Abstract
There is limited evidence to guide clinical decision-making for antiplatelet therapy in peripheral artery disease (PAD) in the setting of lower extremity endovascular treatment. The Ticagrelor in Peripheral Artery Disease Endovascular Revascularization Study (TI-PAD) evaluated the role of ticagrelor versus aspirin as monotherapy in the management of patients following lower extremity endovascular revascularization. The trial failed to recruit the targeted number of patients, likely due to aspects of the design including the lack of option for dual antiplatelet therapy, and inability to identify suitable patients at study sites. In response, the protocol underwent amendments, but these changes did not adequately stimulate recruitment, and thus TI-PAD was prematurely terminated. This article describes the rationale for TI-PAD and challenges in trial design, subject recruitment and trial operations to better inform the conduct of future trials in PAD revascularization.
Introduction
There is a paucity of evidence to guide clinical decision-making for antiplatelet therapy in peripheral artery disease (PAD) in the context of lower extremity endovascular treatment. The original evidence included meta-analyses of aspirin published in 1994 by the Antiplatelet Trialists’ Collaboration, indicating that graft patency and prevention of restenosis after peripheral angioplasty was improved by aspirin.1,2 However, since that time there have been no completed, adequately sized randomized trials addressing the role of contemporary antiplatelet therapy for promoting patency and long-term outcomes of peripheral endovascular procedures. In the context of surgical revascularization, the Clopidogrel and Acetylsalicylic acid in bypass Surgery for Peripheral ARterial disease (CASPAR) trial evaluated dual antiplatelet therapy (DAPT) after below-knee bypass grafting and failed to show a benefit for clopidogrel added to aspirin compared with aspirin alone. 3 Given this overall lack of evidence for antiplatelet therapy following lower extremity revascularization, current guidelines provide mixed recommendations for the use of antiplatelet agents in the setting of endovascular procedures. The 2011 update of the ACCF-AHA guidelines provides a Class IIb, level of evidence B recommendation for the combination of aspirin and clopidogrel for patients with prior lower extremity revascularization (endovascular or surgical). 4 However, the 2012 CHEST antithrombotic guidelines in effect at the time of the trial outlined in this report recommend aspirin alone and not DAPT after an endovascular procedure. 5 The newer guidelines for PAD published in 2016 by the American Heart Association/American College of Cardiology were not available at the time the trial described herein was designed and conducted, and have a level IIb recommendation stating that DAPT with aspirin and clopidogrel may be reasonable after lower extremity peripheral vascular intervention (PVI), but acknowledge the sparseness of supporting data for this recommendation. 6
Based on the absence of good evidence, the Ticagrelor in Peripheral Artery Disease Endovascular Revascularization Study (TI-PAD) was designed to address the role of ticagrelor versus aspirin as monotherapy in the management of patients with PAD following an endovascular procedure, with the primary endpoint of improved exercise performance. Several challenges in study design and study execution led to premature discontinuation of TI-PAD, despite amendments to the protocol designed to stimulate recruitment. This article describes the rationale for TI-PAD and the challenges encountered in trial design and subject recruitment to better inform the conduct of future trials in this area.
Hypothesis and aims
The Ticagrelor in Peripheral Artery Disease Endovascular Revascularization Study (TI-PAD I EVR, or TI-PAD for short) was a Phase 2 multicenter, randomized, controlled, double-blind, active comparator study of antiplatelet monotherapy in patients with PAD. The study tested the hypothesis that monotherapy with ticagrelor 90 mg BID would improve a functional endpoint, peak walking time (PWT), after endovascular revascularization, also referred to as PVI, in patients with PAD as compared with aspirin 100 mg daily. This hypothesis was based on the increased potency of ticagrelor for platelet inhibition compared with aspirin and the anticipation that this might result in improved patency and hence better maintenance of walking performance post procedure. In addition, ticagrelor has adenosine-like vasodilatory properties, unlike aspirin, which is postulated to improve function and exercise performance. The primary aim of the study was to compare the effect of ticagrelor versus aspirin on change in PWT from 1 to 26 weeks after endovascular revascularization. Secondary endpoints included change in claudication onset time, time course of change in peak walking time over the course/duration of the study, time course of change in limb hemodynamics, quality of life measures, Rutherford classification over the course of the study, major cardiovascular events (fatal and non-fatal myocardial infarction, stroke and vascular death), and limb events (acute limb ischemia, above-the-ankle amputations, lower extremity revascularizations). The primary safety endpoint was adjudicated bleeding events using the Thrombolysis in Myocardial Infarction Study Group (TIMI) and Platelet Inhibition and Patient Outcomes (PLATO) criteria; bleeding events were also adjudicated using Bleeding Academic Research Consortium (BARC) and International Society on Thrombosis and Haemostasis (ISTH) definitions.
