Abstract
Lower extremity arterial thromboembolism is associated with significant morbidity and mortality. We sought to establish temporal trends in the incidence, management and outcomes of lower extremity arterial thromboembolism within the Veterans Affairs Healthcare System (VAHS). We identified patients admitted to VAHS between 2003 and 2014 with a primary diagnosis of lower extremity arterial thromboembolism. Medical and procedural management were ascertained from pharmaceutical and administrative data. Subsequent rates of major adverse limb events (MALE), major adverse cardiovascular events (MACE), and mortality were calculated using Cox proportional hazards models. From 2003 to 2014, there were 10,636 patients hospitalized for lower extremity thromboembolism across 140 facilities, of which 8474 patients had adequate comorbid information for analysis. Age-adjusted incidence decreased from 7.98 per 100,000 patients (95% CI: 7.28–8.75) in 2003 to 3.54 (95% CI: 3.14–3.99) in 2014. On average, the likelihood of receiving anti-platelet or anti-thrombotic therapy increased 2.3% (95% CI: 1.2–3.4%) per year during this time period and the likelihood of undergoing endovascular revascularization increased 4.0% (95% CI: 2.7–5.4%) per year. Clinical outcomes remained constant over time, with similar rates of MALE, MACE and mortality at 1 year after adjustment. In conclusion, the incidence of lower extremity arterial thromboembolism is decreasing, with increasing utilization of anti-thrombotic therapies and endovascular revascularization among those with this condition. Despite this evolution in management, patients with lower extremity thromboembolism continue to experience high rates of amputation and death within a year of the index event.
Introduction
Peripheral artery disease (PAD) affects a large segment of the population, with an age-adjusted prevalence ranging from 12% to 20%. 1 The clinical manifestations of symptomatic PAD are broad, ranging from intermittent claudication to critical limb ischemia. A number of observational cohorts have defined the prevalence of these conditions in various populations, as well as their association with adverse clinical outcomes.2–5 The data evaluating the management and clinical outcomes of an acute thrombosis of the lower extremity vasculature, however, is less well defined and could have important implications for optimal patient care.
Lower extremity arterial thromboembolism encompasses acute limb ischemia (ALI) as well as severe exacerbations of critical limb ischemia (CLI) and is associated with significant morbidity and mortality. Adverse outcomes in this population remain unacceptably high, with limb loss approaching 15–50% within 5 years after the index presentation.6–9 Previous research has sought to better understand the medical and procedural management of lower extremity arterial thrombosis with the hope of improving outcomes. However, these studies have been limited by small sample sizes and the inability to capture longitudinal outcomes.6–9 A comprehensive evaluation of the temporal trends in management and clinical outcomes is thus needed to inform clinicians on opportunities for improved care.
With this in mind, the present project sought to establish the temporal trends in the incidence, management, and outcomes of lower extremity arterial thromboembolism within the largest integrated healthcare system in the United States: the Veterans Affairs Healthcare System (VAHS). This analysis provides insight into the clinical impact of this condition, as well as its evolving medical and procedural management.
Methods
Population
Administrative, demographic, billing data for all patients receiving care within the VAHS are included in an institutionally maintained registry, the Veterans Affairs National Patient Care Database (NPCD). 10 The present project identified adult (> 18 years) patients enrolled in this registry between October 1, 2002 and September 30, 2014 with a primary diagnosis of lower extremity arterial thrombosis, based on the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes. The diagnostic codes for this condition (444.X) have been previously used and validated in alternative administrative datasets.11,12 The present project was reviewed and approved by the Colorado Multiple Institutions Review Board (COMIRB), with a waiver of informed consent.
Measurements
The present analysis leveraged the longitudinal data available from the VA Corporate Data Warehouse (CDW) to obtain patient demographic information as well as patient comorbidities, laboratory data, and medication prescription refills. Anti-platelet agents included clopidogrel, prasugrel, ticagrelor, ticlopidine, and vorapaxar, while anti-thrombotic therapies were comprised of apixaban, dabigatran, rivaroxaban, and warfarin. Procedural management of lower extremity arterial thromboembolism was determined using ICD-9 Procedure Codes for both endovascular and surgical therapies, similar to prior analyses. 11 In addition, Current Procedural Terminology (CPT) codes validated in previous analyses were used to identify additional peripheral bypass procedures as detailed in Supplemental Table 1. 5
Validation
The diagnostic codes used to identify the primary diagnosis and procedural interventions were validated in a subpopulation of patients. For this process, 100 patients were selected at random for chart review by an experienced interventional cardiologist. The patient was deemed to have true lower extremity arterial thromboembolism leading to ALI if the case met the definition previously published by the Trans-Atlantic Society Consensus II: a sudden decrease in limb perfusion that causes a potential threat to limb viability secondary to a thrombotic or embolic event. 13 The procedure characteristics were determined based on a review of the operative report in the electronic medical record. As summarized in Supplemental Table 2, the billing codes for lower extremity arterial thromboembolism had a poor positive predictive value for the diagnosis of ALI (0.41, 95% confidence interval (CI): 0.31–0.51). The procedural codes for endovascular interventions were more sensitive and specific, with reasonable positive (0.83, 95% CI: 0.63–0.95) and negative (0.95, 95% CI: 0.87–0.99) predictive values. Similarly, the procedural codes for surgical interventions had excellent positive (1.00, 95% CI: 0.92–1.00) and negative (0.94, 95% CI: 0.85–0.99) predictive values after chart review.
