Abstract
The burden and predictors of hospitalization over time in community-based patients with peripheral artery disease (PAD) have not been established. This study evaluates the frequency, reasons and predictors of hospitalization over time in community-based patients with PAD. We assembled an inception cohort of 1798 PAD cases from Olmsted County, MN, USA (mean age 71.2 years, 44% female) from 1 January 1998 through 31 December 2011 who were followed until 2014. Two age- and sex-matched controls (n = 3596) were identified for each case. ICD-9 codes were used to ascertain the primary reasons for hospitalization. Patients were censored at death or last follow-up. The most frequent reasons for hospitalization were non-cardiovascular: 68% of 8706 hospitalizations in cases and 78% of 8005 hospitalizations in controls. A total of 1533 (85%) cases and 2286 (64%) controls (p < 0.001) were hospitalized at least once; 1262 (70%) cases and 1588 (44%) controls (p < 0.001) ≥ two times. In adjusted models, age, prior hospitalization and comorbid conditions were independently associated with increased risk of recurrent hospitalizations in both groups. In cases, severe PAD (ankle–brachial index < 0.5) (HR: 1.25; 95% CI: 1.15, 1.36) and poorly compressible arteries (HR: 1.26; 95% CI: 1.16, 1.38) were each associated with increased risk for recurrent hospitalization. We demonstrate an increased rate of hospitalization in community-based patients with PAD and identify predictors of recurrent hospitalizations. These observations may inform strategies to reduce the burden of hospitalization of PAD patients.
Introduction
Lower extremity peripheral artery disease (PAD) affects 8–10 million individuals in the United States, making it a major public health problem1–3 with attendant high costs 4 attributed primarily to hospitalization. 5 Ascertaining the burden and risk factors for hospitalization is a necessary first step to formulate practice policies that may reduce the clinical and economic consequences of PAD.
The high cost of PAD has been previously demonstrated in a Medicare population 6 as well as among participants of the Reduction of Atherothrombosis for Continued Health (REACH) registry. 7 The latter study highlighted the high cost of hospitalization for cardiovascular indications in PAD patients, 7 although hospitalizations for non-cardiovascular reasons or recurrent hospitalizations were not evaluated. In other studies evaluating hospitalizations, the study groups were restricted to PAD patients who were hospitalized at study entry for lower extremity bypass surgery 8 or critical limb ischemia.9,10 Additionally, in these prior studies, length of follow-up was limited, ranging from 30 days to a maximum of 6 months.8,10 Hence, a knowledge gap exists in regard to rates, reasons and predictors for all-cause hospitalizations in PAD patients from the community setting. To address these knowledge gaps, we identified PAD cases as well as matched controls from a community setting and evaluated the frequency, reasons and predictors for recurrent hospitalizations over time.
Methods
Setting
We used the resources of the Rochester Epidemiology Project (REP) to assemble an observational PAD case–control cohort in Olmsted County, Minnesota, from 1 January 1998 through 31 December 2011. Health care institutions participating in the REP include Mayo Clinic and the Mayo Clinic Hospitals (St Marys and Rochester Methodist hospitals), and Olmsted Medical Center and its affiliated hospital. 11 The REP matches medical records of participating institutions to specific individuals and enables linked health records to be retrieved.11,12 This study was approved by the institutional review boards of the participating institutions and patients agreed to participate.
We excluded all persons with non-atherosclerotic vascular disease such as vasculitis, Buerger’s disease, trauma and arterial embolism. We also excluded incarcerated individuals and persons who refused or did not provide authorization for use of their medical records in research. After completion of training, two independent abstractors ascertained PAD case status in a random sample of 20 persons with agreement in all but one case. Both abstractors were blinded to PAD status.
PAD cases and controls
PAD cases were ascertained based on ICD-9 (International Classification of Diseases, Ninth Revision) codes as well as vascular laboratory criteria. We used REP resources to identify persons with at least one PAD-related billing code (n = 11,782) (Figure 1) and applied our previously validated billing code algorithm to this dataset. The code-based algorithm 13 assigns an integer score to each PAD-related billing code, and the score for each person is the sum of the integer scores for PAD-related codes. Based on previously demonstrated discriminatory performance, persons with an algorithm score ≥ 8 were considered potential PAD cases. 13 In addition, the following established vascular laboratory criteria were used to define the presence of PAD: ankle–brachial index (ABI) ≤ 0.9 at rest or 1 minute after exercise or presence of poorly compressible arteries (PCA; based on an ABI ≥ 1.40 or ankle blood pressure ≥ 255 mmHg).4,14,15 Severe PAD was defined as a resting ABI < 0.5. 15
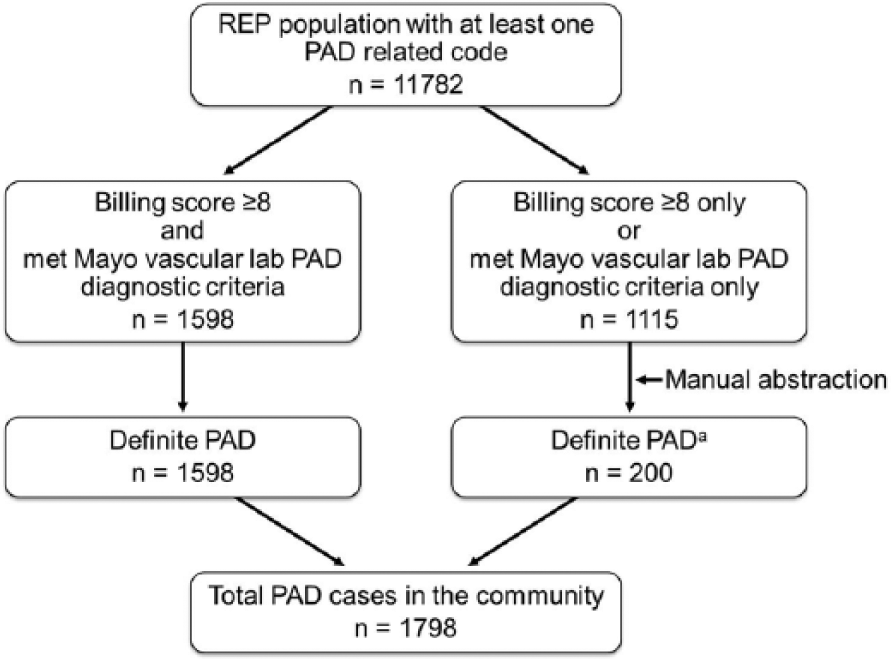
Ascertainment of PAD cases in Olmsted County, Minnesota, USA.
