Abstract
Patients with peripheral artery disease (PAD) are at heightened risk of cardiovascular morbidity and mortality. We sought to evaluate the risk of concomitant coronary artery disease (CAD) in patients with symptomatic PAD versus PAD without diagnosed CAD, and whether ticagrelor was superior to clopidogrel in reducing that risk. The EUCLID trial randomized 13,885 patients with PAD to antithrombotic monotherapy with ticagrelor or clopidogrel. CAD was defined as prior myocardial infarction (MI), percutaneous coronary intervention (PCI), or coronary artery bypass graft (CABG) surgery. Median follow-up was 30 months. Among 4032 (29%) patients with PAD and CAD, 63% had prior MI, 54% prior PCI, and 38% prior CABG. After adjustment for baseline characteristics, patients with PAD and CAD had significantly higher rates of the primary endpoint (cardiovascular death/MI/stroke, 15.3% vs 8.9%, hazard ratio (HR) 1.50, 95% CI: 1.13–1.99; p=0.005), but no statistically significant increase in acute limb ischemia (HR 1.28, 95% CI: 0.57–2.85; p=0.55) or major bleeding (HR 1.10, 95% CI: 0.49–2.48; p=0.81) versus PAD without CAD. Among patients with PAD and CAD, there was no differential treatment effect between ticagrelor versus clopidogrel for the primary efficacy endpoint (HR 1.02, 95% CI: 0.87–1.19; p=0.84), acute limb ischemia (HR 1.03, 95% CI: 0.63–1.69; p=0.89), or major bleeding (HR 1.06, 95% CI: 0.66–1.69; p=0.81). There was a statistically significant interaction between prior coronary stent placement and study treatment (p=0.03) with a numerical reduction in the primary efficacy endpoint with ticagrelor versus clopidogrel (13.8% vs 16.8%, HR 0.82, 95% CI: 0.65–1.03; p=0.09). Patients with PAD and prior CAD had higher composite rates of cardiovascular death, MI, and ischemic stroke versus PAD without diagnosed CAD. There were no significant differences between ticagrelor and clopidogrel in cardiovascular events or major bleeding.
Keywords
Introduction
Patients with peripheral artery disease (PAD) are at heightened risk of cardiovascular morbidity and mortality and impaired quality of life. Many patients with PAD have clinical evidence of concomitant coronary artery disease (CAD) or a prior history of coronary revascularization and are defined as having polyvascular disease.1,2 Patients with polyvascular disease have a worse prognosis compared with patients with disease in a single arterial bed. 3 Accordingly, intensive secondary prevention strategies are a mainstay of treatment.
Antiplatelet therapy reduces the risk of myocardial infarction (MI), stroke, and cardiovascular death in patients with PAD and CAD. 4 Among patients with PAD, CAD, or prior stroke in the CAPRIE (Clopidogrel versus Aspirin in Patients at Risk of Ischemic Events) trial, monotherapy with clopidogrel versus aspirin reduced the risk of cardiovascular events, with the PAD subgroup showing the most favorable response to clopidogrel. 5 Ticagrelor is an oral, reversibly bound inhibitor of the P2Y12 receptor that has a greater and more consistent inhibition of platelet aggregation than clopidogrel. 6 Ticagrelor was shown to be more effective than clopidogrel in the setting of acute coronary syndrome (ACS) and versus placebo in patients with prior MI.7,8 Subgroup analyses of patients with CAD (ACS and prior MI) and PAD suggest that the absolute benefit of ticagrelor versus clopidogrel 9 or placebo 10 was amplified in patients with concomitant CAD and PAD.
The effect of ticagrelor versus clopidogrel was most recently evaluated in the EUCLID (Effects of Ticagrelor and Clopidogrel in Patients With Peripheral Artery Disease) trial, 11 which enrolled patients with symptomatic PAD. In EUCLID, monotherapy with ticagrelor was not shown to be superior to clopidogrel for the reduction of cardiovascular events. Sensitivity analysis of the primary endpoint revealed a nominally significant interaction (p=0.03) in the subgroup of patients with coronary stent implantation prior to randomization. 11 Patients with prior coronary stent implantation appeared to derive a marginal benefit from ticagrelor versus clopidogrel. 11 In this report, two major hypotheses were tested: (1) the risk of cardiovascular, limb, and bleeding events are higher in patients with PAD and CAD than PAD without diagnosed CAD, and (2) monotherapy with ticagrelor would be superior to clopidogrel in the subgroup of patients with PAD and CAD included in the EUCLID trial.
Methods
EUCLID was a prospective, multicenter, randomized, double-blind, event-driven study. It was approved by the institutional review committee of each participating site as well as by appropriate national ethics committees. All patients gave written informed consent. The details of the trial design 12 and the main results have been published previously. 11 Briefly, 13,885 patients ≥ 50 years of age with lower extremity PAD were randomly assigned to receive monotherapy with either ticagrelor (90 mg twice daily) or clopidogrel (75 mg once daily) and followed for a median of 30 months.