Outcomes
Clinical outcomes were derived from administrative and clinical data, including fee-based data for care received outside the VA system. Major adverse limb events (MALE) included major amputation above the ankle (ICD-9 Procedure Codes: 84.13–84.18) or repeat revascularization of the index limb (endovascular or surgical) after the initial hospitalization. Major adverse cardiovascular events (MACE) included readmission for myocardial infarction (ICD-9: 410.01, 410.00, 410.91, 410.90, 410.81, 410.80, 410.71, 410.70, 410.61, 410.60, 410.51, 410.50, 410.41, 410.40, 410.31, 410.30, 410.21, 410.20, 410.11, 410.10), readmission for stroke (ICD-9: 346.63, 346.62, 346.61, 346.60, 997.02, 434.91, 434.11, 434.01, 433.81, 433.31, 433.21, 433.11, 433.01, 431, 433.91) or a death of any cause. Mortality was ascertained from the VHA Vital Status File. This resource includes data from the Beneficiary Identification Records Locator Subsystem (BIRLS) Death File, VA Medicare Vital Status File, VA Patient Treatment File, and the Social Security Administration Master File. 14
Analysis
Temporal trends in incidence
A quasi-Poisson model of incident hospitalizations was created with a non-linear function of fiscal year, accounting for age and allowing for over-dispersion. Counts of patients who are enrolled and using the VAHS were included as the offset, stratified by age group (< 40, 40–44, 45–49, 50–54, 55–59, 60–64, 65–69, 70–74, 75–79, 80–84, and 85+) and fiscal year. Hospitalizations were reported per 100,000 VAHS users, assuming a distribution of users across age groups representing the population from 2010.
Temporal trends in medical management
Medications issued at discharge (anti-platelet, anti-thrombotic or statins) were fit as a function of year, using modified Poisson methods. Generalized estimating equations were utilized to account for hospital-level clustering with exchangeable working correlation structure. Hospitals with less than 10 patients were excluded (38 hospitals, 1.47% of patients). The modified Poisson model was subsequently fit separately for anti-platelet and anti-thrombotic medications.
Temporal trends in procedural management
Use of an endovascular or surgical procedure during index hospitalization was fit as a function of year, using modified Poisson methods. Generalized estimating equations were utilized to account for hospital-level clustering with exchangeable working correlation structure. Hospitals with less than 10 patients throughout the study period were excluded (38 hospitals, 1.47% of patients). Other sites within the integrated healthcare system did not contribute any patients. The modified Poisson model was subsequently fit separately for endovascular and surgical procedures.
Temporal trends in outcomes
To evaluate temporal trends in clinical outcomes following hospitalizations between 2005 and 2014, Cox proportional hazards models of time to event post-discharge were fit as a function of the year of hospitalization. Models were fit separately for three different clinical outcomes: MALE, MACE, and death. Frailty models were utilized to account for hospital-level clustering.
Deaths in the year following discharge were considered as a competing risk for MALE. Sensitivity analyses evaluated the potential bias induced by treating death as an independent censoring event in a cause-specific analysis. This included re-analyses under two extreme assumptions. 15 The first assumption was that patients who died within the year following discharge experienced a limb event at that time. The second assumption was that patients who died within the year following discharge would have remained event-free for the duration of follow-up. We also considered a subdistribution hazards model (cumulative incidence), which included in the risk set all patients free of a limb event at any given time, regardless of whether a death had occurred. This contrasts with the cause-specific model above, which includes in the risk set all patients free of a limb event and still alive at any given time. 16 Finally, we fit the previously proposed shared frailty model 17 of the joint hazards for major adverse limb event and death. This model allows for and explicitly estimates the dependence between semi-competing risks, such as limb events and death.
All statistical analyses were performed with SAS software, version 9.4 (SAS Institute, Cary, NC, USA and R 3.4.0 (R Core Team, 2017, https://www.R-project.org/). A p-value < 0.05 was considered statistically significant.
