Abstract
Calf claudication is a significant cause of walking limitation for patients with peripheral artery disease (PAD). Ankle-foot orthoses (AFO) are leg devices that can reduce the physical demands on the calf muscles during ambulation. The purpose of this study was to determine the efficacy of AFO on walking ability in patients with PAD. This was an open-label, interventional trial including 15 patients with calf claudication who were fit with AFO. Patients completed graded treadmill testing, followed by 12 weeks of unstructured community-based walking using the AFO ad libitum. Comparison of peak walking time (PWT) at baseline versus 12 weeks was the primary outcome. A secondary outcome was claudication onset time (COT) assessed during graded treadmill tests. Change in walking ability of AFO group patients was also compared to outcomes from a historical PAD control group (n = 10) who received upfront advice to walk at home. Patients in the AFO group significantly improved their walking ability from baseline to 12 weeks (mean ± SD) (PWT: 7.8 ± 5.1 to 9.3 ± 5.4 min, p = 0.049; COT: 3.0 ± 2.3 to 4.8 ± 2.7 min, p = 0.01). Change in PWT for AFO group patients when tested without using the devices was not significantly greater compared to historical controls (+1.4 ± 2.4 vs +0.1 ± 2.6 min, p = 0.16) but it was for COT (+1.8 ± 2.5 vs −0.6 ± 2.2 min, p = 0.02). This study found that AFO used during community-based walking improved the primary outcome of PWT in patients with PAD. Further, using AFO delayed claudication onset, indicating patients may be able to increase their walking activity. Large-scale, randomized controlled trials are needed to further explore the use of AFO for PAD.
Keywords
Introduction
Peripheral artery disease (PAD) is a major public health problem, currently affecting 8.5 million US adults. 1 PAD results from atherosclerotic plaque accumulation in the arteries of the lower limbs, leading to a decreased blood flow and potential claudication. 2 Claudication most commonly presents in the calves,3,4 and often results in impaired walking ability and functional performance as well as a lower quality of life.5,6
Treatment of calf claudication includes exercise training to improve the walking ability of those with PAD. 7 Although structured community-based exercise training has shown promise for improving outcomes in those with claudication,8–10 these programs have primarily been limited to research trials. Thus, patients with PAD typically only receive upfront advice from healthcare providers to increase walking activity while unsupervised at home. 11 A significant barrier to the success of community-based exercise programs is the calf claudication that patients have during walking, 12 as there are few options to attenuate the pain they experience. Novel approaches to reducing patients’ calf pain during exercise are needed to improve their health-related outcomes.
Carbon fiber devices called ankle-foot orthoses (AFO) have led to improvements of walking ability in several patient populations with impaired ambulation (e.g. those with stroke or cerebral palsy).13,14 These devices store elastic energy early in a footstep, followed by a release of the stored energy during the propulsive phase of walking. AFO may provide an augmentation of calf muscle performance, reducing claudication during ambulation and thereby improving the walking ability of patients. Thus, the aim of this study was to examine the effect of AFO used by patients with PAD during an unstructured community-based walking program. We hypothesized that patients participating in unstructured exercise in community settings, but enhanced by AFO, would improve the primary outcome of peak walking time (PWT) assessed with a graded treadmill from baseline to post-intervention. A secondary aim compared change in PWT for patients who received AFO to that of a historical control group of patients with PAD, who were only provided upfront advice to walk at home. 8
Methods
Study design and patients
The current study was an open-label, interventional trial with a historical comparator group. Testing was conducted from March 2015 to July 2016 for patients with PAD enrolled in the AFO arm of the trial, and August 2010 to July 2013 for patients in the historical control group. 8 Patients enrolled in the AFO arm included male and female patients ⩾ 40 years of age diagnosed with PAD. Patients were included if they had an ankle–brachial index (ABI) of ⩽ 0.90 and calf pain as the primary rate-limiter to walking. Patients were excluded based on the following: (1) lower extremity amputation(s) interferring with walking, (2) critical limb ischemia, (3) non-atherosclerotic PAD, (4) major surgeries contraindicating exercise or coronary artery bypass graft within 6 months of screening, (5) severe chronic obstructive pulmonary disease (COPD), (6) symptoms of angina, (7) severe symptomatic heart failure, (8) myocardial infarction, stroke or transient ischemic attack within 3 months of screening, (9) acute coronary syndrome diagnosis prior to screening, (10) significant ischemia documented on the 12-lead electrocardiogram, (11) left bundle branch block or sustained ventricular tachycardia of > 30 seconds during screening, (12) uncontrolled hypertension (⩾ 180 systolic or ⩾ 100 diastolic resting blood pressure) or poorly controlled diabetes (glycated hemoglobin > 12%), and (13) anemia (women: hemoglobin < 11 g∙dL−1; men: < 10 g∙dL−1). Inclusion/exclusion criteria were similar for patients in the historical control group. 8 However, recruitment criteria for the historical controls included patients who had (1) claudication originating in any muscle group and an abnormal ABI, and/or (2) previously received peripheral endovascular revascularization 4–6 weeks before baseline testing. The local institutional review board approved the protocol and patients signed written informed consent prior to any study-related procedures.