This report includes the 4032 (29%) patients with CAD who were identified in the EUCLID study. To fulfill criteria for CAD, patients had a history of prior MI, prior percutaneous coronary intervention (PCI), or prior coronary artery bypass graft (CABG) surgery as reported by the site investigator on the case report form.
Endpoints
The primary efficacy endpoint was time from randomization to first occurrence of any event in the composite of adjudicated cardiovascular death, MI, or ischemic stroke. Secondary endpoints included the primary composite endpoint plus acute limb ischemia (ALI), cardiovascular death alone, MI, all-cause mortality, the primary composite endpoint including all-cause stroke, ALI alone, time to lower extremity revascularization, and time to any revascularization. The primary safety endpoint was Thrombolysis in Myocardial Infarction (TIMI) major bleeding. The components of the primary efficacy endpoint, the primary safety endpoint, and all hospitalizations for ALI were adjudicated by an independent clinical events classification committee.
Hospitalization for ALI was not considered a standard endpoint at the time of protocol development. However, the protocol was amended in December 2013 to collect source data for all hospitalizations for PAD, peripheral revascularization, and amputation. Trained adjudicators, while blinded to treatment assignment, reviewed all information to determine whether patients had ALI defined as a hospitalization involving a rapid or sudden decrease in limb perfusion and either (1) a new pulse deficit, rest pain, pallor, paresthesia, or paralysis; or (2) confirmation of arterial obstruction by limb hemodynamics (ankle or toe pressure), imaging, intra-operative findings, or pathological evaluation.
Statistical analysis
The primary and secondary efficacy endpoints were analyzed using the intention-to-treat approach and were performed on the full analysis set consisting of all patients randomized to the study drug, irrespective of study drug adherence or withdrawal of consent. Patients who withdrew consent to participate in the study were included up to the date of their study termination with the exception of an analysis of all-cause mortality that used information from all patients for whom vital status could be determined via publically available records. The safety analysis set included all patients who received at least one dose of study drug while on treatment, defined as within 7 days of the last dose of study drug.
