Abstract
Keywords
Introduction
Endovascular treatment (EVT) for patients with peripheral artery disease (PAD) has long been established as a feasible and effective strategy. 1 However, clinical trials with long-term patency and reintervention for devices used to treat symptomatic PAD are typically limited to 12- and 24-month outcomes. Following outcomes beyond 2 years provides much needed clinical evidence to assist in device selection, particularly for the more complex real-world lesions. Further, long-term patency data are especially important given that the goal of stent devices is to reduce stenosis 1 and primary patency measures are more indicative of device performance.
The GORE VIABAHN Endoprosthesis with Heparin Bioactive Surface (W.L. Gore & Associates, Inc., Flagstaff, AZ, USA; hereafter the Viabahn stent graft) has been established as treatment for patients with symptomatic PAD in femoropopliteal (FP) lesions.2 –7 In these clinical trial settings, the Viabahn stent graft was shown to maintain durable patency rates for up to 2 years, low rates of revascularization for up to 5 years, superiority over bare metal stenting, and efficacy in particularly complex patient populations.2 –7 However, these study results are limited to the clinical trial setting and evaluation in a real-world setting are lacking.
To provide a more complete assessment of the durability of the Viabahn stent graft, longer-term studies are needed. Building on the 12-month outcomes, 8 which allowed inclusion of more complex patients, the purpose of this current study was to continue assessing, in a real-world setting, the safety and effectiveness outcomes of the Viabahn stent graft as a treatment for patients with symptomatic PAD in longer FP lesions, for up to 5 years.
Methods
Study design
This prospective, multicenter, postmarket surveillance study evaluated the 5-year outcomes of the Viabahn stent graft as treatment in patients with symptomatic PAD in the FP lesions. The study was conducted in Japan across 64 sites from August 2016 to May 2017. The 12-month outcomes from this study were reported previously. 8 This report describes outcomes through 5 years.
This study was sponsored by W.L. Gore & Associates and is registered with ClinicalTrials.gov (ClinicalTrials.gov Identifier: NCT04706273). This surveillance study was regulated by the Japanese Ministry of Health, Labour and Welfare; complies with the Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices and Welfare; and was conducted in accordance with Japanese Good Post-Market Surveillance Practice (GPSP) regulations. Per the Japanese Ministry of Health, Labour and Welfare and Japanese GPSP regulations, informed consent is determined by each institution’s ethical committee policy to specify whether informed consent was necessary or outcome data could be abstracted while protecting patients’ rights without requiring individual patient consent. Informed consent was not required.
Patients with symptomatic FP lesions ⩾ 10 cm and reference vessel diameters ranging from 4.0 to 7.5 mm were eligible for enrollment. However, the surveillance design of the study did not exclude patients who may have had lesions or reference vessel diameters outside the range of enrollment eligibility.
Study device
All patients were treated with the GORE VIABAHN Endoprosthesis with Heparin Bioactive Surface (W. L. Gore & Associates, Inc.). The stent graft is a flexible, self-expanding endoluminal endoprosthesis consisting of an expanded polytetrafluoroethylene (ePTFE) lining with an external nitinol support extending along its entire length. The devices were available in diameters of 5, 6, 7, and 8 mm and length sizes of 2.5, 5.0, 7.5, 10.0, 15.0, and 25.0 cm. The device was approved in 2016 in Japan for use in superficial femoral artery lesions.
Study procedure
Baseline assessments included anatomic characteristics, ankle–brachial index (ABI), and Rutherford classification. 9 Sizing and implantation of the stent graft were evaluated with quantitative vascular angiography and/or intravascular ultrasound (IVUS) and performed at the physician’s discretion. Technique or vessel preparation were further described in the device instructions for use (IFU). Additional treatment or postprocedural therapies were also at the physician’s discretion, though the stent graft IFU recommends dual antiplatelet therapy for a minimum of 6 months.
Follow-up visits were scheduled at 30 days, 1 year, and annually to 5 years and assessments included ABI, Ruther-ford classification, and duplex ultrasound patency evaluation. Starting at 1 year, stent fracture evaluation by x-ray was performed annually through 5 years. Suspected stent fractures were confirmed by a core laboratory.
Endpoints
Endpoints were evaluated for the overall patient population through 5 years and included primary patency (PP), primary-assisted patency (PAP), secondary patency (SP), and freedom from mortality, freedom from target lesion revascularization (fTLR), acute limb ischemia (ALI), and major adverse limb event (MALE), changes in mean ABI and Rutherford classification, and occurrence of device- or procedure-related serious adverse events (SAEs), and stent fractures.