Ankle-foot orthoses fittings
Prior to outcome assessments, commercially available AFO (WalkOn Reaction, Ottobock, Austin, TX, USA) (Figure 1) were individually fit to both legs of patients by certified orthotists. Elements of the AFO design included: (1) dual straps immediately below the knee and at the lower calf, and (2) a medial positioned strut to connect the carbon fiber shin and full-length footplates, with the latter fitting beneath the insole of the shoe. An initial skin screening for areas of diminished sensation was conducted and soft foam materials were used to cushion any areas of skin-to-device contact. Patients received training for appropriate adjustment of the straps to ensure the devices remained attached to the lower limbs, but not constricting at a level to reduce blood flow. Patients then walked in the clinic using the AFO, navigating turns at varying speeds. Any gait or balance issues when patients walked with the devices were addressed by the orthotists. An information handout outlining a 1-week familiarization schedule for the AFO was provided to patients to ensure optimal conditioning to the devices. Instructions included (1) using the AFO for no more than 30 minutes on the first day, (2) adding 30 minutes of wear time each day thereafter, and (3) monitoring of lower limb skin three to four times per day when using the AFO to identify any redness. Patients were asked to wear long socks that extended above the proximal portion of the shin plate, or short socks that were below the distal end of the plate (to prevent pressure seams from developing). Patients were informed to contact the orthotists for adjustments to the devices if redness did not dissipate within 30 minutes. Patients were also provided with detailed instructions on appropriate nail care and were recommended to use shoes with a wide and deep toe box.

The Ottobock WalkOn Reaction ankle-foot orthosis used for the current trial (image used with permission).
Outcome assessments
Following the 1-week AFO familiarization period, patients completed two baseline outcome visits within 1 week. Walking ability and functional performance outcomes were assessed with patients (1) wearing the AFO and (2) not wearing the AFO on separate days for the two visits. The same procedures were used for the post-12-week outcome assessments. The order of device usage was randomized using a computer-based permuted block allocation scheme (held by unblinded personnel). Study personnel who conducted the outcome assessments were blinded to whether patients were or were not using the AFO. To accomplish this, unblinded personnel placed cloth coverings over the patients’ lower limbs prior to outcome assessments. The historical control group only completed assessments once at baseline and once at follow-up (14 weeks).
Walking ability and cardiovascular performance endpoints
Graded exercise tests were conducted on a treadmill using a modified Gardner protocol, 15 as previously described. 8 PWT (min) was recorded as the maximal time the patient could walk on the treadmill before stopping secondary to severe leg pain. Claudication in the calves was measured at each test stage using the Claudication Symptom Rating Scale as previously described. 16 Claudication onset time (COT; min) was obtained with the patient pointing to the scale at the initial presentation of calf pain for AFO group patients. An open-circuit respiratory-metabolic system was also used to measure peak oxygen consumption (VO2peak) (mL∙kg−1∙min−1).
Functional performance, patient-reported outcomes, and leg circumferences
To assess the functional performance of AFO group patients, the 6-minute walk test was completed in a 15.24-meter corridor at the hospital following previously reported procedures. 17 The peak walking distance (PWD; m) achieved in 6 minutes was recorded for both device usage/non-usage, in addition to claudication onset distance (COD; m) verbalized by the patient during testing. Patient-reported outcomes were assessed prior to the walking ability tests at baseline and 12 weeks using the Walking Impairment Questionnaire (WIQ) 18 and Medical Outcomes Study Short Form 36-item (SF-36) questionnaire. 19 Bilateral leg circumferential measurements were also conducted prior to walking ability tests at the thigh and shin per previously published guidelines. 20 Historical controls did not complete the 6-minute walk tests or leg circumference assessments.