The primary and secondary endpoints were analyzed in the population of patients with a medical history of CAD in time-to-event analyses in a Cox proportional hazards model with a factor for treatment group. Unadjusted p-values and confidence intervals (CIs) for the hazard ratios (HRs) were based on the Wald statistic. Kaplan–Meier estimates of the cumulative proportion of patients with events were calculated and plotted.
The p-values for differences between baseline characteristics were calculated using chi-squared (cell size ≥ 5) and Fisher’s exact (cell size < 5) tests for categorical variables, and Wilcoxon’s test for continuous variables if the assumption for normality of distribution of the continuous variable was not met. Otherwise, continuous measures were tested using the t-test for two groups. Adjusted analyses for the primary composite endpoint, components of the primary composite endpoint, ALI, and major bleeding (using baseline variables shown in Table 1) were performed to determine whether patients with a medical history of CAD had higher rates of cardiovascular and acute limb events when compared with patients who did not.
Baseline characteristics.
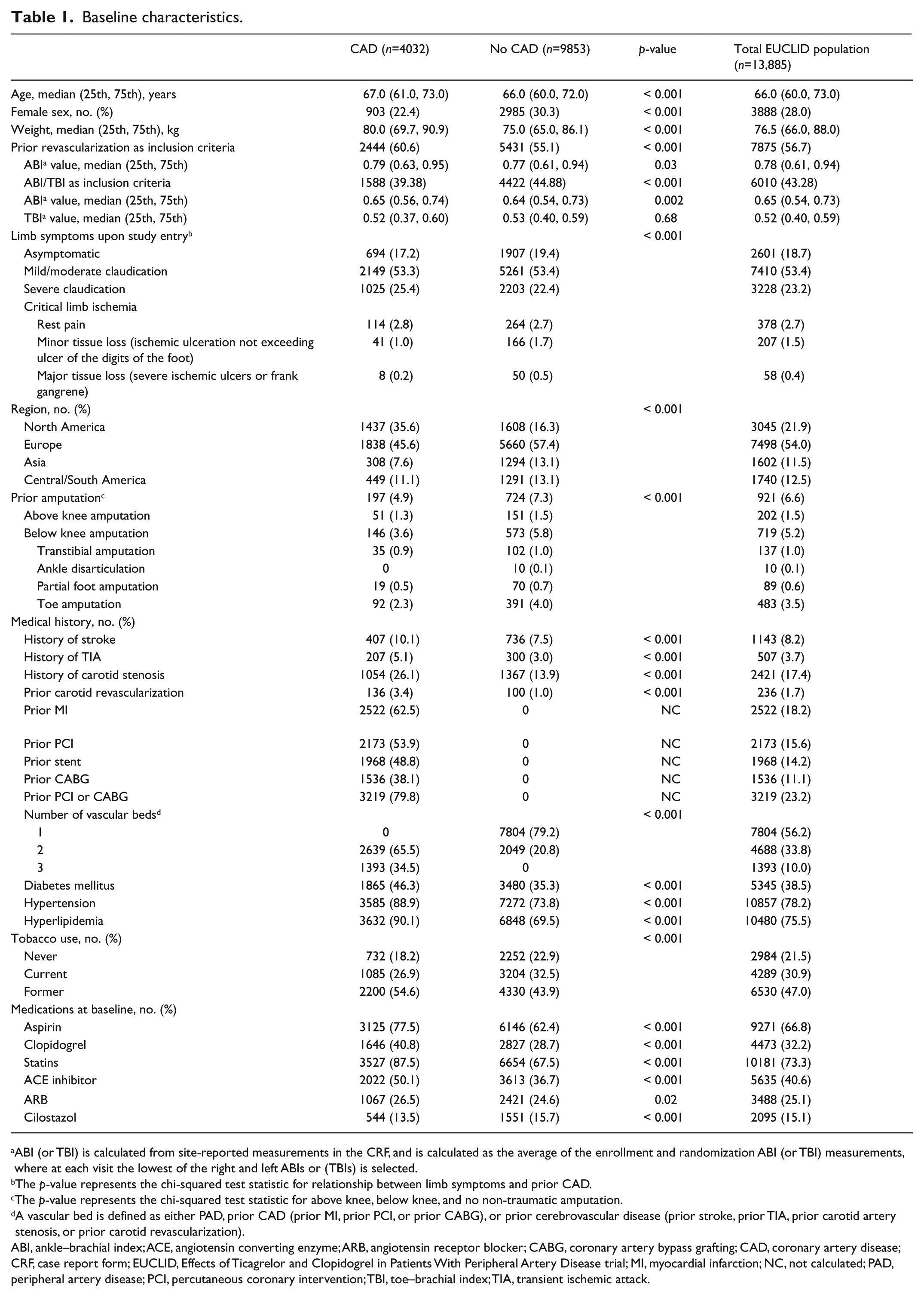
ABI (or TBI) is calculated from site-reported measurements in the CRF, and is calculated as the average of the enrollment and randomization ABI (or TBI) measurements, where at each visit the lowest of the right and left ABIs or (TBIs) is selected.
The p-value represents the chi-squared test statistic for relationship between limb symptoms and prior CAD.
The p-value represents the chi-squared test statistic for above knee, below knee, and no non-traumatic amputation.
A vascular bed is defined as either PAD, prior CAD (prior MI, prior PCI, or prior CABG), or prior cerebrovascular disease (prior stroke, prior TIA, prior carotid artery stenosis, or prior carotid revascularization).
ABI, ankle–brachial index; ACE, angiotensin converting enzyme; ARB, angiotensin receptor blocker; CABG, coronary artery bypass grafting; CAD, coronary artery disease; CRF, case report form; EUCLID, Effects of Ticagrelor and Clopidogrel in Patients With Peripheral Artery Disease trial; MI, myocardial infarction; NC, not calculated; PAD, peripheral artery disease; PCI, percutaneous coronary intervention; TBI, toe–brachial index; TIA, transient ischemic attack.
SAS version 9.4 (SAS Institute, Inc., Cary, NC, USA) was used for all analyses and all tests were two-sided.
Results
A total of 13,885 participants with symptomatic PAD were randomized in the EUCLID trial, including 4032 (29%) participants with concomitant CAD (Figure 1). Of these, 2522 (62.5%) had prior MI, 2173 (53.9%) prior PCI, and 1536 (38.1%) prior CABG.
