Abstract
Patients with peripheral artery disease (PAD) are at high risk for ischemic cardiovascular complications. While single antiplatelet therapy (SAPT), predominantly aspirin, has long been the standard antithrombotic treatment in stable PAD, there have now been greater than 40,000 PAD patients randomized to varying antiplatelet and/or anticoagulant regimens. In this review, we provide a summary of the current evidence for antithrombotics in stable PAD, focusing on the rates of major adverse cardiovascular events (MACE), major adverse limb events (MALE), and major bleeding. SAPT has a limited role in the treatment of asymptomatic PAD, particularly in the absence of concomitant coronary artery disease. In symptomatic PAD, SAPT is effective in preventing MACE, though treatment with a thienopyridine appears marginally superior to aspirin. Dual antiplatelet therapy (DAPT) suggests benefit over SAPT in reducing MACE and MALE, though studies to date are not conclusive and/or are associated with excess major bleeding. Combining moderate to high intensity vitamin K antagonists with antiplatelet therapy does not reduce MACE or MALE and increases life-threatening bleeding. Rivaroxaban 2.5 mg BID in addition to aspirin reduces the incidence of both MACE and MALE as compared to aspirin alone, without increasing life-threatening bleeding. This regimen is associated with a reduced severity of MALE when it does occur. Comparisons across antithrombotic trials in PAD are challenging given the heterogeneity of patient populations and the differing assessment of outcomes. The vascular medicine practitioner can reduce ischemic cardiac and limb events, as well as minimize life-threatening bleeding, by choosing the optimal antithrombotic regimen in their PAD patients.
Keywords
Introduction
More than 200 million people live with peripheral artery disease (PAD) worldwide, 1 including nearly 15% of patients over the age of 70 years. 2 Patients with PAD have higher cardiovascular event rates than those with coronary artery disease (CAD) or cerebrovascular disease, 3 with double the total mortality and cardiovascular mortality than predicted by their risk factors alone. 4 Health-related quality of life is significantly reduced in patients with symptomatic PAD, and may result in greater quality of life impairment than other serious illnesses such as chronic lung disease and some cancers. 5
Despite this, the optimal antithrombotic regimen for long-term use in stable PAD patients remains unclear. Stable PAD refers to patients without recent (6 months) endovascular or surgical peripheral artery procedures and without progressive signs of critical limb ischemia. While single antiplatelet therapy (SAPT) has been recommended as first line therapy in guidelines for patients with symptomatic PAD,6,7 the choice of antiplatelet, consideration of dual antiplatelet therapy, and/or the use of anticoagulants varies widely among practitioners. 8 In the past two decades, greater than 40,000 patients with stable PAD have been randomized to varying antiplatelet and/or anticoagulant regimens (Table 1). There is now a large body of evidence which can inform clinicians’ treatment decisions for PAD patients. However, comparisons across trials are sometimes challenging, particularly when distinct types of PAD patients have been enrolled, when background treatment with respect to secondary prevention therapies differ, and when definitions of outcome events vary. In this review, we assess the key trials conducted in patients with stable PAD for three important outcomes – major adverse cardiovascular events (MACE), major adverse limb events (MALE), and major bleeding – to clarify the evidence to assist clinicians with their choice of antithrombotic therapies.
Antithrombotics for prevention of cardiac events and limb events in patients with known PAD.
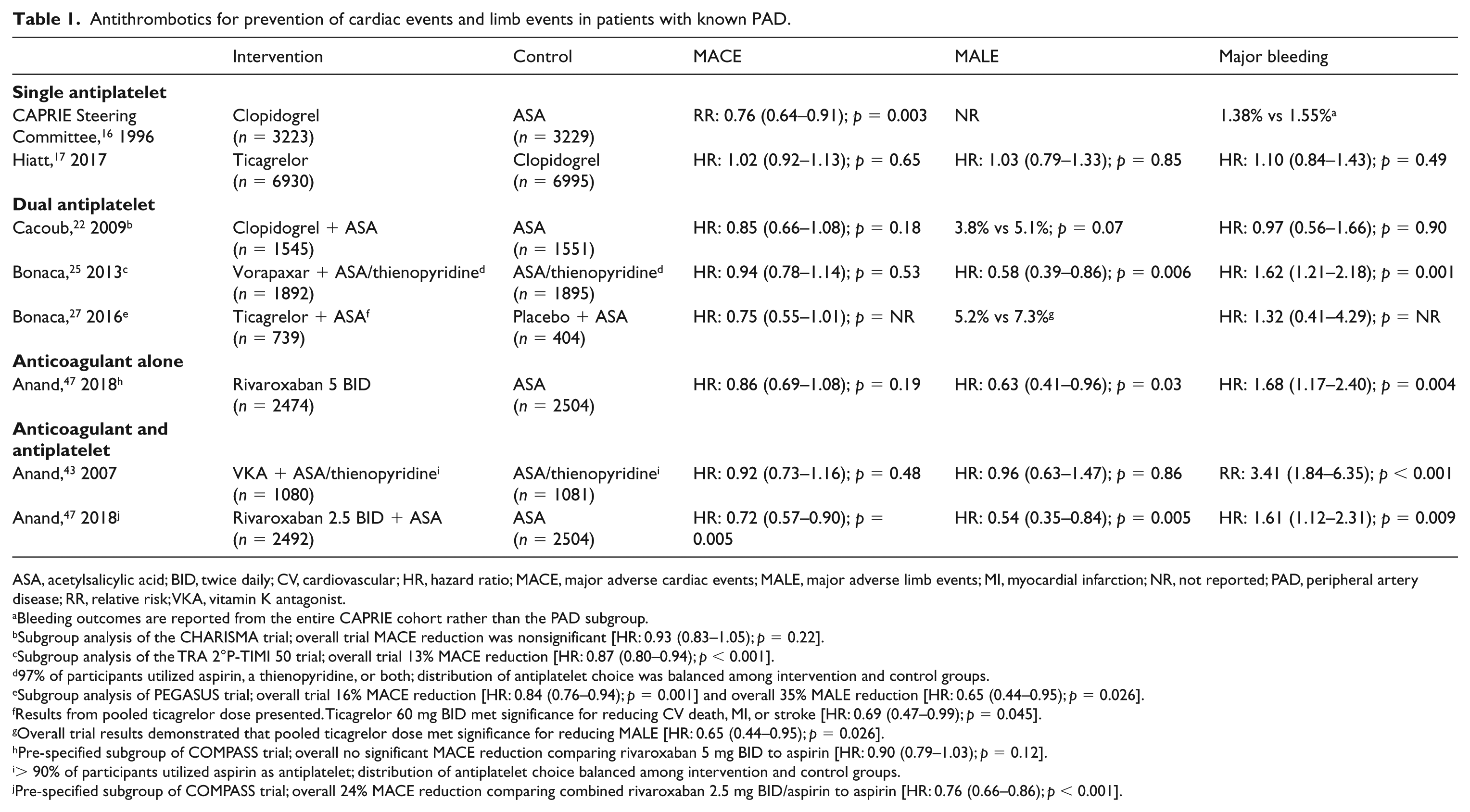
ASA, acetylsalicylic acid; BID, twice daily; CV, cardiovascular; HR, hazard ratio; MACE, major adverse cardiac events; MALE, major adverse limb events; MI, myocardial infarction; NR, not reported; PAD, peripheral artery disease; RR, relative risk; VKA, vitamin K antagonist.
