Abstract
Intensive antithrombotic therapy reduces major adverse cardiovascular events (MACE) and major adverse limb events (MALE) in patients with peripheral artery disease (PAD). Recent studies have suggested heterogeneity in risk and benefit in those with and without concomitant coronary artery disease (CAD) and peripheral revascularization. We evaluated the risk of MACE and MALE in patients with PAD stratified by history of concomitant CAD and prior peripheral revascularization and whether the efficacy and safety of vorapaxar were similar in these subgroups. The TRA 2°P-TIMI 50 trial randomized 26,449 patients with prior MI, ischemic stroke, or PAD to vorapaxar or placebo. This analysis examined the effect of vorapaxar in a broad population of 6136 patients with PAD. Overall, vorapaxar significantly reduced MACE (HR 0.85, 95% CI 0.73, 0.99; p = 0.034) and MALE (HR 0.70, 95% CI 0.53, 0.92; p = 0.011) in patients with PAD. The absolute risk reduction (ARR) for MACE was greater in patients with PAD and CAD versus those with PAD alone (–2.2% vs 0.1%: number needed to treat (NNT) 45 vs 1000). Conversely, the ARR for MALE was higher in those with prior lower extremity revascularization (2.5% vs 0.2%: NNT 40 vs 500). Vorapaxar increased major bleeding (HR 1.39, 95% CI 1.12, 1.71; p = 0.003). The net clinical outcome in all patients with PAD was reduced with vorapaxar (HR 0.82, 95% CI 0.72, 0.94; p = 0.004), with benefits driven by reductions in MACE for those with CAD and by reductions in MALE for those with prior peripheral revascularization. Among patients with PAD, vorapaxar resulted in a net clinical benefit; however, the drivers of benefit were heterogeneous, with greater reductions in MACE in those with concomitant CAD and greater reductions in MALE in those with prior lower extremity revascularization, and unclear benefit in patients with neither. These clinical characteristics may be useful in identifying the subgroups of patients with PAD most likely to benefit from potent antithrombotic therapies.
Keywords
Introduction
It is estimated that more than 200 million individuals have peripheral artery disease (PAD) globally. 1 Patients with PAD are at heightened risk of systemic atherothrombotic events characterized as major adverse cardiovascular events (MACE) including cardiovascular (CV) death, myocardial infarction (MI), and stroke. In addition to these systemic risks, by nature of their lower extremity atherosclerosis, patients with PAD are also at heightened risk of major adverse limb events (MALE) including acute limb ischemia (ALI), urgent revascularization for ischemia, and major amputations.2,3
Recent studies have demonstrated that more potent antithrombotic strategies reduce MACE and MALE but increase bleeding in patients with PAD.4–6 In translating these trials to practice, the benefits of these therapies must be weighed against bleeding risk. Recent guidelines weigh ischemic risk and bleeding risk in patients with PAD, overall giving lower levels of recommendations that acknowledge that clinicians must weigh the risks and personalize therapy. Therefore, identifying subgroups of patients with PAD at higher risk of MACE and MALE who derive greater absolute benefits to offset bleeding risk is of clinical interest.
Although validated scores for risk stratification have not been established in PAD, recent studies have observed heterogeneity in risk based on clinical characteristics. Registries have observed that patients with polyvascular disease, particularly the combination of PAD and coronary artery disease (CAD), are at higher risk than patients with symptomatic disease in one territory alone. 3 The EUCLID trial evaluated ticagrelor relative to clopidogrel in patients with PAD. 7 While the overall study showed that ticagrelor was not superior to clopidogrel, there was significant heterogeneity (p-interaction 0.03) in those with concomitant CAD, suggesting a benefit of ticagrelor in this subgroup. 7 In PEGASUS-TIMI 54, patients with prior MI and PAD were at higher risk than those with PAD alone and derived greater absolute benefits for MACE and MALE from the combination of aspirin and ticagrelor relative to those with MI and no PAD. 8 These observations suggest that patients with PAD and concomitant CAD are at higher risk of MACE relative to those with PAD and no CAD and have the potential to derive greater absolute benefits from preventive therapies. 9
Similarly, recent studies have observed heterogeneous risk of MALE, particularly ALI, in patients with PAD based on clinical characteristics. In TRA 2°P-TIMI 50, PEGASUS-TIMI 54, and EUCLID, patients with PAD with prior peripheral revascularization were all observed to have an approximately fourfold risk of ALI relative to those without prior peripheral revascularization.5,7,8 These observations suggest that this feature may be useful for risk stratification and identification of patients at higher risk of MALE.
We therefore evaluated whether risk of MACE and MALE in patients with PAD enrolled in the TRA 2°P-TIMI 50 trial (ClinicalTrials.gov Identifier: NCT00526474) differed on the basis of concomitant CAD and prior peripheral revascularization, and whether the absolute benefits of more intensive antiplatelet therapy with vorapaxar differed by these criteria.
Methods
Study population
The design and primary results of TRA 2°P-TIMI 50 have been reported previously.10,11 All patients provided informed consent, and the protocol was approved by the Institutional Review Boards. In brief, stable patients with atherosclerotic vascular disease, including prior MI, ischemic stroke, or symptomatic PAD, were randomly assigned to treatment with either vorapaxar sulfate 2.5 mg daily or placebo and followed for a median of 30 months. Key exclusion criteria included pending revascularization procedure, history of bleeding diathesis, recent bleeding, and treatment with a vitamin K antagonist, an indication for anticoagulation (e.g. atrial fibrillation or venous thromboembolism) or active hepatobiliary disease.
A total of 26,449 patients were enrolled in the trial with hierarchical stratification according to the qualifying diagnosis (MI, stroke or PAD). To qualify for inclusion based on PAD, patients were required to have a history of claudication in conjunction with an ankle–brachial index (ABI) < 0.85 or previous revascularization of the lower extremity for symptomatic PAD, and no MI or stroke within 12 months. Patients with PAD were included in the MI or stroke cohorts if they met criteria for those subgroups and were only characterized as ‘qualifying PAD’ if there was no MI or stroke in the prior year. Screening ABI was performed in all 26,449 patients using standardized procedures by trained personnel at the participating centers. The results of the trial in patients enrolled with ‘qualifying PAD’ have been previously reported. 4
The current analysis includes all patients with PAD in TRA 2°P-TIMI 50 regardless of stratum. Analyses of the benefit and risk of vorapaxar are restricted to patients with PAD without transient ischemic attack (TIA) or stroke as this is the population approved for clinical use. Concomitant CAD was defined as any of the following at randomization: prior MI, unstable angina, coronary atherosclerosis with ⩾ 50% stenosis, and history of percutaneous coronary intervention or coronary artery bypass grafting. Prior peripheral revascularization was defined as prior endovascular or surgical revascularization for lower extremity ischemia.
End points
The primary efficacy and safety endpoints have been described previously.10,11 In the present analysis, the primary CV efficacy outcome was MACE, which was a composite of CV death, MI, or stroke. The primary limb efficacy outcome was MALE, which was a composite of hospitalization for ALI, urgent peripheral revascularization, or major amputation due to limb ischemia. All ALI outcomes were adjudicated by a Clinical Events Committee (CEC) blinded to treatment allocation using source documentation. Other pre-defined efficacy outcomes were all-cause death, and the additional endpoints of any peripheral revascularization, hospitalization for ALI or major amputation, and a composite endpoint of MACE or MALE.