Unstructured community-based walking exercise
Following the final baseline outcome visit for patients who received AFO, upfront advice to walk as much as possible using the devices was provided, with no further follow-up. Recommendations per published guidelines included walking to induce moderate leg pain in 3–5 minutes, followed by rest until the pain subsided. 21 Walking and resting was advised for a total of 35–50 minutes each session, three times per week for 12 weeks.21,22 In the historical control group, patients were provided the same upfront exercise advice and returned 14 weeks later for follow-up testing.
Statistical analyses
The primary endpoint in the intent-to-treat analysis was the intra-comparison of treadmill PWT without wearing the devices assessed at baseline and 12 weeks for patients in the AFO group. Separate, two-sided, paired sample t-tests were used to compare PWT between baseline and 12 weeks: (1) with patients not wearing the AFO during testing; and (2) while patients were wearing the devices during outcome assessments. The same approach was used for all other secondary outcomes of interest. Separate paired sample t-tests were also performed to assess differences for intra-group outcome change scores from baseline to week 12 between not using the AFO and AFO usage during testing.
To determine whether the AFO provided benefits for patients with PAD who completed an unstructured walking exercise program, outcomes were compared with those of the historical control group of patients. Analyses of covariance with previous peripheral endovascular revascularization pre-specified as a covariate were conducted separately on outcome change scores for historical controls compared to the AFO group when patients (1) were and (2) were not wearing the devices during testing. The same analysis was performed to compare change scores for patient-reported outcomes between AFO group patients and the historical controls. Imputation of outcomes using the expectation maximization algorithm 23 was used for patients lost to follow-up. Prior to analyses, assumptions of parametric testing were performed, and pre-specified logarithmic transformations of data were conducted in the event of non-normality. Significance was defined as α < 0.05. All analyses were conducted with a standard statistical software package (SPSS, version 24; IBM Corp., Armonk, NY, USA).
Sample size calculations for patients receiving AFO were based on data from the previously described clinical trial 8 that randomized patients with PAD to a structured community-based walking exercise program (intervention: n = 10) or to an upfront advice to walk at home group (control: n = 10). The historical study demonstrated near significant differences between intervention and control groups for the primary outcome of change in PWT (p = 0.052). Thus, based on a paired samples t-test, with an effect size of 0.94, a sample of 12 patients was estimated to provide 80% power at α = 0.05 to detect a significant PWT difference between baseline and post-12 weeks within the AFO group of patients. The sample size for those recruited to the AFO arm was increased to 15 to account for a 20% attrition rate.