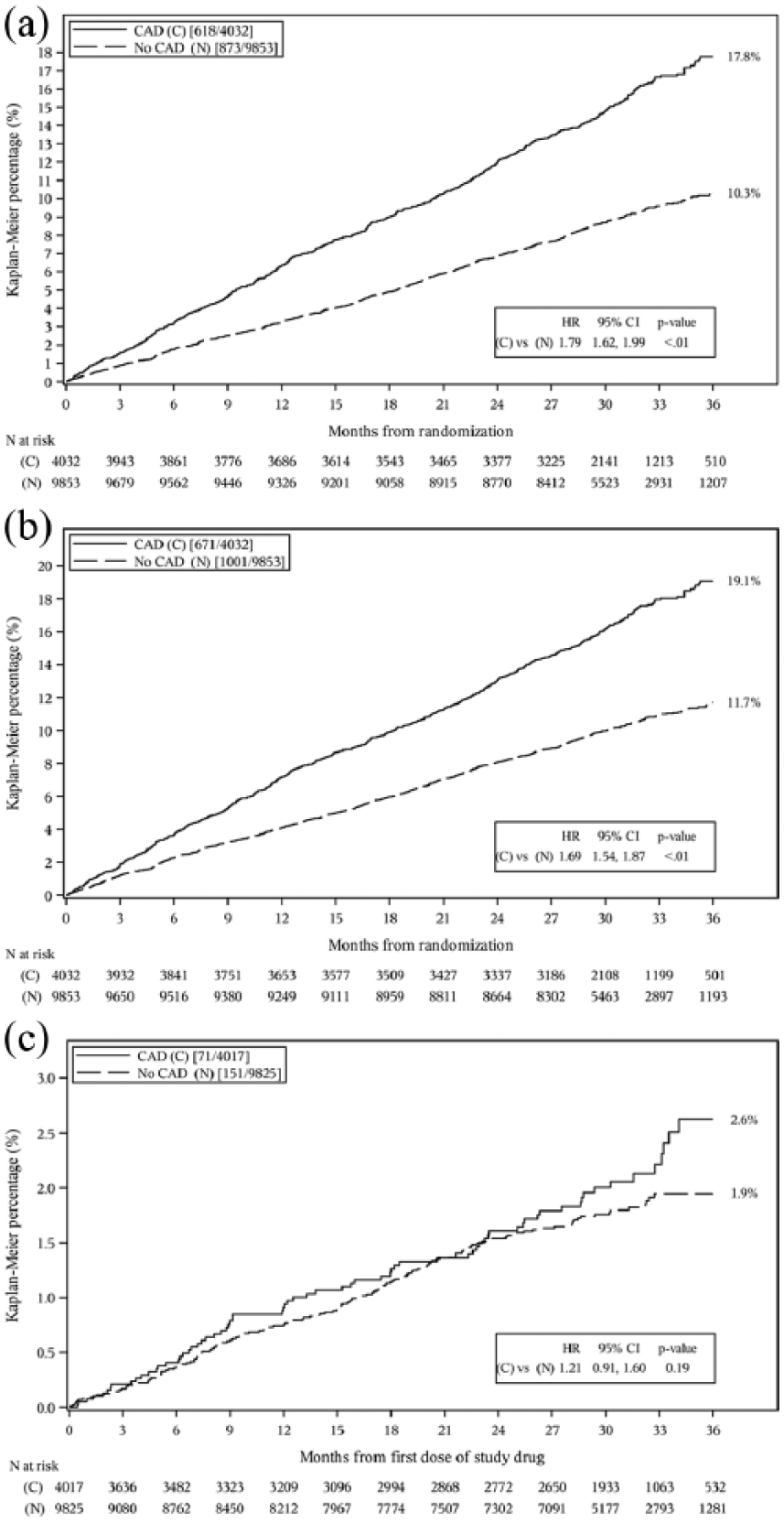
Kaplan–Meier plot of the (a) primary efficacy endpoint (composite of cardiovascular death/MI/stroke); (b) composite of cardiovascular death/MI/stroke/acute limb ischemia; and (c) TIMI major bleeding.
Profile of PAD and CAD
Patients with PAD and CAD were older, more frequently male, and heavier compared with patients without CAD (Table 1). A history of stroke, transient ischemic attack, carotid stenosis, diabetes mellitus, hypertension, and hyperlipidemia were reported more frequently in patients with PAD and CAD compared with PAD without diagnosed CAD. A higher proportion of patients with PAD and CAD were enrolled based on prior lower extremity revascularization (60.6% vs 55.1% with PAD without diagnosed CAD; p < 0.001). Prior amputation at baseline was less common in patients with PAD and CAD than PAD without diagnosed CAD (4.9% vs 7.3%; p < 0.001). Patients with prior CAD were also more commonly treated at baseline with cardioprotective medications including aspirin, clopidogrel, statins, and angiotensin converting enzyme inhibitors or angiotensin receptor blockers prior to participation in the study, as compared with those with no medical history of CAD. Conversely, use of cilostazol was more common in PAD without diagnosed CAD (Table 1).
Outcomes in PAD and CAD
The unadjusted composite rate of cardiovascular death, MI, or ischemic stroke was 15.3% in patients with PAD and CAD and 8.9% in patients with PAD without CAD (HR 1.79, 95% CI: 1.62–1.99; p < 0.001) as were individual components of the composite. ALI occurred in 1.6% of patients with PAD and CAD and 1.7% of patients with PAD without diagnosed CAD (HR 0.92, 95% CI: 0.69–1.22; p=0.55). Major bleeding occurred in 1.8% of patients with PAD and CAD and 1.5% of patients with PAD without diagnosed CAD (HR 1.21, 95% CI: 0.91–1.60; p=0.19) (Figure 1).