The safety endpoint for this analysis was major bleeding, as defined by the International Society on Thrombosis and Haemostasis (ISTH) and was prospectively adjudicated by a blinded CEC. This outcome was chosen to align with other recent trials of antithrombotics in PAD and have not been previously reported. In addition, Global Use of Strategies to Open Occluded Coronary Arteries (GUSTO) classification, intracranial hemorrhage, fatal bleeding, or symptomatic bleeding into a critical organ are also reported. Net clinical outcome was defined as the composite of MACE, MALE, fatal bleeding, or symptomatic bleeding into a critical organ.
Statistical methods
Baseline characteristics of patients with versus without PAD and of patients with PAD and history of concomitant CAD versus PAD alone were compared using the chi-squared test for categorical variables and the Wilcoxon rank-sum test for continuous variables. The efficacy analysis was performed on an intention-to-treat basis. Hazard ratios (HRs) and 95% CIs were calculated using a Cox proportional-hazard model. Safety analyses were performed among all patients who received at least one dose of the study drug and included events through 60 days after premature discontinuation of study drug or 30 days after a final visit in the trial. To test for heterogeneity in the relative effect of vorapaxar in patients with PAD with CAD versus those with PAD alone and in patients with and without prior lower extremity revascularization, we employed the Cox model with a term for the interaction of interest. In addition, qualitative interaction tests for the absolute risk reductions (ARR) in the subgroups were analyzed using a one-sided Gail−Simon test. 12 ARR, absolute risk elevation (ARE), number needed to treat (NNT), and number needed to harm (NNH) to prevent or cause one event at 3 years were analyzed to compare the absolute benefit with vorapaxar across the examined subgroups. Analyses were performed using Stata version 14.2 (StataCorp LP, College Station, TX, USA) and SAS version 9.4 (SAS Institute Inc., Cary, NC, USA). Unless otherwise stated, all reported p-values are two-sided. A p-value of < 0.05 was considered statistically significant.
Results
Baseline characteristics
Of the 6136 patients with PAD in this analysis, 76.3% had known concomitant CAD at baseline and 39.6% had a history of peripheral revascularization. The baseline characteristics in patients with versus without PAD are shown in Table 1. The baseline characteristics of patients with PAD and CAD versus those with PAD alone are shown in online Supplemental Table S1. In this broad population of patients with PAD, approximately half (46.9%) had intermittent claudication and no history of revascularization, 39.6% had prior peripheral revascularization, and 2.9% had history of amputation for limb ischemia. Patients with PAD were older, more often female, and were more likely to have diabetes mellitus, hypertension, hyperlipidemia, renal dysfunction, and history of smoking.
Baseline characteristics of the overall population of patients with PAD versus those without history of PAD.
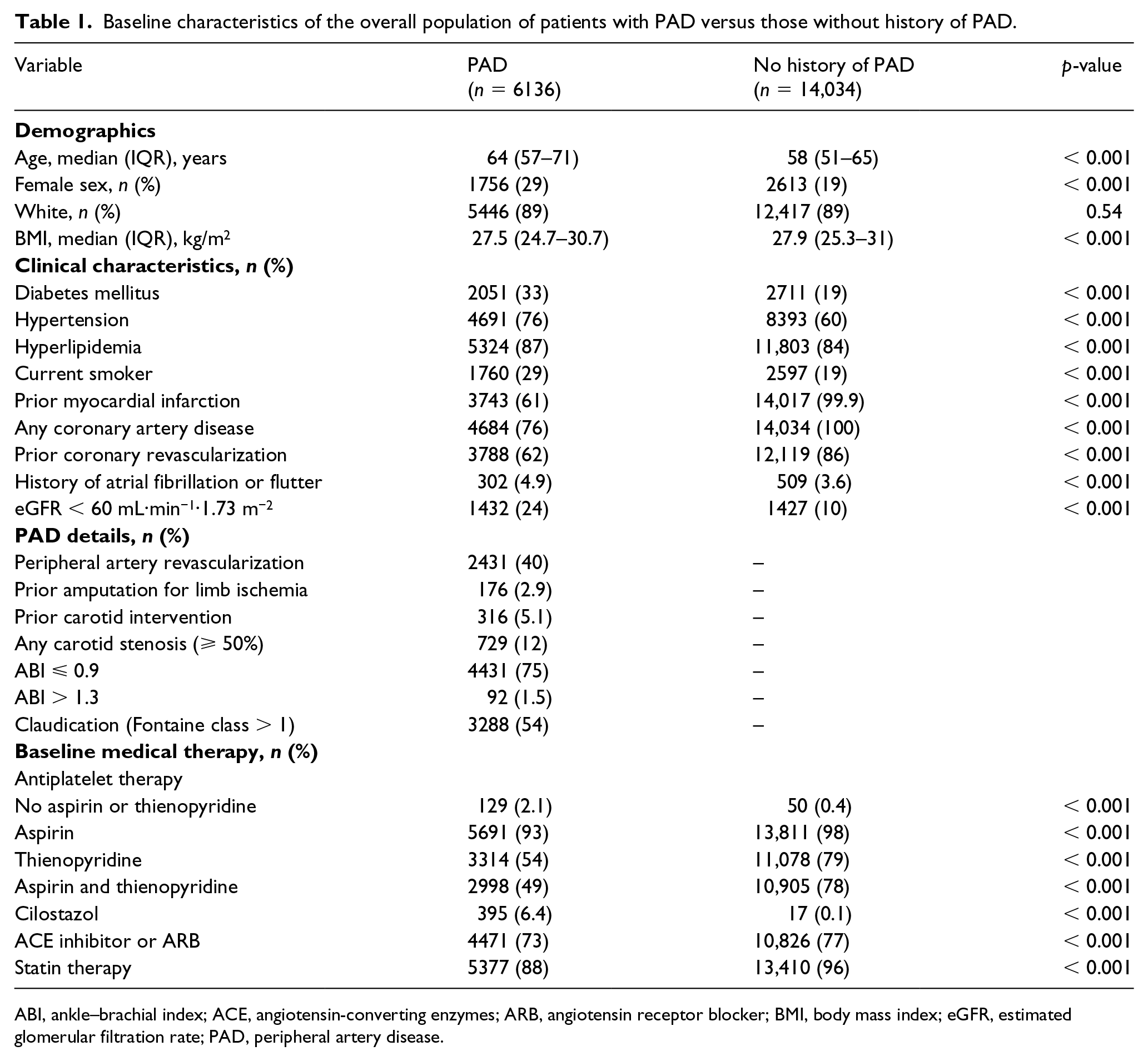
ABI, ankle–brachial index; ACE, angiotensin-converting enzymes; ARB, angiotensin receptor blocker; BMI, body mass index; eGFR, estimated glomerular filtration rate; PAD, peripheral artery disease.