PP, PAP, and SP were defined in alignment with Society for Vascular Surgery guidelines. 10 PP was defined in this study as patients without the need for additional secondary surgical or endovascular procedures (stenosis or occlusion) of the target lesion with < 2.5 peak systolic velocity ratio (PSVR). PAP was defined as hemodynamic confirmation of blood flow through the Viabahn stent graft that did not require target lesion revascularization (TLR) to restore flow after total occlusion (occlusion of the primary treated site has not occurred). SP was defined as hemodynamic confirmation of blood flow through the Viabahn stent graft obtained with the use of an additional or secondary surgical or endovascular procedure after occlusion occurs. Device removal, bypass, or occlusion without successful revision was considered as loss of SP. ALI was defined as a rapid decrease in the lower limb blood flow due to an acute occlusion of the artery. ‘Acute’ was not relative to time postsurgery, but in relationship to how quickly the occlusion occurred (in days or hours) and with occurrence happening at any timepoint. All adverse events were reviewed to understand if the patient is suddenly in pain and their leg generally becomes cold and blue along with the pain, which is an indication of ALI. The occurrence of device- or procedure-related SAEs were collected through 5 years and the relationship to the device or index procedure and seriousness of the event was adjudicated by the physician.
Statistical methods
Kaplan–Meier (KM) survival curve analyses were conducted to assess cumulative PP, PAP, and SP rates and fTLR through 5 years; estimates were associated with a standard error < 10%, unless otherwise indicated. Univariate analyses were conducted to identify factors predictive of loss of PP through 5 years. All statistical analyses were performed using SAS 9.3 (SAS Institute, Inc., Cary, NC, USA). Univariate regression analyses were conducted with JMP Pro 15.1 software (SAS Institute, Inc.). Continuous data are expressed as mean ± SD; categorical data are expressed as number (percentage).
Cox analyses post hoc were conducted on risk factors to determine if there was an effect on PP. The risk factors evaluated were: TransAtlantic Inter-Society Consensus (TASC) II class; lesion lengths (⩾ 20 vs < 20 cm); diabetes mellitus; proximal vessel diameter (5 vs 6 mm); calcification mild/none versus moderate/severe; sex; hemodialysis; chronic total occlusion; and chronic limb-threatening ischemia (CLTI). For evaluation of CLTI and intermittent claudication, the CLTI group was comprised of patients in Rutherford categories 4–6 and the intermittent claudication group were of Rutherford categories 0–3. KM analyses for fTLR and PAP were performed.
Results
Patient population
A total of 321 patients were enrolled at 64 Japanese sites from 2016 to 2017. Baseline patient, anatomic, lesion, and procedural characteristics are presented in Table 1. At baseline, the mean age was 73.9 ± 8.7 years (range 36–97 years). The frequency of diabetes mellitus, end-stage renal disease on dialysis, and CLTI was 54.8%, 23.1%, 26.5%, respectively. Mean lesion length was 23.6 ± 6.6 cm and 86.6% of lesions were TASC II C or D lesions. Twenty-one patients had lesions < 10 cm, most of which were 8–8.5 cm; the range was 3.9–10 cm. Four patients had reference vessel diameters outside the range of 4.0–7.5 mm and included lesion lengths of 3, 8 (n = 2) and 9 mm. Moderate-to-severe calcification was present in 39.8% of 324 limbs and a total of 228 limbs (70.4%) had CTO. A total of 324 limbs were implanted with 562 devices; the median number of devices implanted was 2.0 (range 1–4). As reported previously, 8 technical success was achieved in 96.9% of 324 limbs. The percentage of patients taking antiplatelet medications was collected through 2 years. Two or more antiplatelet medications were used by 244 (77.0%) patients at 1 year and 185 (62.7%) at 2 years. There was a wide variety of patterns in the types of antiplatelet and anticoagulant medications prescribed in this study. Through 24 months of data available, the use of dual antiplatelet therapy ranged from 41.4% to 65.4% of patients (Table 2). The combination of aspirin and thienopyridine-based drugs (clopidogrel) was the most commonly used regimen (online supplemental Table 1) and is considered a narrow-sense dual antiplatelet therapy. However, in this case, all drugs with antiplatelet effects according to the package insert were counted as antiplatelet drugs. On the other hand, ‘other’ drugs were not counted as antithrombotic drugs because their details were unclear. Table 2 describes the regimen for the prescribed antiplatelet agents or anticoagulants, and online supplemental Table 1 further describes the specific medications.
Patient demographics and disease characteristics of all patients at baseline.