Results
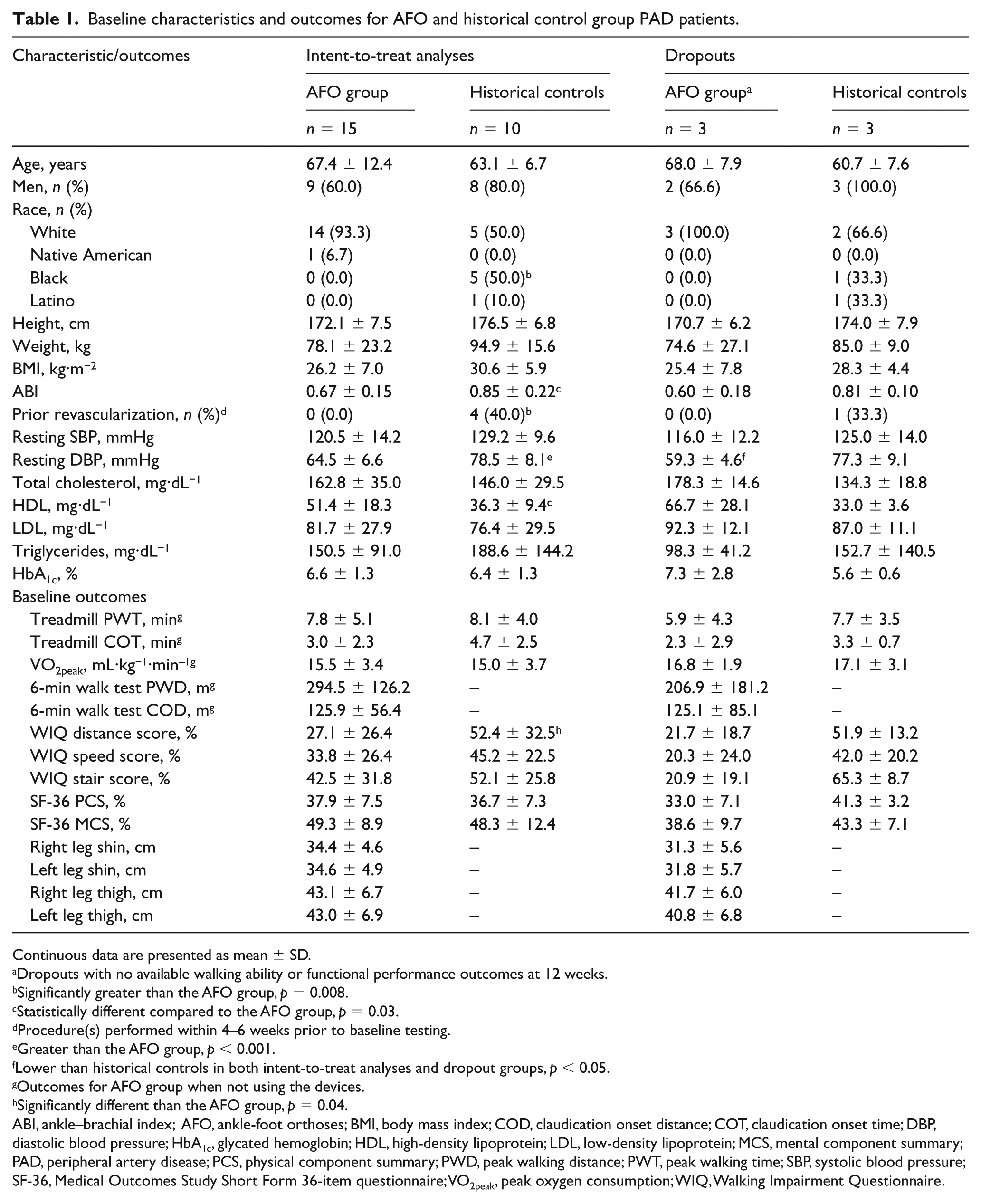
Fifteen patients were initially screened and met the criteria for inclusion in the AFO arm of the study. Three patients were excluded prior to the 12-week time point due to the following: significant chronic respiratory problems (n = 1), inability to achieve a proper fit with the AFO due to an anatomical malformation (n = 1), and diagnosis of end-stage COPD and placement in hospice care (n = 1). One additional patient did not complete the post-12-week outcome assessments using the AFO due to a lower limb skin infection that was exacerbated by the AFO. Prior to imputation for missing data per the pre-specified intent-to-treat design, data were found to be missing completely at random (Little’s MCAR test: χ2(244) = 70.0, p = 1.00). Outcomes for historical controls (n = 10) derived from the previous study 8 consisted of complete data post-randomization. Table 1 depicts baseline characteristics and outcomes for the AFO and historical control groups as well as dropouts. Compared to the AFO group of patients, there were more historical control patients who had previously received peripheral endovascular revascularization (p = 0.008) and were black (p = 0.008). At baseline, patients in the historical group had a higher diastolic blood pressure (p < 0.001), exhibited a higher ABI (p = 0.03), had lower high-density lipoprotein cholesterol (p = 0.03), and had a greater WIQ distance score (p = 0.04). All other characteristics were similar between the two groups at baseline. For dropouts, diastolic blood pressure was lower for patients removed from the AFO arm compared to historical controls (both dropouts and those with complete data, p < 0.05). All other baseline characteristics and outcomes between dropouts and the subsets used in the final analyses were similar (p > 0.05).
Baseline characteristics and outcomes for AFO and historical control group PAD patients.
Continuous data are presented as mean ± SD.
Dropouts with no available walking ability or functional performance outcomes at 12 weeks.
Significantly greater than the AFO group, p = 0.008.
Statistically different compared to the AFO group, p = 0.03.
Procedure(s) performed within 4–6 weeks prior to baseline testing.
Greater than the AFO group, p < 0.001.
Lower than historical controls in both intent-to-treat analyses and dropout groups, p < 0.05.
Outcomes for AFO group when not using the devices.
Significantly different than the AFO group, p = 0.04.
ABI, ankle–brachial index; AFO, ankle-foot orthoses; BMI, body mass index; COD, claudication onset distance; COT, claudication onset time; DBP, diastolic blood pressure; HbA1c, glycated hemoglobin; HDL, high-density lipoprotein; LDL, low-density lipoprotein; MCS, mental component summary; PAD, peripheral artery disease; PCS, physical component summary; PWD, peak walking distance; PWT, peak walking time; SBP, systolic blood pressure; SF-36, Medical Outcomes Study Short Form 36-item questionnaire; VO2peak, peak oxygen consumption; WIQ, Walking Impairment Questionnaire.