Patients with PAD with CAD had a higher prevalence of diabetes mellitus, hypertension, and hyperlipidemia compared with patients with PAD alone. Since patients with PAD alone were enrolled on the basis of symptomatic PAD, they had an approximately twofold higher prevalence of prior lower extremity revascularizations relative to those with concomitant CAD (60.1% vs 32%). Evidence-based medical therapies were more frequently used in patients with concomitant CAD relative to those with PAD alone (online Supplemental Figure S1).
Risk based on concomitant CAD or peripheral revascularization
At 3 years, the rate of MACE in the placebo arm was approximately twofold higher in patients with PAD and CAD as compared with patients with PAD alone (12.8% vs 7.6%; Table 2). At 3 years, the rate of MALE in the placebo arm was more than twofold higher in patients with PAD alone as compared to patients with PAD and CAD (7.2% vs 3.1%; Table 2), with that difference driven by the greater proportion with prior peripheral revascularization. In patients with prior peripheral revascularization, the rate of MALE was more than fourfold higher than in patients with PAD and no prior peripheral revascularization (8.0% vs 2.1%; Supplemental Table S2, Supplemental Figure S2).
Efficacy and safety outcomes in patients with PAD with CAD and in those with PAD alone.
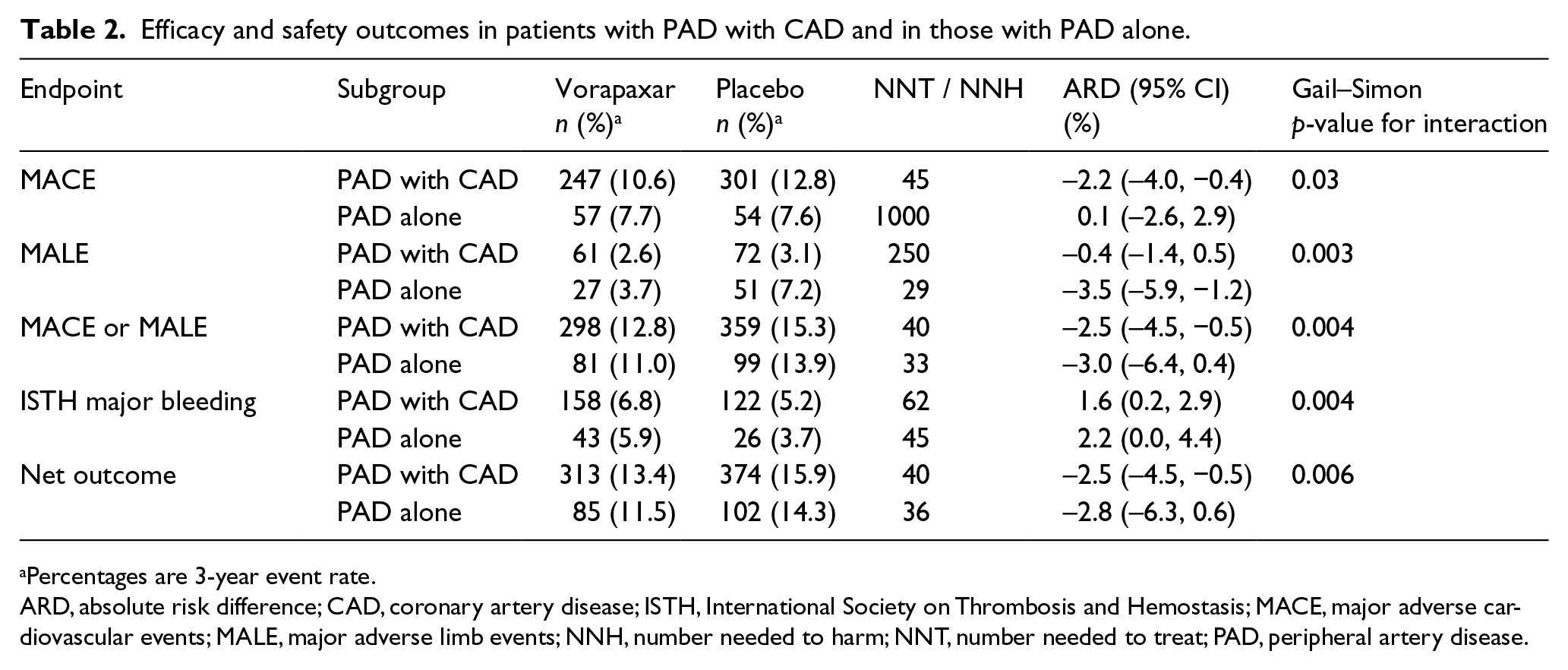
Percentages are 3-year event rate.
ARD, absolute risk difference; CAD, coronary artery disease; ISTH, International Society on Thrombosis and Hemostasis; MACE, major adverse cardiovascular events; MALE, major adverse limb events; NNH, number needed to harm; NNT, number needed to treat; PAD, peripheral artery disease.
Benefit of vorapaxar for MACE in PAD with and without CAD
At 3 years, vorapaxar as compared to placebo resulted in a significant reduction in MACE in the overall population of patients with PAD (9.9% vs 11.6%; HR 0.85, 95% CI 0.73, 0.99; p = 0.03) (Table 3; Supplemental Figure S3). There was apparent heterogeneity, however, on the basis of known concomitant coronary disease. Patients with PAD and CAD appeared to derive a greater absolute benefit with vorapaxar: 2.2% ARR in MACE translating into a NNT over 3 years of 45 (ARD −2.2%, 95% CI −4.0%, −0.4%) compared to those without known CAD (ARD 0.1%, 95% CI −2.6%, 2.9%, NNT > 1000) (Table 2; Figure 1, p-interaction for ARD = 0.03).
Efficacy endpoints in the overall population of patients with PAD.