After adjustment for baseline variables, patients with PAD enrolled with CAD had statistically significantly higher rates of the primary composite endpoint (HR 1.50, 95% CI: 1.13–1.99; p=0.005) and MI (HR 2.19, 95% CI: 1.45–3.32; p < 0.001). No statistically significant difference between groups was seen for rates of ischemic stroke, cardiovascular death, ALI, or major bleeding (Table 2).
Adjusted event rates in patients enrolled with and without concomitant CAD.
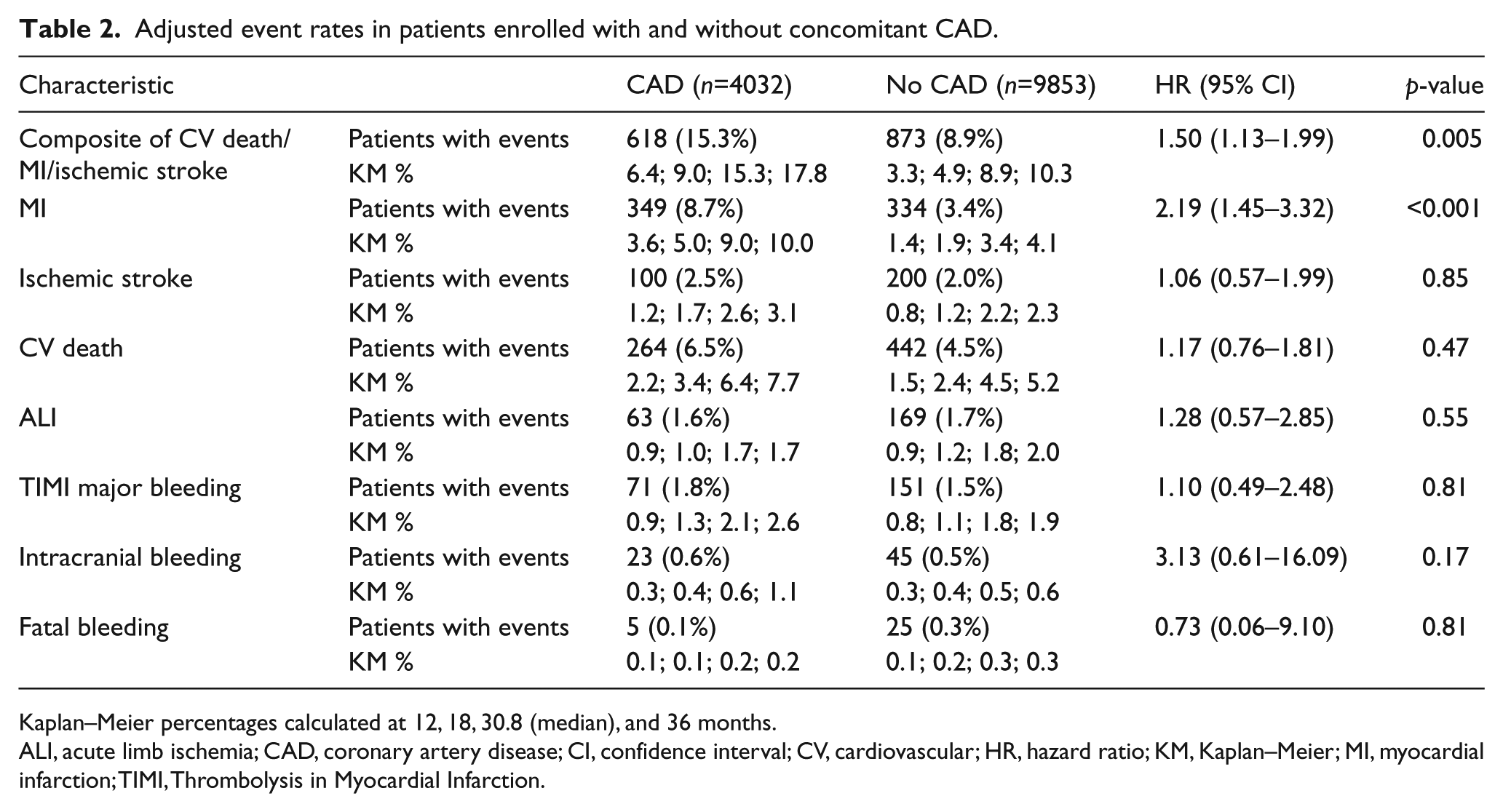
Kaplan–Meier percentages calculated at 12, 18, 30.8 (median), and 36 months.