Results
Population
During the time period under investigation, 10,636 patients were hospitalized for lower extremity arterial thromboembolism across 140 VAHS facilities. The demographic and clinical characteristics of the 8474 patients who were hospitalized between 2005 and 2014 and had adequate comorbidity information for analysis are summarized in Table 1. The population was predominantly caucasian (81%), male (98%), and had a mean age of 65 (standard deviation (SD) 10). Prior diagnosis of peripheral artery disease (76%) was common, as were other significant medical comorbidities including hypertension (79%), diabetes (55%), and congestive heart failure (16%)
Patient demographics.
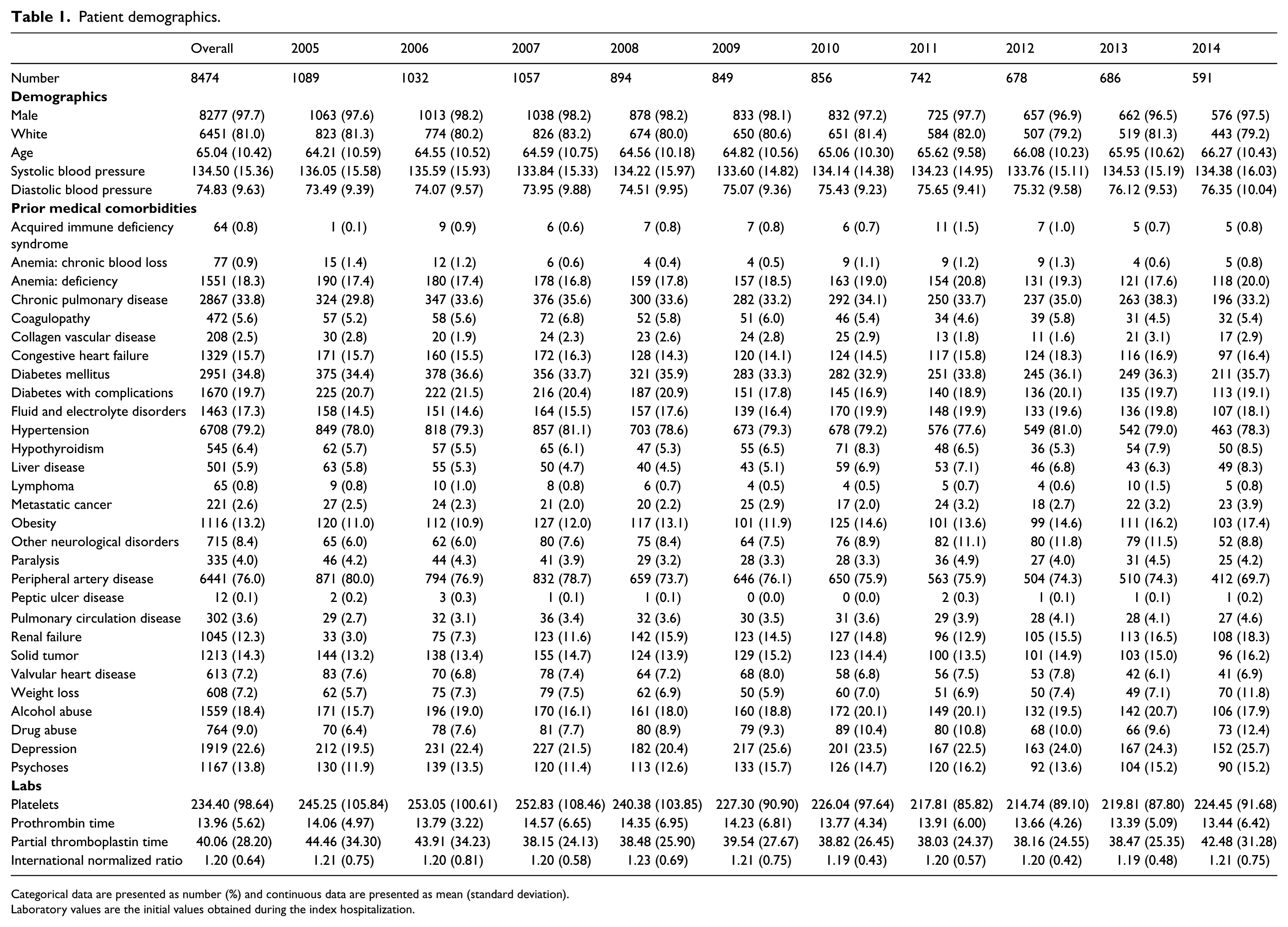
Categorical data are presented as number (%) and continuous data are presented as mean (standard deviation).