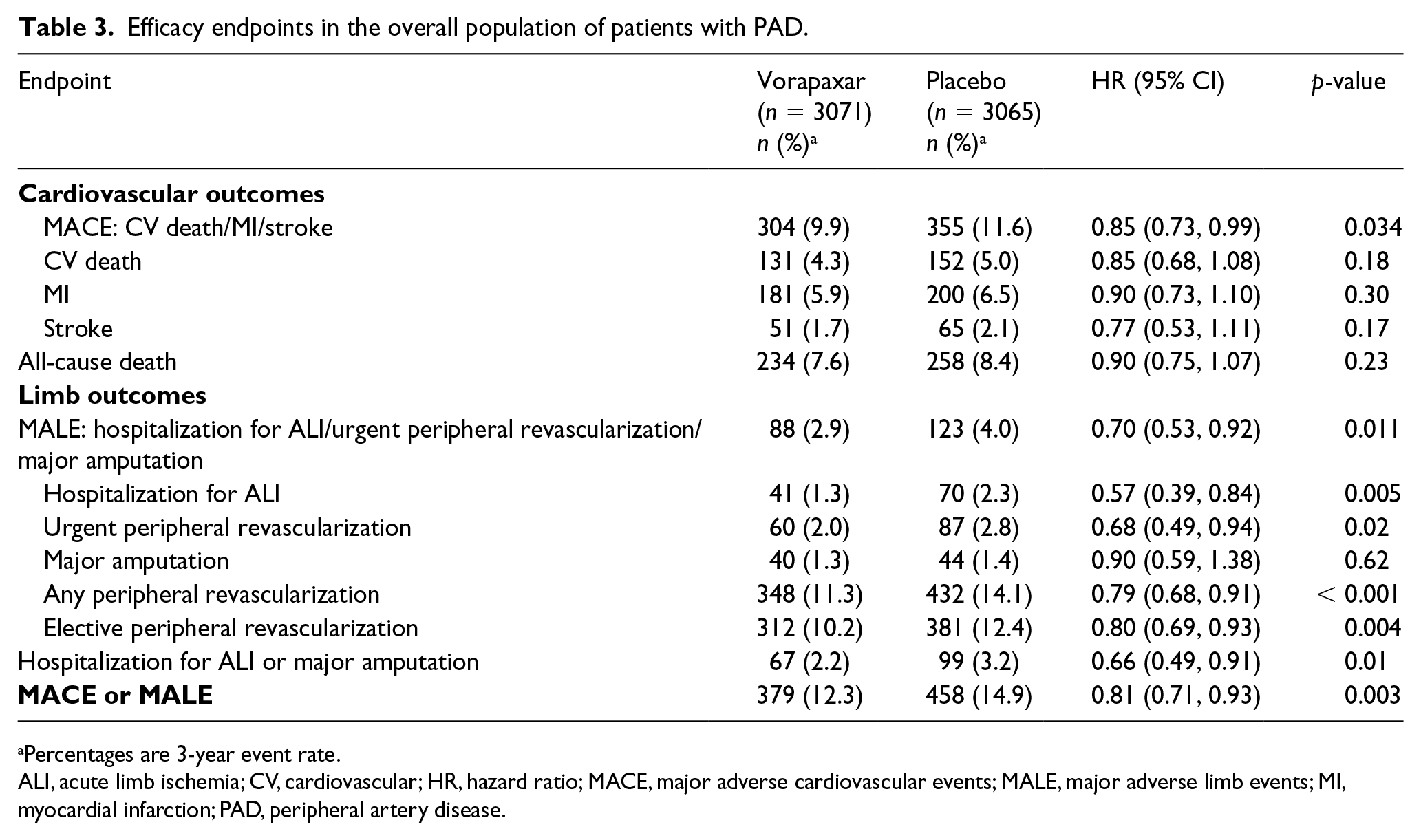
Percentages are 3-year event rate.
ALI, acute limb ischemia; CV, cardiovascular; HR, hazard ratio; MACE, major adverse cardiovascular events; MALE, major adverse limb events; MI, myocardial infarction; PAD, peripheral artery disease.
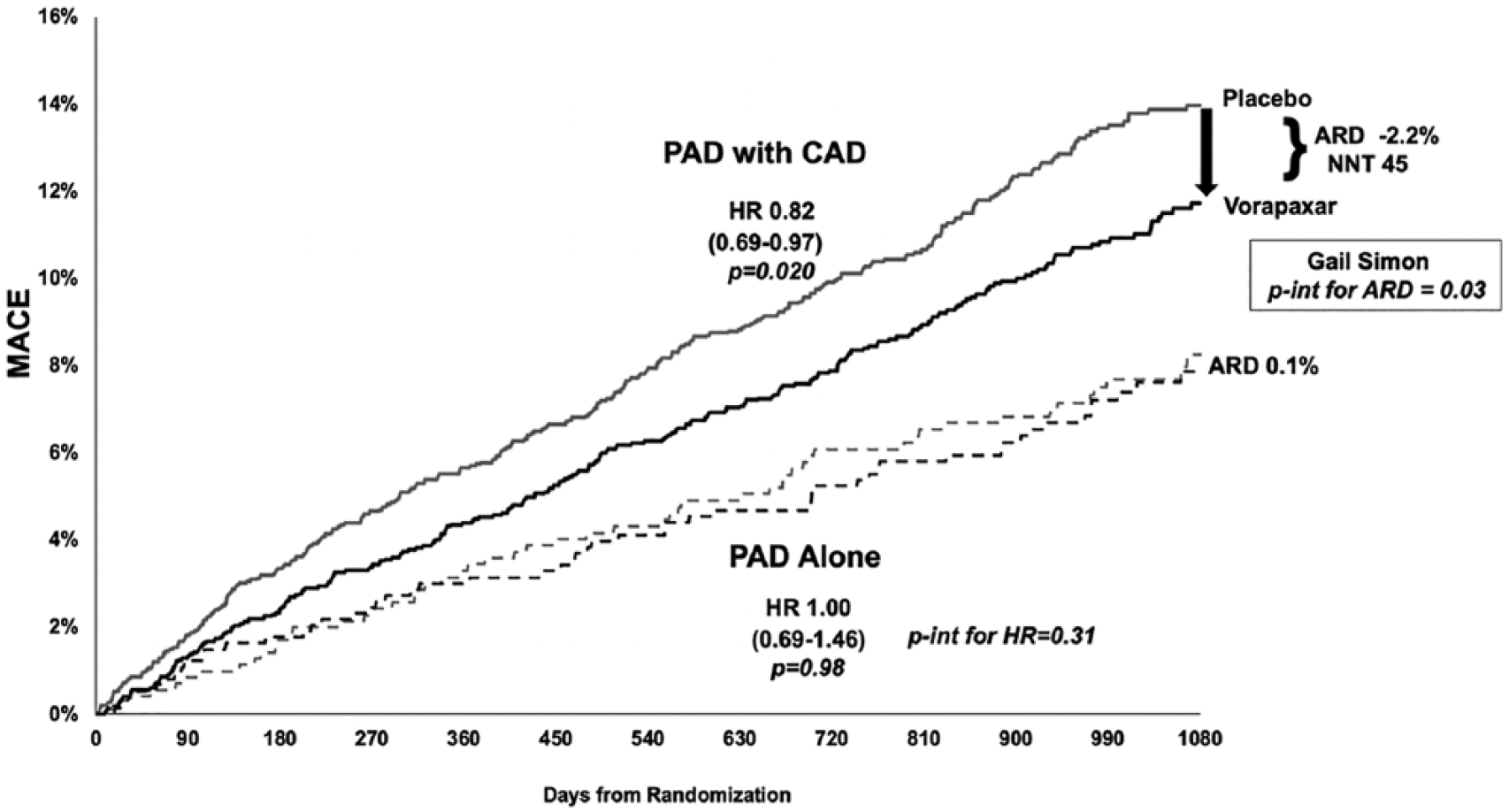
MACE reduction with vorapaxar over 3 years in patients with PAD with CAD and in those with PAD alone.
Effect of vorapaxar on MALE
At 3 years, vorapaxar reduced MALE in the overall population of patients with PAD (2.9% vs 4.0%; HR 0.70, 95% CI 0.53, 0.92; p = 0.011) (Table 3; Supplemental Figure S4) as well as the composites of ALI, urgent peripheral revascularization for limb ischemia, any peripheral revascularization, elective peripheral revascularizations for claudication, and the composite outcome of ALI or major amputation (Table 3; Supplemental Figures S5 and S6). The most important driver of MALE risk and benefit of vorapaxar in patients with PAD appeared to be the history of lower extremity revascularization (Figure 2, Supplemental Table S2, Supplemental Figure S2). When stratifying the overall population of patients with PAD by history of peripheral revascularization, the ARR in MALE was 2.5% (NNT 40) in patients with prior lower extremity revascularization versus 0.2% (NNT 500) in those without prior lower extremity revascularization (p-interaction for ARD = 0.02) (Figure 3; Supplemental Table S2).
