Abstract
Non-valvular atrial fibrillation and venous thromboembolism anticoagulation risk assessment tools have been increasingly utilized to guide implementation and duration of anticoagulant therapy. Anticoagulation significantly reduces stroke and recurrent venous thromboembolism risk, but comes at the cost of increased risk of major and clinically relevant non-major bleeding. The decision for anticoagulation in high-risk patients is complicated by the fact that many risk factors associated with increased thromboembolic risk are simultaneously associated with increased bleeding risk. Traditional risk assessment tools rely heavily on age, sex, and presence of cardiovascular comorbidities, with newer tools additionally taking into account changes in risk factors over time and novel biomarkers to facilitate more personalized risk assessment. These tools may help counsel and inform patients about the risks and benefits of starting or continuing anticoagulant therapy and can identify patients who may benefit from more careful management. Although the ability to predict anticoagulant-associated hemorrhagic risk is modest, ischemic and bleeding risk scores have been shown to add significant value to therapeutic management decisions. Ultimately, further work is needed to optimally implement accurate and actionable risk stratification into clinical practice.
Keywords
Introduction
Medical decision-making around anticoagulation (AC) has become increasingly complex, as new therapeutic agents have been introduced to populations with varying risk profiles and comorbidities. Risk assessment is an important first step in maximizing the beneficial effects of AC while mitigating bleeding events. This process requires knowledge of the indication for AC, the patient’s risk for major and clinically relevant non-major bleeding, anticipated duration of therapy, its expected adherence, and the intrinsic properties of the anticoagulant itself. This review specifically focuses on risk assessment models that are likely to be helpful in evaluating patients with non-valvular atrial fibrillation (NVAF) and/or venous thromboembolism (VTE).
The most commonly used oral anticoagulants include vitamin K antagonists (VKAs) and the direct oral anticoagulants (DOACs). Less often, patients may be treated with parenteral anticoagulation, including unfractionated heparin (UFH), low molecular weight heparin (LMWH), or the pentasaccharide, fondaparinux. Substantial data support long-term AC in patients with NVAF and/or VTE, particularly among those at higher thromboembolic risk.
Antithrombotic and anticoagulant therapies used for non-valvular atrial fibrillation (NVAF) and venous thromboembolism (VTE)
Antiplatelet therapy is less commonly used to reduce thrombotic risk in patients with NVAF or VTE. While aspirin is the most commonly used antithrombotic therapy worldwide and has been shown to reduce the risk of cardioembolic stroke in NVAF by roughly 20%, 1 the 2014 American Heart Association (AHA)/American College of Cardiology (ACC)/Heart Rhythm Society (HRS) atrial fibrillation guidelines limit its use to NVAF patients at lower thromboembolic risk. 2 The value of dual antiplatelet therapy in NVAF was evaluated in the ACTIVE A trial, 3 where the addition of clopidogrel to aspirin reduced the risk of stroke by 27%. This benefit, however, was offset by an almost twofold increased risk of hemorrhage. As such, it is not a recommended therapy for NVAF. 2 Finally, while aspirin is not considered standard therapy for VTE, the American College of Chest Physicians (ACCP) endorses a grade 2B recommendation for its use in patients without contraindication to aspirin who are stopping AC. 4
Substantial evidence exists to support the use of a VKA (e.g. warfarin) in NVAF, with an estimated stroke risk reduction of 68% compared to placebo. 1 Its use, however, is limited by a narrow therapeutic window, delayed pharmacodynamic response, numerous drug–food and drug–drug interactions, and the need for close monitoring. Adverse events with VKAs are also closely related to the quality of AC, with an inverse association between time in the therapeutic range (TTR) and risk of both major bleeding and mortality.5,6
While LMWH represents an alternative to VKA, it is limited by dependence on renal excretion and subcutaneous administration. The DOACs, in contrast, are oral agents which act via direct inhibition of factors II or Xa and have proven efficacy in NVAF, VTE, and stable atherosclerotic cardiovascular disease (coronary artery disease and peripheral artery disease).7–12 These agents are limited in part by cost and can be difficult to implement if monitoring of AC is required.
Given the diversity of agents to choose from, the decision for AC therapy has become increasingly complex. While a nuanced discussion of the risk–benefit differences among VKAs, DOACs, and parenteral anticoagulants is outside the scope of this review, it is important to consider the specific agents for which risk calculators were validated when making decisions about AC.
Variables implicated in prediction of thromboembolism and bleeding in NVAF and VTE
Assessment of thromboembolic risk for NVAF has historically been dependent on the presence of traditional pro-thrombotic risk factors, such as increased age (⩾ 75 years), female sex, and a variety of cardiovascular comorbidities. While risk prediction tools have been heavily weighted by these risk factors, novel markers have been increasingly incorporated to refine predictive accuracy.