Laboratory values are the initial values obtained during the index hospitalization.
Incidence
The age-adjusted incidence of lower extremity arterial thromboembolism was 7.98 hospitalizations per 100,000 VAHS users (95% CI: 7.28–8.75) in fiscal year 2003. Adjusting for fiscal year, the incidence was significantly different across age groups, varying between 0.53 (95% CI: 0.36–0.77) for those less than 40 years of age and 13.10 (95% CI: 11.96–14.36) among those 60–64 years of age in 2003 (Supplemental Figure 1). Over the course of the study period, the incidence of lower extremity arterial thromboembolism hospitalizations demonstrated a non-linear trend, decreasing from 7.98 (95% CI: 7.28–8.75) in 2003 to 3.54 (95% CI: 3.14–3.99) per 100,000 VAHS patients in fiscal year 2014 (Figure 1).
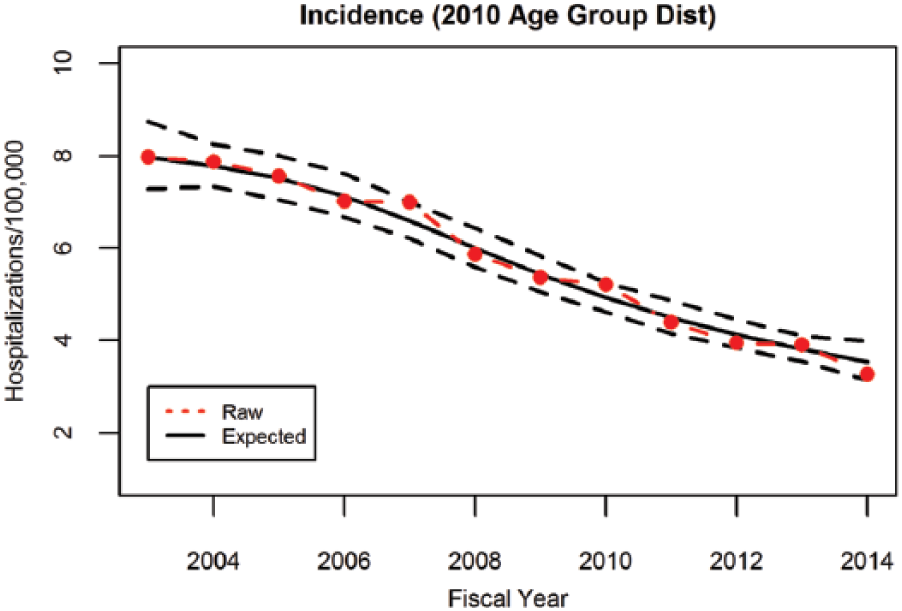
Incidence of lower extremity arterial thromboembolism. There was a significant decline in the number of admissions for this condition over time, decreasing from 7.98 (95% CI: 7.28–8.75) in 2003 to 3.54 (95% CI: 3.14–3.99) in fiscal year 2014.
Medical management
The temporal trend in medical management among patients hospitalized with lower extremity arterial thromboembolism is depicted in Figure 2. As shown, there was a significant increase in administration of anti-platelet or anti-thrombotic medications to patients over the course of investigation (p < 0.001). In 2003, the risk of receiving either anti-thrombotic or anti-platelet medications was 0.55 (95% CI: 0.52–0.58) and this increased to 0.63 (95% CI: 0.60–0.67) in 2014. On average, the risk of receiving either medication increased 2.3% (95% CI: 1.2–3.4%) each year.
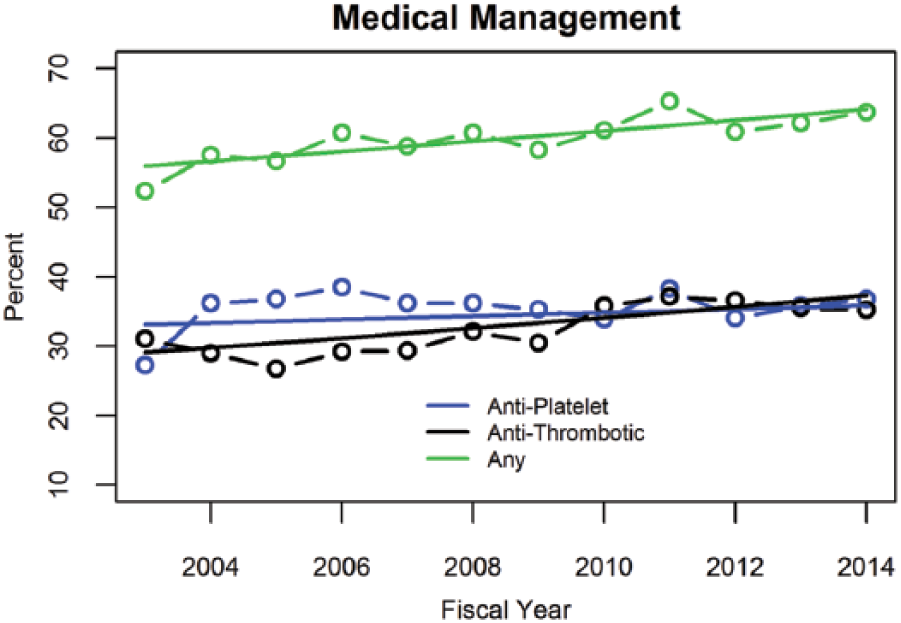
Trends in medical management. The prescription of anti-platelet and anti-thrombotic therapies increased over time, with the average risk of receiving either medication increasing 2.3% (95% CI: 1.2%–3.4%) each year.
