treadmill running to the point of symptom replication), provocative duplex US (rapid tiptoe standing and release at least 20 times, or less if symptomatic sooner), nonprovocative MRI and compartmental pressures (preoperatively), and provocative catheter angiography (plantarflexion and dorsiflexion), with 46 out of 52 limbs showing normalization of provocative ABIs postoperatively after myotomy. 12 The authors in this study recommended hopping at the same rate as the heart rate as a dependable maneuver for obtaining provocative testing. Duplex US was touted as the most helpful examination, but little explanation was provided as to why. On the other hand, Hislop et al. supported a lack of utility of ABI measurements given the inability to interpret findings in the setting of graded compression on plantarflexion. 10 Instead, they presented their own protocol for provocative duplex US using graded compression of at rest, 25% of full plantarflexion, and 50–100% of full plantarflexion. Arterial waveforms, velocities of peripheral arteries, and areas of intimal thickening or fixed arterial disease were assessed. Those subjects who occluded either during rest or 25% of full plantarflexion were considered severe cases of FPAES. Nevertheless, the authors attest that duplex US is not very reliable at effectively eliminating anatomic PAES since occlusion could point to either the functional or anatomic variant. The group also performed MRI/MRA on their patients with the first step consisting of a T1-weighted static image capture to evaluate the musculature and vascular structures in the popliteal fossa followed by T2-weighted image capture on plantarflexion and dorsiflexion, finally concluding with sustained plantarflexion for contrast injection during MRA. One of the primary limitations of MRI/MRA was the inability of the subjects to maintain provocative maneuvers, resulting in associated motion artifact. Chernoff et al. studied the utility of provocative duplex US and MRI in a small cohort of 13 subjects and found concordance in the results between the two imaging modalities supporting their utility for the detection of FPAES. 13 Looking at larger reviews, both Sinha et al. and Lohrer et al. endorse the utility of provocative ABIs, duplex US, and MRI/MRA offering the best current approach to diagnosing PAES, with FPAES studied as a component of it but endorsing the need for specific data collection to develop better guidelines.14,15 Sinha et al. states a sensitivity of 94% with provocative MRI/MRA and a sensitivity of 90% with provocative duplex US coupled with ABIs. 14
Boniakowski et al. described a case report with a relatively new approach in FPAES. The group used intravascular ultrasound in a case of nondiagnostic provocative MRI and digital subtraction angiography (DSA) that showed long segment stenosis without pointing to the exact site of compression. 16 No other studies have reported the utility of this method for FPAES.
