Abstract
We estimated minimal clinically important differences (MCIDs) for small, moderate, and large changes in measures obtained from a standardized treadmill test, a 6-minute walk test, and patient-based outcomes following supervised and home-based exercise programs in symptomatic patients with peripheral artery disease (PAD). Patients were randomized to either 12 weeks of a supervised exercise program (n=60), a home-based exercise program (n=60), or an attention-control group (n=60). Using the distribution-based method to determine MCIDs, the MCIDs for small, moderate, and large changes in peak walking time (PWT) in the supervised exercise group were 38, 95, and 152 seconds, respectively, and the changes in claudication onset time (COT) were 35, 87, and 138 seconds. Similar MCID scores were noted for the home-based exercise group. An anchor-based method to determine MCIDs yielded similar patterns of small, moderate, and large change scores in PWT and COT, but values were 1–2 minutes longer than the distribution approach. In conclusion, 3 months of supervised and home-based exercise programs for symptomatic patients with PAD results in distribution-based MCID small, moderate, and large changes ranging from 0.5 and 2.5 minutes for PWT and COT. An anchor-based approach yields higher MCID values, ranging from a minimum of 73 seconds for COT to a maximum of 4 minutes for PWT. The clinical implication is that a goal for eliciting MCIDs in symptomatic PAD patients through a walking exercise intervention is to increase PWT and COT by up to 4 minutes, which corresponds to two work stages during the standardized progressive treadmill test.
Keywords
Introduction
Peripheral artery disease (PAD) is a highly prevalent condition,1–3 that is both costly4–7 and deadly,8,9 resulting in poor patient-based quality of life.10–13 Between 40% and 75% of those with PAD experience symptomatic leg pain during ambulation that is either typical or atypical of classic claudication, 2 resulting in disability evident by ambulatory dysfunction, 14 impaired physical function which declines over time,15,16 and low daily physical activity. 17
The primary goal in treating symptoms is to improve ambulatory function and quality of life. 18 Treatment options include exercise, pharmacological, and peripheral revascularization interventions to improve ambulatory dysfunction.19–21 While pharmacological and surgical interventions have been associated with measurable functional improvements in patients with PAD,22,23 medically supervised exercise programs can significantly improve ambulation with less cost, morbidity, and mortality. 24 Supervised exercise programs improve objective treadmill-based outcomes such as claudication onset time (COT) and peak walking time (PWT), and have been given a Class IA recommendation supported by multiple randomized controlled trials and meta-analyses.2,25 Home-based exercise programs have recently emerged as an additional method to rehabilitate symptomatic patients with PAD, and offer some advantages to supervised exercise. Contemporary home-based exercise studies, utilizing improved training methodologies, have also demonstrated efficacy in improving COT and PWT.19,26–28
While demonstrating improvements in functional outcomes following an exercise rehabilitation program is important to evaluate efficacy of the intervention, studying the minimal clinically important difference (MCID) has not been addressed in patients with PAD. The MCID represents the smallest threshold change in an outcome measure that patients consider beneficial. 29 In a previous report, the MCID in the 6-minute walk distance was determined by both distribution-based and anchor-based methods in older adults with mobility disability, subacute stroke survivors, and in community-dwelling older adults who were assessed at baseline and at follow-up. 30 The distribution-based method used the distribution of the outcome measure, as well as the variability and reliability to estimate the effect size and the standard error of the measurement. The anchor-based method used two questions assessing physical function quality of life to determine whether there was an improvement by at least one level in the answer selection. Using these methods, small and moderate MCID thresholds in the 6-minute walk distance were found to be approximately 20 meters and 50 meters, respectively; 30 however, the changes for the distribution-based method were calculated by using the baseline standard deviation rather than the change score standard deviation. Furthermore, these distances have been applied to patients with PAD 31 even though they were not originally developed on this population. MCID thresholds in patients with PAD have not been determined for commonly obtained PAD-specific outcome measures, such as the gold-standard variables PWT and COT during a treadmill test, and patient-based outcomes from the walking impairment questionnaire (WIQ) and the Medical Outcomes Study Short Form-36 (MOS SF-36) questionnaire.
Therefore, in an exploratory analysis from our previous randomized controlled trial, 26 the aim of this study was to estimate the MCID for small, moderate, and large changes in outcome measures obtained from a standardized treadmill test, a 6-minute walk test, and from the WIQ and MOS SF-36 questionnaires following both supervised and home-based exercise programs in symptomatic patients with PAD.
Methods
Patients
Approval and informed consent
The procedures of this study were approved by the institutional review board at the University of Oklahoma Health Sciences Center (HSC). Written informed consent was obtained from each patient at the beginning of investigation.
Recruitment
Patients who were not currently exercising were recruited from vascular laboratories and vascular clinics at the University of Oklahoma HSC for possible enrollment into exercise rehabilitation programs to treat leg pain secondary to PAD.19,26
Medical screening through history and physical examination
Patients were evaluated in the morning at the Clinical Research Center at the University of Oklahoma HSC. 26 Patients arrived fasted, but were permitted to take their usual medications. Patients were evaluated with a medical history and physical examination in which demographic information, height, weight, waist circumference, cardiovascular risk factors, comorbid conditions, claudication history, ankle/brachial index (ABI), blood samples, and a list of current medications were obtained.