As shown in Figure 1, 1598 patients had a billing score ≥ 8 and also met Mayo Clinic vascular laboratory diagnostic criteria for PAD. For validation, trained abstractors blinded to PAD status reviewed a random sample of 20 medical records of Olmsted residents from this category and confirmed all as definite PAD cases.
Subsets of patients had either a billing score ≥ 8 or met Mayo Clinic vascular laboratory diagnostic criteria. The medical records of persons in these subsets (n = 1115) were manually reviewed to ascertain PAD status. The manual chart abstraction confirmed PAD in an additional 200 cases, yielding a total cohort of 1798 definite PAD cases (Figure 1).
Using the Olmsted County census data for all county residents, two controls without PAD were randomly identified for each PAD case. Cases (n = 1798) and controls (n = 3596) were matched by birth year and sex. Controls were residents of Olmsted County at the index diagnosis of the PAD case. Controls were identified as subjects without PAD-related codes or a billing score = 0. 13 The abstractors reviewed random samples of 20 medical records from each of these two groups and confirmed all as controls without PAD.
Risk factors, comorbidities, and medications
Risk factors for atherosclerosis (hypertension, hyperlipidemia and current or prior smoking) were ascertained from electronic health records using previously validated electronic algorithms. 16 Previously validated ICD-9 electronic algorithms 17 were applied to ascertain individual comorbidities, which were identified prior to or at the date of PAD diagnosis (for cases) or index date (for controls) and are listed in Table 1.
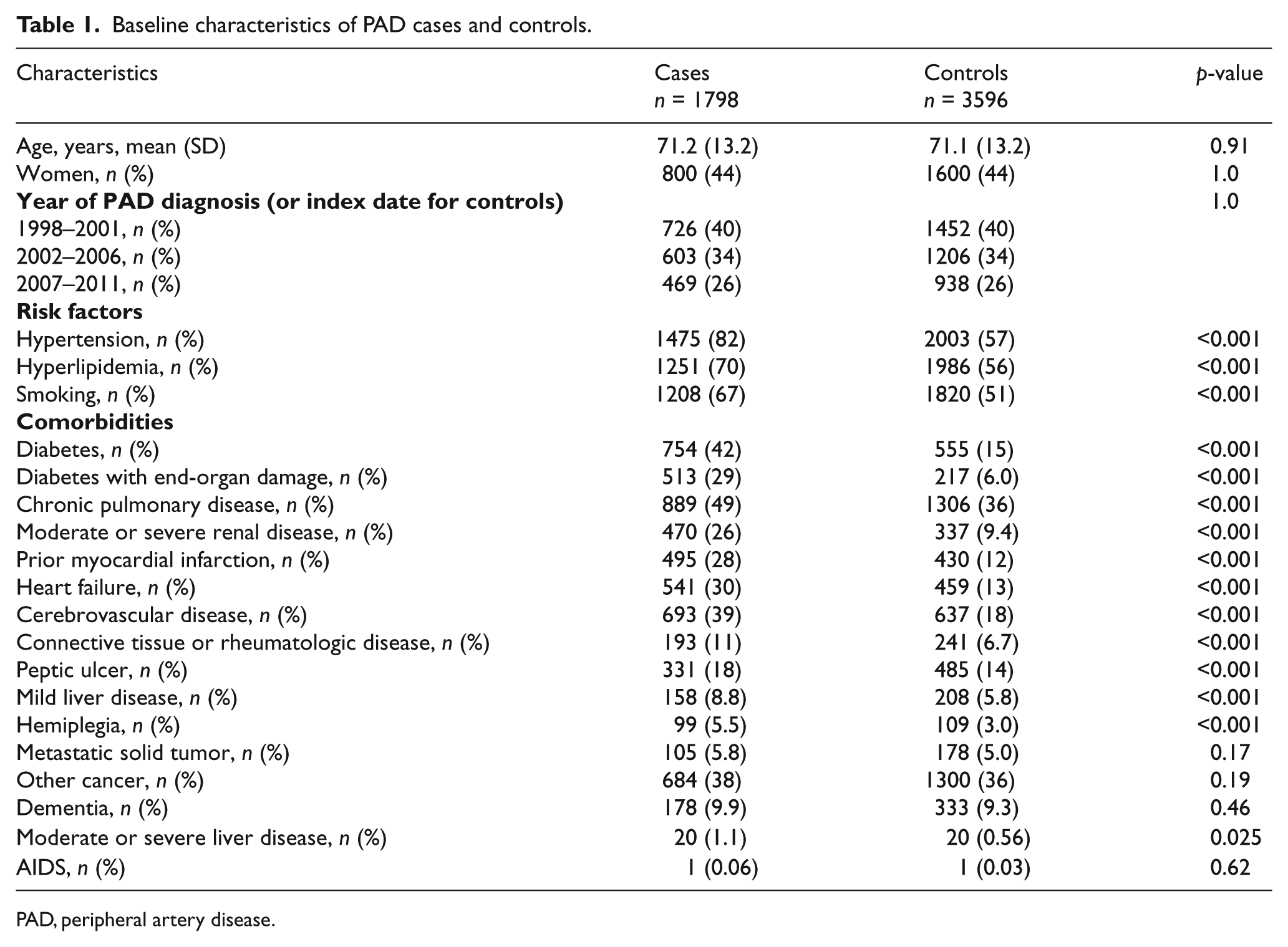
Baseline characteristics of PAD cases and controls.