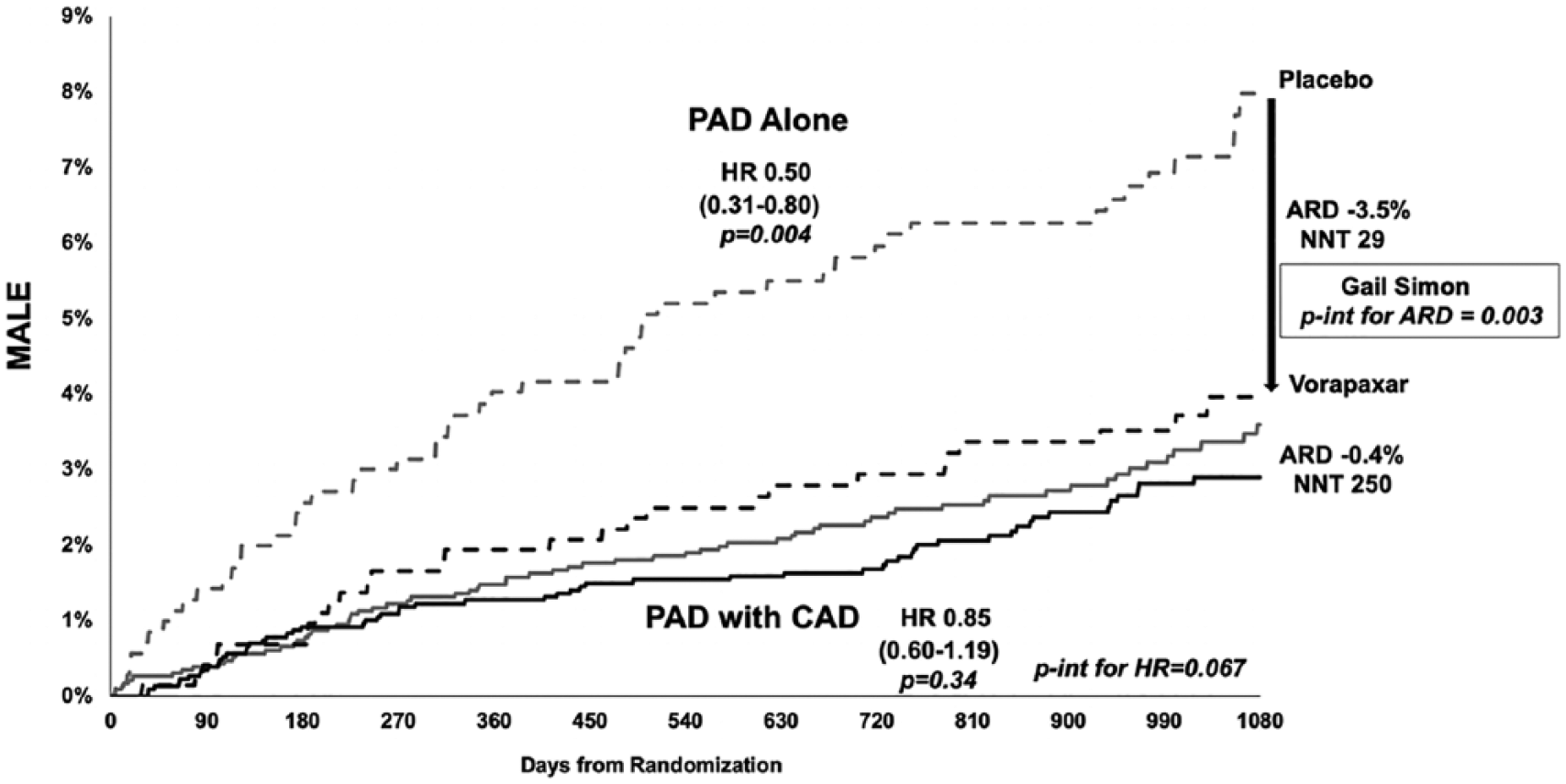
MALE reduction with vorapaxar over 3 years in patients with PAD with CAD and in those with PAD alone.
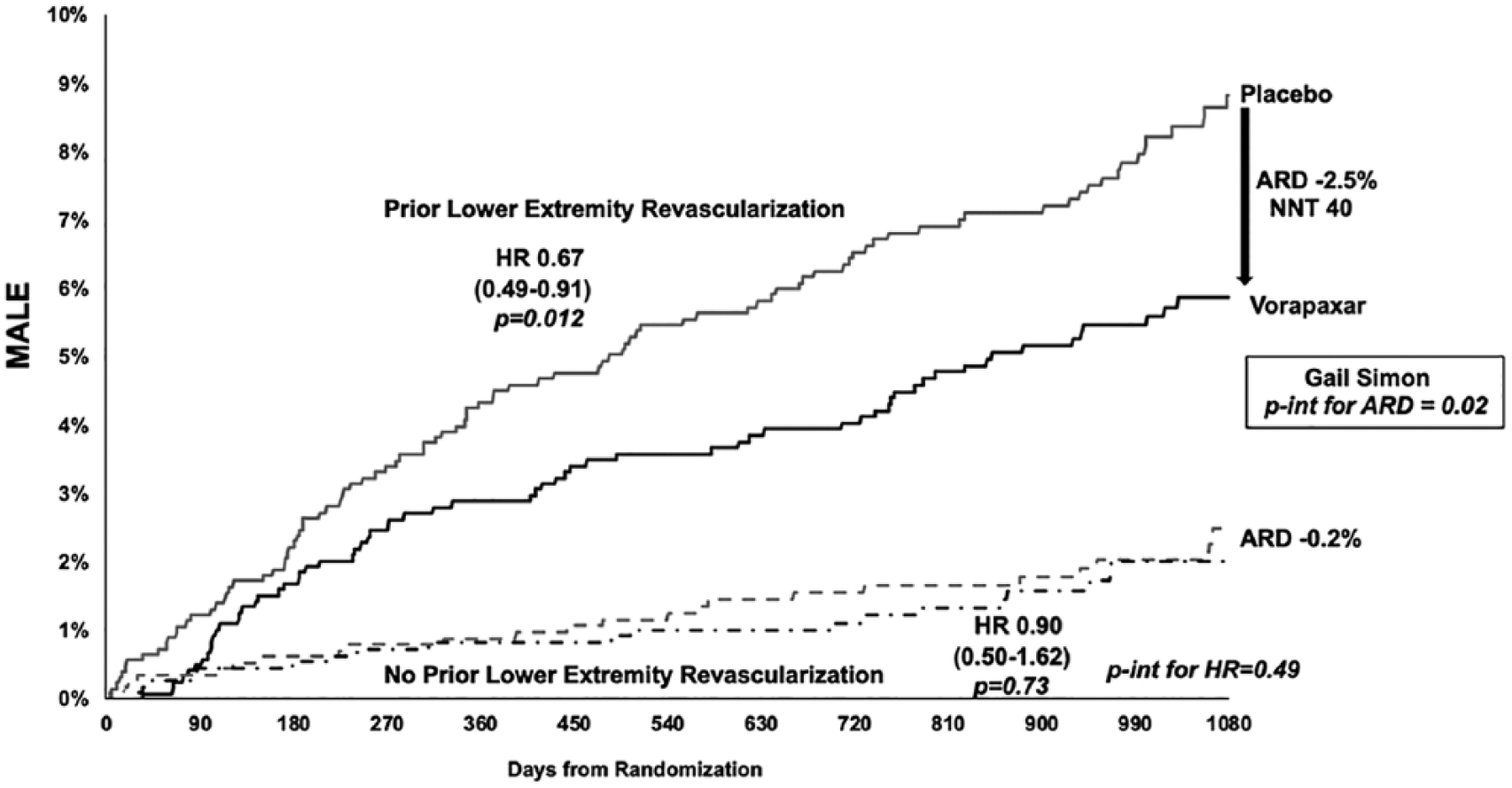
MALE reduction with vorapaxar over 3 years in overall patients with PAD with and without prior lower extremity revascularization.