Peak walking time, claudication onset time, and peak oxygen consumption outcomes
Patients with PAD who used AFO during unstructured community-based walking exercise, had a 35% improvement (+1.4 min, p = 0.049) in PWT from baseline to post-12 weeks when not using the devices during graded exercise testing. When patients performed treadmill tests using the AFO, a 65% improvement (+2.4 min, p = 0.01) was exhibited at 12 weeks. PWT change scores when AFO group patients did not wear the devices compared to when they did wear the devices during treadmill testing were not significantly different (mean ± SD) (+1.4 ± 2.4 vs +2.4 ± 2.9 min, p = 0.13). When AFO group patients did not use the devices during testing, PWT change scores were not significantly greater than the historical control group (+0.1 ± 2.6 min, p = 0.16). However, change in the PWT when patients wore the AFO during testing was significantly different compared to historical controls (p = 0.046). Figure 2 depicts the baseline and post-intervention PWT for the AFO group (both wearing the devices and not wearing the devices during testing) and for the historical control group.
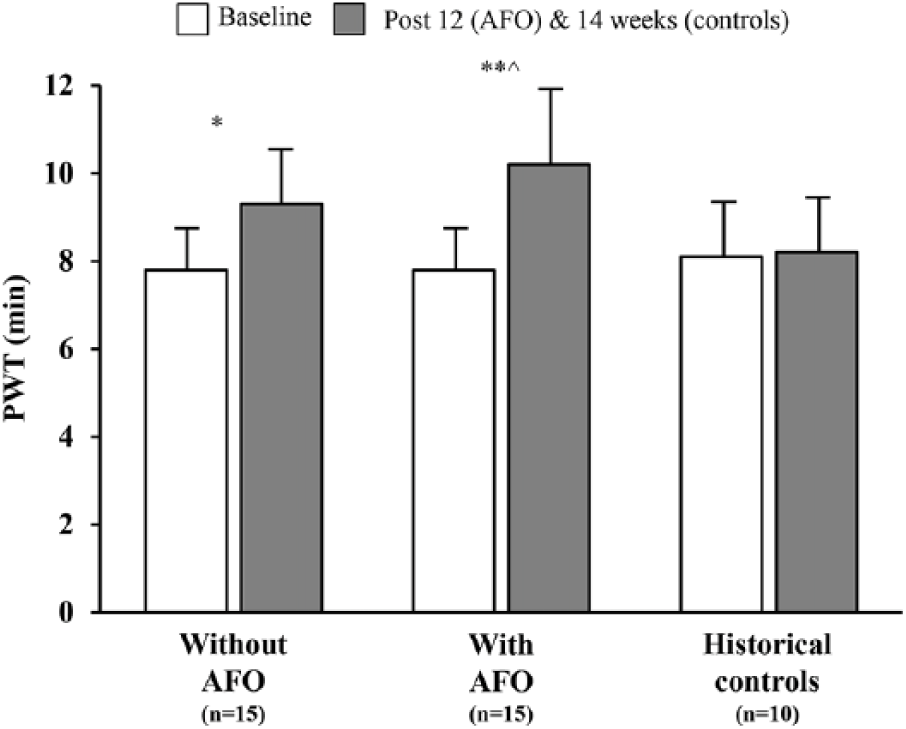
Treadmill PWT for AFO group patients and historical controls.
Analysis of COT for AFO group patients indicated significant improvement from baseline to post-12 weeks, both using the AFO (4.3 ± 4.0 vs 7.0 ± 5.0 min, p = 0.01) and not using the AFO (3.0 ± 2.3 vs 4.8 ± 2.7 min, p = 0.01) during exercise testing. Additionally, significant group mean improvements were demonstrated for COT when compared to the historical controls (–0.6 ± 2.2 min), regardless of whether the devices were used (+2.7 ± 2.9 min, p = 0.02) or not used (+1.8 ± 2.5 min, p = 0.02) during graded treadmill tests. Figure 3 provides a graphical depiction of COT at baseline and post-outcome assessment time points for AFO group patients using the devices and not using the devices, as well as for historical controls.