PAD, peripheral artery disease.
Medications (aspirin, clopidogrel, statins, angiotensin converting enzyme inhibitors (ACE), angiotensin receptor blockers (ARBs) and beta-blockers) administered within 6 months of study entry were ascertained electronically or by manual abstraction. Electronic algorithms for abstraction of medications 16 were applied after 2006, when an electronic system for medication summary became available. Records of medications used prior to 2006 were ascertained by manual chart abstraction. Because controls did not require a clinical encounter to participate in the study, an active medication list within 6 months of the index date was not available for most controls.
Use of guideline-recommended strategies for secondary prevention in PAD
PAD cases were classified according to the number of guideline-recommended strategies they were using within 6 months of PAD diagnosis.2,4,18 These strategies included statin therapy, antiplatelet therapy (aspirin or clopidogrel), ACE or ARB therapy, and smoking cessation.2,4,18
Frequency and rates for hospitalizations
All-cause hospitalizations occurring after PAD diagnosis from 1998 until 2014 were ascertained through the Olmsted County Healthcare Expenditure and Utilization Database, which is a resource of the REP and contains information regarding Olmsted County hospitalizations since 1987. 19 For patients hospitalized at initial PAD diagnosis, only subsequent hospitalizations were analyzed. The primary diagnosis for each hospitalization was assessed using ICD-9 codes. These codes were assigned by trained personnel after hospital dismissal and reflect the main reasons for each hospital admission. ICD-9 codes were not available for 52 (0.31%) hospitalizations, yielding 16,711 hospitalizations for analysis.
All-cause death
The REP captures death information through multiple sources11,12 including electronic Minnesota state death certificates. The REP supplements these data with information obtained quarterly from the national death index. 12 At the end of follow-up, 965 (54%) cases and 1179 (33%) controls had died (p <0.001).
Critical limb ischemia
Patients with ICD-9 codes for atherosclerosis with rest pain (440.22), ulceration (440.23) or gangrene (440.24) were assigned to the critical limb ischemia (CLI) subgroup. 20
Statistical analysis
Baseline characteristics are presented as percentages, mean ± standard deviation (SD) or as median (25th, 75th percentile). Differences in baseline characteristics by PAD status were tested using the chi-squared test for categorical variables or a two-sample t-test for continuous variables. The rate of hospitalization was examined by PAD status and year of PAD diagnosis (or index date for controls) and was calculated as a ratio of total hospitalizations to person-years at risk. Differences in rates were tested by Poisson regression. Trends in the length of stay according to year and differences by PAD status were analyzed by linear regression models and, where appropriate, the chi-squared test.
To identify risk factors for hospitalizations, univariate and multivariable analyses using Andersen–Gill models were conducted. These models account for recurrent events and were used to analyze the recurrent hospitalizations.21,22 Results were summarized by hazard ratio (HR) and 95% confidence intervals (CI). Patients were censored at death or last follow-up. Univariate relationships between the risk of hospitalization and each variable under consideration were assessed first. Candidate variables included in the univariate models for cases and controls were age, sex, prior hospitalization, hypertension, smoking, diabetes, prior myocardial infarction (MI), heart failure (HF), cerebrovascular disease, and pulmonary disease. Variables with univariate p < 0.2 were considered as candidate variables for inclusion in the multivariable models. Forward selection was then used to choose independent variables by multivariable analysis. The proportional hazards assumption was examined visually by plotting residuals versus time. The functional form of continuous covariates was examined by plotting estimated hazard ratio (HR) versus variable values. No strong deviations from assumptions were noted, with the exception of a plateau in the risk when the number of hospitalizations was > 35 (<1% of total observations). Hence, subjects were censored at 35 hospitalizations. Kaplan–Meier analyses evaluating time to first hospitalization were conducted to assess the association of hospitalization with CLI, number of guideline-recommended strategies, and the setting of PAD diagnosis. Analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA) and R software, version 3.0.2 (R Foundation for Statistical Computing, Vienna, Austria). A p-value < 0.05 was considered significant.
Results
Patient characteristics
Baseline clinical characteristics of 1798 cases with PAD and 3596 matched controls are summarized in Table 1. Owing to matching, cases and controls had similar distributions of age, sex, and date of study entry. Cases were more likely to have risk factors for atherosclerosis and more comorbid conditions than controls (Table 1 and Supplemental Figure 1). Although cases were more likely to have individual comorbidities, four comorbidities (metastatic solid tumor, other cancer, dementia, and AIDS) had similar distribution in cases and controls (Table 1). Non-invasive evaluation of the lower extremities revealed severe PAD in 324 cases (18%) and PCA in 395 (22%).
Cases
Frequency and rates for hospitalization
During a median follow-up of 5.6 years (25th, 75th percentile: 2.2, 9.6 years), 8706 hospitalizations occurred in 1798 PAD cases and 8005 in 3596 controls (p < 0.001). A total of 265 (15%) PAD cases and 1310 (36%) controls were never hospitalized. For cases, the number of hospitalizations after PAD diagnosis ranged from 0 to 60 (median = 3) per person. The number of hospitalizations for controls ranged from 0 to 38 (median = 1) per person. A total of 1533 (85%) cases and 2286 (64%) controls (p < 0.001) were hospitalized at least once; 1262 (70%) cases and 1588 (44%) controls (p < 0.001) were hospitalized ≥ two times; 1050 (58%) cases and 1107 (31%) controls (p < 0.001) ≥ three times; and 840 (47%) cases and 777 (22%) controls (p < 0.001) ≥ four times.
The rate of hospitalization in cases (75.5 per 100 person-years; 95% CI: 73.9, 77.1) was significantly higher than in controls (32.9 per 100 person-years; 95% CI: 32.2, 33.7; p < 0.0001). The median length of hospitalization was similar for cases (median 6 days; 25th, 75th percentile: 3, 11 days) and controls (median 5 days; 25th, 75th percentile: 2, 9 days) (p = 0.27). The length of hospital stay remained stable over the study period for cases and controls.