Effect of vorapaxar on the composite of MACE or MALE
At 3 years, vorapaxar significantly reduced the composite of MACE or MALE versus placebo in the overall population with PAD (12.3% vs 14.9%; HR, 0.81, 95% CI 0.71, 0.93; p = 0.003) (Table 3, Supplemental Figure S7). Among patients with PAD and known CAD, vorapaxar resulted in a 2.5% ARR in the composite of MACE or MALE translating into an NNT of 40 (ARD −2.5%, 95% CI −4.5%, −0.5%). This was similar in magnitude to the benefit in those with PAD and no known CAD, where vorapaxar resulted in an ARR of 3.0% and NNT of 33 (ARD −3.0%, 95% CI −6.4%, 0.4%) (Table 2, Figure 4). In patients who had PAD but neither high-risk feature (concomitant CAD or concomitant peripheral revascularization), there was little apparent benefit of vorapaxar (ARD +1.1%).
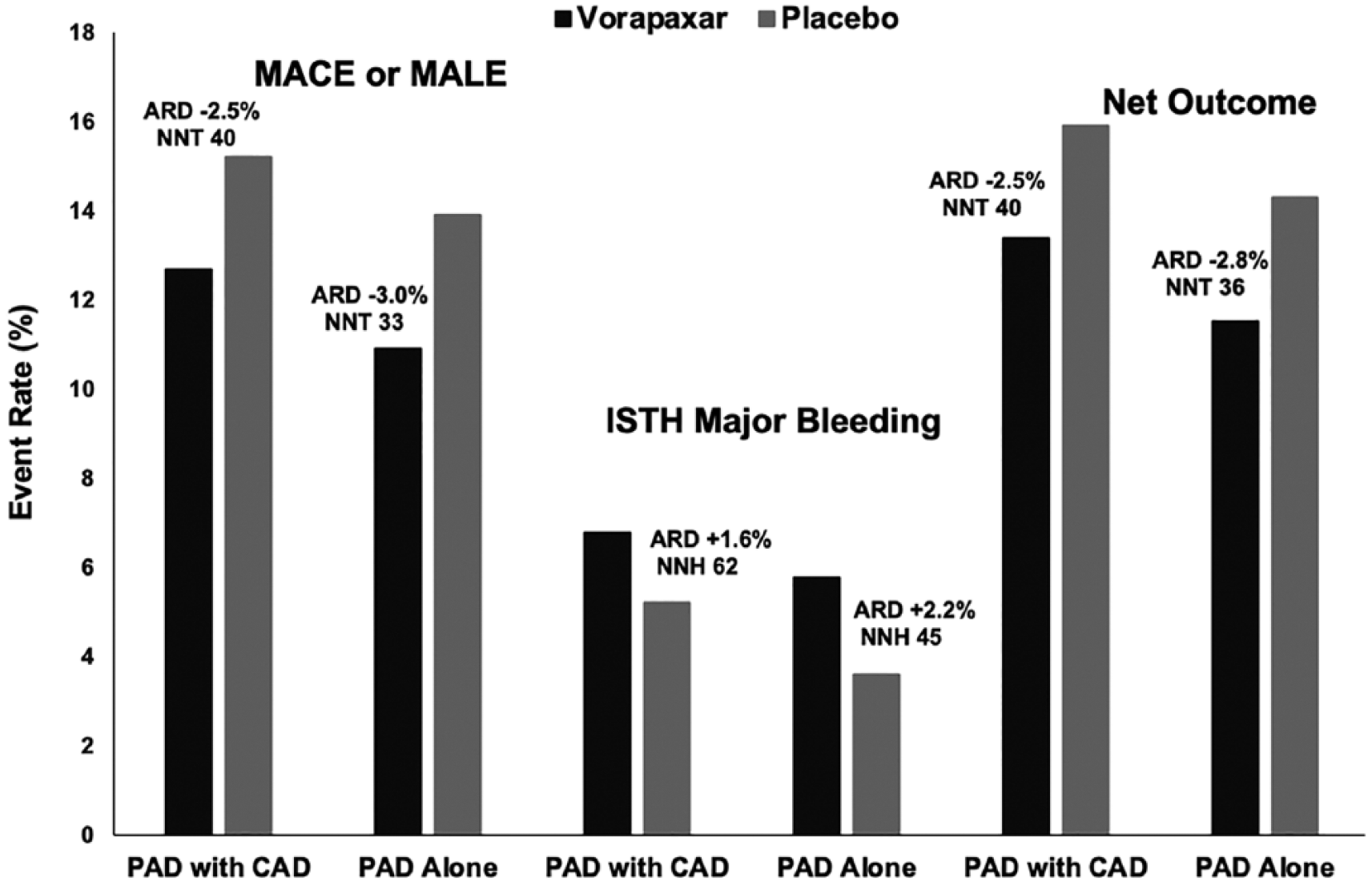
MACE or MALE, ISTH major bleeding, and net clinical outcomes in patients with PAD with CAD and in those with PAD alone.
Effect of vorapaxar on bleeding
At 3 years, vorapaxar significantly increased ISTH major bleeding by 39% (6.6% vs 4.9%; HR 1.39, 95% CI 1.12, 1.71; p = 0.003) (Table 4). There was no significant increase in fatal bleeding, intracranial hemorrhage or symptomatic bleeding into a critical organ with vorapaxar. There was no significant interaction between the risk of ISTH major bleeding with vorapaxar across major subgroups, including those on concurrent antiplatelet therapy with aspirin, thienopyridine or both (p-interaction for HR > 0.05 for each). Bleeding appeared lower in patients with PAD and CAD, where vorapaxar resulted in a 1.6% increase in ISTH major bleeding translating into an NNH of 62 (ARD 1.6%, 95% CI 0.2%, 2.9%) compared to an increase of 2.2% and NNH of 45 (ARD 2.2%, 95% CI 0.0%, 4.4%) in patients with PAD alone (p-interaction for ARD = 0.0045) (Table 2, Figure 4).