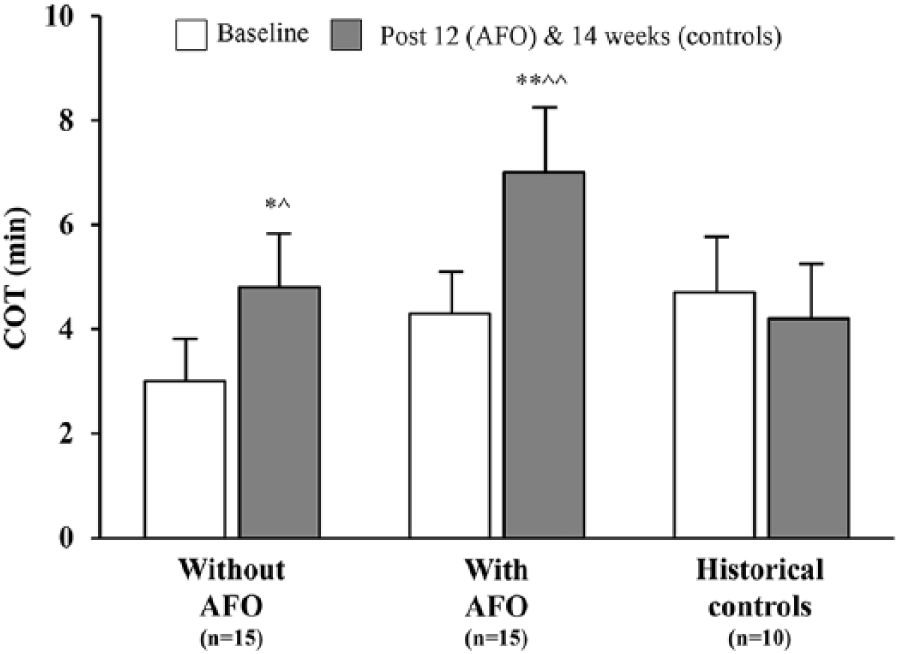
COT during treadmill testing for AFO group patients and historical controls.
No significant differences were found for VO2peak between baseline and 12 weeks within the AFO group of patients (wearing AFO: 15.6 ± 4.0 vs 16.6 ± 3.2 mL∙kg−1∙min−1; not wearing AFO: 15.5 ± 3.6 vs 15.9 ± 3.8 mL∙kg−1∙min−1). The inter-group differences of VO2peak change scores between AFO group patients and the historical control patients also demonstrated non-significant differences (wearing AFO: +1.0 ± 2.4 mL∙kg−1∙min−1; not wearing AFO: +0.4 ± 2.9 mL∙kg−1∙min−1; historical controls: +0.9 ± 3.0 mL∙kg−1∙min−1).
Functional performance, patient-reported outcomes, and leg circumferences
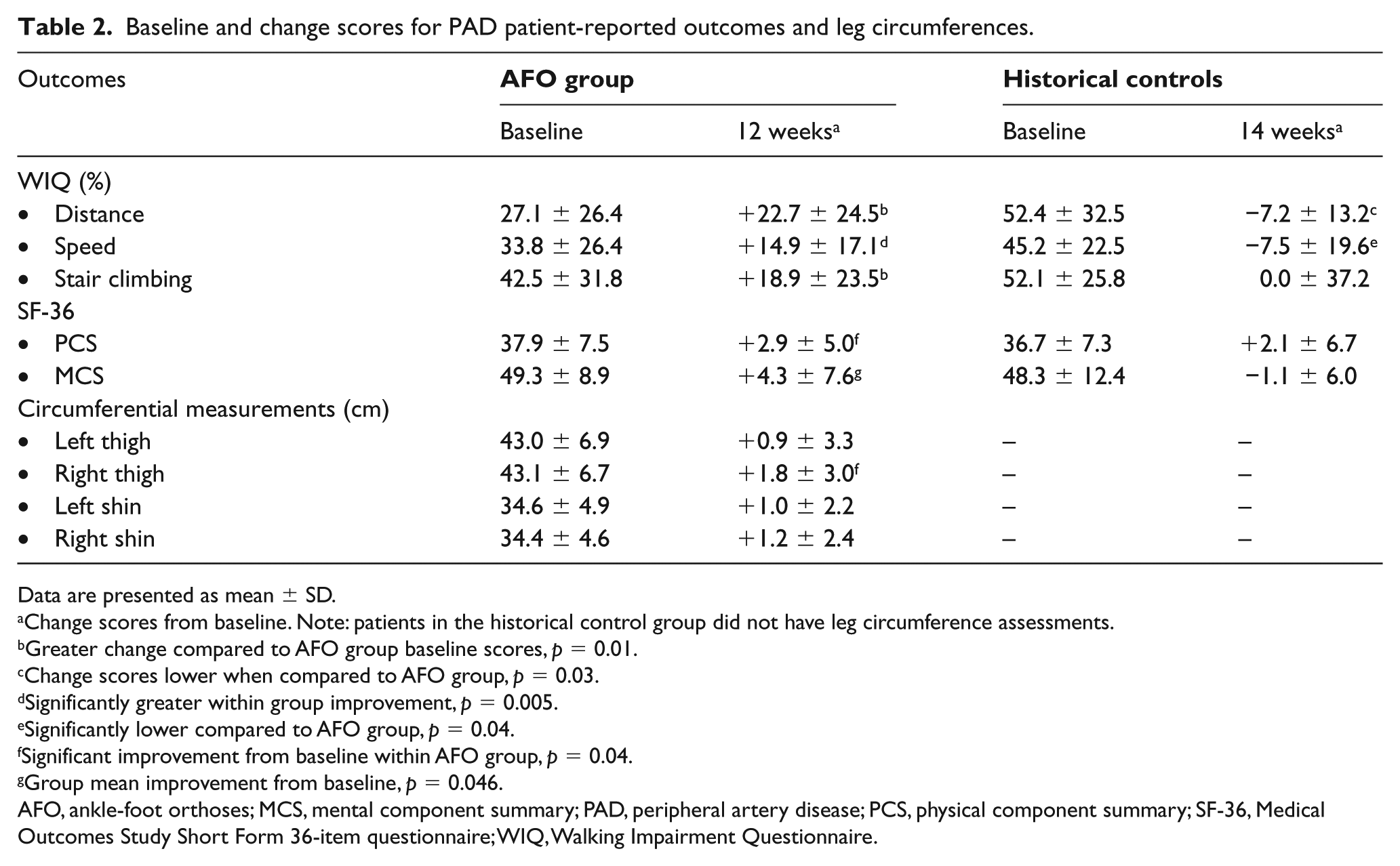
Functional performance as measured by the 6-minute walk test improved significantly over the 12 weeks for patients who received the AFO. When patients did not use the AFO during testing, both PWD and COD increased from baseline to the post-outcome assessments (PWD: 294.5 ± 126.2 to 355.9 ± 95.7 m, p = 0.02; COD: 125.9 ± 56.4 to 182.3 ± 72.9 m, p = 0.01). Additionally, functional performance measured when patients used the AFO during the 6-minute walk tests demonstrated significant improvements in PWD (318.2 ± 108.3 to 354.4 ± 109.1 m, p = 0.01) and COD (164.2 ± 108.5 to 222.2 ± 127.0 m, p = 0.01). Between the two conditions (with vs. without the AFO during testing), there were no significant differences in 12-week change scores for PWD (+36.2 ± 50.2 vs +61.4 ± 62.8 m) and COD (+57.9 ± 57.7 vs +56.4 ± 54.9 m). Overall, 12-week patient-reported outcomes were significantly higher for all domains of the WIQ and the SF-36 when analyzing within the AFO group (p < 0.05). All leg circumferences demonstrated positive numerical trends from baseline to 12 weeks, but only the right thigh was significantly greater (43.1 ± 6.7 to 44.9 ± 6.6 cm, p = 0.04). Table 2 provides patient-reported outcomes for patients in the AFO study and historical controls, as well as circumferential measures for AFO group patients.