Use of guideline-recommended strategies for secondary prevention in PAD
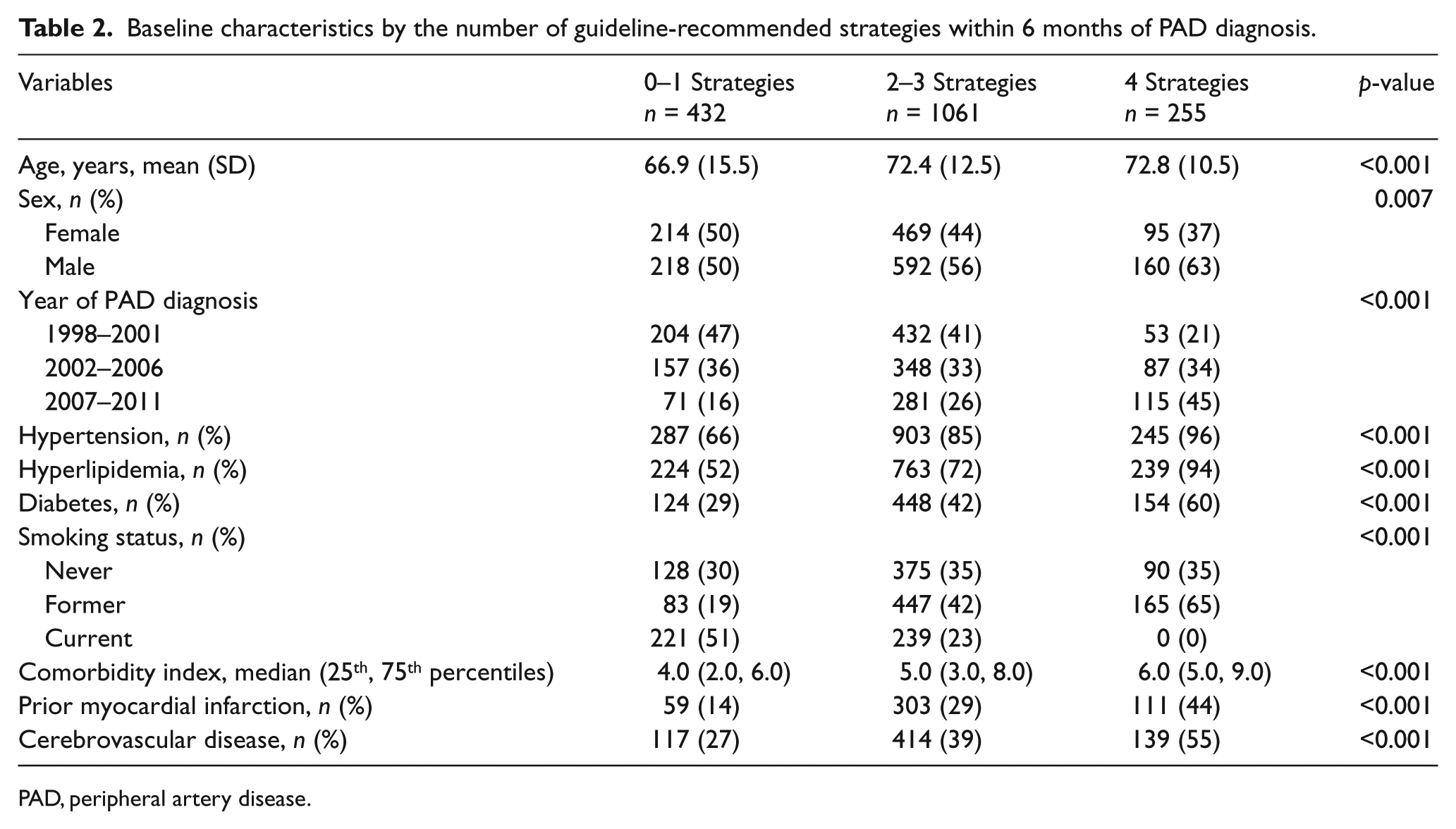
Among cases, 1073 (61%) were on antiplatelet agents (aspirin or clopidogrel), 826 (47%) were on statins, 825(47%) on beta-blockers and 819 (47%) on ACE/ARBs. Although in the overall group there were low proportions of patients using individual guideline-recommended strategies, over the study period there was an increase of proportions of patients on four guideline-recommended strategies and a corresponding decline in proportions of patients on zero to one or two to three strategies (Table 2). In addition, the distributions of older age, proportions of patients with risk factors, and the comorbidity indexes all increased with the higher number of guideline-recommended strategies (Table 2). The proportions of patients with prior MI and cerebrovascular disease also increased (Table 2). As shown in the Kaplan–Meier curve (Figure 2), the higher number of guideline-recommended strategies within 6 months of PAD diagnosis was associated with a higher number of incident hospitalizations.
Baseline characteristics by the number of guideline-recommended strategies within 6 months of PAD diagnosis.
PAD, peripheral artery disease.
