Abstract
In patients with symptomatic peripheral artery disease (PAD), the impact of chronic kidney disease (CKD) on major adverse cardiovascular events has not been fully evaluated. The Examining Use of Ticagrelor In PAD (EUCLID) trial randomized 13,885 patients with PAD to ticagrelor 90 mg twice daily or clopidogrel 75 mg daily. This post hoc analysis compared the incidence of the primary composite endpoint (cardiovascular death, myocardial infarction (MI), or ischemic stroke) in patients with CKD (eGFR < 60 mL/min/1.73 m2) with those without CKD (eGFR ⩾ 60 mL/min/1.73 m2). The primary safety endpoint was thrombolysis in MI (TIMI) major bleeding. A total of 13,483 patients were included; 3332 (25%) had CKD, of whom 237 had stage 4/5 disease. Median follow-up was approximately 30 months. After statistical adjustment, patients with CKD had a higher rate of the primary endpoint compared with those without CKD (6.75 vs 3.72 events/100 patient-years; adjusted hazard ratio (HR) 1.45, 95% CI 1.30–1.63). CKD was not associated with increased risk of hospitalization for acute limb ischemia (ALI) (adjusted HR 0.96, 95% CI 0.69–1.34) or major amputation (adjusted HR 0.92, 95% CI 0.66–1.28). CKD was not associated with a significantly increased risk of major bleeding (adjusted HR 1.21, 95% CI 0.89–1.64), but minor bleeding was significantly increased (adjusted HR 1.51, 95% CI 1.07–2.15). In conclusion, patients with PAD and CKD had higher rates of cardiovascular death, MI, and ischemic stroke, but similar rates of ALI, major amputation, and TIMI major bleeding when compared with patients without CKD.
Keywords
Introduction
Peripheral artery disease (PAD), a manifestation of systemic atherosclerosis affecting the arteries of the lower extremities, is associated with a high risk of cardiovascular and limb ischemic events. 1 Chronic kidney disease (CKD) is commonly associated with cardiovascular disease and worse outcomes in patients with PAD.1–3 However, the effect of CKD and the severity of renal impairment on cardiovascular outcomes and all-cause mortality in patients with PAD has not been fully evaluated. Additional endpoints associated with PAD that may be impacted by renal insufficiency include acute limb ischemia (ALI), which is associated with a high risk of limb loss and risk of cardiac ischemic events, 4 and the risk of major bleeding.
The Examining Use of Ticagrelor In PAD (EUCLID) trial enrolled 13,885 patients with symptomatic PAD but did not show superiority of the potent antiplatelet agent ticagrelor compared with clopidogrel in the prevention of major adverse cardiovascular events. 5 In this event-driven trial, the primary endpoint was a composite of cardiovascular death, myocardial infarction (MI), or ischemic stroke. Secondary endpoints included adjudicated acute limb ischemia and documented major amputations. Creatinine levels were obtained at baseline allowing for the estimation of glomerular filtration rate (eGFR). This post hoc analysis of the EUCLID cohort examined the impact of baseline renal function on the risk of major cardiovascular events and ischemic limb outcomes and risk of bleeding.
Methods
Study design
The design and rationale for EUCLID (NCT01732822) have been previously published. 6 The trial was designed by an independent executive committee. All primary efficacy and safety (bleeding) events were adjudicated by an independent clinical events committee whose members were unaware of treatment assignment. An independent data monitoring committee monitored the trial for safety. The trial results demonstrated that ticagrelor 90 mg twice daily was not superior to clopidogrel 75 mg once daily for the primary endpoint of time to first occurrence of the composite of cardiovascular death, MI, or ischemic stroke. Therefore, the current post hoc evaluation was based on the entire study population (i.e. regardless of treatment arm). The goal was to assess the natural history and outcomes of a population of patients with PAD, including the important subgroup of those with various degrees of renal insufficiency.
Study population
Patients were enrolled with lower extremity PAD defined as either an abnormal ankle–brachial index (ABI) ⩽ 0.80 or toe–brachial index ⩽ 0.60 at screening or a prior revascularization of the lower extremity. This manuscript reports the outcomes of patients with CKD defined as an eGFR < 60 mL/min/1.73 m2. This cutoff is clinically important as this amount of loss of renal function is associated with secondary abnormalities of renal failure (including anemia, acid base disturbances and bone mineral disease, particularly in stage 4/5) and the point at which risk factors for cardiovascular disease become more prevalent. 7 For the primary analysis, eGFR was calculated using the equation developed by the Modified Diet in Renal Disease (MDRD) group. 8 Key exclusion criteria for EUCLID included renal failure requiring renal replacement therapy. Patients were excluded from this analysis if they had an unknown baseline serum creatinine value for calculating eGFR, if the baseline serum creatinine value was < 0.3 mg/dL or > 12 mg/dL, or if baseline eGFR was > 160 mL/min/1.73 m2 as this is physiologically implausible. Normal eGFR is considered to be 130 mL/min/1.73 m2 and 120 mL/min/1.73 m2 in adult men and women, respectively; however, physiologic adaptation and creatinine measurement variation can account for estimates higher than this range. 9 The cutoff of 160 mL/min/1.73 m2 was selected to censor patients whose lab values may have been entered with erroneous units, and it was not possible to reconcile these errors retrospectively.
The study hypothesis was tested on the subgroup of patients with CKD (eGFR < mL/min/1.73 m2) compared with patients without CKD (eGFR ⩾ 60 mL/min/1.73 m2). Patients were further subdivided into the different stages of clinical CKD: stage 4/5 (< 30 mL/min/1.73 m2), stage 3 (⩾ 30 to < 60 mL/min/1.73 m2), stage 2 (⩾ 60 to < 90 mL/min/1.73 m2), or stage 1/no insufficiency (⩾ 90 to ⩽ 160 mL/min/1.73 m2) to assess trends with deteriorating function.
The EUCLID study complied with the Declaration of Helsinki. All patients provided written informed consent and the study protocol was approved by appropriate local ethics committees.