Considering anti-platelet medications alone, there was an increase in administration of anti-platelets, with or without anti-thrombotic medications, between 2003 and 2014 (p=0.289), though this trend was not statistically significant. In 2003, the average risk of being prescribed anti-platelet medication other than aspirin was 0.33 (95% CI: 0.30–0.36) and increased to 0.36 (95% CI: 0.32–0.39) in 2014. On average, the risk of receiving anti-platelet medications increased 0.7% (95% CI: –0.6 to 2.0%) each year. The use of anti-platelet therapies stratified by the type of revascularization modality is shown in Supplemental Figure 2.
Administration of anti-thrombotic medications, with or without anti-platelet medications, also showed a significant increase between 2003 and 2014 (p < 0.001). In 2003, the risk of being administered anti-thrombotics, with or without anti-platelets, was 0.29 (95% CI: 0.26–0.31) and increased to 0.37 (95% CI: 0.34–0.39) in 2014. On average, the risk of receiving anti-thrombotic medications increased 1.4% (95% CI: 0.5–2.4%) each year.
Similarly, statin prescriptions at discharge increased over the time period under investigation. The initial proportion of patients prescribed statins in 2003 was 54%, and increased over the following decade to 71% in 2014 (Supplemental Table 3).
Procedural management
A number of patients underwent major amputation during the index hospitalization, approximating 5.5% of the total cohort. As shown in Supplemental Table 4, the proportion of patients undergoing primary amputation for arterial thromboembolism did not change over time nor did the number of patients who underwent a major amputation during the index hospitalization. The temporal trends in procedural management among the remainder of patients hospitalized with lower extremity arterial thromboembolism are summarized in Figure 3. As shown, there was no change in the risk of receiving a revascularization (endovascular or surgical) procedure during the index hospitalization (p=0.563) between 2003 and 2014. In 2003, the average risk for revascularization was 0.65 (95% CI: 0.61–0.68). There was a modest but not statistically significant increase to 0.66 (95% CI: 0.64–0.69) in 2014.
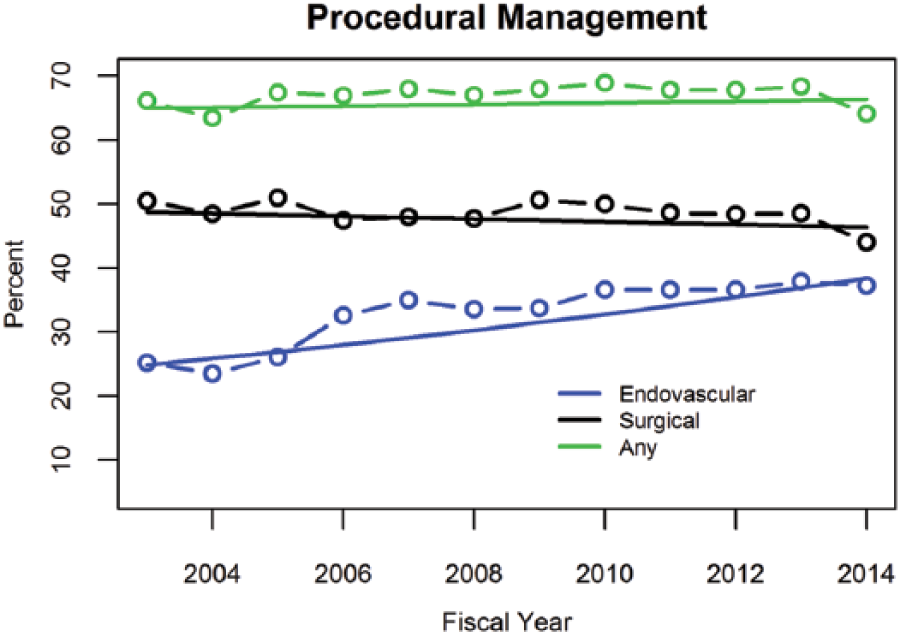
Trends in procedural management. The rate of endovascular revascularization increased 4.0% (95% CI: 2.7–5.4%) each year, while surgical revascularization slightly declined over the same period. Note that patients could undergo both endovascular or surgical revascularization such that those undergoing any revascularization is not the simple sum of the two.