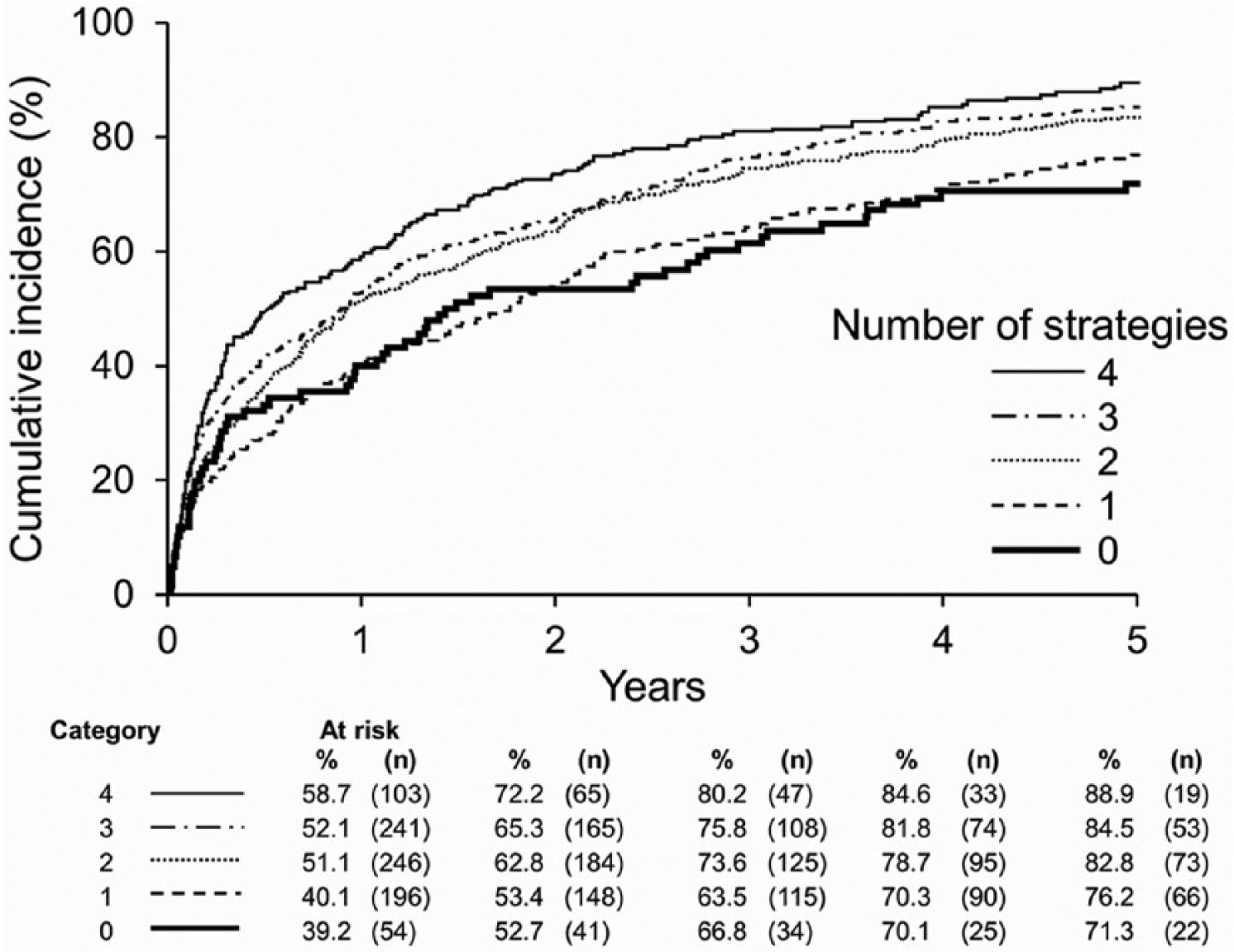
Incidence of first hospitalization by the number of guideline-recommended strategies.
Setting of PAD diagnosis
Most cases were diagnosed with PAD as outpatients (n = 1500, 83%), while only 298 (17%) were diagnosed with PAD as hospital inpatients. Patients diagnosed with PAD as inpatients had a higher rate of future hospitalization (114.4 per 100 person-years; 95% CI: 108.9, 120.2) than patients diagnosed with PAD as outpatients (66.4 per 100 person-years; 95% CI: 64.9, 68.0). During the first year after PAD diagnosis, the cumulative incidence of subsequent hospitalization in patients diagnosed with PAD as inpatients was 67% compared to 47% of patients diagnosed as outpatients (age and sex adjusted p-value < 0.001).
Primary reasons for hospitalizations
The most frequent reasons for hospitalization in cases were non-cardiovascular diagnoses (5913 non-cardiovascular reasons, 68% of 8706 hospitalizations) (Table 3). The most frequent non-cardiovascular reasons were respiratory diagnoses. Nearly one-third of hospitalizations were for cardiovascular reasons (2793 cardiovascular reasons, 32% of 8706 hospitalizations). Among cardiovascular reasons, cases were most likely to be hospitalized for HF and PAD-related diagnosis.