Inclusion criteria for TI-PAD included the following: (1) ambulatory males and females aged 50 years or older; (2) moderate to severe claudication or ischemic rest pain due to distal superficial femoral artery, popliteal, or tibioperoneal disease; (3) patent ‘inflow’; (4) PAD defined as a resting ankle–brachial index (ABI) ≤0.90 or resting ABI >0.90 and ≤1.00 with a reduction of ≥20% in ABI when measured within 1 minute after the Screening Visit exercise treadmill test (ETT), or in patients with an ABI >1.40 (non-compressible vessels), a resting toe–brachial index <0.70; (5) a PWT between 1 and 10 minutes limited only by claudication as assessed by a graded (Gardner protocol) ETT performed at the Screening Visit; (6) being advised of smoking cessation and exercise therapy but no active changes to smoking status or exercise at the time of screening; and (7) a successful peripheral vascular intervention of the distal superficial femoral artery, popliteal, or tibioperoneal arteries. Exclusion criteria included: (1) the need for DAPT within 7 days prior to randomization; (2) the need for an antiplatelet medication other than aspirin or clopidogrel; (3) the need for cilostazol, anticoagulation, a strong CYP3A inhibitor, or a strong CYP3A inducer within 4 weeks prior to randomization; or (4) a disease process other than PAD that would interfere with exercise performance.
The sample-size calculation was based on prior drug trials (cilostazol) and revascularization trials.7,8 Meta-analysis of cilostazol trials demonstrated a net improvement of 25% in PWT over placebo, while revascularization demonstrated a net improvement of 75% over controls (no intervention). The ETT at the Week 1 Baseline Visit served as the baseline for hypothesis testing. From that baseline to the end of the study at week 26, we anticipated a further improvement in patients randomized to ticagrelor due to the adenosine effects. 9 Since this had not been formally tested, the assumption was a nominal 15% improvement, which is less than cilostazol but better than placebo. In contrast, it was anticipated that patients in the aspirin treatment arm would have no improvement given the lack of benefit with aspirin on claudication symptoms or PWT. For example, in patients with asymptomatic PAD, aspirin did not decrease the incidence of new onset claudication. 10 In contrast, more potent antiplatelet agents than aspirin have been shown to improve pain-free walking distance. 11 Thus, there was not anticipated to be any direct benefit of aspirin on PWT. However, it was expected that aspirin treatment would result in loss of patency over time relative to the ticagrelor treatment: evidence from the Platelet Inhibition and Patient Outcomes (PLATO) study demonstrated that ticagrelor was superior to clopidogrel in preventing stent thrombosis by a 25–30% relative risk reduction, 12 whereas patients treated with angioplasty (on a background of aspirin) have shown a loss of PWT over time. 13 Therefore, a conservative estimate was worse patency for patients on aspirin relative to ticagrelor, leading to an overall 15% decrease in PWT on the aspirin treatment. The net difference between groups at the end of 26 weeks would be a 30% difference in PWT. With an assumed common standard deviation of 0.4 on a natural log scale, 60 patients completing the trial per treatment group would have provided 88% power to detect a 30% difference in PWT between groups. Assuming a dropout rate of up to 30%, the final enrollment target was 175 patients to result in 120 evaluable patients.
Trial design
Original trial design
Twenty study sites in the United States were selected for participation in TI-PAD, and all site principal investigators agreed to the study design. The anticipated enrollment rate was 0.6 patients per site per month for an enrollment period of 13 months.
The study was performed in accordance with ethical principles that have their origin in the Declaration of Helsinki and are consistent with the International Conference on Harmonisation Good Clinical Practice Guideline, applicable regulatory requirements, and the AstraZeneca policy on Bioethics and Human Biological Samples. The study protocol and informed consent form were approved by the Institutional Review Board (IRB) at each site. Written informed consent was obtained from each subject prior to any study procedures.
The two treatment arms of the study were: (1) a loading dose of ticagrelor 180 mg and aspirin placebo 300 mg followed by 26 weeks of ticagrelor 90 mg BID and aspirin placebo 100 mg QD versus (2) ticagrelor placebo 180 mg and aspirin 300 mg followed by 26 weeks of aspirin 100 mg QD and ticagrelor placebo 90 mg BID. Visits were scheduled at weeks 1, 4, 13, and 26 post-PVI (Figure 1).