ALI, acute limb ischemia; CAD, coronary artery disease; CV, cardiovascular; HR, hazard ratio; KM, Kaplan–Meier; MI, myocardial infarction; TIMI, Thrombolysis in Myocardial Infarction.
Effect of ticagrelor versus clopidogrel in PAD and CAD
In the subset of 4032 patients with CAD, 2019 were randomized to ticagrelor and 2013 to clopidogrel. The baseline characteristics of these patients in the two randomized arms were well matched (Supplemental Table 1). The composite rate of cardiovascular death, MI, or ischemic stroke was 15.4% in patients treated with ticagrelor and 15.3% in those treated with clopidogrel (HR 1.02, 95% CI: 0.87–1.19; p=0.84), with no statistical interaction for subjects with a prior history of CAD. No difference was observed for the individual endpoints. All-cause mortality (11.4% vs 10.3%, HR 1.11, 95% CI: 0.92–1.34; p=0.27) and ALI (1.6% vs 1.5%, HR 1.03, 95% CI: 0.63–1.69; p=0.89) were not significantly different between the treatment groups. Other key secondary and composite endpoints, including coronary revascularization, were also not significantly different between treatment groups, although there was a significant interaction for ticagrelor for history of coronary or carotid revascularization and for history of coronary stent implantation (Table 3).
Efficacy endpoints for patients with prior CAD by treatment group.
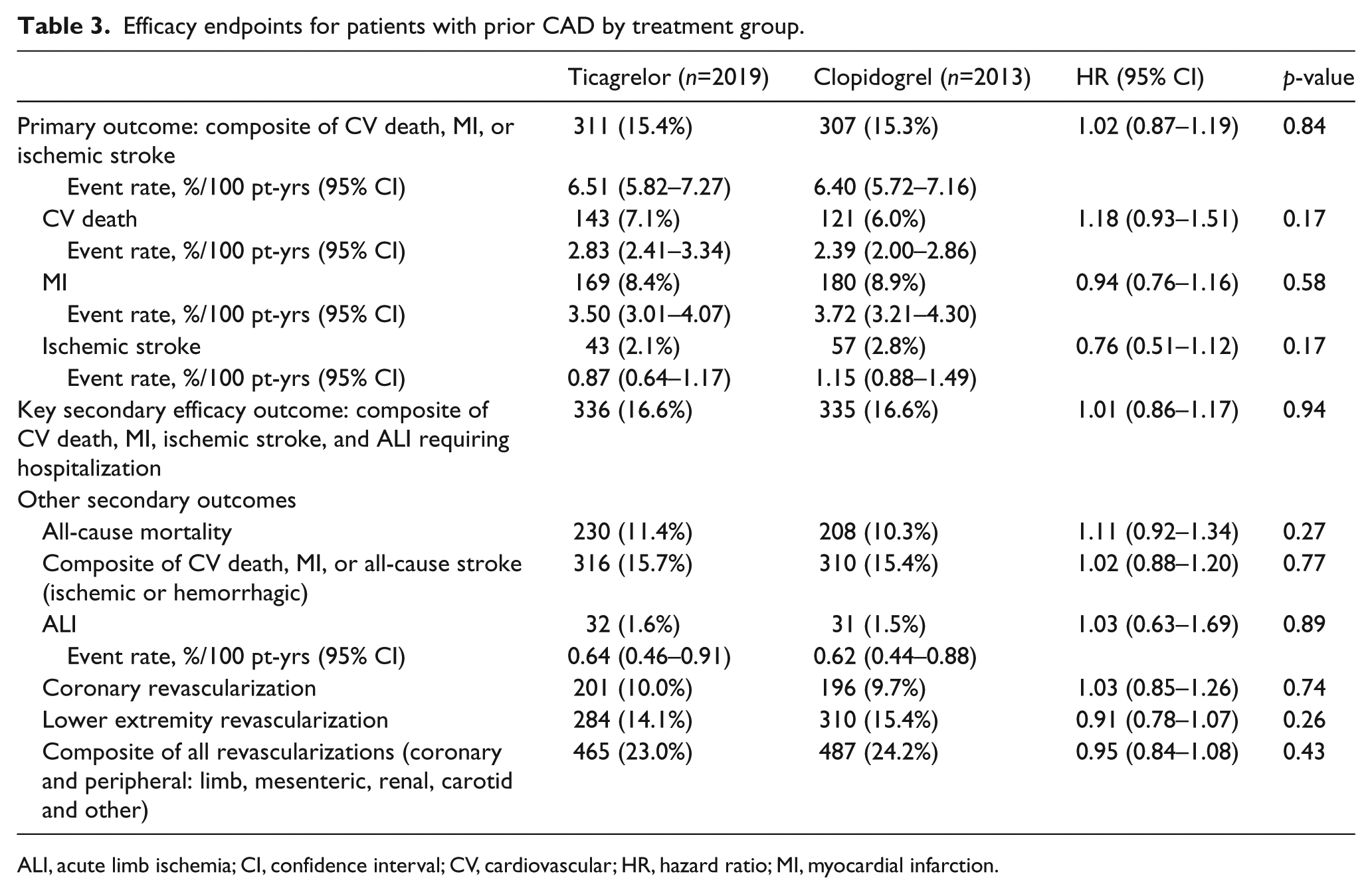
ALI, acute limb ischemia; CV, cardiovascular; HR, hazard ratio; MI, myocardial infarction; pt, patient.