Several biomarkers have been associated with NVAF thromboembolic risk, such as N-terminal pro-brain natriuretic peptide (NT-proBNP) and high-sensitivity cardiac troponin (hs-cTnT) that reflect high atrial stretch, and growth differentiating factor-15 (GDF-15) that reflects oxidative/myocardial stress.13–15 Hs-cTnT and GDF-15 have additionally been associated with bleeding risk in NVAF patients, along with markers of anemia such as hemoglobin and markers of renal dysfunction such as cystatin C and estimated glomerular filtration rate (eGFR).15–17 Several non-specific markers of inflammation, including D-dimer, C-reactive protein (CRP), and interleukin (IL)-6 have additionally been associated with higher bleeding risk,18,19 but have been difficult to integrate into stratification algorithms. Echocardiographic findings such as increased left atrial volume index, abnormal left atrial longitudinal strain, and left atrial appendage pulse-wave velocity have been associated with increased thrombotic risk. 20 Finally, evidence of left atrial fibrosis by cardiac magnetic resonance imaging has also been found to be predictive. 21
Similar to NVAF, there exists substantial interest in the identification of novel biomarkers to increase accuracy in the prediction of VTE and guide appropriateness for extended duration anticoagulant therapy. In addition to D-dimer, biomarkers including soluble P-selectin, coagulation factor VIII, thrombin, CRP, tumor necrosis factor, and interleukins have been evaluated. 22 Some studies have shown niche value, such as the use of baseline P-selectin to evaluate risk of VTE recurrence in patients with cancer-associated VTE. 23 Moving forward, incorporation of biomarkers into VTE prediction models may allow for a more personalized approach to AC management.
It is also very important to recognize that many traditional risk factors and biomarkers that increase thromboembolic risk also simultaneously increase bleeding risk. In addition, risk factors may contribute differentially to risk. For example, the presence of vascular disease in NVAF does not contribute the same attendant risk as other traditional risk factors. 24 Collectively, these factors should be taken into consideration when evaluating risk prediction tools.
Prediction of stroke or systemic embolism in patients with NVAF
Atrial fibrillation is the most common sustained arrhythmia worldwide, 25 and is associated with an increased risk of thromboembolism. The treatment approach (rhythm- vs rate-control) has not been shown to meaningfully impact thromboembolic risk, as demonstrated in the Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) and Rate Control Efficacy in Permanent Atrial Fibrillation (RACE) trials.26,27 Less is known, however, about the relationship between the burden of NVAF and thromboembolic risk. In one study, a burden > 5.5 hours was associated with increased risk of thromboembolism. 28 Current major guidelines do not, however, specify how atrial fibrillation burden should impact decision making for AC.2,29–31
Over time, a number of risk prediction tools have been developed to help identify patients at heightened risk for stroke or systemic embolism for which anticoagulant therapy would be beneficial. Consistent among these tools has been an attempt to divide patients into low-, moderate-, and high-risk strata based on a number of key traditional thrombotic risk factors. In addition, newer calculators have incorporated differential weighting of variables and biomarkers, as well as change in risk over time (Table 1). There has also been a shift to identify lower risk patients who can be successfully treated either with aspirin or no antithrombotic therapy at all.
NVAF stroke risk calculators.
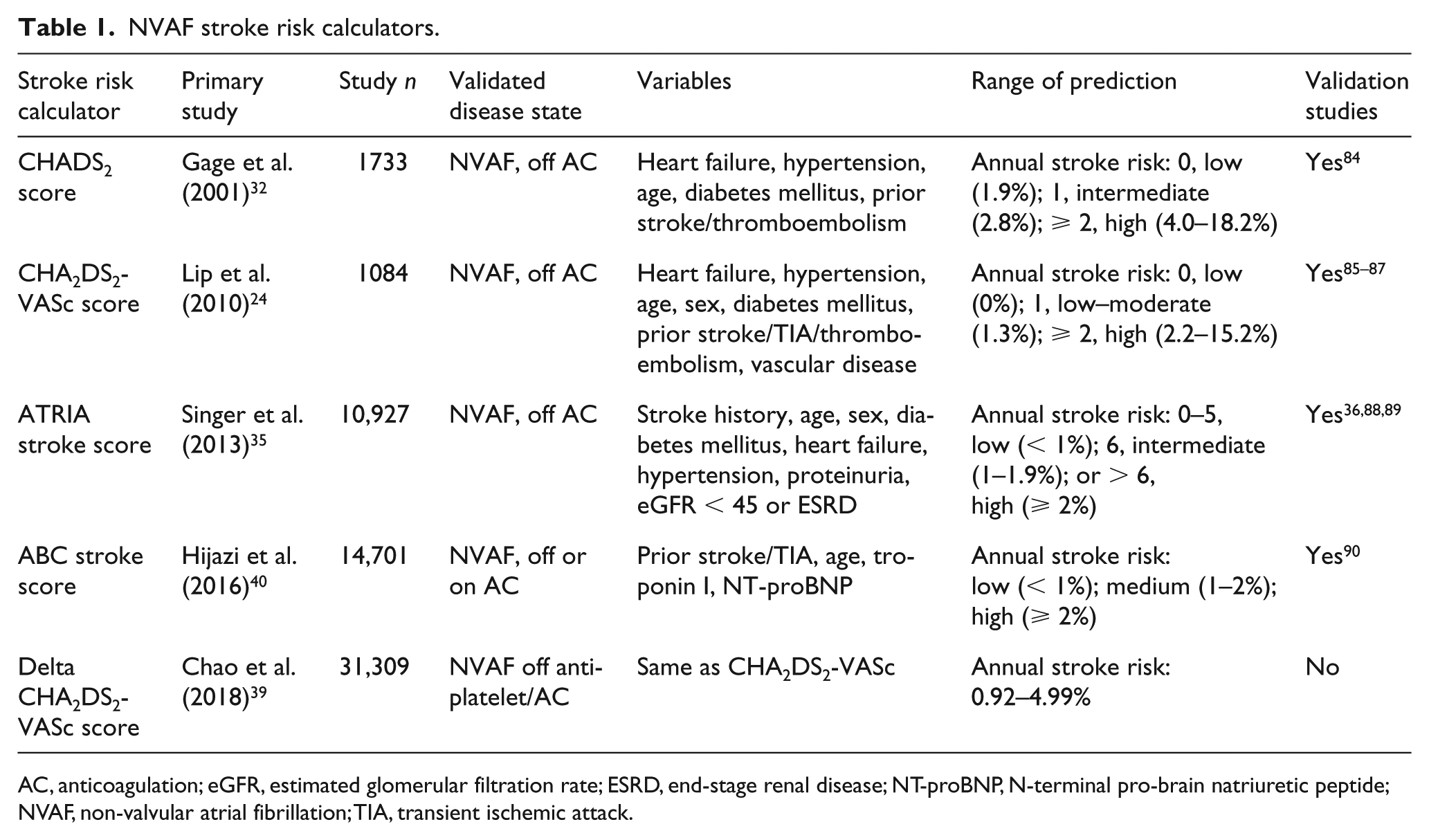
AC, anticoagulation; eGFR, estimated glomerular filtration rate; ESRD, end-stage renal disease; NT-proBNP, N-terminal pro-brain natriuretic peptide; NVAF, non-valvular atrial fibrillation; TIA, transient ischemic attack.