Bleeding outcomes are reported from the entire CAPRIE cohort rather than the PAD subgroup.
Subgroup analysis of the CHARISMA trial; overall trial MACE reduction was nonsignificant [HR: 0.93 (0.83–1.05); p = 0.22].
Subgroup analysis of the TRA 2°P-TIMI 50 trial; overall trial 13% MACE reduction [HR: 0.87 (0.80–0.94); p < 0.001].
97% of participants utilized aspirin, a thienopyridine, or both; distribution of antiplatelet choice was balanced among intervention and control groups.
Subgroup analysis of PEGASUS trial; overall trial 16% MACE reduction [HR: 0.84 (0.76–0.94); p = 0.001] and overall 35% MALE reduction [HR: 0.65 (0.44–0.95); p = 0.026].
Results from pooled ticagrelor dose presented. Ticagrelor 60 mg BID met significance for reducing CV death, MI, or stroke [HR: 0.69 (0.47–0.99); p = 0.045].
Overall trial results demonstrated that pooled ticagrelor dose met significance for reducing MALE [HR: 0.65 (0.44–0.95); p = 0.026].
Pre-specified subgroup of COMPASS trial; overall no significant MACE reduction comparing rivaroxaban 5 mg BID to aspirin [HR: 0.90 (0.79–1.03); p = 0.12].
> 90% of participants utilized aspirin as antiplatelet; distribution of antiplatelet choice balanced among intervention and control groups.
Pre-specified subgroup of COMPASS trial; overall 24% MACE reduction comparing combined rivaroxaban 2.5 mg BID/aspirin to aspirin [HR: 0.76 (0.66–0.86); p < 0.001].
Single antiplatelet therapy
Aspirin monotherapy has long been accepted as the standard of care for patients with symptomatic PAD and it is the most widely used antiplatelet agent for this condition worldwide.6–8 This is largely based on the evidence from the Antithrombotic Trialists’ Collaboration, which reported a 23% reduction in serious vascular events (i.e. cardiovascular death, myocardial infarction (MI), and stroke) with antiplatelet therapy when compared to placebo from 42 randomized trials of 9214 patients. 9 However, more than half the participants within the meta-analysis were included from non-aspirin antiplatelet trials, with a large positive trial of picotamide as compared to placebo in 2304 patients with PAD 10 particularly driving the overall positive results. Thereafter, the Critical Limb Ischemia Prevention Study (CLIPS) reinforced the efficacy of aspirin monotherapy in a population of 366 patients with PAD, predominantly symptomatic (77.6%), demonstrating a decrease in the primary composite outcome of fatal and nonfatal vascular events (MI, stroke, pulmonary embolus) or critical limb ischemia (6.5% vs 15.5%; hazard ratio (HR): 0.42; CI: 0.21–0.83; p = 0.01), albeit with a numerically increased incidence of bleeding (2.2% vs 0.0%; p = 0.99). 11 Berger et al.’s meta-analysis of aspirin in symptomatic PAD patients reported a trend towards reduction in MACE, cardiovascular death, and all-cause mortality, but only a significant reduction in nonfatal stroke. 12 A more recent meta-analysis of antiplatelet agents reported a significant 24% reduction in all-cause mortality, 46% reduction in cardiovascular mortality, and 20% reduction in cardiovascular events, 13 and thus the consensus remains that aspirin is effective in preventing MACE in symptomatic PAD patients.6,7,12
In contrast, there is a limited role for aspirin in patients with asymptomatic PAD, usually defined as a low ankle–brachial index (ABI) in the absence of symptoms. The Prevention of Progression of Arterial Disease and Diabetes (POPADAD) trial randomized 1276 patients with diabetes and an asymptomatic ABI ⩽ 0.99 to aspirin or placebo, finding no difference in death from CAD or stroke, nonfatal MI or stroke, or above-ankle amputation for critical limb ischemia (18.2% vs 18.3%; p = 0.86). 14 Within a more general population, the Aspirin for Asymptomatic Atherosclerosis (AAA) trial randomized 3350 patients with ABIs ⩽ 0.95 and free of clinical cardiovascular disease, to aspirin versus placebo, finding no difference in coronary events, stroke or revascularization (13.7% vs 13.3%; HR: 1.03; CI: 0.84–1.27). 15 Although no additional data are available to distinguish those with severely reduced as opposed to mildly reduced ABIs, the overall lack of efficacy in asymptomatic PAD is convincing.