Data collection
Baseline eGFR was defined from the patient’s creatinine measurement performed at the study site prior to the first administration of study medication. Since creatinine was not measured in a core laboratory, there was no standardized normal reference range. If the baseline creatinine was missing, then any measurement collected within 7 days of first administration of study medication was also included as a baseline. Creatinine values were from local labs and subject to local reference ranges.
Endpoints
The primary endpoint in EUCLID was time from randomization to the first occurrence of any event in the composite of cardiovascular death, MI, or ischemic stroke. Secondary endpoints included the occurrence of any component of the primary composite, hospitalization for ALI, major amputation (above the ankle), and all-cause mortality. Major adverse limb events (MALE) were defined as the first occurrence of either ALI or major amputation. The primary safety endpoint was thrombolysis in myocardial infarction (TIMI) major bleeding. Secondary safety endpoints included intracranial and fatal bleeding. All primary efficacy, safety, and hospitalization data were adjudicated by an independent clinical events committee.
Statistical analysis
The primary analysis was based on the comparison of patients with CKD versus those without CKD. Additional analyses compared each patient group of stage 4/5, stage 3, and stage 2 with patients with stage 1/no insufficiency. All patients with a valid eGFR value at baseline were included in the analysis.
Baseline demographic data were compared between the CKD and no CKD groups; continuous variables were compared using a two-sample t-test and categorical variables using a chi-squared test.
The primary and secondary endpoints were analyzed using time-to-event analyses with a Cox proportional hazards model with a factor for renal subgroup (unadjusted analysis) and separately with a factor for renal subgroup and controlling for baseline characteristics (adjusted analysis; see the online Supplemental Appendix for adjustment variables). Kaplan–Meier estimates of survival probabilities for each event type were calculated out to 36 months, as well as at the median follow-up time for each event type. The p-value, hazard ratio (HR), and 95% CI for the HR were reported; p-values and CIs for the HR were based on the Wald statistic. The Efron method was used for handling ties. 10 Data for patients in whom the event in question had not occurred were censored at either the censoring date for the primary analysis or the last trial contact when all components of the endpoint in question were assessed, whichever came first. To determine whether the results were sensitive to the eGFR equation, a sensitivity analysis was performed using the CKD-EPI (Chronic Kidney Disease–Epidemiology Collaboration) equation. 11
The nature of the association between eGFR as a continuous variable and the composite endpoint was explored using a covariate-adjusted restricted cubic spline regression model with knots placed at clinically informed eGFR values of 30, 60, 90, and 120 mL/min/1.73m2 and a reference eGFR value of 0. A plot of the restricted cubic spline fit was visually assessed to evaluate whether a more simplified transformation (i.e. linear splines) could be applied.
The interaction between renal subgroup and treatment (ticagrelor or clopidogrel) was assessed for the composite endpoint (cardiovascular death, MI, or ischemic stroke) and TIMI major bleeding. For this assessment, the adjusted Cox proportional hazards model for each endpoint was updated to include an interaction term for treatment and renal subgroup. HRs, 95% CIs, and p-values comparing treatment groups within each renal subgroup were estimated.
SAS software, version 9.4 (SAS Institute, Inc., Cary, NC, USA) was used for all analyses and all tests were two-sided.
Results
Baseline characteristics
A total of 13,885 patients were included in the full analysis set, as reported in the primary results of the original study. 5 A total of 402 patients were excluded from this post hoc analysis due either to the absence of a baseline creatinine to calculate eGFR (n = 174), baseline creatinine < 0.3 (n = 103) or > 12 (n = 70), or because calculated eGFR was > 160 mL/min/1.73 m2 (n = 55). Baseline demographics are presented for both the CKD and no CKD groups (Table 1). Baseline demographics are also presented by eGFR stage in online Supplemental Table 1.
Baseline characteristics by renal subgroup (full analysis set). a
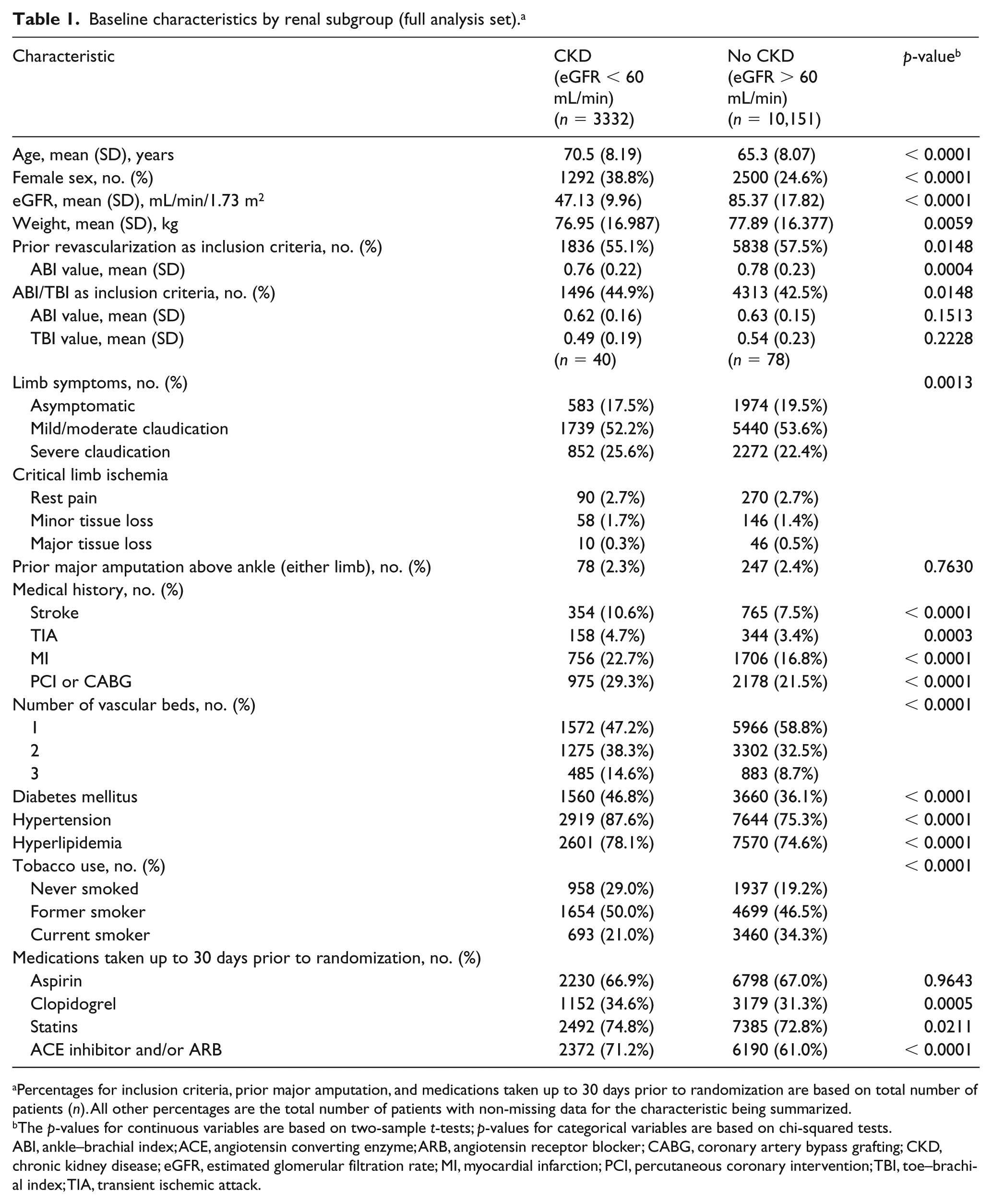
Percentages for inclusion criteria, prior major amputation, and medications taken up to 30 days prior to randomization are based on total number of patients (n). All other percentages are the total number of patients with non-missing data for the characteristic being summarized.