Baseline and change scores for PAD patient-reported outcomes and leg circumferences.
Data are presented as mean ± SD.
Change scores from baseline. Note: patients in the historical control group did not have leg circumference assessments.
Greater change compared to AFO group baseline scores, p = 0.01.
Change scores lower when compared to AFO group, p = 0.03.
Significantly greater within group improvement, p = 0.005.
Significantly lower compared to AFO group, p = 0.04.
Significant improvement from baseline within AFO group, p = 0.04.
Group mean improvement from baseline, p = 0.046.
AFO, ankle-foot orthoses; MCS, mental component summary; PAD, peripheral artery disease; PCS, physical component summary; SF-36, Medical Outcomes Study Short Form 36-item questionnaire; WIQ, Walking Impairment Questionnaire.
Discussion
The current study evaluated the impact of AFO used during unstructured community-based exercise on walking ability for patients with calf claudication. Results indicated PWT improved at the 12-week time point both with and without the use of the AFO during testing. Secondary outcomes of treadmill-based COT, 6-minute walk test PWD and COD, and patient-reported outcomes from the WIQ and SF-36 also improved for those who received AFO during community-based walking. When AFO group patient outcomes were compared to those of a historical control group, results indicated significant improvement in outcomes when using the AFO. This study provides evidence that there may be clinical utility of AFO for PAD in community walking programs.