Choice of antiplatelet agent: With the advent of alternative antiplatelet agents, clinical trial evidence supports that thienopyridines, such as clopidogrel, have greater relative risk reduction of MACE when compared to aspirin in those with PAD.13,16 Thienopyridines work differently than aspirin, selectively targeting the adenosine diphosphate (ADP) receptor on the surface of platelets, inhibiting the activation and aggregation of platelets. The CAPRIE trial investigated clopidogrel in comparison to aspirin for the prevention of cardiovascular events in patients with known, symptomatic atherosclerotic vascular disease, including patients with symptomatic CAD, cerebrovascular disease, and lower limb PAD. The overall trial demonstrated an 8.7% (CI: 0.3–16.5%) reduction in ischemic stroke, MI, or vascular death with clopidogrel therapy. 16 However, this was largely driven by the subgroup of patients included based on PAD. The subgroup of 6452 patients with symptomatic lower limb PAD demonstrated a 23.8% (CI: 8.9–36.2%) reduction in MACE as compared to aspirin, and was the only subgroup of vascular patients to meet significance. 16 No difference in the secondary outcome of ischemic stroke, MI, vascular death or amputation was observed in the overall trial (p = 0.076), and acute limb ischemia events were not collected. Severe gastrointestinal hemorrhage was modestly increased in those taking aspirin (0.71% vs 0.49%; p < 0.05), and this increase was consistent within the PAD patient cohort. More recently, the EUCLID trial conducted in 13,925 symptomatic lower limb PAD patients compared two different thienopyridine agents, ticagrelor and clopidogrel, but yielded no significant difference in MACE (n = 1491; p = 0.65), MALE (n = 232; p = 0.85) or Thrombolysis in Myocardial Infarction (TIMI) major bleeding outcomes (n = 222; p = 0.49). 17
Beyond aspirin: dual antiplatelet therapies
The benefit of dual antiplatelet therapy (DAPT) with a thienopyridine added to aspirin has long been established in patients with coronary vascular disease, since the CURE trial demonstrated that adding clopidogrel to aspirin reduced the composite of cardiovascular death, MI, and stroke (9.3% vs 11.4%; HR: 0.80; CI: 0.72–0.90; p < 0.001) in 12,562 patients with non-ST elevation acute coronary syndrome (ACS). 18 While this trial revolutionized the use of DAPT in those with coronary disease, it did not explore the implications of DAPT in patients with concomitant PAD. 18 As the number 19 and nature 20 of vascular beds affected by atherosclerosis informs patient risk, the study of those with both previous coronary disease and concomitant PAD has been of interest in the ensuing decades.
The combination of clopidogrel and aspirin has subsequently been tested in patients with chronic stable vascular disease in multiple territories. 21 CHARISMA 21 compared clopidogrel and aspirin in combination to aspirin alone in patients at high risk for atherothrombotic events, whether through documented symptomatic atherosclerotic vascular disease or via multiple atheroembolic risk factors. While the overall trial of 15,603 patients reported no significant difference in MACE (6.8% vs 7.3%; HR: 0.93; 95% CI: 0.83–1.05; p = 0.22) or severe bleeding (1.7% vs 1.3%; HR: 1.25; 95% CI: 0.97–1.61; p = 0.09), those included based on symptomatic atherosclerotic vascular disease (n = 12,153) had lower MACE rates in the DAPT group compared to aspirin alone (6.9% vs 7.9%; HR: 0.88; CI: 0.77–0.998; p < 0.05). 21 In the subgroup of 3096 CHARISMA patients included with established PAD, DAPT conferred no significant reduction in MACE (7.6% vs 8.9%; HR: 0.85; 95% CI: 0.66–1.08; p = 0.18), and only trended toward a decrease in peripheral artery bypass surgery (3.8% vs 5.1%; p = 0.07). 22 While patients with PAD randomized to DAPT did have significantly lower rates of MI (HR: 0.63; CI: 0.42–0.96; p = 0.03), and hospitalization for ischemic events (HR: 0.81; CI: 0.68–0.95; p = 0.01) compared to those randomized to aspirin alone, these results should be interpreted with caution as they are secondary endpoints in an overall neutral trial. 22 Overall, the trial results raise the possibility, but do not prove, that DAPT is superior to SAPT in patients with vascular disease.