The primary safety endpoint of TIMI major bleeding occurred in 1.7% of patients treated with ticagrelor versus 1.8% of clopidogrel-treated patients (HR 1.06, 95% CI: 0.66–1.69; p=0.81). Fatal bleeding, intracranial bleeding, and TIMI minor bleeding were all low frequency events and not significantly different between treatment groups (Table 4).
Safety endpoints for patients with a medical history of CAD by treatment group.

Kaplan–Meier percentages calculated at 12, 18, 30.8 (median), and 36 months.
HR, hazard ratio; KM, Kaplan–Meier; NC, not calcluated; TIMI, Thrombolysis in Myocardial Infarction.
There was no heterogeneity of efficacy or safety treatment effects observed in patients enrolled with prior MI, prior PCI, or prior CABG (Supplemental Table 2).
In the primary analyses, a nominally significant interaction was observed for the treatment effect for the primary endpoint in the group of patients with PAD with a prior coronary stent at randomization compared with those without a stent (p for interaction = 0.03). 11 Among 1964 patients with PAD and a history of a coronary stent, there was a trend toward benefit with ticagrelor for the composite rate of cardiovascular death, MI, or ischemic stroke versus clopidogrel (13.8% vs 16.8%, HR 0.82, 95% CI: 0.65–1.03; p=0.09). Other secondary and safety endpoints are presented in Supplemental Table 3.
Discussion
EUCLID enrolled patients with symptomatic lower extremity PAD either based on a prior history of limb revascularization or a low ABI indicating hemodynamic compromise in the lower extremity. In this patient population, 29% had a prior history of CAD defined as a prior MI or coronary revascularization. The subgroup with PAD and CAD had a markedly elevated risk of major cardiovascular events indicating that polyvascular disease is a potent mediator of increased risk, as previously described, despite a higher use of cardioprotective medications compared with those who had PAD only. 3 There was no difference in ALI between groups, suggesting that a history of CAD is not a driver of ALI independent of PAD. The primary efficacy and safety endpoints occurred with similar frequency in patients with PAD and CAD treated with ticagrelor or clopidogrel. This finding was consistent in patients with PAD who entered the study with a prior history of MI, PCI, or CABG.
Patients with PAD without CAD had an annualized cardiovascular death, MI, or stroke incidence of 3.3%, which is considerably elevated compared with a healthy, age-matched population.13,14 Patients with PAD without a history of CAD are often not recognized as having a systemic cardiovascular condition and have been well described as receiving less intensive risk-reduction therapies.15–17 In EUCLID, this phenomenon was also observed: at baseline, patients with PAD and no history of CAD had a lower use of antiplatelet therapies, statins, angiotensin converting enzyme and angiotensin receptor blocker drugs. This observation underscores the need for continued education of both patients with PAD and their providers in the need for appropriate use of secondary prevention strategies. 18
Patients with symptomatic PAD are a high-risk group for cardiovascular morbidity and mortality. The composite endpoint of cardiovascular death, MI, or ischemic stroke occurred in 15.3% of patients with PAD and CAD, a 6.4% absolute increase compared with PAD without CAD. Concomitant CAD increases the rate of MI, ischemic stroke, and cardiovascular death. After adjustment for baseline differences, the risk of MI was more than twofold higher in patients with symptomatic PAD and CAD than PAD without diagnosed CAD. These findings underscore the importance of identifying polyvascular disease in the clinic to inform future cardiovascular risk.