While the criteria used to define which patients with NVAF warrant AC has varied among international guidelines, there has been a tendency to rely upon either the CHADS2 or CHA2DS2-VASc risk calculators.2,29–31 Guide-lines such as the 2016 European Society of Cardiology/European Atherosclerosis Society (ESC/EAS) suggest that biomarker-based risk scores may provide additional prognostic information, but do not offer any specific recommendations for their incorporation into the treatment algorithm. 30
The first NVAF assessment tool to achieve widespread use was the CHADS2 score, which was developed from a cohort of approximately 1,700 Medicare beneficiaries with non-rheumatic atrial fibrillation untreated with AC (warfarin) at hospital discharge. 32 The score ranges from 0 to 6, with scores of zero, three, and six correlating with annual stroke risks of 1.9%, 5.9%, and 18.2%, respectively. A subsequent analysis by Fang et al. found the predicted annual stroke rate to be substantially lower: 0.5% for a CHADS2 score of zero and > 3% for a CHADS2 score ⩾ 3. 33 Although the score is simple to calculate and implement into daily practice, it is limited by inadequate refinement of risk at the low end of the stroke/systemic embolism spectrum, placing up to 50% of patients with a score of one in the intermediate-risk category. 34 This risk stratification tool is currently supported only by a limited number of guidelines. 29
The CHA2DS2-VASc score was created to refine risk stratification among patients with low to intermediate CHADS2 scores by incorporating additional variables, including age of 65–74 years, vascular disease, and female sex. 24 It is superior in discriminating between patients at very low risk, and reclassifies many patients with a CHADS2 score of zero into higher-risk categories. In the original study, patients characterized at lowest risk (CHADS2 score of zero) had a stroke rate of 1.4% during the follow-up period, whereas no strokes were detected in those with a CHA2DS2-VASc score of zero. 24 Critics of both CHADS2 and CHA2DS2-VASc scores note that they both have a less than optimal c-statistic (0.6–0.7).35,36
The CHA2DS2-VASc score is the most commonly utilized score at present, with endorsement in several modern atrial fibrillation guidelines. According to the 2014 AHA/ACC/HRS atrial fibrillation guidelines, aspirin or no antithrombotic therapy is recommended for those with a CHA2DS2-VASc score of zero; no antithrombotic therapy, aspirin, or AC are all reasonable options for a score of one; and AC is recommended for those with a score ⩾ 2. 2 Similarly, the ESC/EAS 2016 guidelines give preference to the CHA2DS2-VASc score, recommending AC in men with a score ⩾ 1 and women with a score ⩾ 2. 30 In spite of this, several other stroke assessment tools have been proposed, with a goal of improving risk stratification afforded by the CHA2DS2-VASc score.
The Anticoagulation and Risk Factors in Atrial Fibrillation (ATRIA) score expands upon the CHADS2 score, incorporating elements such as age, prior stroke, female sex, eGFR < 45 mL/min, and proteinuria. 35 It also differentially weights risk factors depending on the presence of other co-factors. In contrast to the CHADS2 and CHA2DS2-VASc scores, which assign a large number of patients into the intermediate stroke risk range, the ATRIA score has been shown to correctly reclassify more than 8.6% and 20% of CHA2DS2-VASc and CHADS2 intermediate-risk patients, respectively, largely into lower risk categories. 37 These results are complicated by findings in higher-risk patient cohorts which found CHA2DS2-VASc to be superior to ATRIA in terms of accurately classifying patients as ‘low risk’ 38 ; in these cohorts, patients marked as ‘low risk’ by ATRIA had higher risk on average than those marked as ‘low risk’ by CHA2DS2-VASc. To date, no major guidelines have endorsed the use of the ATRIA risk score for NVAF.