Vorapaxar is a novel antiplatelet agent, which blocks protease-activated receptor 1 (PAR-1) on the platelet cell surface, thereby inhibiting thrombin-mediated platelet activation and thrombosis. TRA 2°P-TIMI 50 randomized 26,449 patients with a history of MI, ischemic stroke, or symptomatic PAD to vorapaxar to placebo, with a substantial proportion of patients taking concomitant DAPT with aspirin (administered in 93.5% of patients) and a thienopyridine (administered in 58.0% of patients). 23 The patients with a prior history of stroke had vorapaxar discontinued after a median of 2 years of follow up due to an excess of increased intracranial hemorrhage (2.5% vs 1.0%; p < 0.001) without improvement in cardiovascular death, MI or stroke (13.0% vs 11.7%; p = 0.75) in the vorapaxar group. 24 Among those patients with CAD and PAD who remained in the overall trial, vorapaxar conferred a significant reduction of MACE (9.3% vs 10.5%; HR: 0.87; CI: 0.80–0.94; p < 0.001), but with this came an increase in moderate to severe Global Utilization Of Streptokinase And Tpa For Occluded Arteries (GUSTO) bleeding (4.2% vs 2.5%; HR: 1.66; CI: 1.43–1.93; p < 0.001). 23 In a subgroup of PAD patients (n = 3787), adding vorapaxar to a second antiplatelet agent, namely aspirin (88%), a thienopyridine (37%) or both (29%), resulted in significantly less hospitalization for acute limb ischemia (2.3% vs 3.9%; HR: 0.58; CI: 0.39–0.86; p = 0.006) and peripheral artery revascularization (18.4% vs 22.2%; HR: 0.84; CI: 0.73–0.97; p = 0.017), yet it did not appear to prevent MACE (11.3% vs 11.9%; HR: 0.94; 95% CI: 0.78–1.14; p = 0.53), and was associated with an increase in GUSTO moderate or severe bleeding (7.4% vs 4.5%; HR: 1.62; CI: 1.21–2.18; p = 0.001). 25 Therefore, vorapaxar seemingly has benefits with respect to preventing ischemic limb events, but when used as part of DAPT, is associated with substantial bleeding, and at present is only rarely used in clinical practice.
The PEGASUS trial evaluated ticagrelor, at both 90 mg BID and 60 mg BID doses, in addition to aspirin compared to aspirin alone, in patients 1–3 years following MI. 26 The overall trial demonstrated a significant reduction in MACE at both the 90 mg (7.85% vs 9.04%; HR: 0.85; CI: 0.75–0.96; p = 0.008) and 60 mg (7.77% vs 9.04%; HR: 0.84; CI: 0.74–0.95; p = 0.004) doses, 26 as well as MALE (acute limb ischemia or peripheral revascularization for ischemia) when the two doses were pooled (0.46% vs 0.71%; HR: 0.65; CI: 0.44–0.95; p = 0.026); 27 however, in the smaller subgroup of 1143 patients with known PAD, only the 60 mg BID ticagrelor dose decreased MACE (14.1% vs 19.3%; HR: 0.69; CI: 0.47–0.99; p = 0.045) when added to aspirin, while pooled dosing did not reach significance. Rates of MALE were numerically lower in patients treated with ticagrelor combined with aspirin, analogous to the overall cohort, but did not reach statistical significance. 27 Similarly, TIMI major bleeding was significantly increased in the overall trial (90 mg: HR: 2.69; CI: 1.96–3.70; p < 0.001 and 60 mg: HR: 2.32; CI: 1.68–3.21; p < 0.001), but was only numerically higher, and did not meet significance, in the PAD subgroup (pooled dose relative major bleeding HR: 1.32; CI: 0.41–4.29). 27 However, the subgroup of PAD patients was small in comparison to the overall trial population, and was underpowered. Moreover, all patients within the PEGASUS PAD subgroup had established CAD as well, a pertinent consideration given that polyvascular disease increases future vascular risk. 20 Once again, PEGASUS, like CHARISMA, raises the possibility that treating PAD patients with DAPT may be more effective than SAPT in reducing cardiovascular and limb events, but the result is not conclusive.