While the overall average risk of revascularization did not change, the revascularization modalities utilized did differ over the study period, with a significant increase in the rate of endovascular revascularization. As shown in Figure 3, there was a significant increase in utilization (p < 0.001) of in-hospital endovascular procedures from 0.24 (95% CI: 0.21–0.27) in 2003 rising to 0.37 (95% CI: 0.34–0.40) in 2014, for a relative increase of 4.0% (95% CI: 2.7–5.4%) annually during the study period. In contrast, surgical revascularizations decreased during index hospitalizations between 2003 and 2014; however, the trend did not reach statistical significance (p=0.337). In 2003, the risk of patients undergoing surgical revascularization was 0.49 (95% CI: 0.45–0.53), while in 2014 it was 0.47 (95% CI: 0.44–0.50).
Clinical outcomes
Major adverse clinical outcomes
A small proportion of clinical outcomes were recorded outside the VA Healthcare System (Supplemental Table 5). There were no temporal patterns in MALE, MACE or mortality as demonstrated by the Kaplan–Meier survival curves (Figure 4A–C). The rate of major limb events at 1 year was 24.8% in 2005 and 26.2% in 2014. Similarly, the rate of major cardiac events at 1 year was 17.4% in 2005 and 18.4% in 2014. Finally, the 1-year mortality remained constant throughout the study period at 15.8% in 2005 and 16.2% in 2014. In addition, there was no association between year of initial hospitalization and time to MALE, MACE, or mortality before or after risk adjustment.
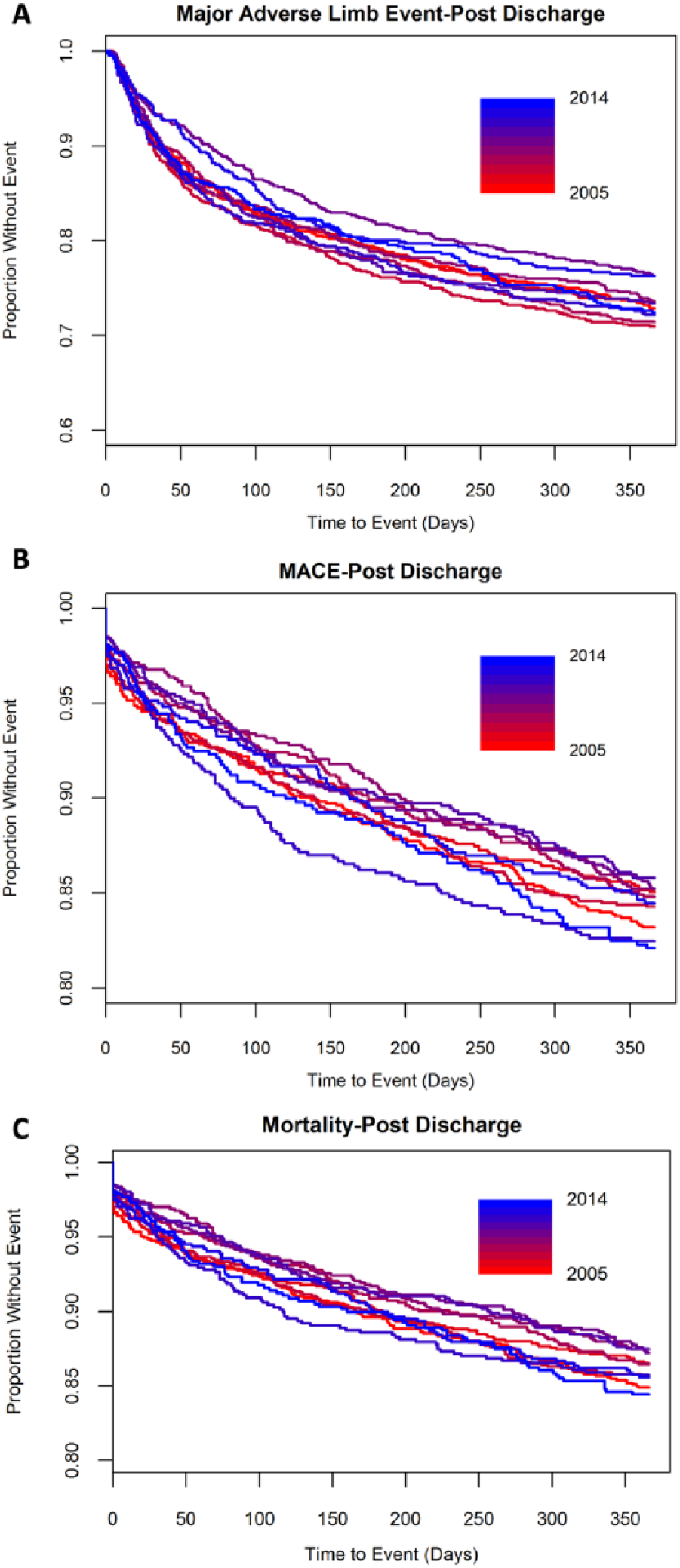
Trends in clinical outcomes. The rate of MALE and MACE remained constant over time, despite changes in the medical and procedural management. The large number of events at time zero in all years reflects an in-hospital adverse event.