Bleeding endpoints and net clinical outcomes in a broad population of patients with PAD.
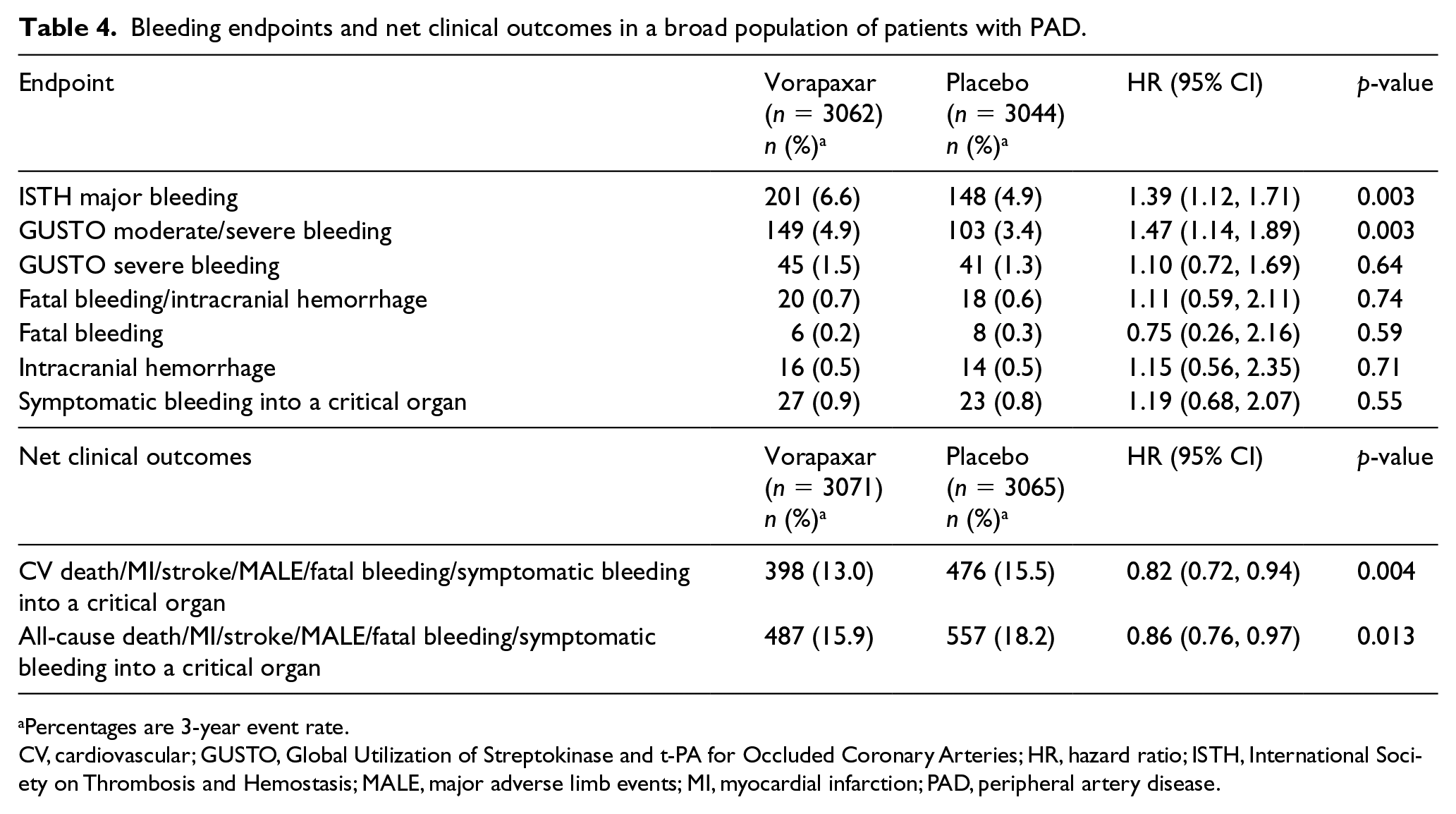
Percentages are 3-year event rate.
CV, cardiovascular; GUSTO, Global Utilization of Streptokinase and t-PA for Occluded Coronary Arteries; HR, hazard ratio; ISTH, International Society on Thrombosis and Hemostasis; MALE, major adverse limb events; MI, myocardial infarction; PAD, peripheral artery disease.
Effect of vorapaxar on the net clinical outcome
The net clinical outcome comprising of MACE, MALE, fatal bleeding, or symptomatic bleeding into a critical organ was reduced with vorapaxar in patients with PAD (13.0% vs 15.5%; HR 0.82, 95% CI 0.72, 0.94; p = 0.004) (Table 4, Supplemental Figure S8). Among patients with PAD and CAD, vorapaxar reduced the net outcome by 2.5%, translating into an NNT of 40 (ARD −2.5%, 95% CI −4.5%, −0.5%). There was a similar net benefit in those with PAD and no CAD, with an ARR of 2.8% and NNT of 36 (ARD −2.8%, 95% CI −6.3%, 0.6%) (Table 2, Figure 4). In patients with PAD and no high-risk feature (neither concomitant CAD nor prior peripheral revascularization), there was no apparent net benefit of vorapaxar (ARD 1.5%, 95% CI −3.7%, 6.7%).
Discussion
The current analysis describes risk of MACE and MALE as well as the efficacy and safety of intensive antithrombotic therapy in a broad cohort of well-characterized patients with PAD enrolled in a randomized trial. Similar to other studies, it was observed that the clinical characteristics of concomitant CAD and prior peripheral revascularization were associated with higher risk of MACE and MALE, respectively. A novel observation in this study, however, is the significant heterogeneity in the magnitude of benefit of more potent antithrombotic therapy in those with and without these characteristics. A key related finding is that there was no apparent benefit in the subgroup of patients with symptomatic PAD but no concomitant coronary disease or history of peripheral revascularization.
The overall effects of vorapaxar on MACE and MALE in this population are consistent with those described in previous reports of patients with PAD in TRA 2°P-TIMI 50 and FDA approval of vorapaxar for this population.4,5,13 These benefits, however, come at the cost of increased bleeding; this observation is noted in current clinical guidelines stating a IIb recommendation and noting that the net benefit for patients with PAD overall is uncertain. 14 Therefore, the ability to risk stratify and identify subgroups of patients that derive greater benefit is of clinical utility.
Registries and recent clinical trials have described polyvascular disease, the combination of multiple symptomatic vascular territories, as a potent marker of MACE risk. 15 The observation of higher risk in those with PAD and CAD versus PAD alone in this analysis supports those observations. The current analysis, however, expands on this observation by evaluating the absolute benefits of vorapaxar in patients with PAD based on this characteristic. It was found that those with PAD and CAD had a significantly greater magnitude of benefit for MACE reduction relative to those with PAD alone. Similarly, prior peripheral revascularization has been identified as a potent and independent predictor of MALE in patients with PAD in several datasets. This risk is observed in the current analysis as well, with the novel finding now that the magnitude of benefit for MALE with vorapaxar was significantly different based on this characteristic. In patients with PAD with either of these characteristics, there was a net benefit of vorapaxar.