Alternative approaches to risk prediction in NVAF have been proposed that take into account temporal changes in risk factors and specific biomarkers. In patients with an initially low CHA2DS2-VASc score, recent evidence provides support for the delta CHA2DS2-VASc, which assesses change in risk factors over time to predict thromboembolic risk. 39 Most patients (90% of the cohort tested) will newly develop one or more comorbidities (most commonly hypertension) prior to developing an ischemic stroke, suggesting broad applicability. Reported hazard ratios for ischemic stroke for a delta CHA2DS2-VASc score of one, two, three, and four (calculated 1 year from baseline) are 1.56, 1.94, 2.39, and 2.88, respectively. The delta CHA2DS2-VASc score has been shown to be a better predictor of incident stroke than either the initial or subsequent absolute risk score. 39
A new biomarker-based risk tool called the ABC stroke risk score incorporates age, biomarkers, and a history of stroke/transient ischemic attack. The ABC stroke risk score has been shown to be superior to the CHA2DS2-VASc in both its derivation and external validation cohorts for predicting ischemic stroke. 40 The ABC stroke risk score uniquely utilizes two biomarkers, NT-proBNP and hs-cTnT, to improve predictive value. The addition of inflammatory (CRP, IL-6), oxidative stress (GDF-15), and coagulation (D-dimer) biomarkers to the risk model did not improve prognostic accuracy. 40
Modern NVAF risk calculators are subject to a number of limitations. While some calculators have begun to take a weighted approach to risk factors as reflected in epidemiologic data, the most commonly used scores are over-simplistic and binary in terms of how factors are weighted in the calculations. Co-morbidities associated with thromboembolic risk represent a range and do not equally contribute to stroke risk. 41 Additionally, duration and frequency of NVAF episodes are not taken into consideration, which may have an impact on thromboembolic risk. 42 Finally, these models take into account risk of thromboembolism, but not bleeding risk, so a comprehensive risk–benefit discussion requires incorporation of a separate bleeding risk assessment.
Prediction of progressive or recurrent thromboembolic complications in patients with VTE
Unlike NVAF, risk prediction for VTE is primarily used to define the duration of AC and whether long-term secondary prophylaxis with AC is warranted to reduce the risk of recurrent events. Generally, patients require a minimum of 3 months of AC for an acute, proximal, deep venous thrombosis (DVT) provoked by surgery or a proximal DVT provoked by a non-surgical, transient risk factor. In contrast, lifelong therapy is indicated for patients with a second unprovoked DVT or pulmonary embolism (PE).
According to the 2016 ACCP guidelines, decision-making about whether or not to extend AC treatment in a non-provoked VTE should be dictated by risk of bleeding. 4 In general, after 3 months of therapy, AC is continued indefinitely in patients at low or moderate bleeding risk. In contrast, those at high bleeding risk will generally stop AC.
Currently, no risk assessment tools exist to acutely guide the duration of secondary prophylaxis in patients hospitalized for VTE. Instead, current risk assessment models are usually carried out after a defined period of AC treatment. Risk scores, however, do exist to predict risk of VTE among predisposed individuals hospitalized for other indications. In fact, hospitalization is a known risk factor for VTE, associated with an eightfold increased risk of VTE due, in part, to factors such as immobility. 43 The Padua prediction score for VTE was created to stratify patients into low- or high-risk groups for acute VTE, primarily to determine if prophylactic AC is indicated. 44 The score is based on presence of active cancer, previous VTE, reduced mobility, known thrombophilia, recent trauma/surgery, age, cardiac/respiratory failure, acute myocardial infarction/ischemic stroke, presence of acute infection, presence of a rheumatologic disorder, and obesity. For scores < 4, prophylactic doses of AC with subcutaneous heparin or LMWH should be considered on a case-by-case basis. For scores ⩾ 4, prophylactic AC is routinely recommended among non-pregnant patients without contraindications.
After the first VTE, risk of recurrence generally decreases and risk of AC-related bleeding increases over time.45,46 Accordingly, the need for secondary prophylaxis should be serially reassessed based on individual risk factors at discrete follow-up intervals. Evidence from randomized controlled trials such as Rivaroxaban or Aspirin for Extended Treatment of Venous Thromboembolism (EINSTEIN CHOICE) and Apixaban for Extended Treatment of Venous Thromboembolism (AMPLIFY-EXT) suggest that extended-duration thromboprophylaxis with reduced dose rivaroxaban and apixaban are superior to aspirin in terms of preventing recurrent VTE.47–49
A variety of risk calculators have been created to assess the need for therapeutic AC for secondary VTE prophylaxis after completion of acute VTE treatment, including the Vienna Prediction Model, DASH score, and HER DOO2 score (Table 2). The Vienna Prediction Model can assess recurrence of unprovoked VTE at any time 3 weeks to 15 months after AC is discontinued.50,51 An update to the prediction model utilizes serial D-dimer levels at multiple random time points to calculate risk of recurrent VTE. 51 The DASH prediction score utilizes similar risk factors, but includes hormonal use at VTE onset, which helps adjust for provoked VTE. 52 This model indicates that in patients with low risk of recurrence (score ⩽ 1), long-term AC can be avoided, so that roughly 50% of patients with unprovoked VTE may obviate extended therapy. The HER DOO2 recurrent VTE assessment identifies patients at low risk for recurrence who can safely discontinue AC after 5–7 months of therapy. 53 The annual risk of recurrent VTE was higher among men and those with signs and symptoms of post-thrombotic syndrome. No men in the low-risk subgroup could be identified who could safely discontinue AC, but women with 0 or 1 risk factors were found to have a low annual risk of VTE recurrence (1.6%), thus allowing for discontinuation of AC. Although these three scores are not specifically endorsed for secondary VTE prophylaxis management in the most recent 2016 ACCP ‘Antithrombotic Therapy for VTE Disease’ guidelines, they have independent predictive ability, and may be used to quantify risk of recurrent VTE at various treatment time points to better facilitate AC risk–benefit assessment. 4
NVAF bleeding risk calculators.