Inclusion and exclusion criteria
According to criteria previously described, 26 patients with symptomatic PAD were included in this study if they met the following criteria: (a) a history of ambulatory leg pain, (b) ambulatory leg pain confirmed by treadmill exercise, 14 and (c) an ABI ≤ 0.90 at rest2 or ≤ 0.73 after exercise. 32 Patients were excluded for the following conditions: (a) absence of PAD (ABI > 0.90 at rest and ABI > 0.73 after exercise), (b) non-compressible vessels (ABI ≥ 1.40), (c) asymptomatic PAD, (d) use of medications indicated for the treatment of claudication (cilostazol or pentoxifylline) initiated within 3 months prior to investigation, (e) exercise limited by other diseases or conditions, (f) active cancer, (g) end stage renal disease defined as stage 5 chronic kidney disease, (h) abnormal liver function, and (i) failure to complete the baseline run-in phase within 3 weeks.
Intervention and control groups
Home-based exercise rehabilitation program
The home-based exercise program consisted of 3 months of intermittent walking to mild-to-moderate claudication pain at least 3 days per week at a self-selected pace, in which exercise duration was progressively increased from 20 to 45 minutes per session, as previously described.19,26 Patients wore a step activity monitor during the exercise sessions to accurately record the duration and cadence of ambulation on a minute-to-minute basis during each exercise session.
Supervised exercise rehabilitation program
Exercise sessions in our supervised, on-site, treadmill exercise program were performed while also wearing a step activity monitor.19,26 The supervised program consisted of 3 months of intermittent treadmill walking to mild-to-moderate claudication pain 3 days per week at a speed of approximately 2 mph (3.22 km/h) and at a grade equal to 40% of the highest workload achieved during the baseline maximal treadmill test. 33 Exercise sessions progressively increased during the program from 15 to 40 minutes. The volume of exercise performed, expressed as metabolic equivalent (MET)-minutes, was matched for the home-based and supervised exercise programs. 26
Attention-control, light resistance program
Light resistance training was performed three times per week, without any walking exercise, using a Pro-Form Fusion 6.0 LX weight system (ICON Health and Fitness, Inc., Logan, UT, USA). 26 Resistance training consisted of performing both upper and lower extremity exercises. One set of 15 repetitions was performed for each exercise. Patients wore a step activity monitor during each exercise session to quantify the total time of their visits, which was designed to match the supervised exercise program on the amount of exposure patients had with the research staff.
Outcome measurements
Graded maximal treadmill test
Patients performed a progressive, graded treadmill protocol to determine study eligibility, as well as to obtain outcome measures related to exercise performance. 14 The COT, defined as the walking time at which the subjects first experienced pain, and the PWT, defined as the walking time at which ambulation could not continue due to maximal pain, were both recorded to quantify the severity of claudication. Peak oxygen uptake was measured by oxygen uptake obtained during the peak exercise workload with a Medical Graphics VO2000 metabolic system (Medical Graphics Inc., St Paul, MN, USA).
6-Minute walk test
On a separate day, typically within 1 week from the treadmill test, patients performed an over-ground, 6-minute walk test in which two cones were placed 100 feet (30.48 meters) apart in a marked corridor, as previously described. 34 The time and distance to onset of claudication as well as the total distance walked were recorded.
Walking impairment questionnaire (WIQ)
Patient-based ambulatory ability was obtained using the validated WIQ instrument for PAD patients that assesses ability to walk at various speeds and distances, and to climb stairs. 35
Medical Outcomes Study Short-Form 36 (MOS SF-36 – RAND Version 1.0)
The self-reported physical function subscale of health-related quality of life was assessed from this instrument. 36 We previously found that the physical function subscale was the most impaired subscale in patients with symptomatic PAD compared to national norms. 37 Thus, we selected the physical function subscale as a primary outcome measure for the distribution-based method to determine MCIDs.
The physical function subscale was also selected as the anchor for the anchor-based method to determine MCIDs for the other outcome measures. The physical function scale is comprised of 10 questions about performing daily activities with answer selections that include the following: limited a lot, limited a little, and not limited at all. A change in one level of a question (e.g. a pre-test response is limited a lot and the post-test response is limited a little) is equal to a 5% change in the total physical function scale. We considered a one-level change in any one of the 10 questions for the physical function scale to represent a small MCID of 5%. Based on our data, we selected a 25% change and a 40% change in the physical function scale to represent moderate and large MCIDs, respectively. These changes are equivalent to five levels of improvement among the 10 questions, in any combination, for a moderate MCID, and eight levels of improvement for a large MCID.
Statistical analyses
The data are presented as means and standard deviations (SD) for continuous variables, and frequencies with proportions for categorical variables. The normality assumption for continuous variables was checked based on Shapiro–Wilk tests. The p-values for multiple group comparison were obtained from the ANOVA tests and Pearson’s chi-squared tests for continuous and categorical variables, respectively. The equality of variance for pairwise group comparison is evaluated based on F-tests. The magnitudes of meaningful changes were calculated based on a distribution-based method by using the effect size with the formula
Results
The baseline clinical characteristics of the three groups are shown in Table 1. The groups consisted of older, overweight, symptomatic patients with mean ABI values reflecting moderately severe PAD. Additionally, the groups had a concomitantly high burden of comorbid conditions and cardiovascular risk factors, particularly hypertension, dyslipidemia, and metabolic syndrome. None of the baseline characteristics was significantly different among the groups. As described in the original investigation, 26 156 patients completed the trial and 24 did not.
Baseline clinical characteristics of each group.