The p-values for continuous variables are based on two-sample t-tests; p-values for categorical variables are based on chi-squared tests.
ABI, ankle–brachial index; ACE, angiotensin converting enzyme; ARB, angiotensin receptor blocker; CABG, coronary artery bypass grafting; CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate; MI, myocardial infarction; PCI, percutaneous coronary intervention; TBI, toe–brachial index; TIA, transient ischemic attack.
Patients with CKD were older and had a higher proportion of female patients as compared with the no CKD group. Patients in the CKD group were less likely to have a history of a prior lower extremity revascularization. Established cardiovascular risk factors were more prevalent including diabetes, hypertension, and hyperlipidemia; however, tobacco exposure (both current and never smoked) was lower as compared with the group without CKD (Table 1). Concomitant coronary disease (history of MI, percutaneous coronary intervention, or coronary artery bypass grafting), stroke or transient ischemic attack, and polyvascular disease (two or three vascular beds affected) were more common in patients with CKD than in patients without. In the eGFR subgroups, as CKD became more severe, baseline demographic data demonstrated a progressively increased preponderance of cardiovascular risk factors and concomitant coronary and cerebrovascular disease (online Supplemental Table 1). Patients with CKD were more likely than those without to be taking a statin or angiotensin converting enzyme (ACE) inhibitor/angiotensin receptor blocker (ARB). Aspirin use at baseline was similar in both groups.
Clinical outcomes
The primary composite endpoint occurred at a rate of 6.75 events/100 patient-years in patients with CKD and 3.72 events/100 patient-years in those without CKD (Table 2). This resulted in an unadjusted HR of 1.81 (95% CI 1.63–2.02; p < 0.0001); after adjustment, the HR was 1.45 (95% CI 1.30–1.63; p < 0.0001). Figure 1 shows the Kaplan–Meier curves for the primary endpoint in the CKD group (cumulative probability 18.28%) compared with the group without CKD (cumulative probability 10.74%). The individual components of the primary endpoint had similar increased HRs with the exception of ischemic stroke, which was not significantly increased (Table 2). All-cause mortality was also increased, with an event rate of 5.41/100 patient-years in the CKD group compared with 3.02/100 patient-years in the no CKD group (adjusted HR 1.40, 95% CI 1.24–1.59; p < 0.0001). The risk of hospitalization for ALI (adjusted HR 0.96, 95% CI 0.69–1.34; p = 0.8074), major amputation (adjusted HR 0.92, 95% CI 0.66–1.28; p = 0.6371), and the MALE composite (adjusted HR 0.99, 95% CI 0.77–1.26; p = 0.9082) were not increased in patients with CKD as compared with those without CKD. Given the significant risk of all-cause mortality in CKD, a competing risk analysis was performed for MALE events. The competing risks analysis (results not shown) did not significantly alter the HR for MALE, and the Kaplan–Meier curves were generally superimposable.
Event rates by renal subgroup (full analysis set).