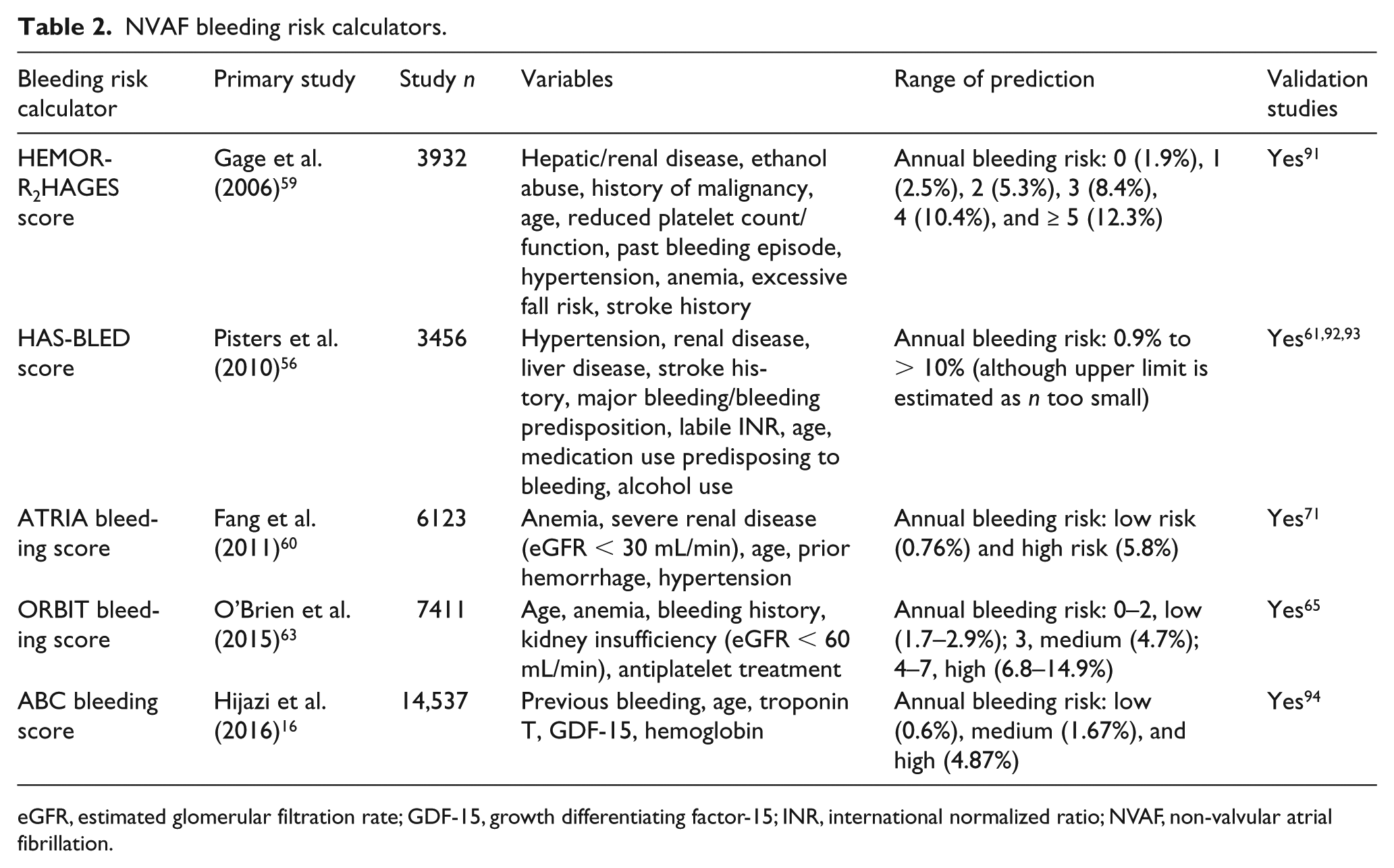
eGFR, estimated glomerular filtration rate; GDF-15, growth differentiating factor-15; INR, international normalized ratio; NVAF, non-valvular atrial fibrillation.