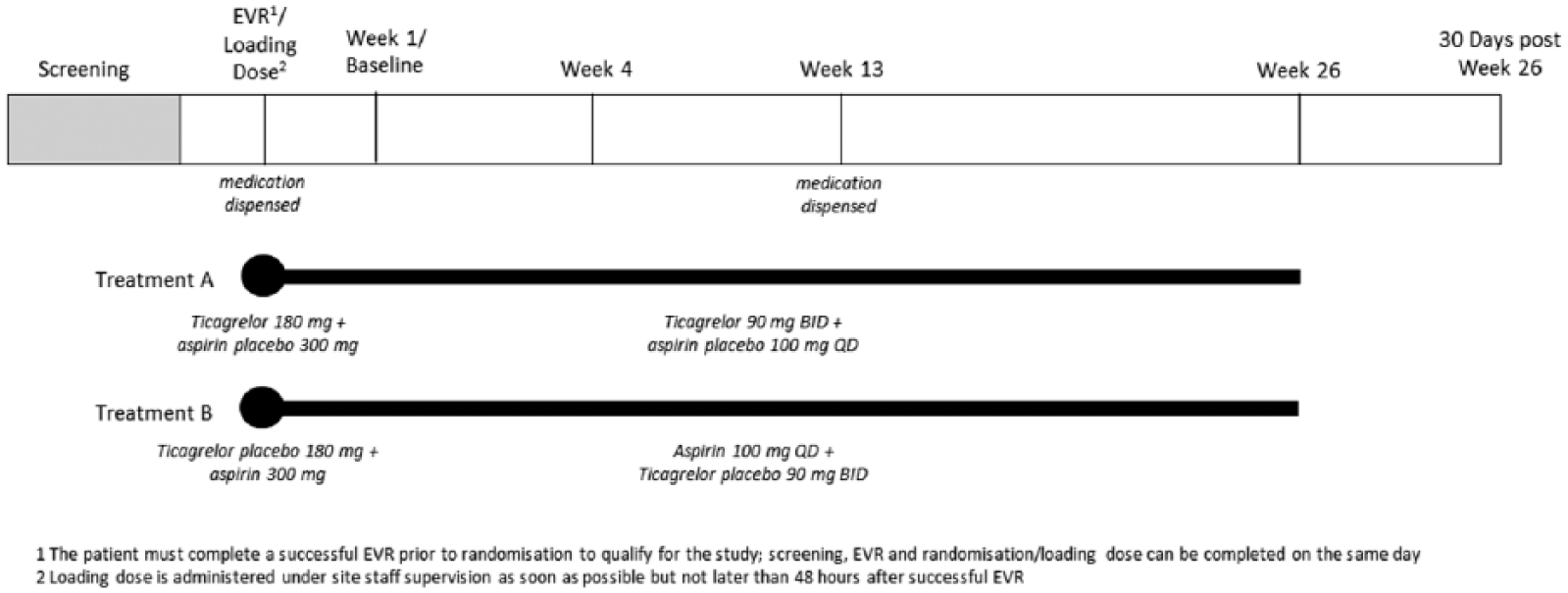
Flow chart of study design. After the initial screening phase, patients underwent endovascular revascularization, and were randomized once this was deemed to be successful. Study medication was started immediately after randomization with a loading dose, and patients were followed on study medication for 26 weeks, with study visits at weeks 1, 4, 13, and 26, along with a 30-day follow-up visit.
The initial design of TI-PAD involved screening potential subjects within 1 week prior to the planned PVI. Screening included obtaining informed consent, collecting baseline demographic and medical information, performing a pertinent physical exam, obtaining laboratory studies, and implementing quality of life questionnaires. An ABI was performed, as was an electrocardiogram and an exercise treadmill test (ETT). Baseline medical therapy was also assessed. Prohibited medications that could not be taken within 4 weeks prior to screening included mono- or dual-antiplatelet therapy, a requirement for more than 7 days of anticoagulation, or strong inducers or inhibitors of CYP3A enzymes. A 4-week washout period for these medications was required starting from the time of the Screening Visit, with reconsideration for enrollment after the washout for subjects on antiplatelet or anticoagulant therapies.
If the subject met all of the inclusion and none of the exclusion criteria, a randomization visit was scheduled for the 24–48 hour window prior to the PVI. At this visit, study medication was dispensed and the first (loading) dose of medication was observed. Subsequent to the randomization visit, a Baseline Visit was scheduled for 1 week post-PVI, but could occur between 7 and 14 days after the PVI. In summary, this initial protocol design involved four separate visits (including the PVI) within a 2–3-week period (Figure 1).
Exercise treadmill testing was performed by trained study personnel according to the Gardner protocol after participant familiarization and a 10-minute rest period. PWT was determined for each instance of the graded ETT (Baseline/Week 1 post-PVI, Week 4, Week 13 and Week 26, or at the early termination visit). Claudication was determined using a claudication rating scale at least two times per minute throughout the ETT, and verbal prompts were used to encourage subjects to continue walking until they felt unable to walk due to claudication, or were limited by other symptoms. PWT was defined as the maximum time in minutes and seconds walked on the treadmill until severe claudication symptoms forced the cessation of exercise. Since the PVI may significantly improve walking ability and reduce claudication symptoms, it was possible that patients would not be limited by claudication pain during the ETTs performed subsequent to the PVI. In those cases PWT was expected to be limited by other symptoms, such as shortness of breath or exertional fatigue. It was considered acceptable for the post-PVI ETTs to be terminated due to these symptoms, in which case PWT was defined as the maximum time walked on the treadmill until severe symptoms other than claudication forced cessation of exercise.