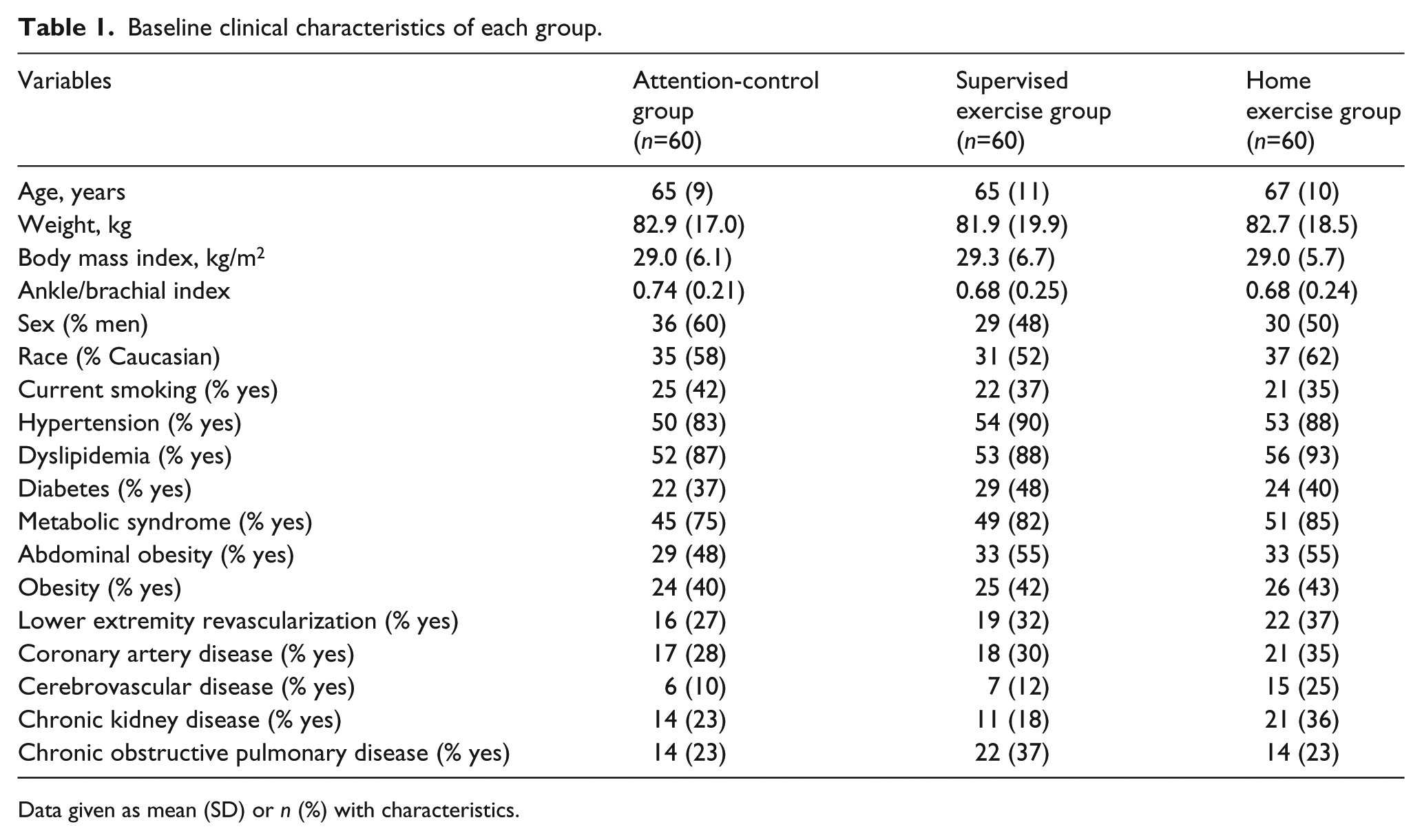
Data given as mean (SD) or n (%) with characteristics.
The group change scores of outcome measures obtained from a standardized treadmill test, a 6-minute walk test, and from the WIQ and MOS SF-36 questionnaires are shown in Table 2. The change scores were different among the three groups for COT (p = 0.020), PWT (p = 0.037), and the 6-minute walk distance (p < 0.001). These change scores were significantly greater than zero for the home-based and supervised exercise groups (p < 0.001).
Group change scores of outcome measures following 3 months of intervention.