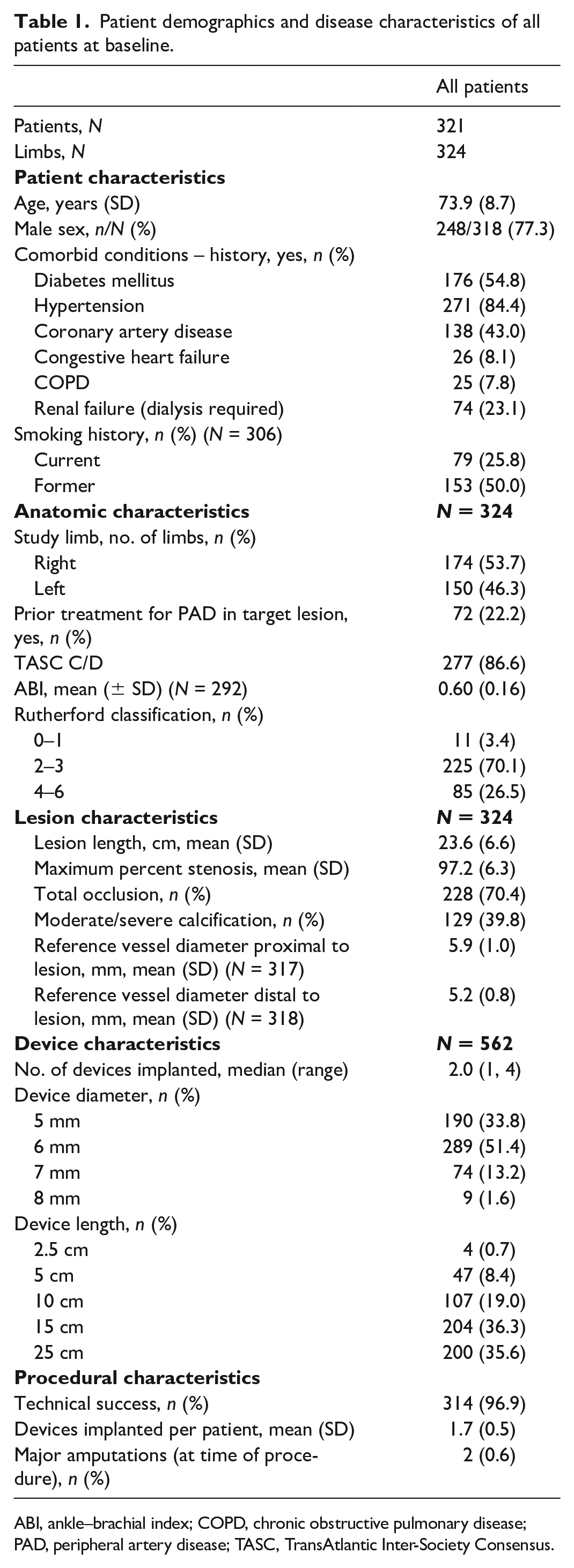
ABI, ankle–brachial index; COPD, chronic obstructive pulmonary disease; PAD, peripheral artery disease; TASC, TransAtlantic Inter-Society Consensus.
Antithrombotic therapy regimen across 2 years.
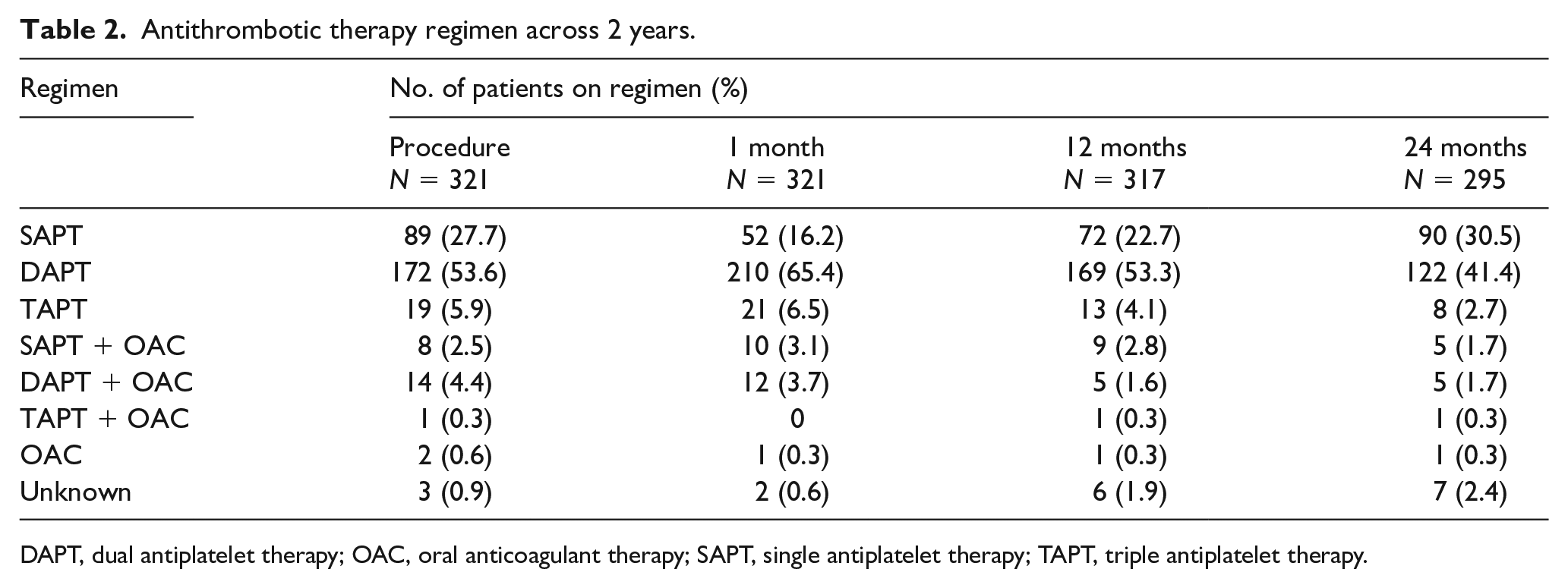
DAPT, dual antiplatelet therapy; OAC, oral anticoagulant therapy; SAPT, single antiplatelet therapy; TAPT, triple antiplatelet therapy.