ABIs were calculated from the higher average of two brachial pressures in each arm and the higher average of two dorsalis pedis and posterior tibial pressures for each extremity. These measurements were obtained after subjects were supine for 10 minutes.
Challenges in execution of the initial trial design
Half of the study sites were open for enrollment in the first month, and 90% were open by the fourth month. However, initial enrollment in TI-PAD occurred at an average of only 0.04 patients per site per month, which was substantially less than originally projected. Therefore, the Steering Committee evaluated the major barriers to recruitment, and found that three exclusion criteria and one of the inclusion criteria accounted for most of the non-enrollment, with a disproportionately high number accounted for by the clinical necessity to use restricted concomitant medication within 4 weeks prior to screening and the requirement for a 4-week washout period. Other important criteria preventing eligibility included the presence of lower extremity gangrene or ischemic ulcer, a disease process other than PAD limiting the ability to reach the primary endpoint of the study, and the requirement for a PWT between 1 and 10 minutes limited only by claudication as assessed by the graded (Gardner protocol) ETT performed at the Screening Visit. Sites reported logistical challenges related to multiple study visits within a short span of time, and completion of the Screening ETT. The requirement for antiplatelet monotherapy after an intervention was a major barrier: practice patterns favored DAPT for most patients in the setting of percutaneous revascularization despite clear evidence to support that initial treatment strategy. 2 However, the key design feature requiring the use of antiplatelet monotherapy was not modifiable without compromising the original trial design. After discussion with site investigators, several other aspects of the protocol were changed by the trial Steering Committee as follows:
The prevalence of concomitant clinical coronary artery disease in the TI-PAD study population (symptomatic PAD) was relatively high (45%). Therefore, at least antiplatelet monotherapy was indicated. Eleven of the 40 (28%) enrolled patients had a history of prior coronary stent placement; as such, DAPT may have been indicated. Regardless, either mono- or dual-antiplatelet therapy was indicated in a high percentage of potential subjects, and investigators were uncomfortable stopping antiplatelet agents, even for a short period of time, especially when this therapy was prescribed by another clinician. In addition, during start-up and recruitment for this trial, practice patterns began moving toward a higher percentage of patients undergoing PVI with drug-coated devices, with an associated increase in the use of DAPT despite a lack of strong clinical evidence to support this practice.