An important question remaining regards treatment strategies to reduce risk and event rates in this population. In trials enrolling patients with ACS or prior MI, more potent antiplatelet therapy is more effective at decreasing cardiovascular events. The PLATO (PLATelet inhibition and patient Outcomes) trial used a dual antiplatelet strategy (in contrast to EUCLID in which monotherapy was investigated) and demonstrated a similar benefit of ticagrelor versus clopidogrel in patients with ACS with and without PAD. 9 However, owing to the higher risk of cardiovascular events in the PAD subgroup, a greater absolute benefit was observed in patients with versus without PAD. 9 In the PEGASUS-TIMI 54 (Prevention of Cardiovascular Events in Patients With Prior Heart Attack Using Ticagrelor Compared to Placebo on a Background of Aspirin–Thrombolysis In Myocardial Infarction 54) trial, patients with a prior MI and PAD experienced a robust absolute risk reduction of 4.1% at 3 years with ticagrelor versus placebo, in which both groups were taking aspirin, relative to 1% for those without PAD. 10 In a post hoc analysis of patients with PAD enrolled in the CHARISMA (Clopidogrel for High Atherothrombotic Risk and Ischemic Stabilization, Management, and Avoidance) trial, dual antiplatelet therapy with aspirin plus clopidogrel versus aspirin alone did not reduce the composite of cardiovascular events. 19 With this background in mind, the neutral effect of ticagrelor versus clopidogrel observed in the current analysis, even in patients with a prior history of CAD, indicates that potent platelet inhibition with monotherapy targeting the P2Y12 pathway did not result in additional clinical benefit in an outpatient population with PAD with or without CAD.
Study limitations
The EUCLID trial was not designed to specifically evaluate the use of ticagrelor in patients with symptomatic PAD and concomitant CAD. The limited statistical power available and the well-known potential confounding of post hoc analyses require caution and demonstrate the need for adequately designed and powered studies in this high-risk cohort. Differences in outcomes between patients with PAD and CAD versus PAD without diagnosed CAD should be interpreted within the context of between-group differences (Table 1) and multiple comparisons between groups. The EUCLID trial was an antiplatelet monotherapy trial; patients with PAD requiring dual antiplatelet therapy were excluded from this trial. Clopidogrel was an active comparator and has potent antiplatelet effects. Finally, patients who were homozygous for loss-of-function alleles to clopidogrel were excluded before randomization.
Conclusions
In patients with symptomatic PAD, concomitant CAD is associated with a heightened risk of cardiovascular events, even after adjusting for their extensive comorbidities. In these patients, ticagrelor did not reduce the primary composite endpoint of cardiovascular mortality, MI, or ischemic stroke compared with clopidogrel.
Supplemental Material
VMJ775594_Supplementary_material – Supplemental material for Ticagrelor versus clopidogrel in patients with symptomatic peripheral artery disease and prior coronary artery disease: Insights from the EUCLID trial
Supplemental material, VMJ775594_Supplementary_material for Ticagrelor versus clopidogrel in patients with symptomatic peripheral artery disease and prior coronary artery disease: Insights from the EUCLID trial by Jeffrey S Berger, Beth L Abramson, Renato D Lopes, Gretchen Heizer, Frank W Rockhold, Iris Baumgartner, F Gerry R Fowkes, Peter Held, Brian G Katona, Lars Norgren, W Schuyler Jones, Marcus Millegård, Juuso Blomster, Craig Reist, William R Hiatt, Manesh R Patel and Kenneth W Mahaffey in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Berger: advisory board fees from Janssen, Merck, and Takeda. Abramson: none. Lopes: research grants from Bristol-Myers Squibb, GlaxoSmithKline, Medtronic, and Pfizer; consulting or advisory board fees from Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, GlaxoSmithKline, Medtronic, Merck, Pfizer, and Portola Pharmaceuticals. Heizer: none. Rockhold: none. Baumgartner: research grants from Abbott Vascular, Amgen, Boston Scientific, Cook Medical, Optimed, Promedics, and Terumo Medical; advisory board fees from Bayer and Sanofi. Fowkes: consulting fees from Bayer and Merck. Held: employee of AstraZeneca at the time of the study. Katona: employee of AstraZenca. Norgren: research grants from AnGes MG and Mitsubishi Tanabe Pharma; consulting or advisory board fees from AnGes MG, Bayer, Cesca Therapeutics, Mitsubishi Tanabe Pharma, and Pluristem Therapeutics. Jones: none. Millegård: employee of AstraZeneca. Blomster: employee of AstraZeneca at the time of the study. Reist: none. Hiatt: none. Patel: research grants from AstraZeneca, Bayer, HeartFlow, Janssen, Medtronic, and NHLBI; consulting fees from AstraZeneca, Bayer, and Janssen. Mahaffey: research grants from Amgen, Daiichi Sankyo, Johnson & Johnson, Medtronic, Merck, St Jude Medical, and Tenax Therapeutics; consulting fees from BAROnova, Bayer, Bio2 Medical, Boehringer Ingelheim, Bristol-Myers Squibb, Cubist, Eli Lilly, Epson, Forest Laboratories, GlaxoSmithKline, Johnson & Johnson, Medtronic, Merck, Myokardia, Omthera Pharmaceuticals, Portola, Purdue Pharma, The Medicines Company, Theravance, Vindico, and WebMD; equity in BioPrint Fitness.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work and the EUCLID trial were supported by AstraZeneca. The sponsor had no role in the study design, data analysis, or interpretation of results.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