Of the 321 patients in the study, 136 (42.4%) patients completed the 5-year follow-up and 185 (57.6%) patients withdrew from the study. The reasons (number, percentage) for withdrawal from the study were: death (n = 85; 26.5%), lost to follow-up (n = 42; 13.1%), withdrew consent (n = 27; 8.4%), changed hospitals (n = 22; 6.9%), surgical bypass (n = 3; 0.9%), other reasons not specified (n = 3; 0.9%), amputation that removed the study device (n = 2; 0.6%), and investigator decision (n = 1; 0.3%).
Endpoints
Of the patients eligible for follow-up, 76.5% (192/251) returned for the 3-year follow-up and 65.9% (122/185) returned at 5 years. Illustrated in Figure 1 are the KM estimates of PP, PAP, and SP annually through 5 years. At 3 and 5 years, the KM-estimated PP was 69.8% and 62.4%, respectively; PAP was 82.6% and 74.1%, respectively; and SP was 88.9% and 82.3%, respectively.
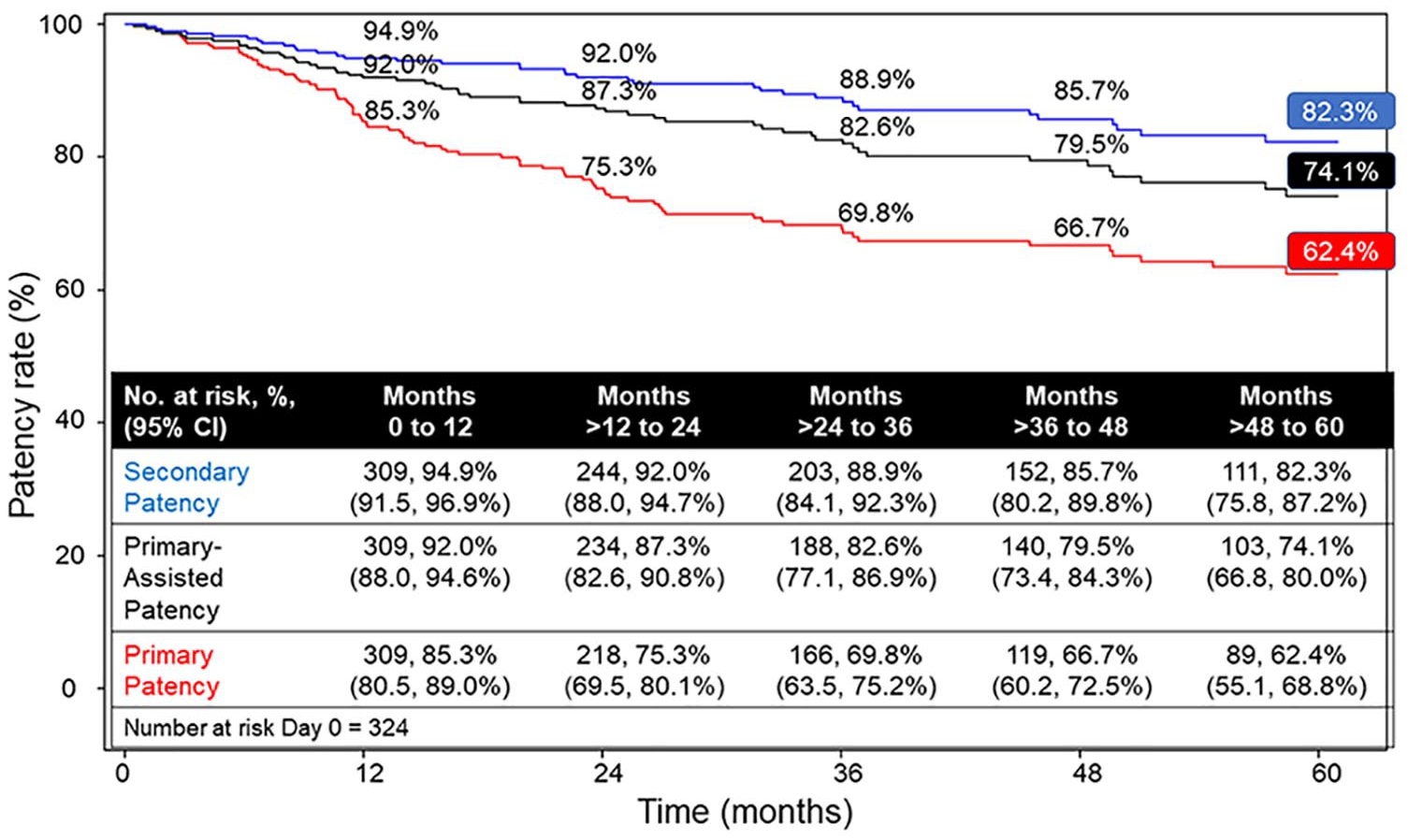
Primary, primary-assisted, and secondary patency Kaplan–Meier estimates.