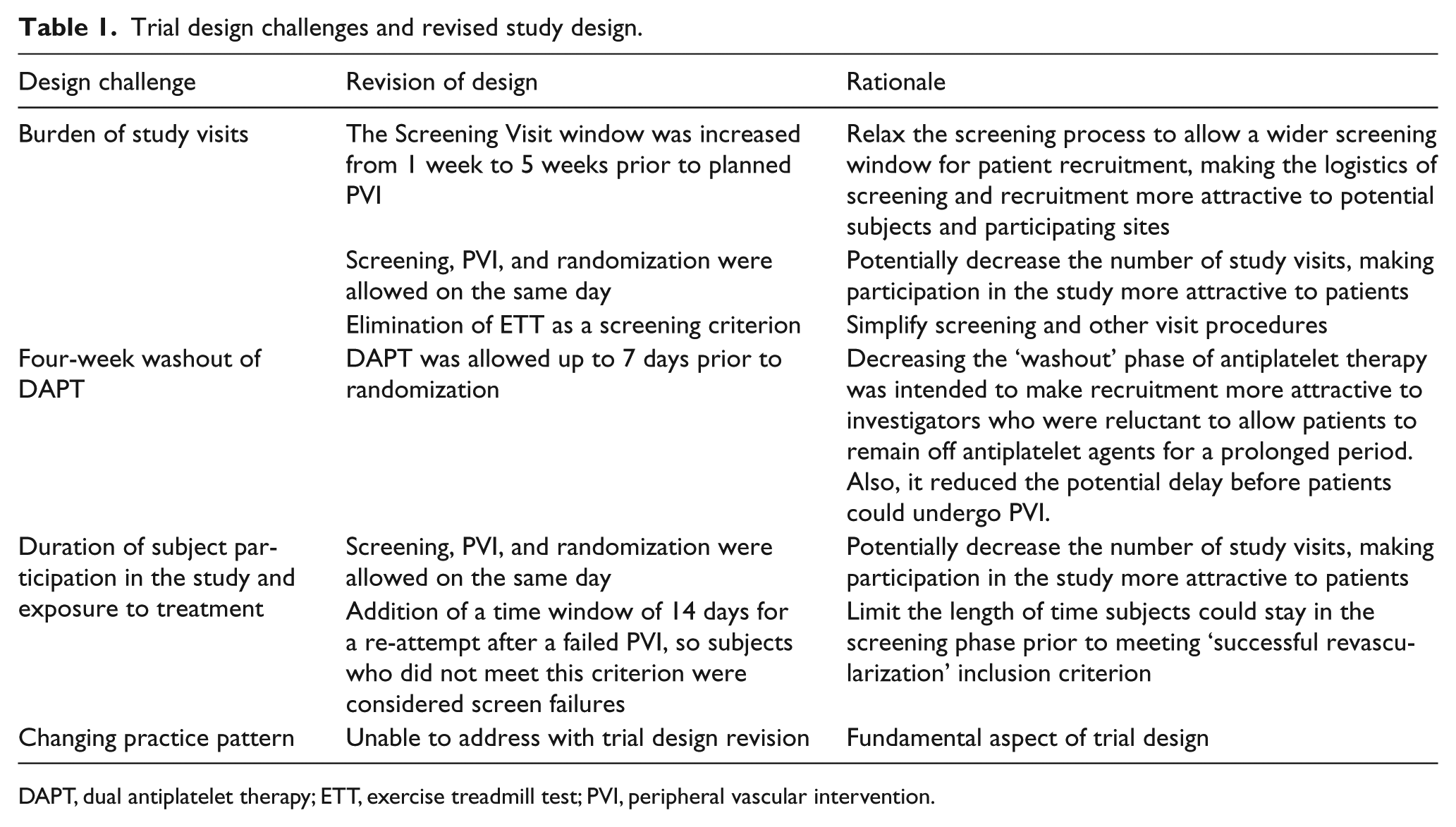
Rationale for the revised design (Table 1)
The aforementioned challenges hindered patient recruitment and greatly limited the enrollment into TI-PAD. As such, these challenges formed the basis for an amended protocol. These are detailed in Table 1, and changes made to the original protocol are described below:
Timing of randomization was moved to as soon as possible but within 48 hours after a technically successful PVI. This change decreased the potential delay between taking the study drug and undergoing a successful PVI. In addition, a time window of 14 days for a re-attempt after a failed PVI was added, and any subjects who did not meet this criterion were considered screen failures.
The Screening Visit was allowed up to 5 weeks before planned PVI, rather than 1 week. This change relaxed the screening process to allow for a wider screening window for patient recruitment, making the logistics of screening and recruitment more attractive to potential subjects and participating sites.
Screening, PVI, and randomization were allowed on the same day if all of these procedures could be completed successfully. This change potentially decreased the number of study visits, making participation in the study more attractive to patients.
Elimination of ETT as a screening criterion. This change simplified the screening procedures.
DAPT was allowed up to 7 days prior to randomization. Decreasing the ‘washout’ phase of antiplatelet therapy was intended to make recruitment more attractive to investigators who were reluctant to allow patients to remain off antiplatelet agents for a prolonged period. This change also reduced the potential delay before patients could undergo PVI.
Trial design challenges and revised study design.
DAPT, dual antiplatelet therapy; ETT, exercise treadmill test; PVI, peripheral vascular intervention.
Results
TI-PAD subject disposition
Seventy-one subjects were screened at 16 US centers; four of the originally planned 20 centers either did not screen any subjects or did not complete the activation process. A total of 40 patients were randomized at 11 centers: 16 were randomized to ticagrelor while 24 were randomized to aspirin. Subjects were randomized 1:1 utilizing blocked randomization at the site level. The imbalance in overall study treatment allocation was due to the small number of subjects enrolled, resulting in incomplete blocks enrolled at individual sites. No randomization strata were used. Prior to study entry, 10 (25%) patients were taking clopidogrel, three (7.5%) were on cilostazol, one (2.5%) patient was on warfarin, and 16 (40%) were taking aspirin. Ten patients were not taking any antithrombotic agents prior to study entry. Baseline characteristics of randomized patients are in Table 2.
Baseline characteristics.