Primary reasons for hospitalizations in PAD – cases.
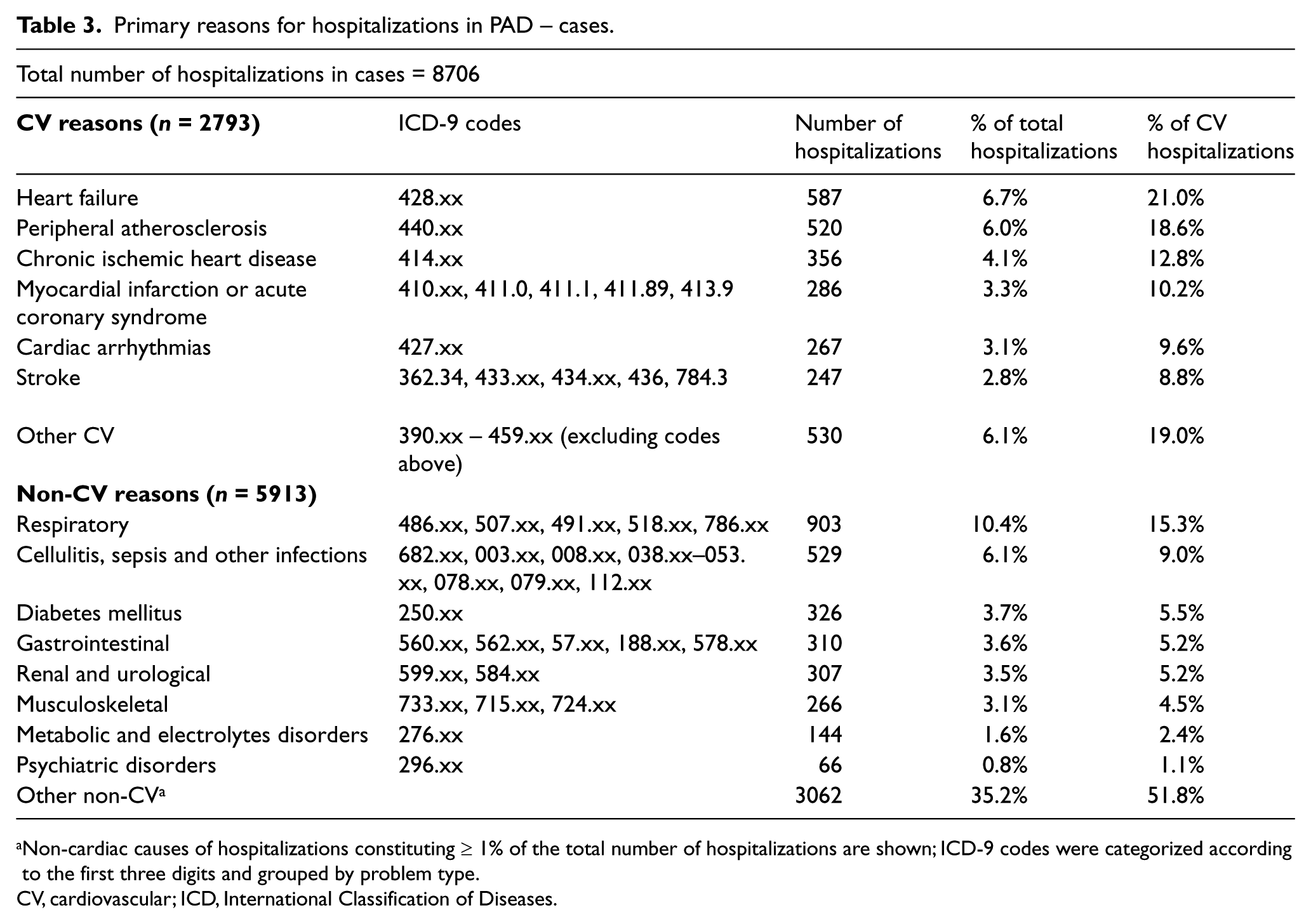
Non-cardiac causes of hospitalizations constituting ≥ 1% of the total number of hospitalizations are shown; ICD-9 codes were categorized according to the first three digits and grouped by problem type.
CV, cardiovascular; ICD, International Classification of Diseases.
Predictors for recurrent hospitalizations
In all models (unadjusted and adjusted), prior hospitalization was an independent predictor for recurrent hospitalizations in cases (Table 4). Additionally, in multivariable model-1, the older age groups, hypertension and four comorbidities (diabetes, prior MI, HF and pulmonary disease) had independent associations with recurrent hospitalizations (Table 4). After further adjustments for PCA or severe PAD (model-2), the same variables remained independent predictors for recurrent hospitalizations. Both severe PAD and PCA were associated with increased risk of recurrent hospitalizations (Table 4).
Predictors of recurrent hospitalizations – cases.
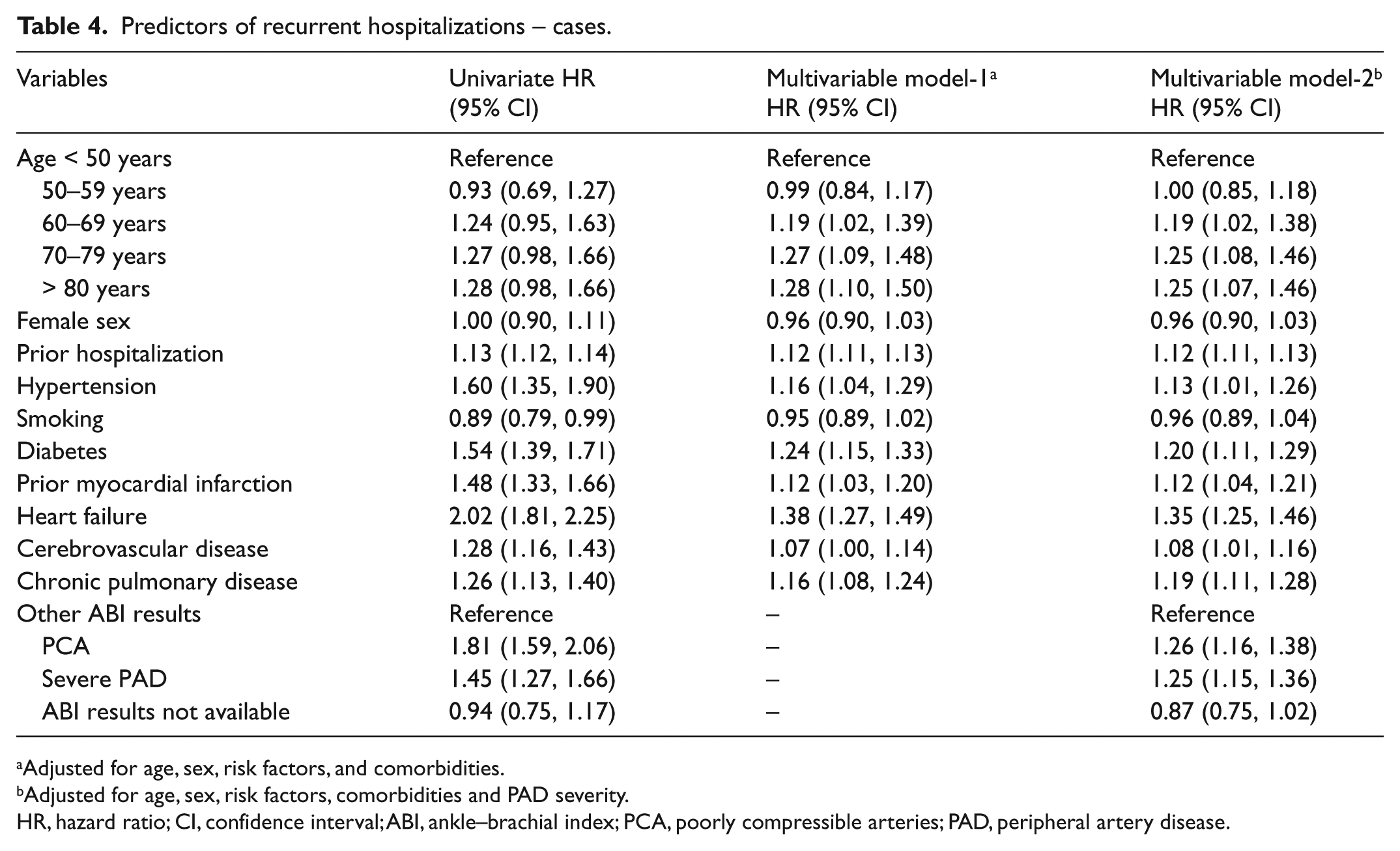
Adjusted for age, sex, risk factors, and comorbidities.