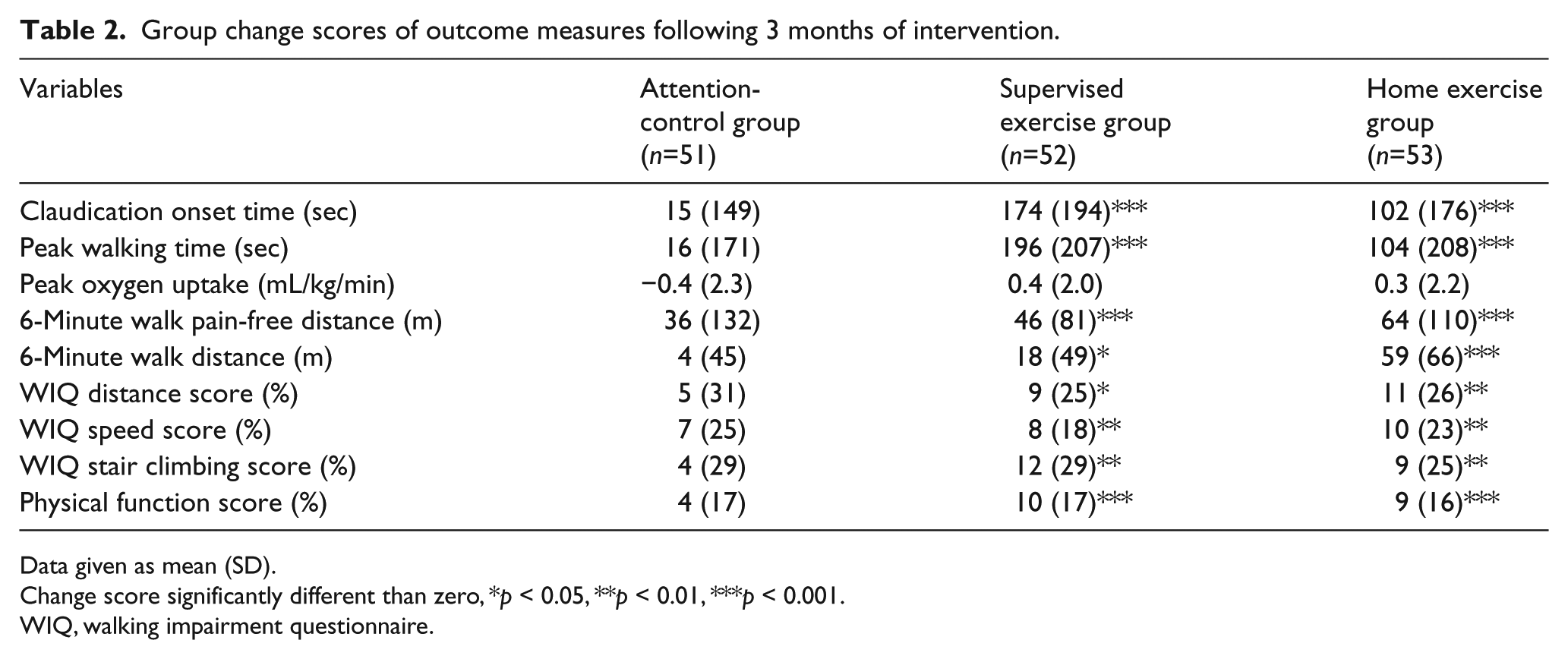
Data given as mean (SD).
Change score significantly different than zero, *p < 0.05, **p < 0.01, ***p < 0.001.
WIQ, walking impairment questionnaire.
Based on effect size calculations, the distribution-based method to determine MCIDs for small, moderate, and large changes following 3 months of intervention are shown in Table 3. Of particular interest, the MCIDs for small, moderate, and large changes in the treadmill-based outcome measure of PWT in the supervised exercise group were 38, 95, and 152 seconds, respectively, which represents one or two additional work stages completed during the treadmill test. The changes in COT were 35, 87, and 138 seconds. Furthermore, the MCIDs for small, moderate, and large changes in the 6-minute walk distance in the supervised exercise group were 9, 24, and 38 meters, respectively. The MCID change scores for the remaining variables in the supervised group, and for all of the variables in the home-exercise group, are displayed in Table 3. Similar change score values were noted between the supervised and home-based exercise groups.
Distribution-based method to calculate the MCID for small, moderate, and large changes following 3 months of intervention.
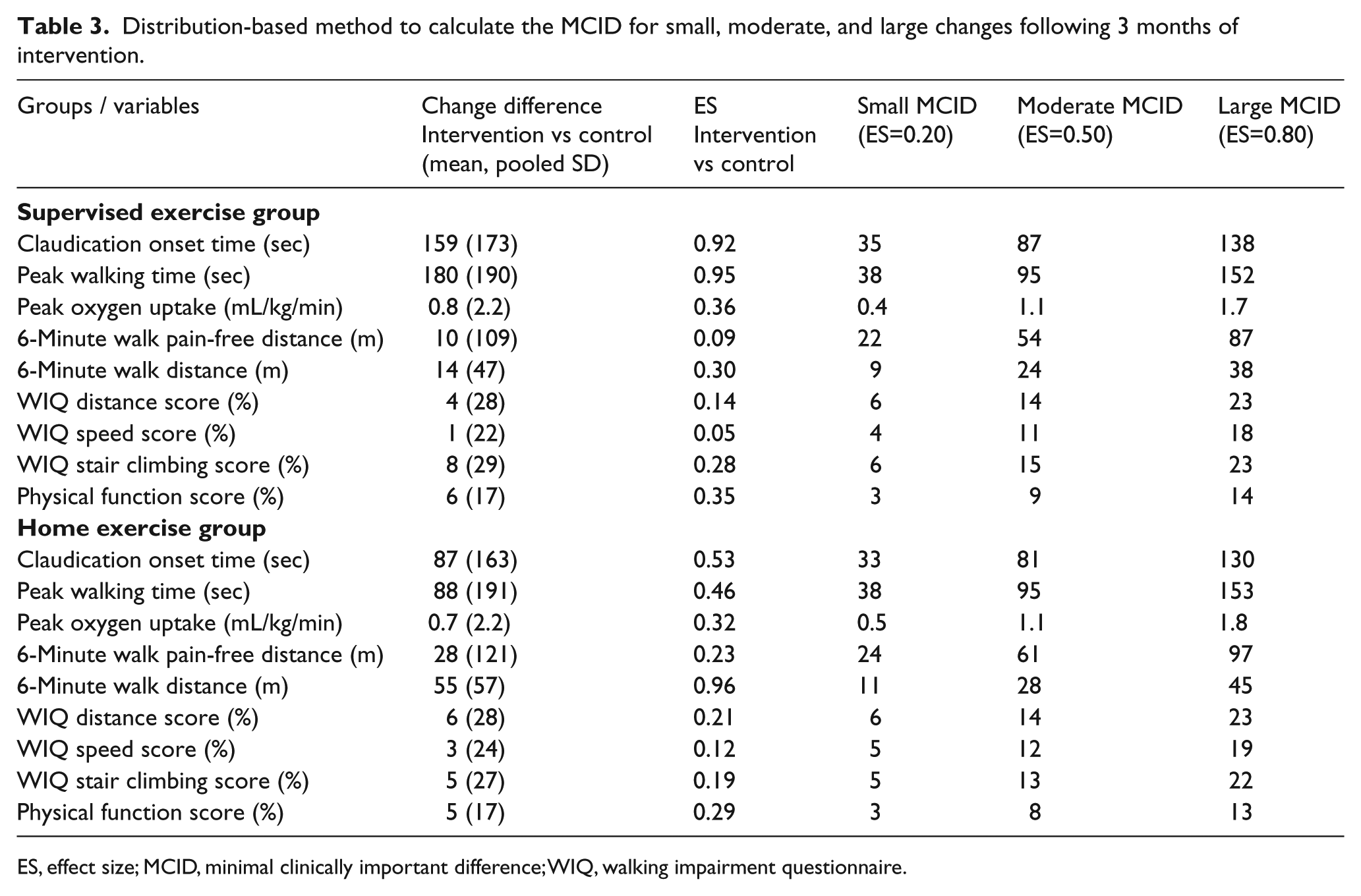
ES, effect size; MCID, minimal clinically important difference; WIQ, walking impairment questionnaire.