Discussion
The present study sought to evaluate the temporal trends in incidence and management of lower extremity arterial thromboembolism in a large integrated healthcare system. As the data demonstrate, the rate of hospitalizations for this condition peaked over a decade ago and now continues to decline. Medical treatment has also changed, with increasing proportions of patients prescribed anti-platelet and anti-thrombotic therapy over time. Further, revascularization of acutely ischemic limbs has shifted toward more minimally invasive endovascular procedures rather than open surgical thrombectomy and arterial bypass. Despite these changes, however, the clinical outcomes among patients presenting with ischemic limbs demonstrate high rates of adverse limb and cardiovascular events in the first year after the index presentation. These findings have important implications for the overall health of this vulnerable patient population.
Several investigations have demonstrated a widely varying incidence of lower extremity arterial thromboembolism, with rates ranging from 14 per 100,000 in the United Kingdom18,19 to 26 per 100,000 in a United States Medicare population. 20 Current consensus estimates the incidence among the US general population at 1.5 per 100,000. 21 In the present analysis, we have demonstrated that the incidence is considerably higher among Veterans in the largest integrated healthcare system in the United States. Further, the data demonstrated a significant decline in incidence over the study period with a final reported rate of 3.54 per 100,000. This temporal decline is consistent with prior reports, and likely attributable to a multitude of factors including increased use of statin therapy 22 and declining use of tobacco products. 23 The increased recognition and evolving treatment of PAD in the primary care setting may also be playing a role. 24 The significant variation in administrative codes used to document this condition may also be contributing, as demonstrated by the low positive predictive value for the diagnostic codes for lower extremity arterial thromboembolism to truly represent ALI. Despite the declining incidence of this condition, lower extremity arterial thromboembolism is associated with significant morbidity and mortality.
Patients with lower extremity arterial thromboembolism have a high rate of adverse limb and cardiovascular events in the year following their index presentation. The present study demonstrates that most patients survive their sentinel event (30-day mortality, 4.8%), though the 1-year mortality remains as high as 15%. Other data sets report even greater rates of mortality (30–40%), suggesting that a successful intervention may be associated with improved outcomes.25,26 Regardless, the high mortality rate in this population is likely a reflection of the multiple comorbidities and overall illness of the patients. In addition to mortality, lower extremity arterial thromboembolism carries significant morbidity, with 8% of patients suffering an amputation or repeat revascularization within the first 30 days and 25% of patients suffering the same fate in the first year after presentation. The data suggest that these high rates of morbidity and mortality have not significantly changed over the last decade, highlighting the continued need to identify the most effective treatment modality for this condition.
The variability of medical management in this population may reflect the heterogeneity of data guiding physicians.13,27 Changing procedural techniques with increasing numbers of endovascular and hybrid procedures28–30 has also likely affected medical management, leading to the adoption of additional anti-platelet agents previously reserved for coronary interventions. Interestingly, the use of anti-platelet medications initially declined with a nadir in 2007 before increasing through the end of the study period. The low rates of non-aspirin anti-platelet agents runs counter to prior data, demonstrating lower adverse cardiovascular events among patients with PAD using these agents.31,32 The population studied had a high rate (76%) of peripheral artery disease, yet a low rate of anti-platelet therapy usage. The low prescription of P2Y12 inhibitors as mono-therapy is likely confounded by concomitant coronary disease and the ambiguity of the guideline recommendations which allow practitioners to choose aspirin or P2Y12 inhibitors. 13 This may suggest that there are persistent gaps in the medical management of lower extremity arterial thromboembolism that could be improved, with hopes of improving the clinical outcomes for this population.