Reviewing the literature presented so far on the diagnosis of FPAES (summarized in Table 1), provocative testing is essential in order to detect this condition in patients. ABIs and duplex US are the first step in the workup, presenting as reliable, noninvasive testing modalities for the detection of baseline vascular disease and a primary evaluation of the contents of the popliteal fossa as well as any intimal vascular disease in the lower limb arterial branches. MRI/MRA presents as a judicious next step in order to more concretely evaluate the anatomy of the popliteal fossa, determine the extent of vascular pathology, and inform operative planning. The primary drawback of all these testing methods is the lack of standardization of provocative maneuvers between the studies conducted so far, which clouds result interpretation and consistent utility of these methods. Furthermore, the ability of patients to maintain certain maneuvers during sensitive tests such as MRI/MRA can be subject to motion artifact. Conventional angiography is often performed as a confirmatory test prior to consideration of surgical correction or if duplex and/or MRI/MRA is equivocal.
Diagnostic modalities.
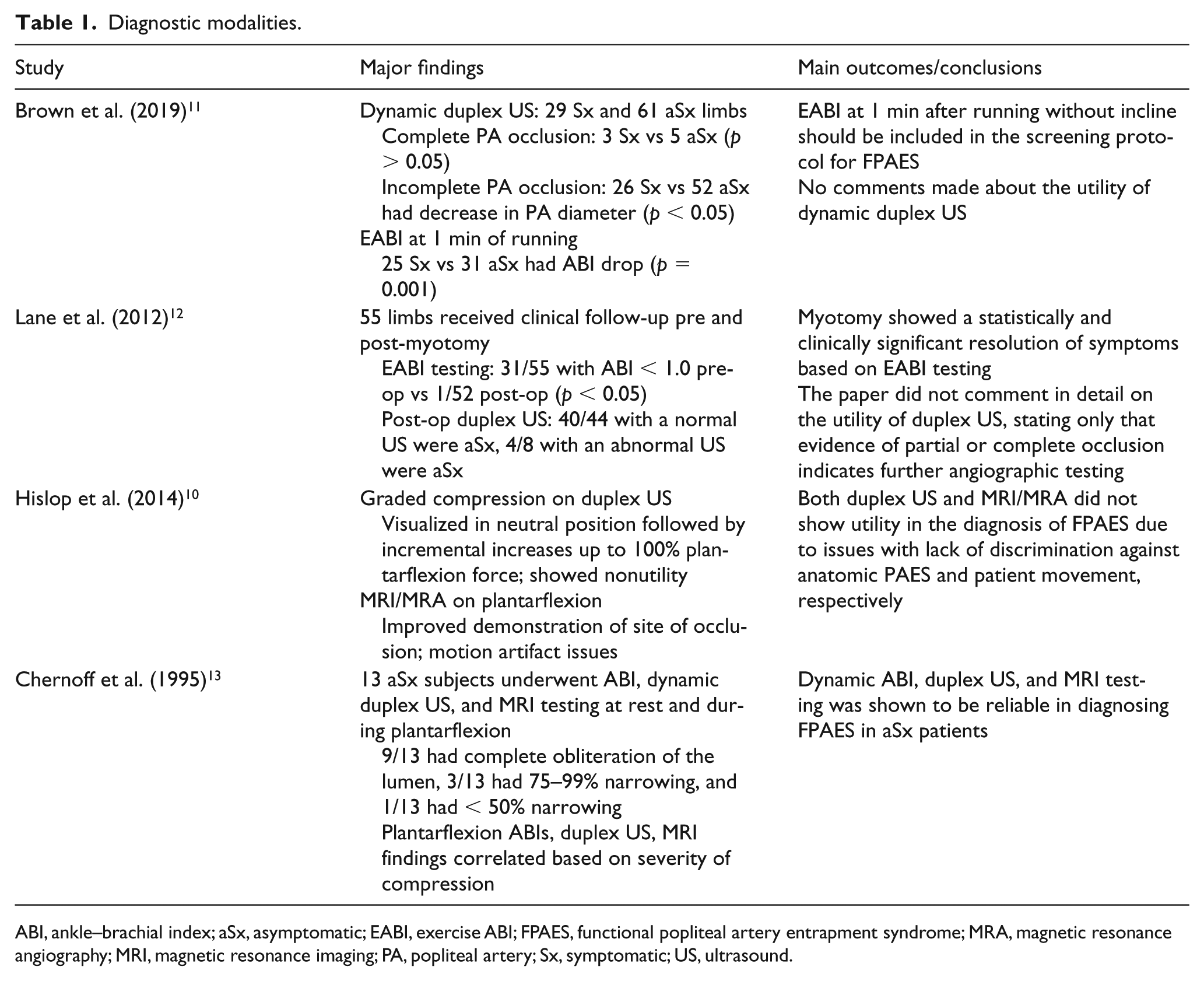
ABI, ankle–brachial index; aSx, asymptomatic; EABI, exercise ABI; FPAES, functional popliteal artery entrapment syndrome; MRA, magnetic resonance angiography; MRI, magnetic resonance imaging; PA, popliteal artery; Sx, symptomatic; US, ultrasound.
Management approaches
While the treatment for anatomic PAES has been largely agreed upon as open surgical intervention, the treatment for functional PAES is debatable. If an open intervention is attempted, the site and extent of myotomy to ensure resolution of symptoms is not always known. Historically, the literature (summarized in Table 2) reports successful symptom treatment in only 77% of patients, with a failure rate of 27.5% with attempted arterial reconstructive procedures. 17 Turnipseed et al. showed successful surgical intervention through compartment release, involving soleal and gastrocnemius fascial attachments on the medial aspect along with resection of the distal third of the plantaris muscle. 9 In their study, 43 patients with functional PAES were treated, with the resulting morbidity of 4.6% due to seromas and 2% due to wound infections, 38% of patients had new CRECS complaints, and there was no need for redo compartment release with a mean follow-up of 46 months. Causey et al. showed the utility of intraoperative ultrasound for guiding the extent of myotomy in a small series of three patients. 18 The authors state that clinical assessment of the extent of resection is not very accurate and intraoperative ultrasound can be used to guide the extent of myotomy. Clemens et al. conducted a retrospective analysis of 25 limbs treated surgically at a military tertiary referral center, showing better improvement in symptoms for patients with identifiable muscle slips as opposed to in patients without one, overall demonstrating the utility and effectiveness of surgical intervention for FPAES. 19
Management modalities.