Freedom from mortality was 100% at time of treatment, 83.1% at 3 years, and 67.7% at 5 years. Freedom from TLR was 81.1% at 3 years and 75.9% at 5 years (Figure 2). Of the 63 patients who had a first reintervention (Table 3), the plain old balloon angioplasty was the most common reintervention (69.8%) and most commonly the location was distal to stent implantation (65.4%). Of the 63 patients with first reintervention, 21 patients had more than one reintervention. Freedom from ALI was 100% through 5 years. Freedom from MALE and ALI was 97.9% at 3 years and 97.4% at 5 years (Figure 3).
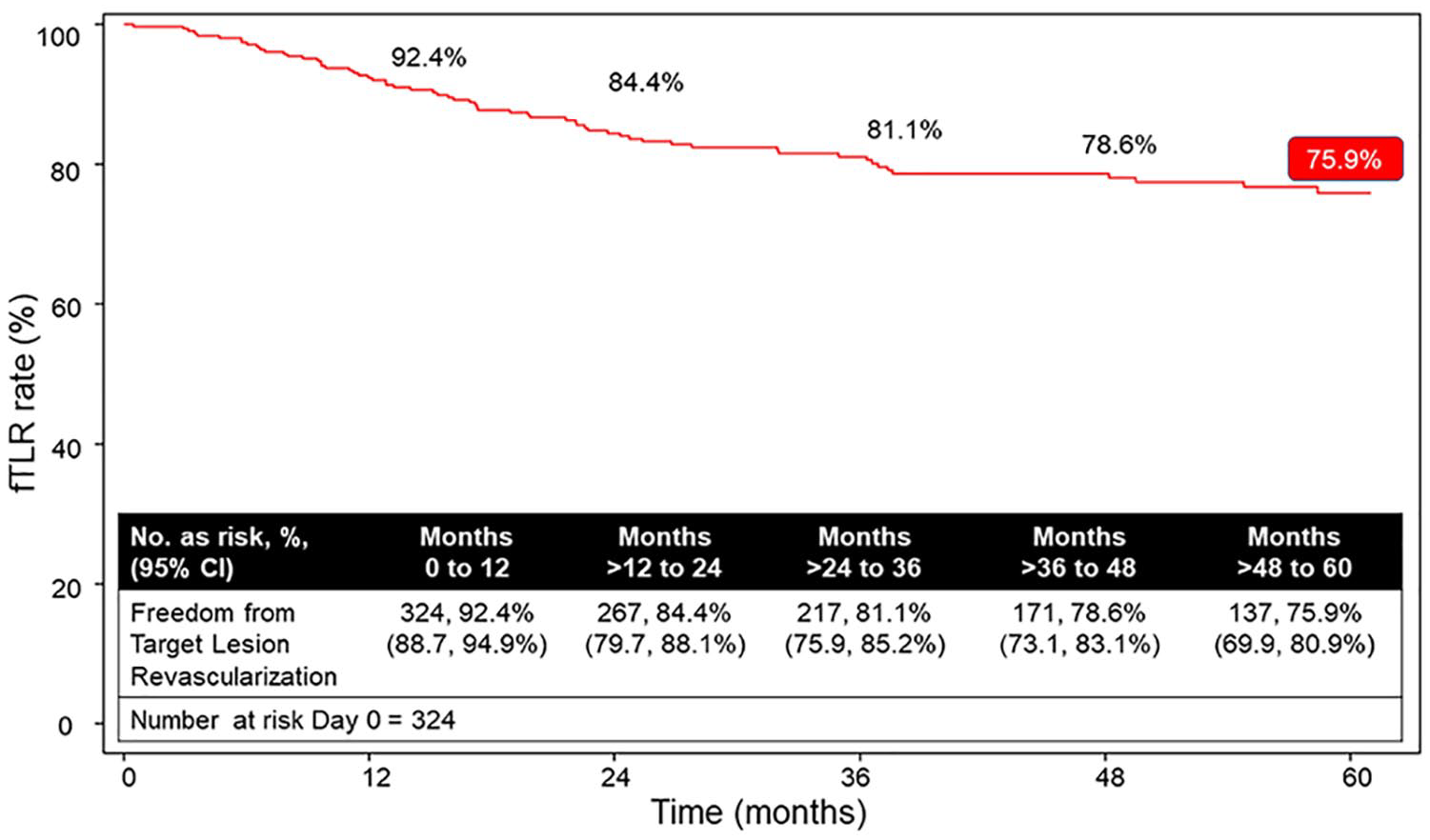
Freedom from target lesion reintervention Kaplan–Meier estimates.