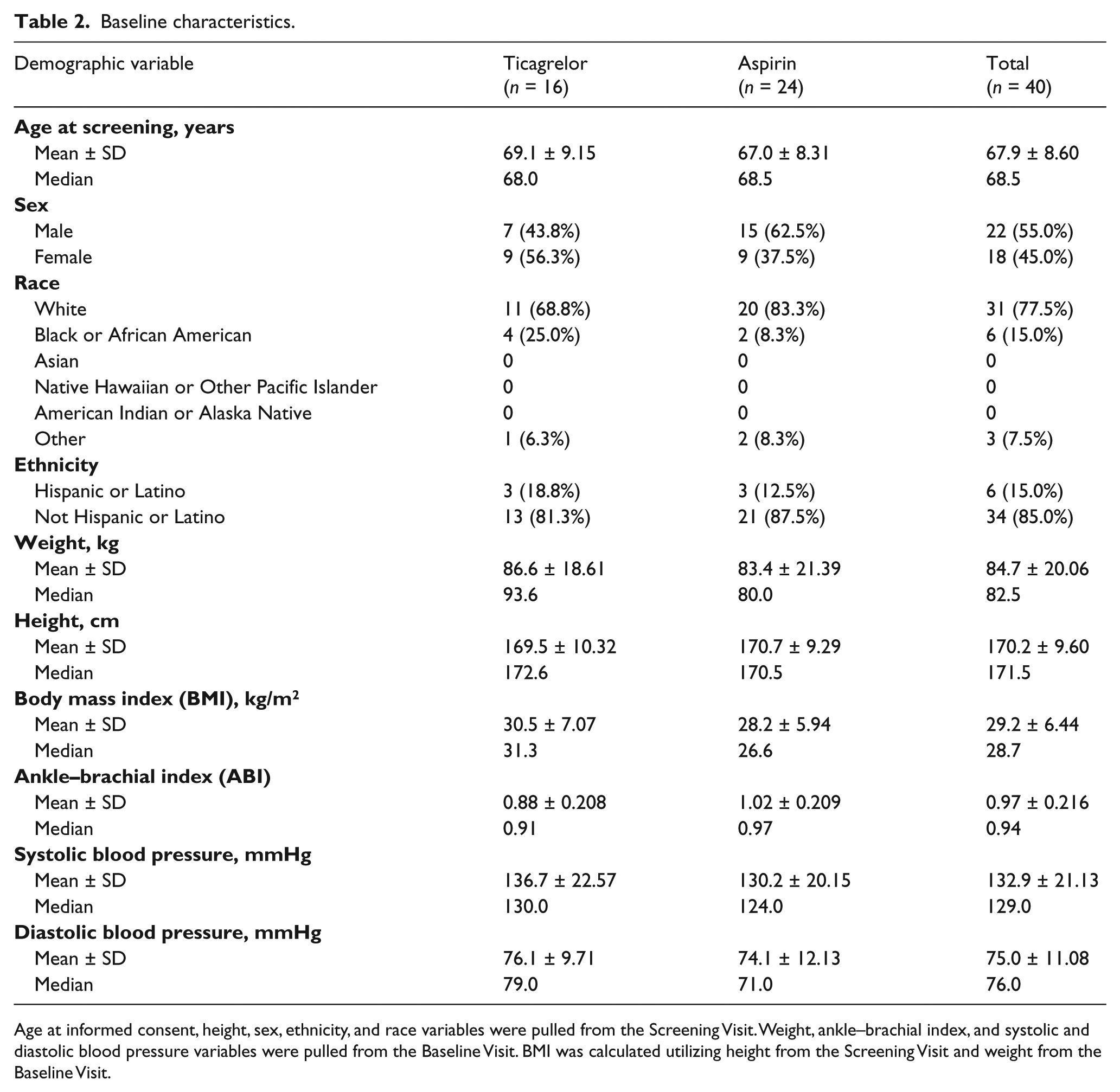
Age at informed consent, height, sex, ethnicity, and race variables were pulled from the Screening Visit. Weight, ankle–brachial index, and systolic and diastolic blood pressure variables were pulled from the Baseline Visit. BMI was calculated utilizing height from the Screening Visit and weight from the Baseline Visit.
The disposition of subjects considered for TI-PAD is detailed in Figure 2. Of the 31 screened patients not randomized, 20 subjects did not meet the eligibility criteria, two patients withdrew consent prior to randomization, and nine subjects were not randomized for other reasons, the most common of which was inability to attend the 48-hour post-PVI randomization visit. A total of six subjects terminated the study early: two subjects (12.5%) randomized to ticagrelor were withdrawn from the study (one was lost to follow-up; another withdrew after an adverse event) and four subjects (16.7%) randomized to aspirin were withdrawn from the study. Of the 40 randomized patients, six subjects (37.5%) in the ticagrelor arm withdrew and/or discontinued treatment versus seven subjects (29.2%) randomized to aspirin (Figure 2). Subjects who permanently discontinued use of study medication were encouraged to remain in the study. Therefore, the number of subjects completing the study was greater than the number of subjects who completed treatment.