To date, only one trial has evaluated the effect of brace-like devices on walking impairments of patients with PAD. In the cross-sectional study by Honet et al., 24 patients with either bilateral or unilateral calf claudication (with a contralateral above-knee amputation) were fit with ‘short-leg braces’ that prevented ankle motion. Patients completed individualized constant speed and grade treadmill tests with and without the braces. Results indicated a 40% (+1.3 min) increase in PWT when patients used the fixed braces versus testing without the braces (p = 0.02). This classic study provided promising findings but is not without limitations. Our trial employed modern, commercially available AFO that limited ankle motion but did not completely immobilize the joints. We also demonstrated that the devices could improve a patient’s walking ability when used as an adjunct to community-based walking. Outcome change scores were comparable regardless of whether the devices were used or not used during testing in the AFO group. It appears patients were not dependent on the AFO continuously to demonstrate benefit. Additionally, minimal clinically important differences (MCIDs) have been calculated for patients with PAD following walking-based interventions, and the current study demonstrated that both PWT and COT showed small to large meaningful change following AFO use. 25 However, results should be interpreted with caution given the preliminary nature of the study and the suggestion that a learning effect may have occurred before any benefit was derived.
The rationale for further evaluation of AFO to treat patients with PAD was also supported by the overall changes found in functional performance. Large meaningful change at 12 weeks when patients did not use the devices during testing was observed (+61.4 m), regardless of the method used to calculate the MCIDs. 25 Patients also self-perceived improvement in an ability to walk faster, farther, and climb stairs in addition to their quality of life. This is important for the potential acceptance of these devices by patients to treat calf claudication. Finally, a common clinical perception is that bracing a limb may result in a lower usage of the muscles, thus resulting in weakness and/or atrophy.26,27 The thigh and shin cross-sectional measures did not decrease following 12 weeks of using the AFO in patients with PAD. This has some implication that using the devices over an extended period of time may not adversely attenuate muscle size.
Supervised exercise programs in hospital settings for PAD have recently been approved for reimbursement. 28 However, due to their firm qualification criteria, resource intensity, and the requirement for those with claudication to travel to clinics for training, they are not accessible to some patients. As an alternative, patients are often relegated to the standard of care for exercise therapy, which is community-based exercise on their own. A mitigating factor for the completion of community programs is claudication experienced during ambulation. 29 Because claudication in the calf is the most common location, the ability of the AFO to delay COT and improve walking distance may improve the adoption and implementation of walking exercise in community settings. The costs of AFO are currently covered by the Centers for Medicare and Medicaid for ambulatory beneficiaries who may derive functional benefit. However, until now, the feasibility and tolerability of AFO have not been examined in modern trials. The proof of concept study serves as a starting point for further investigation into orthoses for PAD.
Limitations
There were several limitations to the current study. We employed an open-label, interventional trial but future studies should conduct randomized controlled trials to determine the impact of AFO on walking outcomes using a control group that (1) only experiences calf claudication and (2) uses sham devices to address the placebo effect. The historical controls we used as our comparator to the patients who received the AFO were limited by different inclusion criteria; thus, secondary aim results should be interpreted carefully. Although our study lays the groundwork for additional studies of AFO, it is imperative that controls are prospectively assigned in lieu of using historical patients for analyses. Our dataset also lacked ambulatory monitoring; thus, future studies should use wearable activity monitors (e.g. accelerometers) to determine if the volume of walking exercise increases in patients with PAD who are provided AFO.
Conclusion
In conclusion, the significant improvements in walking outcomes from the current study indicate a signal of benefit for patients with PAD who use AFO. It is important to determine the ease of use and how well patients can tolerate any new device to treat vascular disease. AFO for PAD would not be considered an off-label therapy and may target a primary barrier to exercise for those with PAD: calf claudication. Our study may serve as the basis for large-scale, randomized controlled trials that establish AFO as an effective means to improve the walking ability, functional performance, and quality of life of patients with PAD.
Footnotes
Acknowledgements
The authors thank the International Heart Institute’s clinical and research staff for their assistance in the trial. The authors would like to especially thank Randy Rosenquist and Cliff and Robin Creekmore at Momentum Prosthetics Clinic (Missoula, MT, USA) for their help with the orthotics component of the trial.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the study was funded by the National Institutes of Health (NIH), National Institute of General Medical Sciences, Mountain West Clinical Translational Research – Infrastructure Network under award U54GM104944. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