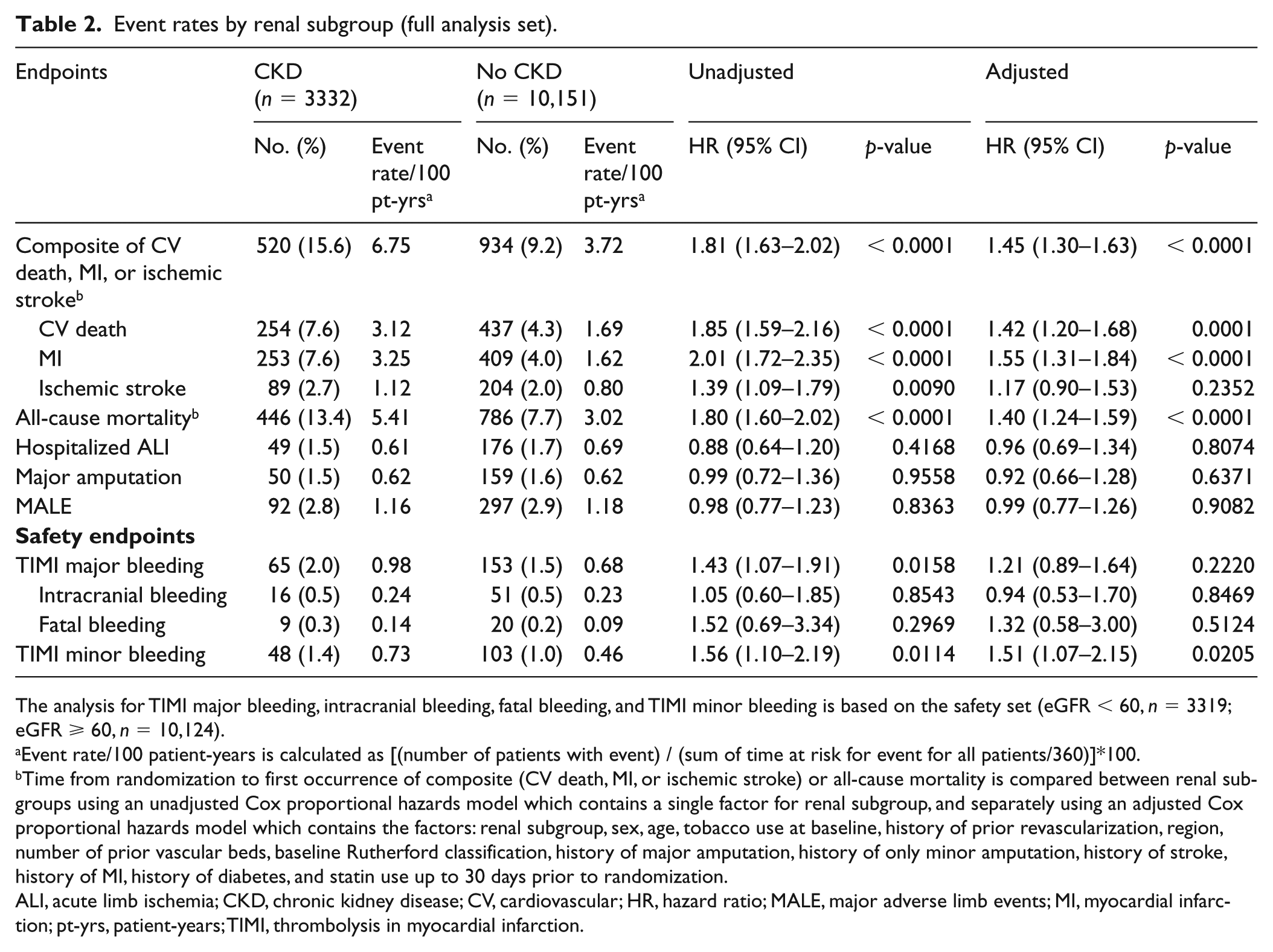
The analysis for TIMI major bleeding, intracranial bleeding, fatal bleeding, and TIMI minor bleeding is based on the safety set (eGFR < 60, n = 3319; eGFR ⩾ 60, n = 10,124).
Event rate/100 patient-years is calculated as [(number of patients with event) / (sum of time at risk for event for all patients/360)]*100.
Time from randomization to first occurrence of composite (CV death, MI, or ischemic stroke) or all-cause mortality is compared between renal subgroups using an unadjusted Cox proportional hazards model which contains a single factor for renal subgroup, and separately using an adjusted Cox proportional hazards model which contains the factors: renal subgroup, sex, age, tobacco use at baseline, history of prior revascularization, region, number of prior vascular beds, baseline Rutherford classification, history of major amputation, history of only minor amputation, history of stroke, history of MI, history of diabetes, and statin use up to 30 days prior to randomization.
ALI, acute limb ischemia; CKD, chronic kidney disease; CV, cardiovascular; HR, hazard ratio; MALE, major adverse limb events; MI, myocardial infarction; pt-yrs, patient-years; TIMI, thrombolysis in myocardial infarction.
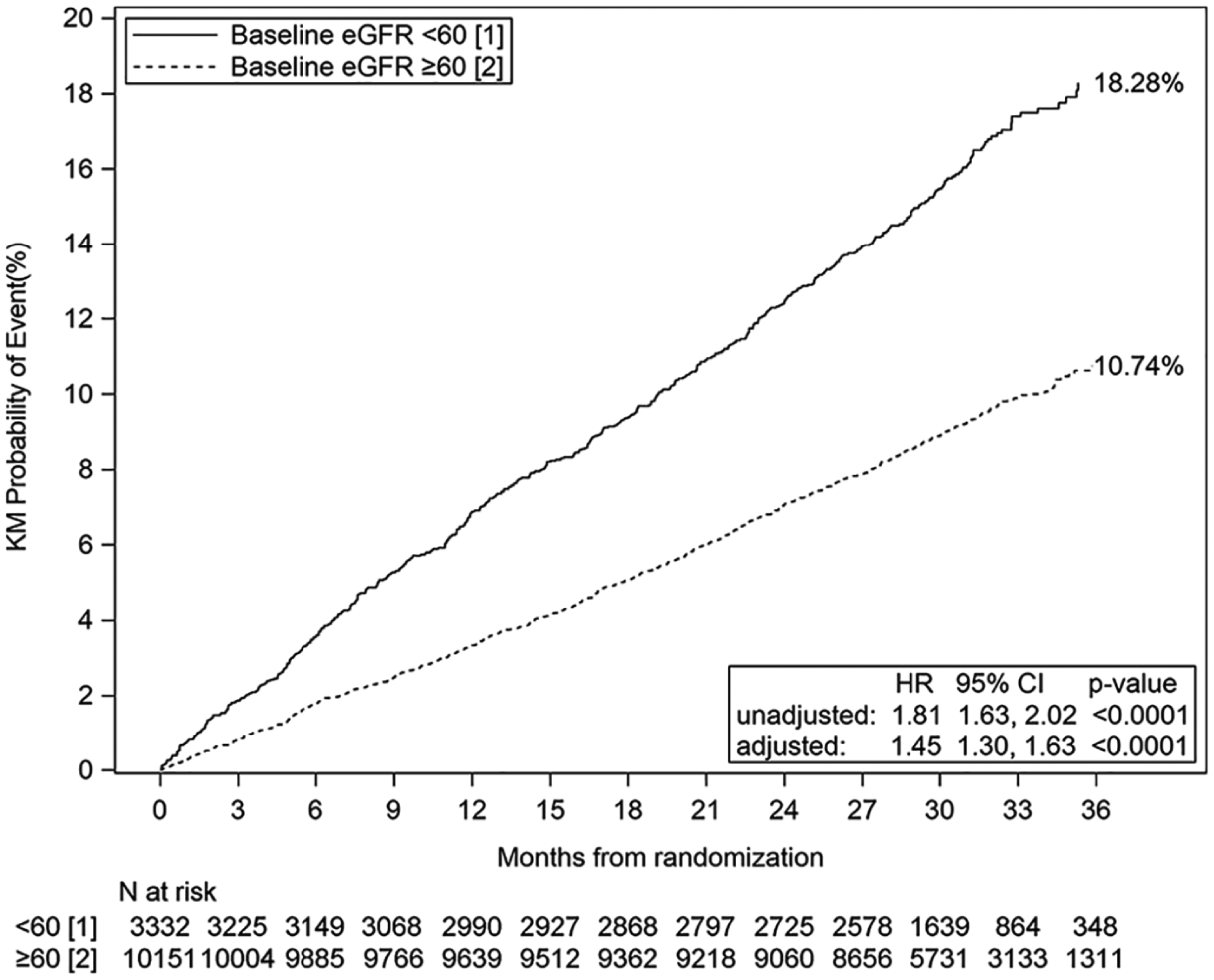
Kaplan–Meier plot of composite of cardiovascular death, MI, or ischemic stroke by primary renal subgroup (full analysis set).