The anchor-based method to calculate MCIDs for small, moderate, and large changes following 3 months of intervention is displayed in Table 4. To have a larger sample size for the anchor-based method we pooled the patients from the supervised and home-based exercise groups to obtain more stable exercise estimates. The anchor-based MCIDs for small, moderate, and large changes in treadmill-based outcomes were approximately 1–2 minutes longer than the distribution-based results, as the MCIDs for PWT were 121, 141, and 241 seconds. These MCID values represent one or two additional work stages accomplished during the treadmill test after intervention compared to baseline. The anchor-based MCIDs for small, moderate, and large changes in the 6-minute walk distance were similar to the distribution-based results, as the MCIDs were 12, 32, and 34 meters, respectively. The anchor-based MCID values for the WIQ outcomes tended to be higher than the distribution-based results.
Anchor-based method to calculate the MCID for small, moderate, and large changes following 3 months of exercise intervention.
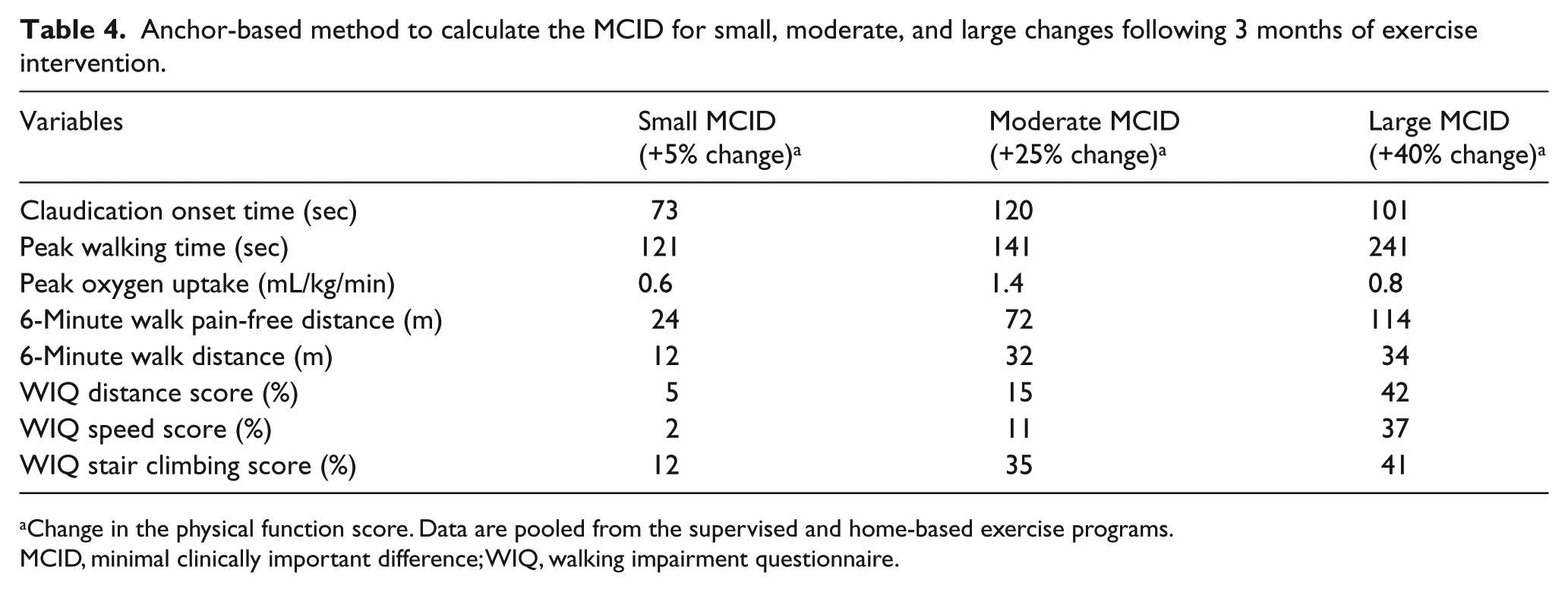
Change in the physical function score. Data are pooled from the supervised and home-based exercise programs.
MCID, minimal clinically important difference; WIQ, walking impairment questionnaire.