Dual antiplatelet therapy has been evaluated in patients with ACS and PAD. The PLATO trial evaluated ticagrelor (180 mg load with 90 mg BID thereafter) versus clopidogrel (300–600 mg load with 75 mg daily thereafter), on a background of aspirin, in 18,624 patients admitted to hospital for ACS. 28 After 12 months of therapy, the combination of ticagrelor and aspirin reduced the primary composite outcome of vascular death, MI or stroke (9.8% vs 11.7%; HR: 0.84; CI: 0.77–0.92; p < 0.001), reduced vascular death alone (4.0% vs 5.1%; HR: 0.79; CI: 0.69–0.91; p = 0.001), and reduced all-cause mortality as well (4.5% vs 5.9%; HR: 0.78; CI: 0.69–0.89; p < 0.001), all without a significant increase in major bleeding (11.6% vs 11.2%; HR: 1.04; CI: 0.95–1.13; p = 0.43). 28 In a subgroup of 1114 patients in PLATO with lower limb PAD, as defined by symptomatic claudication with prior objective lower limb vascular disease, the primary MACE outcome, vascular death, and all-cause mortality were all similarly reduced in magnitude when compared to the overall trial. 29 While these results did not meet significance, potentially due to small sample size, no significant interaction between PAD status and vascular outcome was noted. 29
It is also informative to review trials that evaluated extended DAPT following coronary stenting, as PAD patients commonly have concomitant CAD. PRODIGY randomized 1970 patients with recent bare metal or drug eluting stents to either 6 months or 24 months of clopidogrel in addition to aspirin. 30 Prolonged clopidogrel use did not confer a significant reduction in the primary MACE endpoint of MI, stroke, or all-cause mortality (10.1% vs 10.0%; HR: 0.98; CI: 0.74–1.29; p = 0.91), nor did it meet significance for any of the secondary efficacy endpoints. 30 Prolonged clopidogrel use did result in an increase in TIMI major bleeding (1.6% vs 0.6%; HR: 0.38; CI: 0.15–0.97; p = 0.041). 30 Analyzing the 246 PRODIGY patients enrolled with intermittent claudication, previous lower limb arterial revascularization or previous vascular amputation, 24 months of clopidogrel significantly decreased the incidence of MACE compared to a 6-month course (16.1% vs 27.3%; HR: 0.54; CI: 0.31–0.95; p = 0.03), mainly driven by a decrease in overall death (10.2% vs 21.1%; HR: 0.45; CI: 0.23–0.88; p = 0.02). 31 The composite of major or minor bleeding in the PAD subgroup was not increased in the prolonged DAPT versus shorter DAPT groups (1.8% vs 3.5%; HR: 0.50; CI: 0.09–2.74; p = 0.43). The discordant results of the PAD subgroup compared with the overall trial may be attributed to the higher baseline risk of those with polyvascular disease, 19 though the results should be interpreted with caution given the very small sample size of the PAD subgroup. The ‘DAPT study’ investigated 30 months of thienopyridine (clopidogrel or prasugrel) versus 12 months of thienopyridine, in addition to aspirin, in patients receiving drug eluting stents. 32 Thirty months of DAPT reduced the composite of death, MI, and stroke (4.3% vs 5.9%; HR: 0.71; CI: 0.59–0.85; p < 0.001), as well as the co-primary outcome of definite or probable stent thrombosis (0.4% vs 1.4%; HR: 0.29; CI: 0.17–0.48; p < 0.01), but came at the expense of increased GUSTO moderate to severe bleeding (2.5% vs 1.6%; HR: 1.61; CI: 1.21–2.16; p = 0.001). 32 A subgroup of 649 patients in this trial with a history of PAD did not demonstrate a similar reduction in MACE (12.04% vs 11.26%; HR: 1.06; CI: 0.67–1.67; p = 0.81) nor a statistically significant reduction in stent thrombosis (0.71% vs 2.20%; HR: 0.30; CI: 0.06–1.44; p = 0.11) with prolonged DAPT, although again, conclusions are limited due to the small sample size. 33
Anticoagulants
Oral anticoagulants have been used since the 1960s to prevent limb and cardiovascular events in PAD patients, although the evidence supporting its use is mixed.34–41 Building upon the observation that anticoagulation prevented cardiovascular events in patients with CAD, 42 oral anticoagulants combined with antiplatelet therapy was evaluated in patients with PAD. The DUTCH BOA trial evaluated the use of vitamin K antagonists (VKAs) targeting a moderate to high intensity (INR 3–4.5) as compared to aspirin among patients who had undergone an infrainguinal bypass surgery. No reduction in the primary outcome of MACE or graft occlusion was observed and there was an excess in severe bleeding, including increased intracranial hemorrhage. 