Figure 2 shows the Kaplan–Meier rates for the primary endpoint by eGFR subgroup. The event rates for the primary endpoint were very similar between the stage 1/normal (3.52 events/100 patient-years) and stage 2 (3.83 events/100 patient-years) groups (adjusted HR 1.00, 95% CI 0.87–1.15; p = 0.9834) (online Supplemental Table 2). The primary endpoint event rates and HRs progressively increased for the stage 3 (adjusted HR 1.41, 95% CI 1.20–1.66; p < 0.0001) and stage 4/5 (adjusted HR 2.31, 95% CI 1.67–3.19; p < 0.0001) groups. All-cause mortality followed a similar trend with increasing HRs as CKD progressed to more severe stages (online Supplemental Table 2).
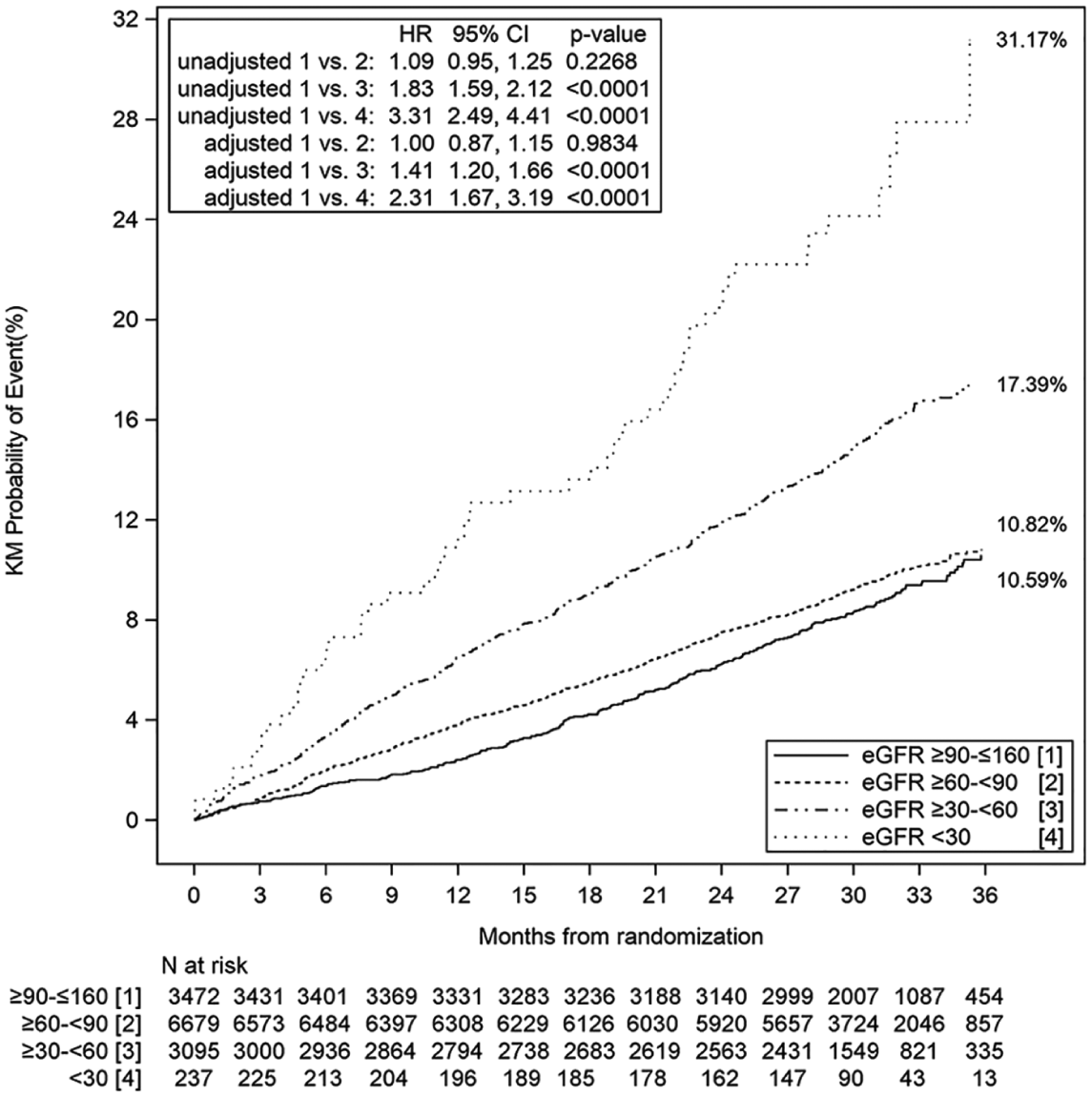
Kaplan–Meier plot of composite of cardiovascular death, MI, or ischemic stroke by eGFR subgroups (full analysis set).