The number of patients needed per group to detect meaningful changes in a between-group comparison with the non-exercise control group with 80% power is shown in Table 5. The small, moderate, and large meaningful changes in PWT in the supervised exercise group require group sample sizes of 318, 52, and 21 patients, respectively. For all of the outcomes in the supervised group, a sample size near or above 300 patients was required for a small MCID change to be statistically significant, whereas moderate and large changes required near or above 50 and 20 patients. Similar sample size calculations for small, moderate, and large MCID changes were determined for the home-based exercise group.
Patients needed per exercise group for 80% power in a between-group comparison with the non-exercise control group for 3 months of intervention.
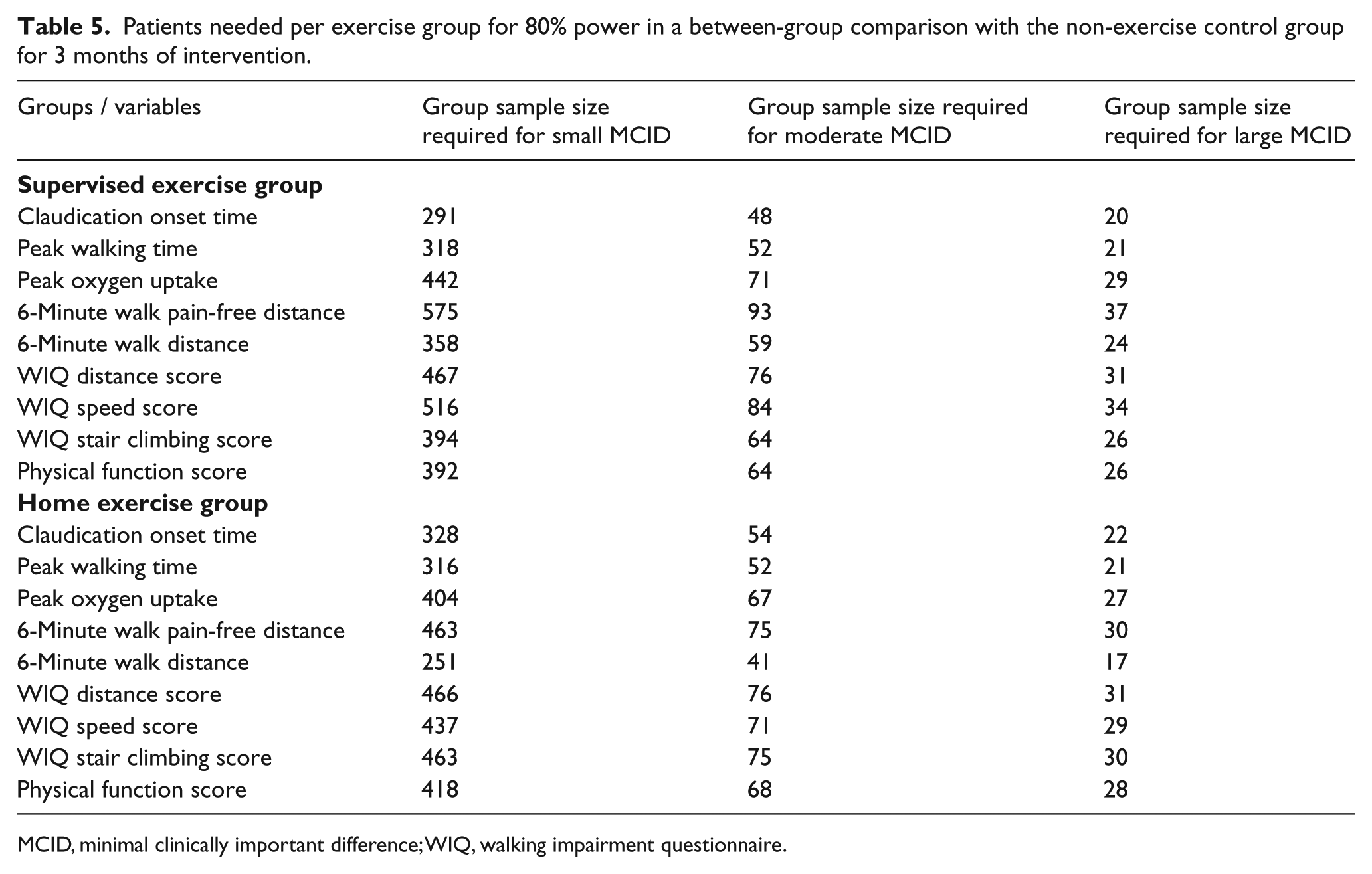
MCID, minimal clinically important difference; WIQ, walking impairment questionnaire.
To examine the robustness of the MCID calculations for a walking exercise intervention to treat claudication, we analyzed individual patient data using the distribution-based method from a previous on-site, supervised walking program 39 shown in (Table 6) to directly compare to MCID values with our distribution-based method in the current study (Table 3). The previous investigation was double in length (6 months) to the current study, and had equal or greater effect sizes for the treadmill and 6-minute walk outcomes. Significant improvements were observed in the supervised exercise group for the objective measurements during the treadmill test and 6-minute walk test in the previous report, and these changes were different than the non-exercise control group. 39 The MCIDs for small, moderate, and large changes in PWT following 6 months of supervised exercise were 48, 119, and 191 seconds, respectively. These values for PWT are approximately 10–40 seconds greater than the shorter 3-month intervention of the current study, and a similar trend for COT was noted. The MCIDs for small, moderate, and large changes in the 6-minute walk distance following 6 months of supervised exercise were 13, 33, and 53 meters, respectively. These values are approximately 4–15 meters greater than the current study. A similar trend was observed with the WIQ variables, as slightly greater MCID values were found with the previous longer study.