An important associated observation is that there may be a subgroup of patients with PAD that do not benefit from more potent antithrombotic therapy. In the current analysis, patients with symptomatic PAD but with no known CAD and no history of peripheral revascularization, had no apparent benefit with vorapaxar but did have bleeding risk. The notion that some patients with PAD may not derive benefit from intensive antithrombotic therapy may help clinicians in personalizing therapy in PAD.
Our findings should be viewed in the context of other studies of antithrombotic therapy in patients with PAD. Antiplatelet monotherapy is the standard of care in PAD.14,16 More potent antiplatelet strategies, however, have shown mixed results. Ticagrelor, a potent P2Y12, was not superior as monotherapy to clopidogrel in the Examining Use of Ticagrelor in Peripheral Artery Disease (EUCLID) trial for reducing MACE; 7 however, only ~30% of patients in the EUCLID trial had CAD. The subgroup of patients with prior PCI or CABG showed a statistically significant heterogeneity (p for interaction < 0.05), suggesting ticagrelor may be more beneficial than clopidogrel in those with concomitant CAD or polyvascular disease. This concept was further supported in the Prevention of Cardiovascular Events in Patients With Prior Heart Attack Using Ticagrelor Compared to Placebo on a Background of Aspirin—Thrombolysis In Myocardial Infarction 54 (PEGASUS-TIMI 54) trial, 8 in which relative to those with CAD alone (ARR of 1.0%, NNT 100), patients with PAD and concomitant CAD had higher atherothrombotic risk as well as greater reductions in MACE (ARR of 5.2%, NNT 19 with ticagrelor 60 mg twice daily) and a significant reduction in mortality (ARR 5.8%, p = 0.007). Furthermore, ticagrelor reduced MALE in patients with PAD by 35% (p = 0.026). 8 This observation was further confirmed in the Prolonging Dual-Antiplatelet Treatment After Grading Stent-Induced Intimal Hyperplasia Study (PRODIGY), 9 which showed greater benefits for dual antiplatelet therapy (DAPT) in PAD and CAD versus CAD alone. Taken together, these data suggest that symptomatic coronary disease may be an important identifier of patients who will benefit from more potent antiplatelet therapy.
Although different in design and with a different therapeutic mechanism than that in TRA 2°P-TIMI 50, PEGASUS-TIMI 54, and PRODIGY, the recently reported COMPASS trial demonstrated the benefit of dual-pathway therapy with aspirin and rivaroxaban in patients with CAD and PAD. 6 As part of the inclusion, this trial was enriched for polyvascular disease and revascularization with approximately 65% of patients in the PAD subgroup having concomitant CAD. 6 This stands in contrast to < 30% in the EUCLID trial. Based on the current observation, it is possible that the COMPASS population, enriched for polyvascular disease, is particularly responsive to more potent antithrombotic therapy.
Limitations
This analysis should be interpreted in the context of several limitations. First, this was an exploratory analysis within a randomized trial. Second, as vorapaxar is not approved for use in patients with a history of stroke due to an increased risk of intracranial bleeding, we excluded these patients from our analysis. Thus, our findings are not generalizable to patients with PAD who have a history of stroke. Finally, although permitted by the protocol, very few patients received potent P2Y12 inhibitors, ticagrelor, and prasugrel; thus, the efficacy and safety of vorapaxar in combination with these agents cannot be determined from our analysis.
Conclusions
In patients with PAD, vorapaxar added to aspirin and/or clopidogrel significantly reduces the rate of MACE or MALE and increases ISTH bleeding but not fatal bleeding or intracranial hemorrhage, resulting in overall positive net outcomes. The risk of MACE and MALE in patients with PAD was greater in patients with concomitant CAD and peripheral revascularization, respectively. Patients with either of these characteristics appeared to derive a net benefit from the addition of vorapaxar, while those with neither did not appear to benefit. These clinical characteristics may be useful in a selection of patients who will benefit from more potent antithrombotic therapies.
Clinical implications
Among patients with PAD, those with concomitant CAD or prior peripheral revascularization are at an increased risk of thrombotic cardiovascular and limb events, respectively. Because of their elevated risk, these patients may derive more clinical benefit from potent antithrombotic therapies.
Supplemental Material
10.1177_1358863X19892690_Supplementary_tables_and_figures – Supplemental material for Effect of vorapaxar on cardiovascular and limb outcomes in patients with peripheral artery disease with and without coronary artery disease: Analysis from the TRA 2°P-TIMI 50 trial
Supplemental material, 10.1177_1358863X19892690_Supplementary_tables_and_figures for Effect of vorapaxar on cardiovascular and limb outcomes in patients with peripheral artery disease with and without coronary artery disease: Analysis from the TRA 2°P-TIMI 50 trial by Arman Qamar, David A Morrow, Mark A Creager, Benjamin M Scirica, Jeffrey W Olin, Joshua A Beckman, Sabina A Murphy and Marc P Bonaca in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Qamar is supported by the NHLBI postdoctoral training grant T32 HL007604. Dr Morrow has received support through research grants to Brigham and Women’s Hospital from Abbott Laboratories, Amgen, AstraZeneca, Eisai Co., GlaxoSmithKline, Medicines Company, Merck & Co., Inc., Novartis, Pfizer, Roche Diagnostics, and Takeda Pharmaceutical Co.; and consulting fees from Abbott Laboratories, Aralez Pharmaceuticals, Bayer, Merck & Co., Inc., Peloton, Roche Diagnostics, and Verseon Corp. Dr Creager reports no relationships relevant to the content of this paper to disclose. Dr Scirica has received institutional grant support to Brigham and Women’s Hospital from AstraZeneca, Poxel, Gilead, Eisai Co., and Novartis; and personal fees from Lexicon, AstraZeneca, St Jude’s Medical, Biogen Idec, Elsevier Practice Update Cardiology, Reddy’s Laboratory, Boehringer Ingelheim (BI), Covance, GlaxoSmithKline, Merck, NovoNordisk, Sanofi, Biogen Idec, Eisai Co., and Health [at] Scale. Dr Olin reports no relationships relevant to the content of this paper to disclose. Dr Beckman has served as a consultant to Abbott Vascular; and is a board member of VIVA Physicians group. Ms Murphy reports receiving grants from Abbott Laboratories, Amarin, Amgen, AstraZeneca, Critical Diagnostics, Daiichi Sankyo, Eisai Co., GlaxoSmithKline, Intarcia Therapeutics, Merck & Co., Roche Diagnostics, Takeda, Gilead, Poxel, Novartis, MedImmune, Janssen Research Development, and Genzyme. Dr Bonaca has received institutional grant support to Brigham and Women’s Hospital from AstraZeneca, MedImmune, Merck, and Pfizer; and consulting fees from Aralez, AstraZeneca, Bayer, Janssen, Johnson & Johnson, and Merck.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This TRA 2°P-TIMI 50 trial was supported by a grant from Merck & Co. Dr Qamar and Dr Bonaca are supported by the American Heart Association Strategically Focused Research Network in Vascular Disease under award numbers 18SFRN3390085 (BWH-DH SFRN Center) and 18SFRN33960262 (BWH-DH Clinical Project). The content is solely the responsibility of the authors and does not necessarily represent the official views of the American Heart Association.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