The results of the restricted cubic spline analysis indicated a non-linear relationship between continuous eGFR and the composite endpoint (Figure 3). The plot of eGFR versus the log hazard suggested a simpler piecewise linear spline might fit the data equally well. As a result, a variety of knot points for a linear spline were explored within a plausible range as indicated by the plot and for eGFR values in intervals of five. Of these, a knot positioned at 70 mL/min/1.73 m2 provided the best fit of the data (i.e. model-2 log likelihood [with covariates; linear spline at 70] < model-2 log likelihood [with covariates; restricted cubic spline]). Above this value, changes in eGFR showed no association with the composite endpoint. Below this value, a 10-unit change (decrease) in eGFR was associated with a 20% increase in the risk of the composite event HR 1.20 (95% CI 1.15–1.26; p < 0.0001).
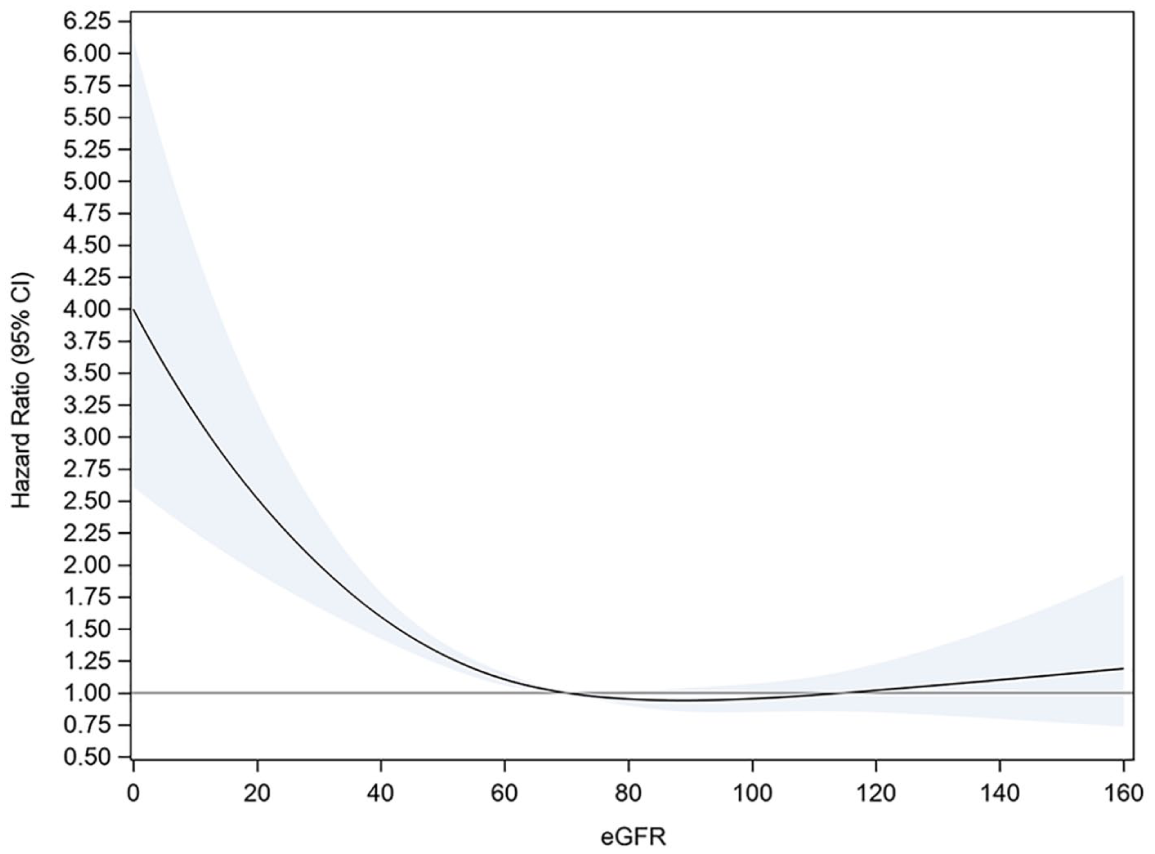
Plot of HR (95% CI in the shaded area) relative to an eGFR value of 70 from a restricted cubic spline model for the composite of cardiovascular death, MI, or ischemic stroke (full analysis set).
TIMI major bleeding was not significantly increased in the CKD group (adjusted HR 1.21, 95% CI 0.89–1.64; p = 0.2220) (Table 2). Adjusted HRs for intracranial and fatal bleeding were 0.94 (95% CI 0.53–1.70; p = 0.8469) and 1.32 (95% CI 0.58–3.00; p = 0.5124), respectively; there were few of these severe bleeding events in either group, making the upper bounds of the CIs large. There were no significant trends for bleeding risk across the eGFR subgroups (online Supplemental Table 2). TIMI minor bleeding was significantly increased, with an adjusted HR of 1.51 (95% CI 1.07–2.15; p = 0.0205) in patients with CKD.
In a sensitivity analysis using the CKD-EPI equation, the number of patients classified as having CKD was slightly smaller (263 less patients); however, the occurrence of the primary composite was similar to the primary analysis (adjusted HR 1.49, 95% CI 1.33–1.68; and adjusted HR 1.42, 95% CI 1.25–1.61, respectively).
A potential interaction was observed between renal subgroup and treatment (ticagrelor or clopidogrel) in a sensitivity analysis of the composite primary endpoint (p = 0.0014). Patients with CKD had a higher composite event rate on ticagrelor (17.3%) compared with clopidogrel (13.8%), whereas patients without CKD had a similar but slightly higher composite event rate on clopidogrel (9.6%) compared with ticagrelor (8.7%). There was no significant interaction between renal subgroup and treatment for all-cause mortality (p = 0.2561).
Discussion
EUCLID enrolled 13,885 patients with PAD, 3332 had CKD. Patients with PAD and CKD were older and had a higher prevalence of cardiovascular risk factors and more polyvascular disease compared with those without CKD. With adjustment for the prevalent risk factors and concomitant disease, patients with CKD had a 45% increased risk of the primary endpoint (adjusted analysis), with a similar increased risk of the individual components and for all-cause mortality as compared with those without CKD. When evaluated by eGFR subgroup, these risks dramatically increased with an eGFR < 60 mL/min/1.73 m2. An exploratory analysis examining eGFR as a continuous variable also showed incremental risk for the primary endpoint: every decrease in eGFR of 10 mL/min/1.73 m2 below 70 mL/min/1.73 m2 corresponded to a 20% increase in the HR. HRs of 4.0 were observed at the lowest eGFR values when compared with an eGFR of 70 mL/min/1.73 m2 or greater.