Adjusted for age, sex, risk factors, comorbidities and PAD severity.
HR, hazard ratio; CI, confidence interval; ABI, ankle–brachial index; PCA, poorly compressible arteries; PAD, peripheral artery disease.
Critical Limb Ischemia
There were 430 patients in the CLI subgroup and the rate of hospitalization was 113.7 per 100 person-years (95% CI: 109.2, 118.7), whereas for the subgroup of 1368 patients without CLI the rate of hospitalization was significantly lower at 63.7 per 100 person-years (95% CI: 62.2, 65.3) (p-value <0.05). Patients with CLI (Table 5) were older, with a higher comorbidity index, and were more likely to have diabetes and HF than patients without CLI. Distributions of prior MI, cerebrovascular disease and pulmonary disease were similar among these subgroups.
Characteristics of patients with CLI compared to patients without CLI.
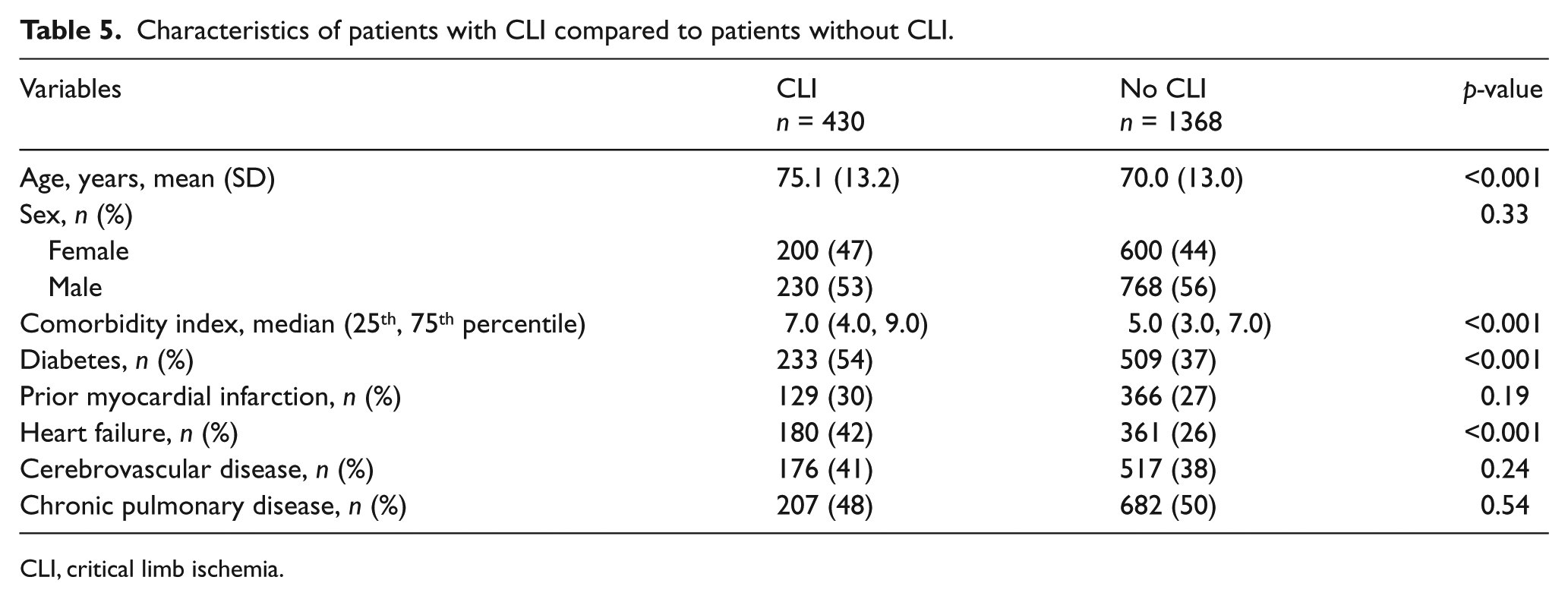
CLI, critical limb ischemia.
In multivariable models adjusted for age and sex, independent predictors for recurrent hospitalization in patients with CLI were prior hospitalization (HR: 1.09; 95% CI: 1.08, 1.11) and four comorbidities (diabetes (HR: 1.20; 95% CI: 1.04, 1.38), HF (HR: 1.16; 95% CI: 1.21, 1.33), prior MI (HR: 1.16; 95% CI: 1.01, 1.33) and pulmonary disease (HR: 1.15; 95% CI: 1.01, 1.31)). Smoking, hypertension and cerebrovascular disease were not independent predictors for recurrent hospitalizations in CLI cases.
Controls
Primary reasons for hospitalizations
In controls, 78% of the hospitalizations were for non-cardiovascular reasons (6209 of 8005 hospitalizations) and 22% were for cardiovascular reasons (1796 of 8005 hospitalizations). The most frequent non-cardiovascular reasons were respiratory diagnoses (Supplemental Table 1). Among cardiovascular reasons the most frequent diagnoses were cardiac arrhythmias or HF (Supplemental Table 1).
Predictors for recurrent hospitalizations
In controls, the independent predictors of recurrent hospitalization were age, prior hospitalization, hypertension, smoking, and five comorbidities (diabetes, prior MI, HF, cerebrovascular disease and pulmonary disease) (Supplemental Table 2). The effect of age was more pronounced in controls than cases. In ancillary analysis, the independent predictors for hospitalizations adjusted for time period of hospitalization were similar in both cases and controls.