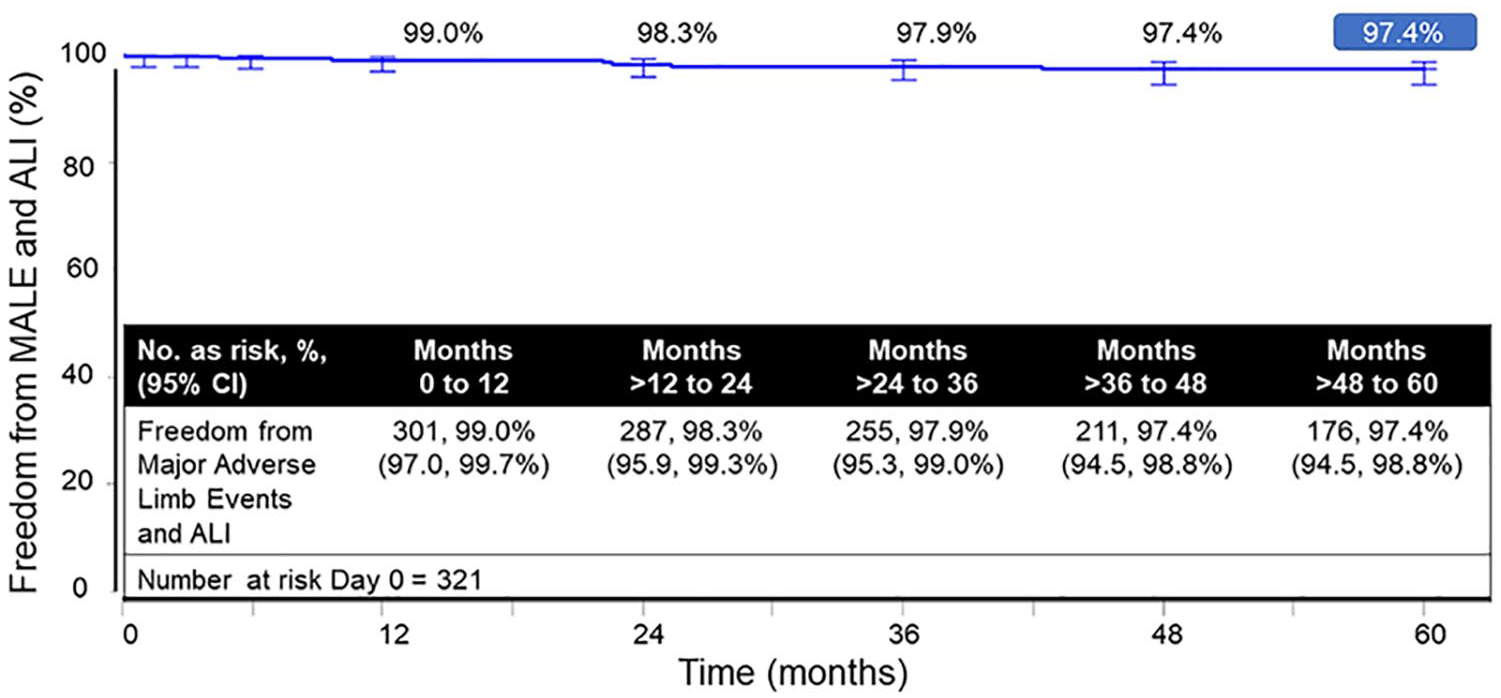
Freedom from combined major adverse limb events and acute limb ischemia Kaplan–Meier estimates.
Summary of first reinterventions (N = 63 patients).

Those patients who required additional stenting with bare-metal stent, drug-eluting stent, or Viabahn.
BMS, bare-metal stent; DES, drug-eluting stent; POBA, plain old balloon angioplasty.
The mean ABI at index procedure was 0.60 ± 0.16 and for patients available at the 5-year follow-up (n = 103) was 0.92 ± 0.15; mean change was 0.31 ± 0.19. At 5 years, 77.6% (90/116) of patients were of Rutherford category 0, 17.2% (n = 20) were category 1 or 2, and 1.7% (n = 2) for each category 4, 5, and 6. No patient was classified as category 3. The mean change from index procedure to 5 years was 2.3 ± 1.4 (Figure 4).
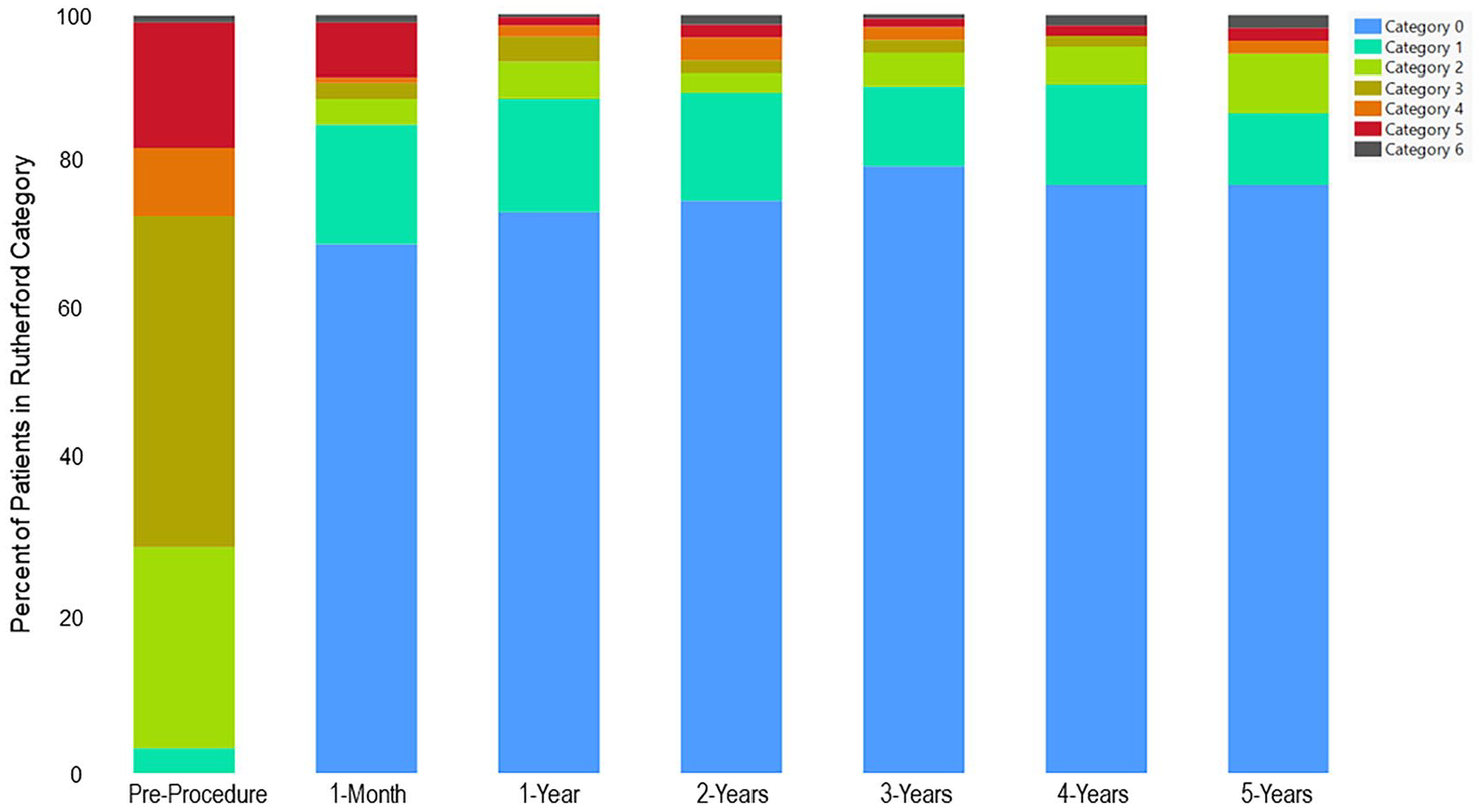
Rutherford category classification – percentage of patients in categories from preprocedure through 5 years. The percentage of patients classified as categories 0 (blue), 1 (turquoise), or 2 (lime green) combined was 29.9% at preprocedure. At 1-month postprocedure, a majority of patients (89.0%) were in categories 0, 1, or 2, and by 5 years nearly all patients (94.9%) remained in these categories.
Safety
Through 5 years, 87 device- or procedure-related SAEs were reported in a cumulative number of 64 patients (19.9%); between 1 and 5 years, only 9.3% of patients experienced a device- or procedure-related SAE (online supplemental Table 2). Events reported in that timeframe of the patients available (N = 321) included: device occlusion (n = 25), vascular stent graft stenosis (n = 9), vessel occlusion (n = 1), in-stent arterial restenosis (n = 1), and lower-limb (nonacute) ischemia (n = 1). No stent fractures were observed by the core lab (X-ray evaluation) through 5 years.
From index procedure through 5 years, two (0.6%) patients required a major amputation (study device was removed). Both had leg amputations and one was reported at the 12-month and the other at the 4-year interval. In six patients with preexisting ulceration or gangrene on the study limb, one minor and five major amputations were reported.