CKD did not significantly increase the risk for TIMI major bleeding. However, with only 65 major bleeding events in this group there were broad confidence interval estimates for bleeding risk. Thus, the TIMI major bleeding adjusted HR of 1.21 and the upper bound of the 95% CI of 1.64 could indicate an increased bleeding risk. Finally, CKD did not increase the risk of ALI, major amputation, or their composite (MALE), noting that there were few patients enrolled with baseline critical limb ischemia.
The association of renal insufficiency and cardiovascular disease has been increasingly recognized; however, this information has mainly been extrapolated from literature focusing on coronary artery disease. The prevalence of PAD increases with advancing severity of renal insufficiency, and further increases in the dialysis-dependent population.12,13 Liew and colleagues reported that the combination of CKD and PAD infers a greater risk of mortality than having only one of the diseases alone. 14 Furthermore, O’Hare et al. described an association with increasing severity of renal impairment and mortality in patients presenting with critical limb ischemia. 15 All-cause mortality was significantly higher in patients with severe renal insufficiency, and this is consistent with findings from other studies.2,14 Lüders et al. looked at incident hospitalizations in patients with PAD and found that patients with PAD and CKD had a 2.5 times higher risk for myocardial infarction and mortality was three times more likely compared with those without CKD. 16 The EUCLID trial provides a large population of patients with stable PAD to evaluate the association of renal insufficiency with major adverse cardiovascular events, and the results contribute to a growing body of literature describing this population.
It is not surprising that patients with PAD who also have CKD have a greater risk for cardiovascular events given the greater preponderance of cardiovascular risk factors and prevalence of polyvascular disease in this group at baseline. However, tobacco use was less common in the CKD group as compared with the group without CKD. Prior statin and ACE inhibitor/ARB use was higher in this group, all of which did not mitigate the marked increased risk for the primary endpoint in patients with CKD as compared with those without.
Patients with stable PAD and CKD had more severe limb symptoms, particularly severe claudication at baseline. Despite this, CKD did not increase the risk of ALI, major amputation, or MALE. Multiple retrospective studies have suggested that renal insufficiency increases the risk of ischemic limb events in patients undergoing a lower extremity revascularization (both open surgical and endovascular).17,18 Thus, in EUCLID, renal insufficiency was a risk for major adverse cardiovascular events but not MALE, which does not exclude a risk for MALE in the setting of revascularization. The higher mortality rate in patients with CKD may have created a competing risk for ALI and major amputation, meaning that these patients could have died before developing a MALE event. These findings contradict the study by Lüders, which demonstrated that while patients with CKD and PAD underwent less revascularization, the amputation rate was doubled in patients with CKD compared with those without. 16 MALE events are relatively frequent in the PAD population, with a main risk factor of prior lower extremity revascularization. 19 Patients with CKD may have been high risk for a procedure and therefore could have had fewer lower extremity revascularizations, which could also explain why the rates for MALE were similar for PAD patients with or without CKD.
The lack of a significantly increased TIMI major bleeding risk with decreasing renal function may be difficult to interpret given how few incidents occurred. Post hoc analyses of the PLATO trial demonstrated increased bleeding risk with decreasing eGFR and this was numerically higher in patients receiving ticagrelor compared with clopidogrel. 20 In EUCLID, there were numerically few major bleeding events and with broad confidence intervals the HR of 1.21 for TIMI major bleeding suggests an increased risk could exist. TIMI minor bleeding was significantly increased, consistent with the concept that moderate/severe renal insufficiency is associated with excess bleeding. Decreased renal function has long been known to increase bleeding risk, and this is thought largely to be a multifactorial process. For example, as clearance of uremic toxins decreases, platelets are thought to lose functionality through an inability to aggregate and interact with endothelium. 21 While these bleeding mechanisms are most closely associated with severe renal insufficiency and end-stage renal disease, no studies have definitively defined the point at which risk of platelet dysfunction begins. In EUCLID, the addition of a treatment group (ticagrelor versus clopidogrel) to the multivariable models for the primary endpoint and TIMI major bleeding did not change the HR estimates, indicating no clinically meaningful effect of either drug on those endpoints.
A strength of the EUCLID study was that patients with symptomatic and stable PAD were carefully selected and all primary efficacy, bleeding events, and MALE were prospectively adjudicated by a clinical events committee. In addition, the results were unaffected by the equation used to calculate eGFR. The MDRD equation was selected mainly to remain consistent with the original study. An alternate formula (CKD-EPI) was identified for sensitivity analyses because that equation may perform better in the setting of near normal renal function. 11 Sensitivity analyses indicated there were no meaningful changes in any of the outcomes or associations.
Limitations
This post hoc analysis has several limitations, including that it represents a secondary analysis of existing data. A serum creatinine was obtained in most patients, which allowed for calculation of eGFR at a single point in time, but there were missing data for some patients. This analysis did not attempt to examine how kidney function changed post-randomization. Given the primary intent of EUCLID, other key renal parameters were not collected, such as urinary albumin. In addition, more than 300 patients had undergone amputation at baseline and given that serum creatinine relies on muscle mass, these patients may have been misclassified. However, baseline amputations appeared to be well balanced among the patients with moderate/severe renal insufficiency and those without. While the lack of association between CKD and MALE events was a surprising finding in the study, this could have been influenced by a combination of a relative short follow-up time and lack of power due to a low number of limb events.
This analysis assigned patients to subgroups based on renal function in cross section and could not censor patients whose renal function was not in steady state at the time of randomization. With respect to the restricted cubic spline modeling, the authors recognize that the number and placement of clinically informed knots used in the analysis (four knots placed at 30, 60, 90, and 120 mL/min/1.73 m2) may not have represented the most optimal knot solution for the data. The introduction of a quadratic or cubic term to the model may have resulted in a better fitting model than what was ultimately obtained. Specifying a reference eGFR value other than zero may have resulted in the exploration of a different set of linear splines to simplify the interpretation of the analysis. As such, the results of this exploratory analysis should be interpreted in light of the methods used.