41 The Warfarin Antiplatelet Vascular Evaluation (WAVE) trial in 2180 patients with stable PAD demonstrated that combining VKAs targeting moderate intensity anticoagulation (INR 2–3) and antiplatelet therapy as compared with antiplatelet therapy alone did not reduce MACE (12.2% vs 13.3%; p = 0.48) or severe ischemia leading to a coronary or peripheral intervention (3.9% vs 4.1%; p = 0.86). 43 Importantly, the WAVE trial also demonstrated that VKA plus antiplatelet therapy was associated with significantly more life-threatening bleedings than antiplatelet therapy alone (relative risk (RR): 3.41; CI: 1.84–6.35; p < 0.001). 43 Thus, the DUTCH BOA and WAVE trials, put in the context of other PAD trials of warfarin,41,44,40 have prompted guideline recommendations against anticoagulation after peripheral vascular surgery or in stable PAD.6,7
Following successes in the treatment of venous thromboembolic disease, atrial fibrillation, and ACS, 45 direct oral anticoagulants (DOACs), which target a single factor in the coagulation cascade, have been tested among patients with PAD. The COMPASS trial enrolled patients with CAD and/or PAD, randomizing to aspirin alone, rivaroxaban 5 mg BID alone, or the combination of rivaroxaban 2.5 mg BID in addition to aspirin. 46 The dosing regimen was based on the ATLAS-2 trial, investigating the addition of rivaroxaban following ACS. 45 The overall trial demonstrated significantly decreased MACE in those on combination rivaroxaban 2.5 mg BID with aspirin as compared to aspirin alone (HR: 0.76; 95% CI 0.66–0.86; p < 0.001), but increased major bleeding as defined by the modified International Society on Thrombosis and Haemostasis (ISTH) criteria (HR: 1.70; 95% CI: 1.40–2.05; p < 0.001). 46 Rivaroxaban alone at 5 mg BID as compared to aspirin did not confer significant cardiovascular benefit, and increased major bleeding. 46 The two rivaroxaban therapies were compared to aspirin for both MACE and MALE outcomes in a pre-specified subgroup analysis of patients with PAD, defined as PAD of the lower extremities (previous peripheral bypass surgery or angioplasty, limb or foot amputation, intermittent claudication with objective evidence of PAD), prior carotid artery disease (previous carotid artery revascularization or asymptomatic carotid artery stenosis of at least 50%), or CAD with an ABI of less than 0.90. 47 Concordant with the overall trial, the combination of rivaroxaban 2.5 mg BID with aspirin decreased both the composite of cardiovascular death, stroke, and MI (HR: 0.72; CI: 0.57–0.90; p = 0.005), as well as the limb composite of acute limb ischemia, and chronic limb ischemia including major vascular amputation (HR: 0.54; CI: 0.35–0.84; p = 0.005). 47 The combined reduction of MACE or MALE in PAD patients treated with rivaroxaban 2.5 BID and aspirin was 31% (95% CI: 0.56–0.85; p < 0.001). 47 Combination therapy increased modified ISTH major bleeding (HR: 1.61; CI: 1.12–2.31; p = 0.009), though without significant increases in intracranial or fatal bleeding. 47 It is prudent to consider, however, that patients at high risk of bleeding were excluded from this trial, perhaps underestimating the true bleeding risk in the general PAD population. Considering the totality of MACE, MALE and fatal or critical bleeding in this population, there was a persistent net clinical benefit in utilizing rivaroxaban 2.5 BID and aspirin (HR: 0.75; 95% CI: 0.60–0.94; p = 0.01). 47 While the inclusion of a heterogenous population of PAD patients (from patients with previous lower limb revascularization to those with asymptomatic carotid stenosis) lends to high statistical power and broad generalizability, it’s heterogeneity may limit inferences regarding subgroups of study. This limitation is minimized given the consistent effects, of similar magnitude, across all subgroups included in COMPASS-PAD. A subsequent analysis demonstrated that following a MALE, PAD patients suffer a high rate of amputation (absolute rate: 20.5%; relative HR of MALE: 197.5), as well as hospitalization and death (absolute rate: 8.3%; relative HR of MALE: 3.23) within the year following MALE; 48 furthermore, these outcomes are substantially worse in patients who received aspirin alone, compared to rivaroxaban and aspirin, suggesting that the severity of MALE suffered may be reduced in patients receiving combination therapy (Figure 1). 48
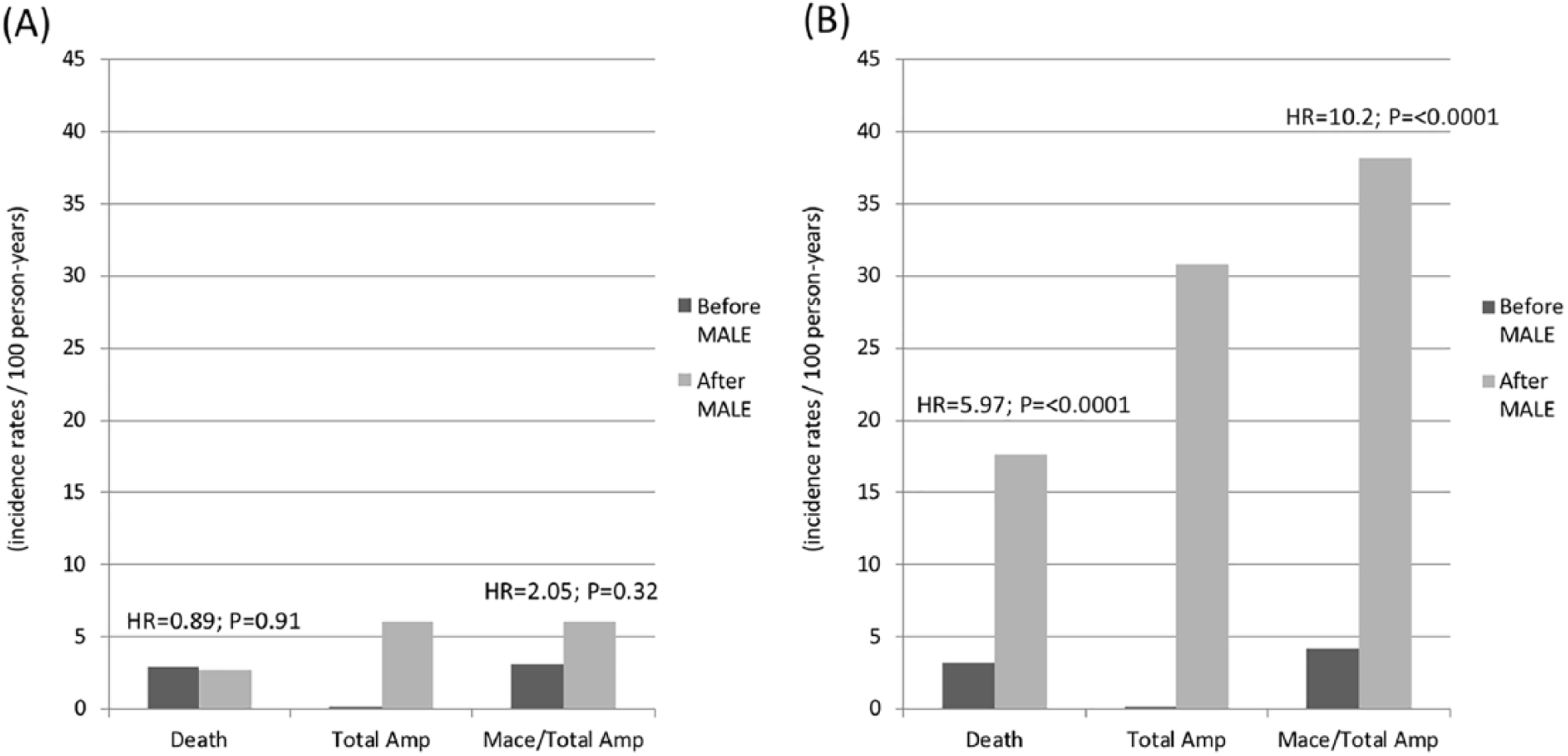
Outcomes before and after a MALE in PAD patients randomized to and continued on rivaroxaban 2.5 mg BID with aspirin (A) or aspirin alone (B) within the COMPASS trial 48 .