Surgical bypass conversion was reported in five (7.9%) patients. Of the five patients with events, three were arterial bypass (two at the 1-year interval, one at the 2-year interval, and two at the 3-year interval), one was FP (2-year interval) who also experienced two additional bypasses that were disease-related, and one was arterial stenosis (12-month interval). Four of the five patients had total occlusion and one had 90% stenosis.
Subgroup analyses
Uni- and multivariable subgroup analyses stratified by length, CTO, CLTI, and TASC revealed that factors associated with loss of PP were not found (online supplemental Table 3). Comparison of PP outcomes in subgroups of patients with potential associated factors were not significantly different for any of the comparisons. The PP rates for the subgroup comparisons for CTO and no CTO were 63.4% versus 59.6%; < 20 cm and ⩾ 20 cm lesion lengths were 63.4% versus 62.0%; CLTI and intermittent claudication were 59.9% versus 62.7%; and TASC A/B and TASC C/D were 68.4% versus 62.3%.
Discussion
This study assessed the long-term outcomes of the GORE VIABAHN stent graft in Japanese patients PAD and complex FP lesions in a real-world setting. The PP rate at 5 years was 62% with an acceptable safety profile and no observed stent fractures. Further, improvements in the ABI and Rutherford classification observed at 1 year were maintained at the 5-year mark. Subgroup analysis revealed 5-year PP was not affected by patient comorbidities and lesion severity.
The favorability of these outcomes is notable as the study population enrolled contained patients with complex FP disease and significant comorbidity burden, and the most severe manifestation of FP intervention. The durability of the device at 5 years was demonstrated by respectable rates of PP (62%), PAP (74%), and SP (82%). The rates of freedom from combined acute limb ischemia and MALE were around 97% and further support the long-term device performance. Clinically, patient quality of life was improved as represented by the asymptomatic status by Rutherford classification of about 77% of patients at 5 years.