While anti-platelet agents use remained relatively stable, the use of anti-thrombotic therapy was dynamic over our study. Initially there was a decline through 2006, followed by a resurgence of anti-thrombotic use through 2010, at which time the percent usage of anti-platelets and anti-thrombotics became near equal. Changing utilization over time was likely multifactorial and closely related to evolving endovascular techniques and medical therapy, demonstrating potential benefits for novel anti-thrombotic therapies, such as rivaroxaban. The changing tide of medical treatment again exemplifies the need for better established guidelines and randomized control trials to guide the medical therapy of ALI.
Contrary to medical therapies, rates of revascularization remained constant over time. While the rate of revascularization did not change, we did observe a significant evolution in procedural approaches employed by providers. Data showing equal efficacy of percutaneous therapies with thrombolysis compared to surgical intervention33,34 is reflected in our population, with a trend towards decreased surgical revascularization at the time of index hospitalization. While surgical procedures decreased, endovascular interventions rapidly grew, accounting for only 20% of revascularizations in 2003 to nearly 40% in 2014, mirroring trends in revascularization for claudication and CLI. 35 Finally, changes in surgical and endovascular rates may be a reflection of the current state of medical education. There is a continual increase in trainee exposure to endovascular technique in vascular surgery, interventional radiology, and interventional cardiology programs. This has led to a higher number of practitioners who are comfortable employing these techniques, leading to alternative therapeutic options for patients with this condition.36–38
These findings highlight the enormous room for the growth and development of new interventions for patients presenting with lower extremity arterial thromboembolism. In patients with stable atherosclerotic disease, the rates of a subset of this condition have been shown to be significantly reduced when patients were treated with vorapaxar. 39 Additionally, recent studies have begun to evaluate the use of low-dose rivaroxaban in patients with vascular disease, with a promising reduction in adverse events. 40 Our study encompasses a time period where rivaroxaban was prohibitively expensive and vorapaxar largely unavailable, which likely reduced its use in the present analysis. Further analyses are needed, as vorapaxar and rivaroxaban may prove to be novel therapies in the post-procedural management of lower extremity arterial thrombotic disease.
Limitations
The present project should be interpreted in the context of several limitations. First, the current analysis is derived from linked clinical and administrative databases used for clinical care. Data entry errors are thus possible, though several interventions have been deployed to increase the validity of the data. The validation of billing codes performed in this study demonstrates that lower extremity arterial thromboembolic disease does not always represent true ALI, and a focused evaluation for this condition is not possible with the current dataset. Widespread adoption of more specific administrative codes (ICD-10) may ameliorate this issue in the future. Second, a small proportion of patients treated within the VAHS receive additional care from outside providers. Fee-based data, which includes information regarding care Veterans received outside the VA that is paid for by the VA, was utilized to capture readmissions to outside facilities and mitigate this limitation. Any deficiencies in capturing repeat readmissions are unlikely to be differential across time and would only serve to underestimate the morbidity associated with this condition. Finally, the current dataset includes predominantly male patients treated within the VAHS. The results thus may not be generalizable to other populations with more gender or ethnic diversity.
Conclusion
In conclusion, the incidence of lower extremity arterial thromboembolism continues to decrease among a national population of Veterans. Further, the medical and procedural therapy of this condition is evolving with greater utilization of anti-thrombotic medications and endovascular revascularization. Despite this, the clinical outcomes of this patient population remain poor, with consistently high rates of amputation and mortality 1 year after the index event. These data highlight the need for continued advances in the medical and procedural therapies for this vulnerable patient population.
Supplemental Material
VMJ793210_supplementaltable – Supplemental material for Temporal trends in the management and clinical outcomes of lower extremity arterial thromboembolism within a national Veteran population
Supplemental material, VMJ793210_supplementaltable for Temporal trends in the management and clinical outcomes of lower extremity arterial thromboembolism within a national Veteran population by Andrew F Prouse, Paula Langner, Mary E Plomondon, P Michael Ho, Javier A Valle, Anna E Barón, Ehrin J Armstrong and Stephen W Waldo in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Armstrong is a consultant to Abbott Vascular, Boston Scientific, Cardiovascular Systems Incorporated, Medtronic, and Spectranetics. Dr Ho serves on a Steering Committee for a clinical trial sponsored by Janssen, Inc. Dr Waldo receives research support to the Denver Research Institute from Abiomed, Cardiovascular Systems Incorporated, and Merck Pharmaceuticals. The other authors have no industry relations to disclose. The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs or the United States Government.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the present project was funded from a research grant from the Merck Investigator Initiated Studies Program (MISP # 54744) to the Denver Research Institute.
Supplementary material
The supplementary material is available online with the article.
Dr Bruce Gray served as Guest Editor for this manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