Discussion
In this community-based study, prior hospitalization was a strong independent risk factor for recurrent hospitalizations. In contrast, other recent studies have been limited to subgroups of PAD patients who were hospitalized at study entry and followed for readmission within 30 days8,9 or 6 months. 10 One previous study of PAD patients only considered events during the hospitalization as the primary outcome for analysis. 23 Another prior study of hospitalized PAD patients from The Netherlands 24 focused only on mortality after first hospitalization. Hence, the generalizability of the findings from our study are broader as the observations were not limited to PAD patients who were previously hospitalized.
In our study, PAD patients were hospitalized nearly once per year, a rate which is comparable to that previously reported for patients with HF in Olmsted County during a similar era. 25 Moreover, one-third of PAD cases in our study had co-existing HF (i.e. HF as comorbidity) and a previous study demonstrated that among HF patients concomitant PAD increased risk for hospital readmission. 26 Cases were also more likely than controls to have risk factors for atherosclerosis and comorbidities associated with established systemic atherosclerosis, including prior MI, cerebrovascular disease, and hemiplegia. Our findings are also consistent with a previous registry study showing that the majority of PAD patients have cardiovascular comorbidities (prior MI or prior stroke) at the time of PAD diagnosis. 27 As PAD is a manifestation of systemic atherosclerosis, coronary heart disease and cerebrovascular disease frequently co-exist in affected patients. Studies that incorporate time-to-event analyses are needed to evaluate the incidence of stroke or MI on follow-up of community residents with clinically diagnosed PAD.
Severe PAD
Severe PAD was an independent risk factor for recurrent hospitalizations. Previous studies have reported a strong association between disease severity (as measured by the ABI) and MI, stroke, and MI/stroke 28 and that the presence of severe PAD predicts cardiovascular morbidity and mortality.14,29 In our study, PCA was another risk factor for recurrent hospitalization. We have previously shown that the presence of PCA was associated with high risk for mortality. 14 The present study adds to this knowledge by demonstrating that severe PAD and PCA were both independent predictors for recurrent hospitalizations in PAD patients from the community.
Reasons for hospitalization
The most frequent reasons for hospitalization in PAD patients were non-cardiovascular disorders. Greater numbers of comorbidities have been associated with an increased need for hospitalization in patients with other chronic diseases including HF 25 and gastrointestinal cancer. 30 A previous study of PAD patients demonstrated that comorbid conditions are risk factors for mortality 31 , but whether comorbidities are risk factors for recurrent hospitalizations in PAD patients was previously unknown.
Quality indicators
Overall, the proportion of patients on guideline-recommended strategies was low, though these proportions were comparable to a prior study. 32 Over the duration of the study, there was an increase in the proportion of patients on four guideline-recommended strategies, with a corresponding decline in patients receiving zero to one or two to three strategies. These observations underscore the need to further emphasize physician and patient awareness of PAD to reduce the gap between guideline recommendations and implementation of these strategies for PAD patients in the community.32,33
In the present study, the higher number of guideline-recommended strategies within 6 months of PAD diagnosis was associated with a higher number of incident hospitalizations. The presence of co-existing comorbidities and risk factors likely contributed to this association, which does not represent a direct causal relationship. The higher number of guideline-recommended strategies could be an indicator for the presence of associated comorbid cardiovascular conditions (e.g. prior MI, or previously diagnosed cerebrovascular disease), which, similar to PAD, are also manifestations of atherosclerotic cardiovascular disease. Similar strategies for secondary prevention have been recommended by practice guidelines for management of these associated cardiovascular comorbidities.34,35 Another explanation is that over time there has been an increase in the number of guideline-recommended strategies used by PAD patients, which may be related to the year of publication (2006) of the first PAD practice guidelines 2 with subsequent adoption of the strategies for secondary prevention.
The high rate of hospitalization in PAD patients was likely driven by multiple combined factors occurring simultaneously in the health system. Potential solutions to lower these rates include health care system redesign with adoption of innovative models for health care (e.g. patient-centered medical home model 36 ) to reorganize the delivery of care for PAD patients. Care coordination 37 and re-engineering of the discharge process 38 could also lower these rates. Importantly, the plan of care for PAD patients should be patient-centered and tailored both to PAD and to each one of the co-existing risk factors for recurrent hospitalization identified by the current study.
Limitations
First, asymptomatic patients with PAD were not included, though all patients with clinically diagnosed PAD in the community who agreed to participate in this cohort study were included. Second, the reasons for not using specific guideline-recommended strategies (e.g. stain-induced myopathy) or adherence to guideline-recommended strategies were not assessed in this observational cohort study. These factors may have influenced the study results. Third, this study identified an association between comorbidities and recurrent hospitalizations. However, because of the observational cohort study design, a causal relationship for increased re-admissions (i.e. comorbidities vs PAD) could not be defined. Finally, the generalizability may be limited to communities with composition similar to the Olmsted County population, which is comprised mostly of white individuals.11,39
Conclusions
This community-based study demonstrated that recurrent hospitalizations were common and more frequent in PAD cases; age, prior hospitalization and comorbid conditions were risk factors for recurrent hospitalizations. Our observations emphasize the need for optimizing management strategies for PAD and associated chronic comorbidities to reduce the burden of hospitalization.
Footnotes
Acknowledgements
We thank Jared Robb, LuAnne Koenig and Cynthia Regnier for data collection; Carin Smith for statistical analysis; and Rebecca M Olson and Tamie Tiedemann for secretarial support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: this study supported by the National Heart, Lung, and Blood Institute of the National Institutes of Health (K01HL124045), the NHGRI eMERGE (Electronic Records and Genomics) Network grants HG04599 and HG006379, and the Rochester Epidemiology Project (National Institute on Aging of National Institutes of Health award R01AG034676). The content is solely the responsibility of the authors and does not necessarily represent official views of the National Institutes of Health.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