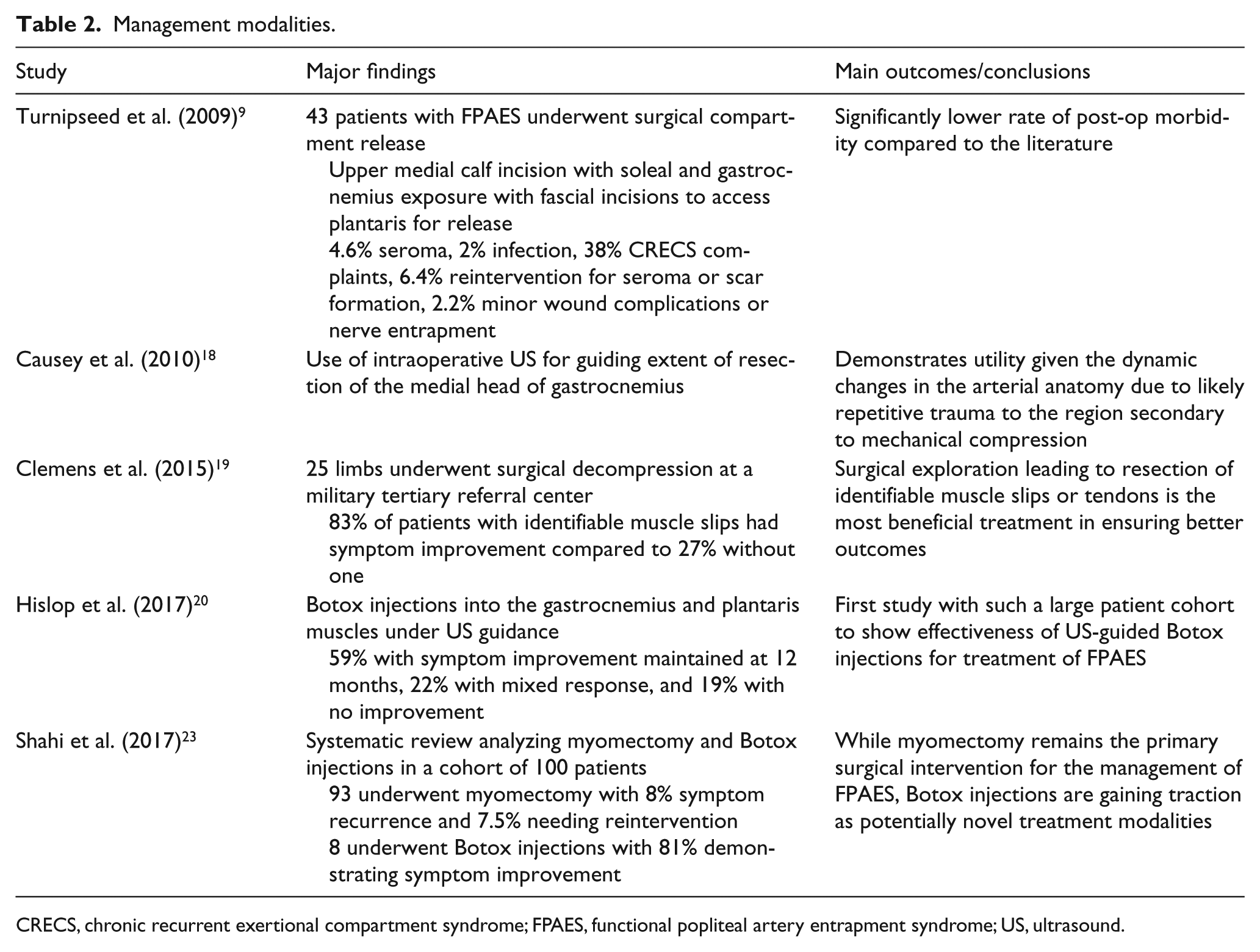
CRECS, chronic recurrent exertional compartment syndrome; FPAES, functional popliteal artery entrapment syndrome; US, ultrasound.
The role of botulinum toxin injections has been considered for the past several years. In a recent study conducted by Hislop et al., among their cohort of 27 patients who received localized 50 U (max 100 U) BTA-X injections into the gastrocnemius and plantaris muscles under ultrasound guidance at the level of the occlusion, 59% reported a symptom improvement maintained at 12 months, 22% relayed a mixed response characterized as an initial response that subsequently reduced over 12 months, and 19% gained no benefit from treatment. 20 No major complications were reported overall. While a few other independent case reports support the utility of Botox injections for functional PAES,21,22 this study by Hislop et al. presents the largest cohort in the management of functional PAES using Botox.
Shahi et al. conducted a systematic review on the management of FPAES. Myotomy was conducted in 93 cases and Botox injections in eight cases. 23 In the myotomy group, eight patients underwent adjunctive vascular reconstruction. On follow-up with duplex US at a mean of 22 months, 8% had a recurrence of symptoms and 7.5% needed revision procedures. In the Botox group, 81% noted symptom improvement at a mean follow-up of 53 days. No duplex US was conducted in this group.
While surgical intervention for functional PAES has been attempted and has shown success in a few studies, the results have yet to be reproduced in large cohorts. Botox presents as a potentially viable new alternative given the findings reported by Hislop et al. However, this remains to be validated in a bigger cohort before being applicable to patients with functional PAES. Nevertheless, the relatively noninvasive nature of Botox injections compared to open surgical interventions and the ability to administer the injections under ultrasound guidance definitely makes it an available intervention for vascular surgeons as well as other providers.
Conclusion
FPAES continues to be a diagnostic and treatment challenge for clinicians attempting to manage this condition in a very young patient population. Provocative ABI and duplex US testing present as reliable first-line tools to detect this condition followed by further characterization using MRI/MRA. While surgery is not always associated with symptom improvement, it remains one of the only methods available today. With Botox injections paving the way for alternative approaches, it remains to be seen what other modalities will be developed in the near future to manage this condition.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