Prediction of bleeding in patients with NVAF
When considering anticoagulant therapy for the treatment of NVAF, it is important to assess the risk of both major and minor bleeding. Outside of surgery, the former has been defined by the International Society on Thrombosis and Haemostasis as fatal bleeding, symptomatic bleeding in a critical area or organ, or bleeding causing a decrease in hemoglobin of ⩾ 2 g/dL or requiring transfusion of ⩾ 2 units of packed red blood cells54,55 (Table 3). For patients with an absolute contraindication to anticoagulation, the decision to hold AC may be clear-cut; however, patients with relative contraindications due to high bleeding risk require a more nuanced approach. The 2014 Canadian Cardiovascular Society atrial fibrillation guidelines recommend implementation of bleeding risk scores (i.e. HAS-BLED) in addition to stroke risk assessment to inform decision-making in complex patients. 29 The 2018 ACCP Guideline and Expert Panel Report on antithrombotic therapy for atrial fibrillation further suggests that patients on a VKA at high bleeding risk (i.e. HAS-BLED score ⩾ 3) warrant more frequent follow-up and attention to modifiable bleeding risk factors. 31 To this end, a variety of new stroke and bleeding risk prediction tools have been subsequently created to further improve risk estimation in clinically challenging patients (Table 2).
The first of these tools is the HAS-BLED score, which takes into account several bleeding factors to calculate yearly bleeding risk. 56 The HAS-BLED score draws attention to several modifiable bleeding risk factors such as uncontrolled hypertension, concomitant use of aspirin or non-steroidal anti-inflammatory drugs, and alcohol use. Furthermore, it takes into account the quality of AC control, reflected by TTR among VKA users, which can be easily assessed via the percentage of time having achieved a therapeutic international normalized ratio (INR) goal. A high HAS-BLED score (⩾ 3) is indicative of the need for regular clinical review and follow-up, but should not be used per se to stop AC.57,58 Similar to other bleeding risk calculators, patients with NVAF and a high HAS-BLED score derive a higher net clinical benefit from AC when balancing the risks of ischemic stroke against intracranial bleeding. The HAS-BLED score has additionally been shown to have good predictive value for risk of intracranial bleeding and bleeding risk associated with bridging therapy.
Several other bleeding risk calculators, such as HEMORR2HAGES and ATRIA, were designed concurrent with HAS-BLED but are less predictive of bleeding overall.59,60 HAS-BLED was found to be superior to ATRIA and HEMORR2HAGES upon retrospective analysis of VKA patients from the ‘Evaluating the Use of SR34006 Compared to Warfarin or Acenocoumarol in Patients With Atrial Fibrillation (AMADEUS)’ study, 61 although all three risk scores were shown to have limited predictive accuracy overall (c-statistics < 0.70). In this comparison, the ATRIA score failed to demonstrate additional benefit over chance alone, whereas HEMORR2HAGES was slightly better than random stratification. The HEMORR2HAGES score had the worst mortality prediction, thought to be due to a larger number of bleeding-oriented factors used in its calculation. A subsequent study showed that HAS-BLED demonstrated superior predictive ability as compared to ATRIA amongst both VKA and non-VKA anticoagulant subgroups. 62 Although the ATRIA tool may be of limited clinical utility, one unique aspect of the tool is that it was the first bleeding risk calculator to weigh risk factors differentially.
Each of the aforementioned bleeding risk scores was primarily designed and validated in those on VKAs. In an attempt to better predict bleeding risk among those on a VKA or DOAC, the ORBIT score was introduced in 2015, taking into account five easily calculable variables to predict bleeding risk: age, anemia, treatment with concurrent antiplatelet agent, renal insufficiency, and prior bleeding history. 63 The ORBIT score notably eliminated ‘labile INR’ as a criterion due to its difficulty to measure and poor applicability to patients on DOACs or those not on prior anticoagulation. The original study cohort included both patients on warfarin and dabigatran, 63 and was further validated in those on rivaroxaban. 64 The ORBIT score was subsequently shown to be superior to HAS-BLED, ATRIA, and HEMORR2HAGES64,65 among patients on VKA or dabigatran.
Subsequent analyses comparing ORBIT to HAS-BLED have demonstrated greater variability in predictive value, with several cohort studies showing HAS-BLED to be more predictive of bleeding risk than ORBIT for those on VKA or DOAC.66–70 In these analyses, the ORBIT score tended to classify a higher percentage of patients as ‘low risk’ 70 as compared to HAS-BLED, but was ultimately less predictive at identifying those at true ‘low risk’ of major bleeding among patients treated with a VKA. 67 It is possible that exclusion of labile INR or an alternative assessment of AC quality may have led the ORBIT score to underestimate risk, with one study showing significant improvement in predictive performance via the addition of ‘time in the therapeutic range’ as an additional variable. 71 Ultimately, we believe that the ORBIT score is not demonstratively superior to HAS-BLED, and favor use of HAS-BLED, given that it is less likely to underestimate risk of bleeding in patients on a VKA or a DOAC.
Reflective of recent research into several new biomarkers correlated with AC-associated bleeding risk, the ABC bleeding score was created. 16 This risk score incorporates hemoglobin, troponin, and GDF-15 in addition to demographic data to improve bleeding risk prediction. The ABC bleeding risk calculator, along with its counterpart ABC stroke risk calculator, were both derived from the 2011 ARISTOTLE study, with results translated into ‘low-’, ‘intermediate-’, and ‘high-’ risk of bleeding or stroke, respectively. The ABC bleeding score outperformed other bleeding scores, such as HAS-BLED and ORBIT, in its validation cohort, and continued to outperform these calculators when GDF-15 was exchanged for the more frequently assessed ‘creatinine clearance’. 16
Use of biomarkers as part of the ABC score also allows for easier monitoring of changes in bleeding risk, which can be helpful in tailoring therapy over time. 72 Prior to the ABC scores, pairing stroke and bleeding scores for overall clinical assessment was difficult given that scores were typically derived from different cohorts and utilized differing variables and clinical endpoints. In addition to being derived from similar cohorts, the ABC scores overlap traditional and biomarker variables (age and troponin) that contribute dually to thromboembolic and bleeding risk. Simultaneous use of these two risk tools may provide a future opportunity for improved tailoring of stroke prevention treatment in patients with atrial fibrillation due to their clinical overlap.