The authors acknowledge that there are additional variables which co-exist with CKD that were not accounted for in the adjusted analysis. The original set of candidate variables considered for inclusion in the adjusted analyses presented in this paper were selected to be consistent with multiple prior analyses conducted on the EUCLID study database. A sensitivity analysis of the primary composite endpoint determined that anemia at baseline, history of congestive heart failure, and history of atrial fibrillation were important covariates to include in the adjusted model; when included, the adjusted HR decreased to 1.35 (95% CI 1.20–1.51; p < 0.0001) compared to when these three covariates were excluded from the adjusted model (adjusted HR 1.45, 95% CI 1.30–1.63; p < 0.0001). The results of this sensitivity analysis did not meaningfully change the conclusions of the primary analysis.
A treatment interaction was observed between study drug and renal subgroup. Given the fact that there is no known renal mechanism of either drug that would cause this difference and the fact that the overall results of the trial were neutral (i.e. no treatment effect was detected for the comparison of ticagrelor to clopidogrel), this potential interaction should be interpreted with caution. Further to this point, the endpoint of all-cause mortality did not show an interaction between renal subgroup and treatment.
There are many potential reasons why CKD increases cardiovascular risk. While statistical adjustments were made for certain conditions that serve as risk factors for both kidney disease and cardiovascular disease, such as diabetes and hypertension, kidney disease itself may modify some of these risk factors. For example, in patients with kidney disease, hypertension is often longer standing, more severe, and more difficult to control with medications. 7 In addition to the traditional risk factors for cardiovascular disease, nontraditional risk factors (hyper-homocysteinemia, oxidative stress, and inflammatory markers) are increasingly recognized as promoting atherosclerosis and are highly prevalent in patients with CKD. 7
Conclusions
Patients with stable, symptomatic PAD and concomitant CKD at baseline had a marked increase in cardiovascular ischemic events and all-cause mortality that progressively worsened with greater degrees of renal impairment. The greater use of statins and ACE inhibitors/ARBs at baseline did not mitigate this risk. However, renal insufficiency was not associated with the occurrence of ALI or major amputation. Additional studies are warranted to address the adverse impact of renal insufficiency in patients with PAD.
Supplemental Material
10.1177_1358863X19864172_Supplementary_tables – Supplemental material for Chronic kidney disease and risk for cardiovascular and limb outcomes in patients with symptomatic peripheral artery disease: The EUCLID trial
Supplemental material, 10.1177_1358863X19864172_Supplementary_tables for Chronic kidney disease and risk for cardiovascular and limb outcomes in patients with symptomatic peripheral artery disease: The EUCLID trial by Charles W Hopley, Sarah Kavanagh, Manesh R Patel, Cara Ostrom, Iris Baumgartner, Jeffrey S Berger, Juuso I Blomster, F Gerry R Fowkes, W Schuyler Jones, Brian G Katona, Kenneth W Mahaffey, Lars Norgren, Frank W Rockhold and William R Hiatt in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Hopley: nothing to report. Kavanagh: consulting fees from Karuna Pharmaceuticals, Tal Medical, ResTORbio, Sonde Health, Commense Health, Grifols Pharmaceuticals, AveXis Pharmaceuticals, and UCB Pharma. Patel: research grants from AstraZeneca, Bayer, HeartFlow, Janssen, Medtronic, and the NHLBI; consulting fees from AstraZeneca, Bayer, and Janssen. Ostrom: nothing to report. Baumgartner: research grants from Abbott Vascular, Cook, Terumo, Amgen, and Boston Scientific; consulting fees from AstraZeneca, Bayer, and Amgen. Berger: consulting fees from Janssen, Merck, and Takeda. Blomster: employee of AstraZeneca at the time of trial conduct. Fowkes: consulting fees from AstraZeneca, Bayer, and Merck. Jones: research grants from the Agency for Healthcare Research and Quality, AstraZeneca, American Heart Association, Bristol-Myers Squibb, Doris Duke Charitable Foundation, and the Patient-Centered Outcomes Research Institute; consulting fees from the American College of Radiology, Bayer, Bristol-Myers Squibb, Daiichi Sankyo, and Janssen. Katona: employee of AstraZeneca. Mahaffey: research grants from Amgen, Boehringer Ingelheim, Daiichi Sankyo, Johnson & Johnson, Medtronic, St Jude, and Tenax; ownership/equity: BioPrint Fitness; consulting fees from Eli Lilly, American College of Cardiology, AstraZeneca, BAROnova, Bayer, Bio2Medical, Boehringer Ingelheim, Bristol-Myers Squibb, Cubist, Elsevier (AHJ), Epson, Forest, GlaxoSmithKline, Johnson & Johnson, Medtronic, Merck, Mt Sinai, MyoKardia, Omthera, Portola, Purdue Pharma, Springer Publishing, The Medicines Company, and Vindico. Norgren: research grants from AnGes and Mitsubishi; consulting fees from AstraZeneca, Bayer, AnGes, Pluristem, CESCA, and Mitsubishi. Rockhold: research grants from AstraZeneca and Luitpold Pharmaceuticals; ownership/equity: GlaxoSmithKline; consulting fees from AstraZeneca, Rho Pharmaceuticals, Eidos Therapeutics, Adverum Biotech, Syneos Health, Janssen Research & Development, and Merck Serono; other: Datavant, Inc. Hiatt: research grants from AstraZeneca, NIH, Bayer, Janssen, and Kowa.
Funding / Role of Sponsor
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work and the EUCLID trial were sponsored by AstraZeneca. AstraZeneca had two non-voting representatives on the EUCLID Executive Committee, which designed the trial, performed the data analyses, and wrote the manuscript. The sponsor had no veto power over publication. Dr Hopley was funded by T32 training grant DK007135 to the University of Colorado School of Medicine, Department of Medicine, Division of Renal Diseases and Hypertension, Aurora, CO.
Supplemental material
The supplementary material is available online with the article
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