Distribution-based method to calculate the MCID for small, moderate, and large changes following 6 months of intervention. 39
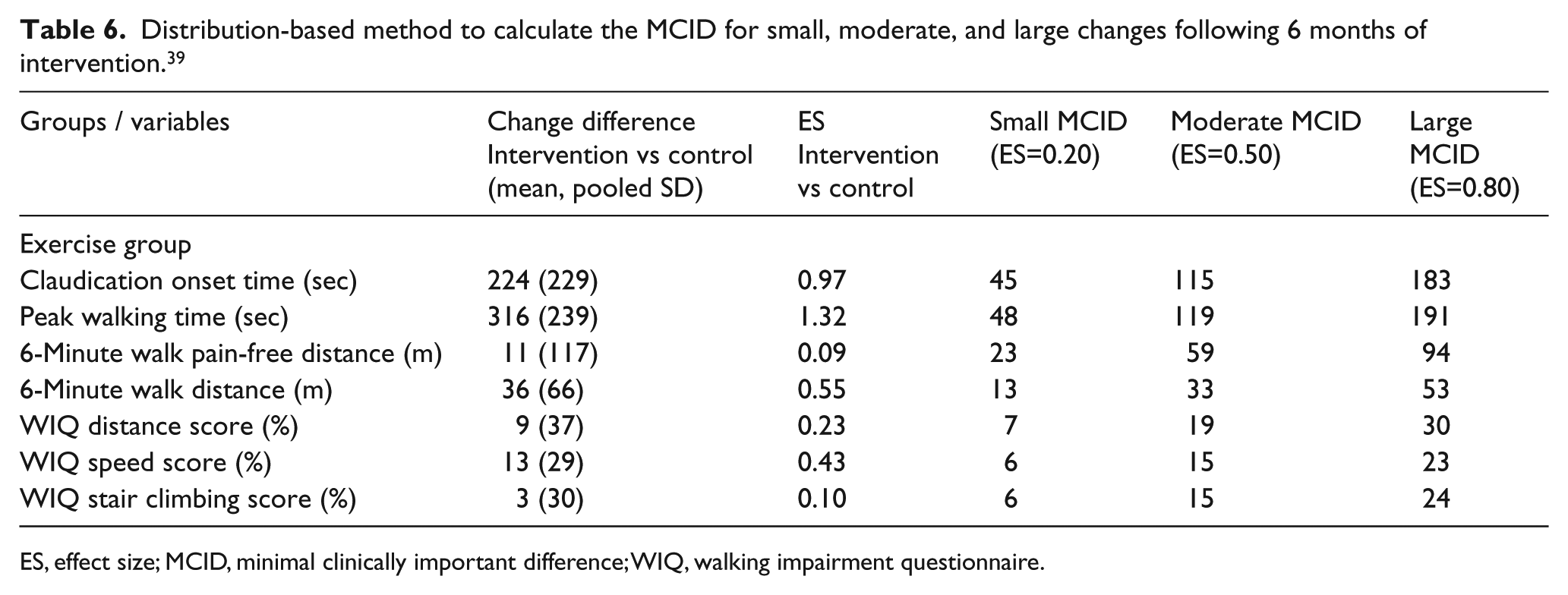
ES, effect size; MCID, minimal clinically important difference; WIQ, walking impairment questionnaire.
Discussion
The primary novel finding was that following 3-month programs of supervised and home-based exercise for symptomatic patients with PAD, the distribution-based method to determine MCIDs for small, moderate, and large changes in the treadmill outcomes of PWT and COT were approximately 0.5, 1.5, and 2.5 minutes, respectively. Furthermore, the respective changes in the 6-minute walk distance were approximately 10, 25, and 40 meters, which are smaller estimates than the previously reported small meaningful change (20 meters) and moderate meaningful change (50 meters) determined in elderly and stroke populations. 30 Compared to the distribution-based method, MCIDs determined from the anchor-based method yielded values that were approximately 1–2 minutes longer for the treadmill outcomes, and values that were similar for the 6-minute walk distance.
MCID for treadmill test outcomes
This is the first study attempting to define the MCID for changes in treadmill-based outcome measures, such as PWT and COT, in patients with PAD. A standardized treadmill test is the most common exercise methodology to assess ambulatory limitations in symptomatic patients with PAD, and is argued to be superior to 6-minute walk tests for this purpose. 18 This is particularly true for graded treadmill protocols that utilize progressive increments in workload.14,18 Yet, despite the preference to use treadmill tests in many clinical settings, a limitation is that the MCID in the treadmill performance of PAD patients has not been previously defined.18,31 Our distribution-based results indicate that the MCIDs for changes in PWT following either an on-site supervised or a home-based exercise program were identical for a small change (38 seconds) and for a moderate change (95 seconds), and was nearly identical for a large change (152 and 153 seconds). Similar distribution-based results were found for MCIDs in COT following both exercise programs, and the values were slightly lower than for PWT. These distribution-based results were confirmed by comparison with results from an earlier study published from our laboratory, although the MCID values were slightly greater, perhaps because the intervention duration of 6 months was twice that of the current 3-month study. An anchor-based approach resulted in higher MCID values for PWT and COT. Thus, compared to the distribution-based results, greater objective changes in the treadmill outcomes were needed for the patients to subjectively report that they experienced improvements in their physical function. However, the differences between the distribution-based and anchor-based methods are not large enough to result in much change in the clinical interpretation of the treadmill test outcomes. For example, small MCID values for PWT and COT determined by both methods range between 33 and 121 seconds, and therefore all values occur during or at the end of one 2-minute work stage during the progressive treadmill test. For moderate MCIDs for PWT, the anchor-based approach resulted in a value of 141 seconds compared to the distribution-based approach of 95 seconds. Thus, the clinical interpretation is that the PWT needs to improve by two treadmill work stages for a MCID if the anchor-based method is selected, whereas PWT only needs to improve by one treadmill work stage for the distribution-based approach.