Ultimately, the decision whether or not to start AC for NVAF in high-risk patients can be aided by risk stratification tools that assist in quantifying estimated bleeding risk. Bleeding risk is not a static process, and patients require re-evaluation at frequent intervals over the course of their follow-up. 66 Importantly, modifiable risk factors for bleeding should be addressed to mitigate the risk of bleeding. Bleeding calculators do not take into account location or severity of bleeding, which are important factors for decision-making that may inform patient and physician preference. Finally, choice of AC may have an impact on risk stratification for these patients, as certain anticoagulants have been shown to have superior safety profiles as compared with VKAs, thus favoring treatment with AC in equivocal risk–benefit cases among those previously classified at lower ischemic risk.10,12,73
Prediction of bleeding in patients with VTE
With regard to calculating bleeding risk in patients with VTE, options include use of NVAF bleeding tools (i.e. HAS-BLED, ATRIA, HEMORR2HAGES) or those specific to VTE (Table 4). The 2010 International Medical Prevention Registry on Venous Thromboembolism (IMPROVE) score was created to assess risk of bleeding with VTE thromboprophylaxis in the inpatient setting, as a counterpart bleeding score to the Padua Prediction Score.74,75 The Kuijer and RIETE scores were similarly derived and developed in VTE-specific populations to calculate the risk of hemorrhage with AC.76,77 Disappointingly, these latter scores suffer from inferior predictive value within the first 3 months of treatment. They are also limited given their derivation from cohorts of patients using VKAs, and accordingly may not translate well to patients not treated with VKAs.
VTE risk calculators.
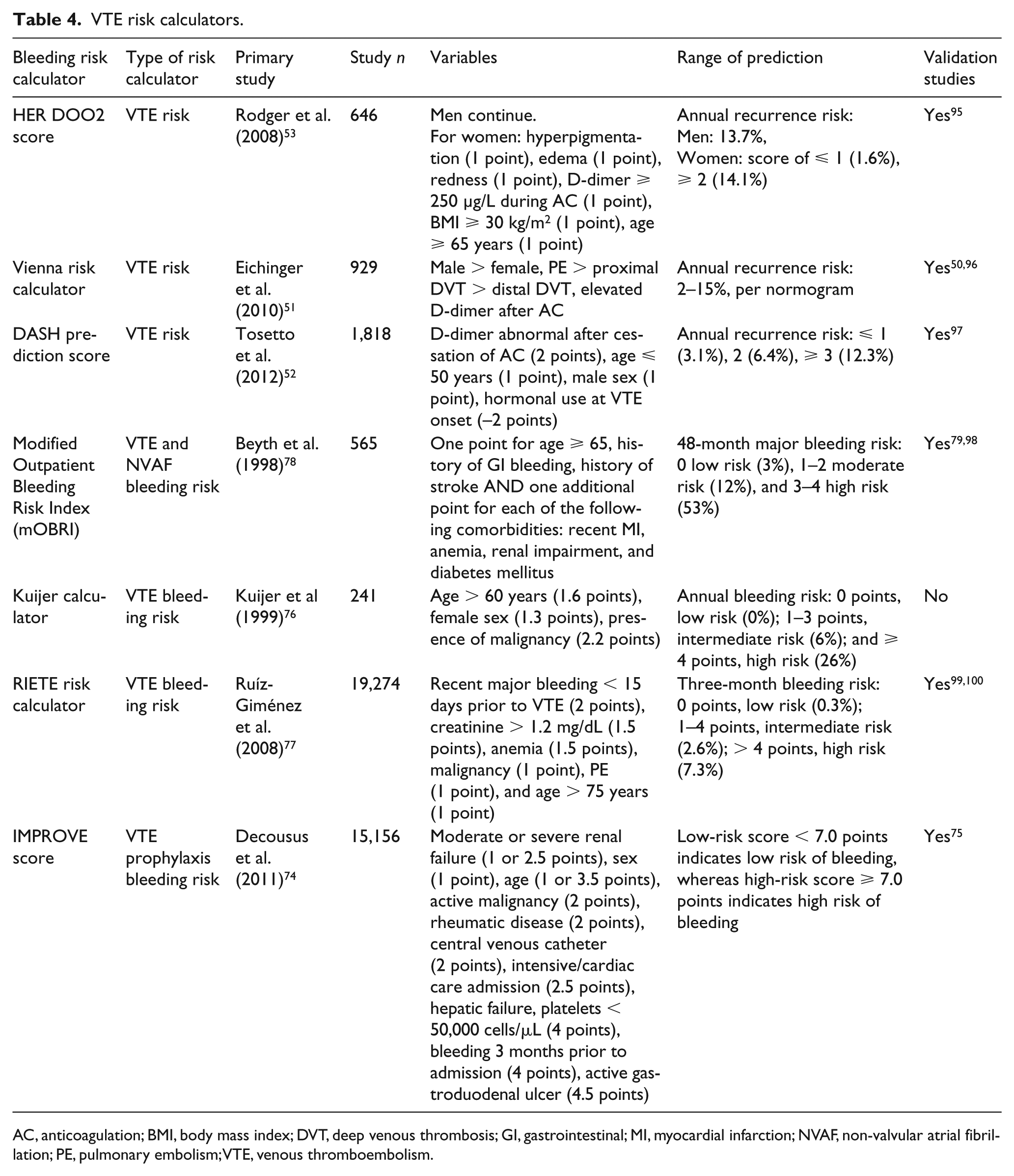
AC, anticoagulation; BMI, body mass index; DVT, deep venous thrombosis; GI, gastrointestinal; MI, myocardial infarction; NVAF, non-valvular atrial fibrillation; PE, pulmonary embolism; VTE, venous thromboembolism.