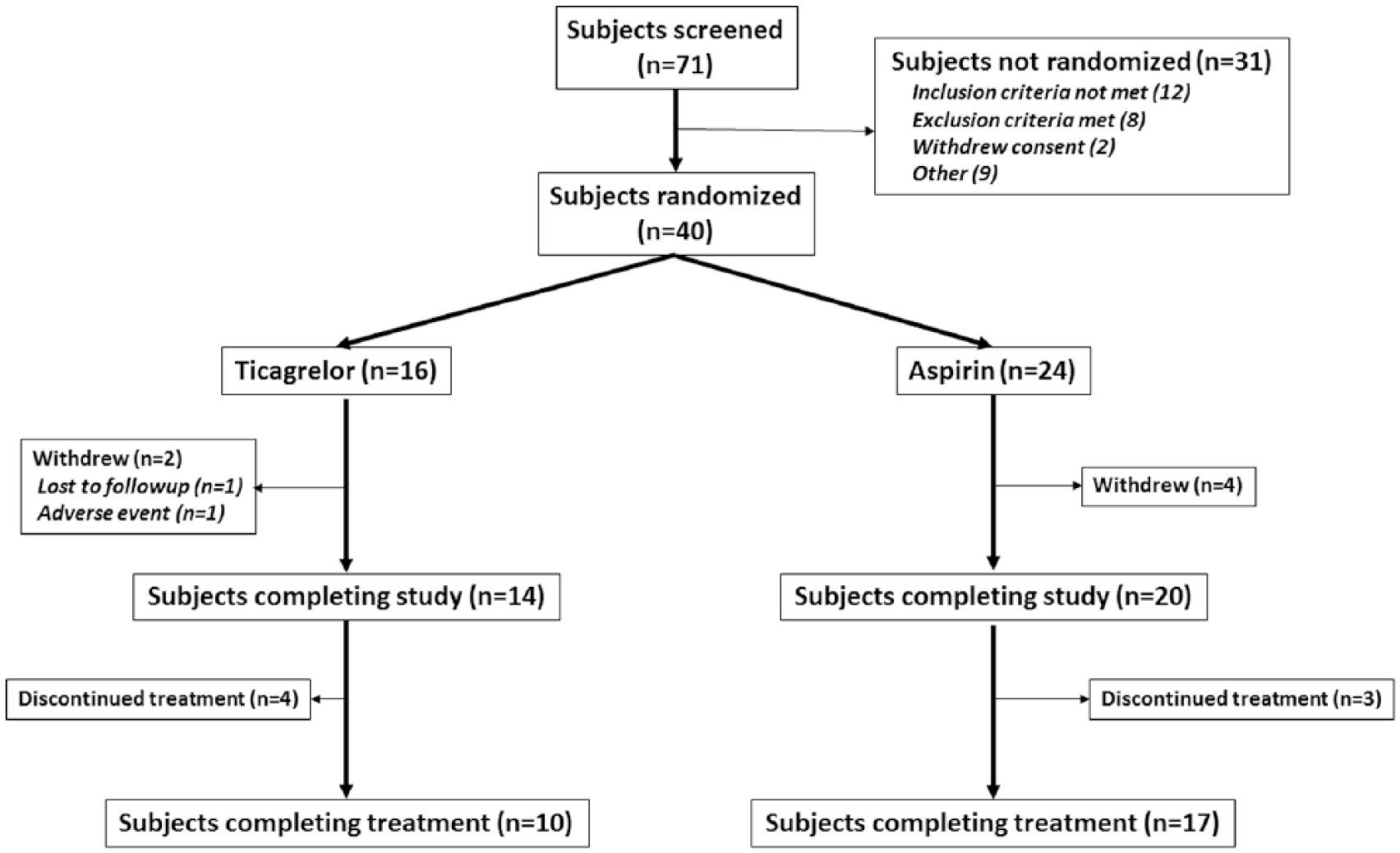
Subject disposition. Seventy-one patients were screened and 40 were enrolled and randomized in TI-PAD. By the time the study was terminated, 14 subjects had been randomized to the ticagrelor group and 20 subjects had been randomized to the aspirin group. A total of 34 subjects completed the study.
A total of 11 treatment emergent serious adverse events occurred in six subjects: four were randomized to ticagrelor and two were randomized to aspirin. No deaths occurred. There were four subjects who had adverse events leading to discontinuation of study medication, two in each group. These included coronary artery disease, hypotension, cellulitis and arterial occlusive disease (Table 3).
Summary of TEAEs leading to discontinuation of study medication.