Benchmarking the long-term PP of the Viabahn stent graft against other devices is difficult as data are sparse, particularly in a real-world population as is reported here. Surgical bypass is a standard treatment for complex lesions. Reports for 5-year patency rates of above-the-knee bypass with vein ranged from 73% to 76% and with PTFE ranged from 18% to 68%.11 –15 A systematic literature review of the 5-year PP rates of native vein and PTFE grafts placed above-the-knee were 69% and 49%, respectively. 15 Patients with complex disease often have concomitantly complex comorbidities. As such, peri- and postoperative complications from bypass are not uncommon and likely lead to prolonged hospitalization. Farhan et al. reported EVT to be a safe and efficacious alternative to surgical bypass therapy with lower early complication rates and shorter length of hospital stay. The PP with EVT at 2 years was 51.2% and was 61.3% for bypass. 16 The incidence of MALE was not significantly different between patients with EVT or bypass. 16
Long-term outcomes of fTLR have been reported5,17,18 as a proxy for device performance; however, this outcome is not reflective of the ideal performance of a device as differing definitions of fTLR can limit the comparisons. The IN.PACT global study of treatment with drug-coated balloons (DCB) for treatment that included long lesions, or CTOs reported of fTLR rates of 67% for long lesions and 70% for CTO. 19 The fTLR rate for this study was 76% and was numerically higher than DCB despite the postmarketing surveillance population that included more complex comorbidities, longer lesions, and total occlusions in 70% of the population. The advantage of the Viabahn stent graft over DCB would be marked in complex lesions.
Alongside reported evidence that EVT is a safe and efficacious alternative to bypass, the results from the current study demonstrated a low complication rate, durable pat-ency, and consistent performance of EVT and use of the Viabahn stent graft. The durability is further supported by subgroup analyses of potential associated factors of common comorbidities in this patient population that did not yield a relationship for loss of PP. This study serves to further support an endovascular approach with the Viabahn stent graft as a first-line treatment strategy for complex FP lesions.
Study limitations
The results from this study are encouraging in this complex patient population and add to the body of literature for long-term outcomes. However, results must be considered under limitations of the study and design. The postmarketing surveillance study design is not as rigorous as a randomized, controlled trial but, conversely, it provides relevant, real-world setting data. The percentage of patients who returned for a 5-year follow-up (66%) was quite respectable. Enrollment limitations of patients with complex FP disease, and significant comorbidity burden, and residing in one country, Japan, narrows the generalizability of these results. Though the severity of the disease may be more reflective of patients in a real-world setting, it limits the generalization to the broader, less severe populations.
Antiplatelet therapy was collected through the 2-year follow-up but was not collected through the 5-year follow-up. The continuation of dual antiplatelet therapy was at the physician’s discretion in this study but was strongly recommended and is standard practice among physicians. The classification of ALI data was not included in the original protocol and therefore data further describing limb viability were not available. This may have had some influence in the low incidence of ALI and thrombotic occlusion.
This study evaluated TLR rather than clinically driven TLR. The study was not designed to collect ABI and Rutherford values for adverse events that were not associated with repeat intervention, making it difficult to discern those which would be considered clinically driven TLR. The limitation of a core lab used only for X-ray analysis while other measurements were performed at each hospital must also be taken into consideration.
Conclusions
The 5-year safety and efficacy outcomes of the Viabahn endoprosthesis were clinically acceptable for treating complex FP lesions in a real-world Japanese practice setting for pateints with PAD.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241233528 – Supplemental material for Five-year outcomes of the GORE VIABAHN Endoprosthesis for the treatment of complex femoropopliteal lesions from a Japanese postmarket surveillance study
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241233528 for Five-year outcomes of the GORE VIABAHN Endoprosthesis for the treatment of complex femoropopliteal lesions from a Japanese postmarket surveillance study by Osamu Iida, Takao Ohki, Yoshimitsu Soga, Nobuhiro Suematsu, Tatsuya Nakama, Terutoshi Yamaoka, Kazuki Tobita and Shigeo Ichihashi in Vascular Medicine
Footnotes
Acknowledgements
The authors thank Chrystal Redding, Yasunori Arida, and Masahiko Kawaguchi (W.L. Gore & Associates, Inc.) for their technical review of the data and manuscript and Millie Hollandbeck for assistance with preparation of the manuscript.
Data were presented in part as an oral presentation at the Vascular InterVentional Advances (VIVA) 2023 Conference; October 30 to November 2, 2023; Las Vegas, NV, USA
Data availability statement
Data will be made available upon reasonable request to the corresponding author.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Funding
W. L. Gore & Associates, Inc. sponsored this study, and medical writing support was provided by Millie Hollandbeck (Phoenix, AZ, USA). W.L. Gore & Associates, Inc. provided funding for the medical writer, unaffiliated with Gore, to assist with preparation of the manuscript. W.L. Gore & Associates, Inc. participated in the study design and collection, analysis, and assisted with interpretation of the data but was not involved in constructing the manuscript contents and the decision to submit the manuscript for publication.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