While rivaroxaban alone at 5 mg BID outperformed aspirin in preventing the limb composite of acute limb ischemia and chronic limb ischemia including major amputation above the forefoot due to vascular cause (HR: 0.63; CI: 0.41–0.96; p = 0.03), it did not reduce MACE and was similarly associated with increased major bleeding (HR: 1.68; CI: 1.17–2.40; p = 0.004), including intracranial hemorrhage. 47 Given the improved bleeding profile and flexible dosing associated with DOACs, there are on-going trials investigating DOAC and antiplatelet combinations for preventing ischemic events in PAD. 49
Discussion
To summarize the evidence to date, antithrombotic agents do not confer cardiovascular benefit in patients with asymptomatic PAD. In patients with stable, symptomatic PAD, clopidogrel alone appears marginally superior to aspirin in reducing adverse cardiac events, although meta-analyses suggest aspirin alone is a reasonable alternative choice and is currently recommended as the first line of therapy. Subgroup analyses suggest some benefit of long-term DAPT over single antiplatelet therapy in reducing MACE and MALE in PAD patients; however, the results from trials are not definitive. DAPT is clearly superior to SAPT in the 12 months following ACS. Combining VKAs which target moderate to high intensity anticoagulation with antiplatelet therapy should not be utilized for the long-term treatment of patients with stable PAD, as it yields no benefit in MACE or MALE, and increases the risk of life-threatening bleeding. On the other hand, recent evidence shows that low-dose DOACs (rivaroxaban 2.5 mg BID) in combination with aspirin reduces MACE as well as MALE, and does not increase life-threatening bleeding. Furthermore, the combination of low-dose rivaroxaban and aspirin is associated with a reduced severity of MALE when it does occur in patients with PAD. 48
Interpreting the published literature of antithrombotics in PAD is challenging given the heterogeneity of patient characteristics, drug combinations, therapeutic doses, and outcome definitions. The definition of PAD varies between trials, with some restricting patient inclusion to lower extremity PAD (CAPRIE, 16 CHARISMA, 22 EUCLID, 17 PEGASUS 27 ) while others expand this definition to include other vascular beds such as carotid, subclavian, and lower extremity PAD (WAVE 43 and COMPASS 47 ). Furthermore, some trials’ enrollment criteria required the presence of clinical CAD (PEGASUS 27 ), whereas others did not (COMPASS, 47 EUCLID 17 ). The number 19 and nature 20 of vascular beds with atherosclerosis may influence patient-specific risks. Because all vascular patients suffer from the same underlying atheroma-forming disease, and are at risk for atherothrombosis,50,51 the relative benefit of antithrombotic therapies are expected to be similar among varying vascular populations. However, it is clear from observational registries (REACH 52 ), and the aforementioned clinical trials, that the patients with symptomatic and more widespread atherosclerosis (i.e. number of vascular beds) have higher MACE event rates, making the absolute risk reduction, and the ability to detect significant benefit, differ between trials.
Comparing across trials between types of antiplatelet agents and/or anticoagulants is also challenging. However, it is reasonable to consider the evidence suggesting that some single antiplatelet agents are more potent than others (CAPRIE 16 ), that dual antiplatelet therapy may confer greater efficacy in MACE and MALE reduction (PEGASUS, 27 CHARISMA, 22 TRA 2°P-TIMI 50 25 ), and that a combination of low-dose DOACs with aspirin consistently reduces MACE and MALE across various subsets of vascular patients, including those with PAD (COMPASS 47 ). Clinicians must also consider the safety profile of antithrombotics, particularly with respect to severe bleeding and bleeding within critical organs. The risks of severe and critical bleeding do increase with more potent DAPT combinations, as well as with antiplatelet–anticoagulant combinations, making the net clinical benefit of MACE/MALE reduction versus the risk of severe bleeding a prudent consideration. For PAD patients, while the full-dose antiplatelet–anticoagulant combination is associated with a high risk of severe bleeding, 43 the lower-dose DOAC regimen used in the COMPASS trial appears to have a favorable net clinical benefit profile, 47 understanding that high bleeding risk individuals were excluded from investigation.