Given that the effect size to increase PWT and COT was approximately 1.0 for the supervised exercise group, and that this compares favorably to the effect sizes ranging between 0.87 and 1.01 with graded treadmill test outcomes following exercise training, 18 the results from this study are representative of many previous studies published in the literature, and it is reasonable to expect these effect size changes to occur in future studies. For the graded treadmill protocol used in this study, the moderate-to-large effect sizes correspond to approximately 1.5 to 2.5 more minutes (distribution-based method) and 4 more minutes (anchor-based method) of walking, which equate to one or two additional increments in work stages during the treadmill test. Therefore, patients who increase PWT and COT by a moderate-to-large MCID in walking duration also achieve a higher maximal work rate of 0.55 to 1.1 METs associated with the greater work stages completed. 40 Thus, meaningful changes in PWT and COT during the treadmill test also indicate changes in metabolic capacity, which is not true for the 6-minute walk test.
MCID for 6-minute walk test outcomes
One advantage that has been touted for performing a 6-minute walk test to assess ambulatory function in patients with PAD is that a MCID has been established for the total distance walked during this test, albeit not in a PAD population. 30 Using a distribution-based method similar to that used in the current study, small and moderate meaningful changes in the 6-minute walk distance were reported to be 20 meters and 50 meters, respectively. 30 However, this work was based on baseline and follow-up observations on older adults with mobility disability, subacute stroke survivors, and community-dwelling older adults in which the baseline SD was used to calculate effect size. Given that the metric of interest in these repeated measures designs is the change score, the pooled SD of the change score is more appropriate to calculate effect sizes and MCIDs. Another concern is that the small and moderate meaningful changes of 20 and 50 meters are only generalizable to the studied populations, and therefore application of these values to patients with PAD 31 may not be valid. Our distribution-based results indicate that PAD-specific determinations of small and moderate meaningful changes are only half of those reported for older adults and stroke patients, as the respective values are approximately 10 meters and 25 meters. Additionally, the large MCID in this report is 38 meters for the supervised exercise program and 45 meters for the home-based exercise program, both of which are smaller than even the moderate change in the other populations. 30 Our anchor-based MCID results for the 6-minute walk distance are similar to the distribution-based values. The smaller meaningful changes in the 6-minute walk distance in the current study of PAD patients are due to the smaller pooled SDs of the change scores. For example, our pooled SDs of the change scores in the 6-minute walk distance for comparison of control versus supervised exercise group and that of control versus home exercise group are only 47 and 57 meters, respectively, whereas our baseline SD for the 6-minute walk distance pooled from three groups is 95 meters.
MCID for patient-based outcomes
Patient-based outcomes are being recognized as increasingly important in evaluating ambulation and health-related quality of life of patients with PAD.41,42 Despite the common measurement of the WIQ and MOS SF-36 questionnaires to quantify the patient-based outcomes, MCID changes have not been previously examined in patients with PAD. This is the first study to use both distribution-based and anchor-based methods to determine MCIDs for small, moderate, and large changes following supervised and home-based exercise programs in patients with PAD. In general, the effect sizes for the distance, speed, and stair climbing scores on the WIQ, and the physical function subscale of the MOS SF-36 questionnaire are much smaller than the objective measurements from the treadmill and 6-minute walk tests. This is an important consideration in planning a study, as determining statistical change to correspond with MCIDs requires a larger sample size. Alternatively, a more intense intervention may be necessary to elicit moderate-to-large changes in the patient-based outcomes that are typically observed with the objective measurements of PWT, COT, and a 6-minute walk distance. For interventions to treat PAD patients with smaller effect sizes, such as those between 0.11 and 0.32 typically observed for medication therapy, 18 large multi-site trials that enroll more than several hundred patients per treatment arm are required for the expected small MCID to also reach statistical significance.
Limitations
Although this study has determined MCIDs in common outcome measures for patients with PAD, particularly treadmill-based outcomes of PWT and COT, several limitations exist. It is possible that there was a self-selection bias related to study participation, as patients were volunteers and may represent those with the highest interest in participating, the best access to transportation, and the best health compared to non-volunteers. Additionally, these results are only applicable to symptomatic PAD patients, and may not generalize to patients with different disease severity. A final limitation is that although patients were randomized into one of the groups prior to intervention, the possibility that those who participated in home exercise were more motivated than other patients cannot be ruled out. However, these results are generalizable to symptomatic patients with PAD who typically have a high prevalence of comorbid conditions.
Conclusion and clinical significance
Three months of supervised and home-based exercise programs for symptomatic patients with PAD results in distribution-based MCID small, moderate, and large changes ranging from 0.5 and 2.5 minutes for PWT and COT. An anchor-based approach yields higher MCID values, ranging from a minimum of 73 seconds for COT to a maximum of 4 minutes for PWT. The clinical implication is that a goal for eliciting MCIDs in symptomatic PAD patients through a walking exercise intervention is to increase PWT and COT by up to 4 minutes, which corresponds to two work stages during the standardized progressive treadmill test.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this research was supported by grants from the National Institute on Aging (R01-AG-24296) and General Clinical Research Center (M01-RR-14467) sponsored by the National Center for Research Resources.