From a pragmatic perspective, the NVAF bleeding scores were derived from separate cohorts and are not easily translated to data from counterpart VTE risk prediction scores. Even assuming one could simultaneously compare risk of subsequent VTE and bleeding from AC at a given time point, there exists no threshold to define what is considered an acceptable risk–benefit ratio or cut-off from which one could make an algorithmic decision in the indeterminate-risk patient.
Precision of bleeding in patients with NVAF and VTE
The first bleeding risk scores, the Outpatient Bleeding Risk Index (OBRI) and modified Outpatient Bleeding Risk Index (mOBRI), did not distinguish the indication for AC when measuring risk of hemorrhage with a VKA. These scores included patients with varied indications for AC (NVAF, VTE, and post-cardiac surgery), with calculated bleeding risk measured independently of etiology of AC. 78 mOBRI was initially developed based on patients discharged from the hospital on long-term warfarin therapy, but was later validated in outpatients initiated on warfarin after hospital discharge with up to a 7-year follow-up.78,79
Subsequent bleeding risk calculators, as described in the sections above, have historically focused on either NVAF or VTE, with minimal clinical overlap. Given the relative paucity of VTE bleeding risk calculators, several of the bleeding risk scores developed in the NVAF population have been evaluated in patients with VTE. For example, HAS-BLED has been shown to be predictive in patients with VTE, such that a score ⩾ 3 is associated with an increased risk of major bleeding. 80
Currently, no large prospective studies exist that assess whether bleeding risk in patients with both atrial fibrillation and VTE varies compared to patients with only one indication for AC. Given that different DOAC dosing may be used for various AC indications, further investigation into the dose-dependent prediction of bleeding risk is needed.
Given a similar mechanism of hemorrhage for those receiving AC for NVAF and VTE, bleeding risk scores have significant potential for overlap. Ultimately, bleeding risk is dependent on the specific anticoagulant and dose, with the need to take these aspects into account to ensure fair comparison.
How to operationalize risk assessment tools in practice
Use of ischemic and bleeding risk scores in patients with either NVAF or VTE has been shown to outperform clinician judgment. Steinberg et al. investigated this question through a prospective, observational study of NVAF patients in the ORBIT-AF registry, assessing systemic embolism and major bleeding risk at 28 months with physician-assessment of risk in addition to CHADS2, CHA2DS2-VASc, ATRIA, and HAS-BLED scores. 81 Physician-based risk assessments for patients with NVAF showed risk stratification comparable to stroke scores, but these assessments were found to be inferior to bleeding scores. Furthermore, combining subjective and objective assessments did not add incremental accuracy as compared to empiric risk assessment alone.
Assessment of stroke risk for NVAF is relatively straightforward, in that stroke risk scores are easily actionable and supported by guidelines to direct therapeutic decisions. In contrast, assessment and implementation of bleeding risk is less straightforward. No prospective trial has demonstrated a benefit to withholding AC in patients identified as high bleeding risk by any criteria. 81 Furthermore, some observational data have suggested that a net clinical benefit of AC persists even among patients at the highest risk of bleeding; thus, most major guidelines do not wholly endorse the use of bleeding score algorithms as the primary basis for treatment decision-making.58,82 Ultimately, both stroke and bleeding assessments provide valuable risk stratification information in NVAF, and further work is needed to delineate maximally accurate and actionable risk stratification.
Management of extended duration AC for VTE is less well defined than NVAF given the limited predictive ability of current assessment tools and lack of firm incorporation of these tools into current guidelines. 83 A combined approach incorporating clinical VTE and bleeding risk calculators, subjective clinician assessment, and patient preference may be reasonable in the majority of cases.
Conclusions
A fair number of thromboembolism and bleeding risk prediction tools have been developed to guide AC in NVAF and VTE. Incorporation of these scores into practice has been challenging in spite of their ability to often improve decision-making. Biomarkers offer the potential promise of more personalized risk assessment over time, and choosing biomarkers that are widely available will be important to greater use. Finally, with increasingly greater use of DOACs in clinical practice, risk scores will need to be further refined.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TJG is a consultant for Boehringer-Ingelheim; VAP, RKK, KK, JWM, and RSB have no relevant disclosures or conflicts of interest to report.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