The heterogeneity in outcomes is also challenging to compare. While the concept of MACE has been well established within cardiovascular investigations, there is no consensus definition for MALE as used in clinical trials. 53 Definitions range from hospitalization for limb ischemia to requirements for peripheral artery interventions and amputations, with varying combinations as well. Many trials have included acute limb ischemia as a key component of MALE; however, even acute limb ischemia definitions differ on integral components such as the findings on imaging, clinical examination, and time and type of intervention. Standardization of the MALE definition will be an asset for the designing of future investigations pertaining to antithrombotics in PAD, and allow for more direct comparisons across clinical trials. 54
Conclusion
Patients with PAD are at high risk for MACE and MALE. 48 For decades, antiplatelet monotherapy was the antithrombotic option with the best evidence for reducing cardiovascular outcomes. After a recent proliferation of randomized clinical trials evaluating antithrombotics in PAD, new options in the prevention of MACE and MALE are available in these high-risk 48 patients (Table 2). Understanding the efficacy and complications of varying antiplatelet and anticoagulant combinations, as delineated by clinical evidence, will allow the vascular medicine practitioner to treat stable PAD safely and effectively.
Factors in considering antithrombotic options in patients with stable, symptomatic PAD.
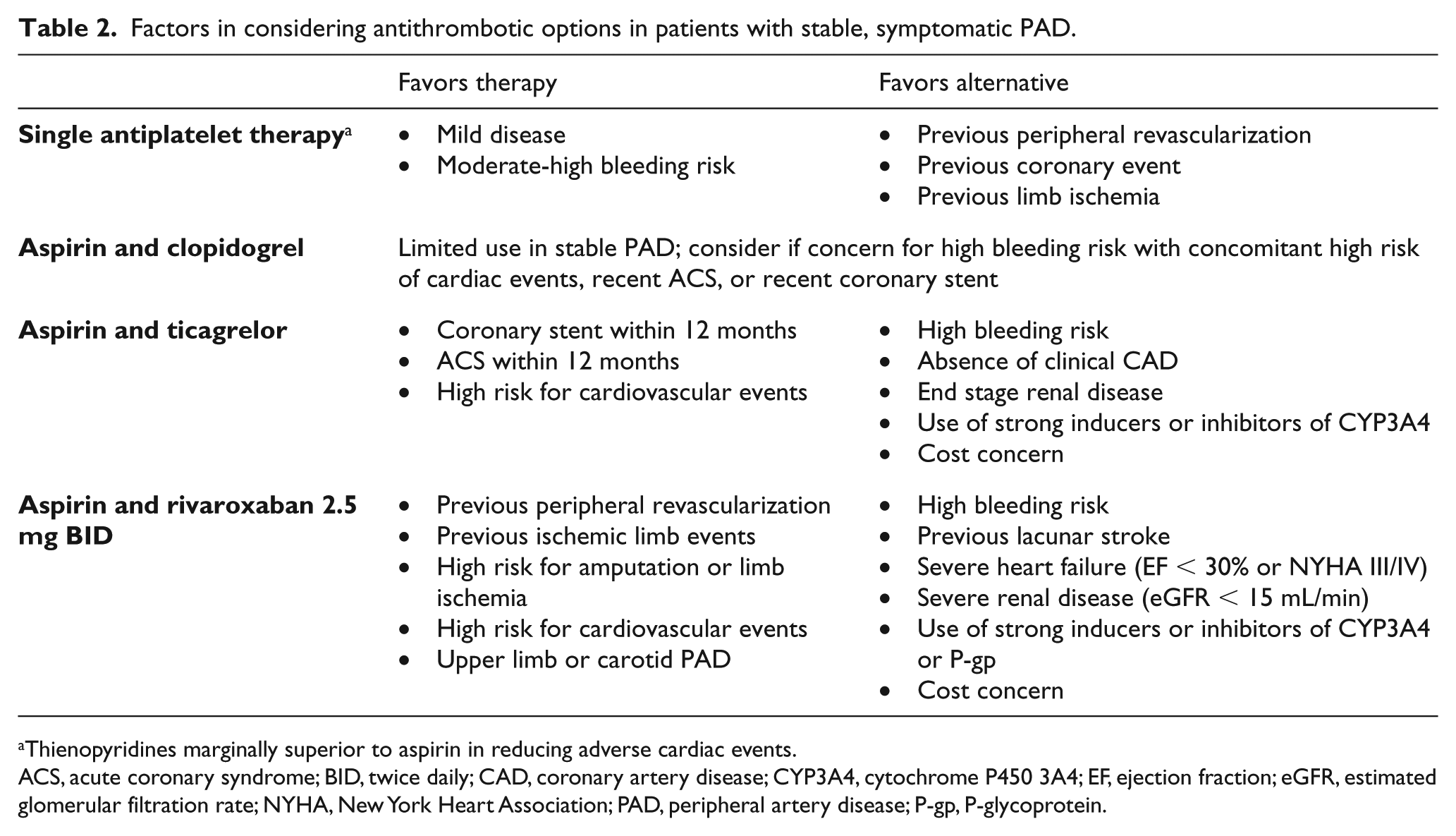
Thienopyridines marginally superior to aspirin in reducing adverse cardiac events.
ACS, acute coronary syndrome; BID, twice daily; CAD, coronary artery disease; CYP3A4, cytochrome P450 3A4; EF, ejection fraction; eGFR, estimated glomerular filtration rate; NYHA, New York Heart Association; PAD, peripheral artery disease; P-gp, P-glycoprotein.
Footnotes
Declaration of conflicting interests
S. Anand receives funding for research, speaking, and consulting from Bayer AG, the maker of rivaroxaban and sponsor of the COMPASS trial.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