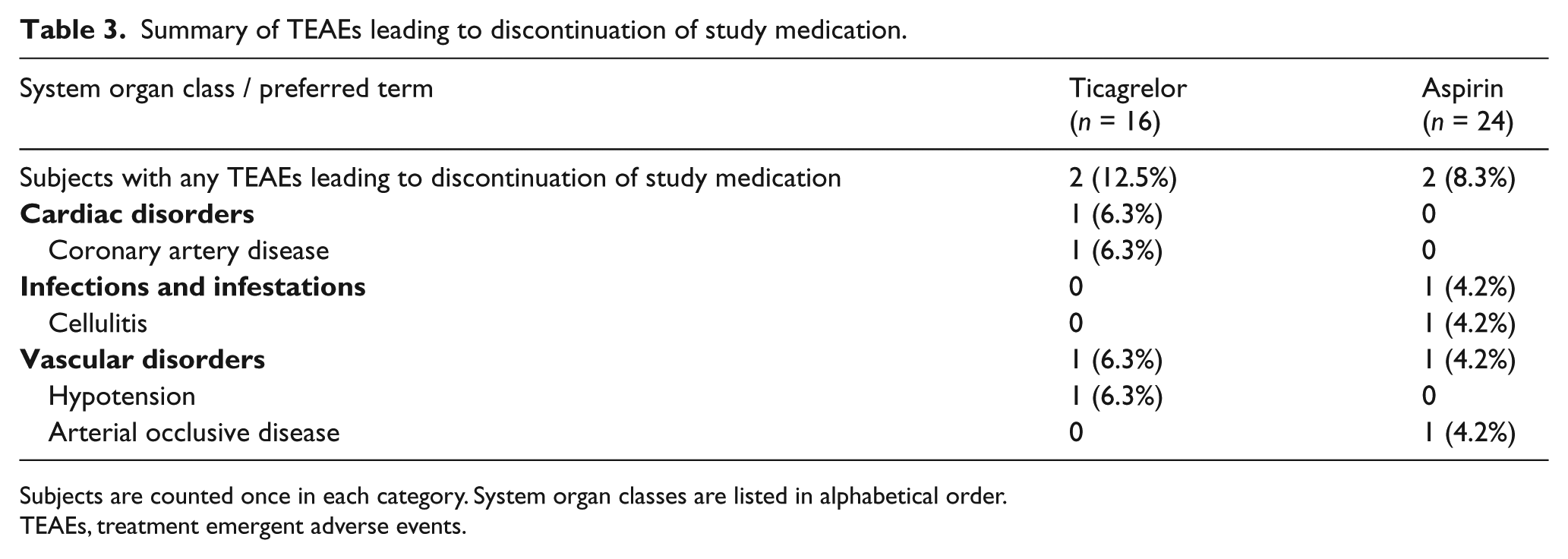
Subjects are counted once in each category. System organ classes are listed in alphabetical order.
TEAEs, treatment emergent adverse events.
Overall, the study was terminated early because of slow recruitment, with 71 subjects screened and 40 of these randomized over a period of 13 months. Sixty patients completing the trial per treatment group would have provided 88% power to detect a 30% difference in PWT. Assuming a dropout rate of up to 30%, the final enrollment target was 175 patients to result in 120 evaluable patients. Of the originally planned cohort for randomization, only 23% were randomized over the course of approximately 13 months. Of the original 20 sites, 16 screened potential subjects and only 11 randomized subjects. There were a maximum of 19 sites open at any one time during the trial, and the overall enrollment rate was 5.5 patients per month, or 0.29 patients per month per site, in contrast to the anticipated enrollment rate of 0.6 patients per month per site. Because of the small sample size and early termination of enrollment, efficacy endpoints could not be reliably evaluated. There were no meaningful imbalances in safety endpoints or adverse events between treatment groups that warranted concern (Table 3).
Discussion
Currently, ticagrelor is indicated to reduce the rate of thrombotic cardiovascular events, including cardiovascular death, in patients with acute coronary syndrome and more recently in patients with a history of myocardial infarction beyond 1 year. 14 The recently published Examining Use of Ticagrelor in Peripheral Artery Disease (EUCLID) trial performed in 13,885 patients with PAD who underwent prior PVI compared monotherapy with ticagrelor (90 mg twice daily) versus clopidogrel (75 mg once daily) and did not show superiority of ticagrelor in reducing cardiovascular events in this population. 15 In patients undergoing endovascular treatment of PAD, there is a lack of evidence regarding the optimal antiplatelet therapy. Therefore, TI-PAD was carefully designed to determine the efficacy of a P2Y12 platelet inhibitor as antiplatelet monotherapy to improve limb outcomes (defined as peak walking time) after lower extremity revascularization, in comparison with low-dose aspirin. Strengths of the trial methodology included the use of a randomized, double-blind, placebo-controlled design, 16 the absence of an upper age limit allowing enrollment of elderly patients, 17 and the choice of a functional endpoint – peak walking time – as the primary outcome. The assumption was that a successful and durable revascularization would translate to an improvement in functional status as measured by the graded treadmill test.
The rationale for TI-PAD was based on a lack of data relevant to antiplatelet therapy in the context of endovascular treatment of patients with PAD.18–20 The Clopidogrel and Aspirin in the Management of Peripheral Endovascular Revascularization (CAMPER) trial was the first trial we are aware of to evaluate DAPT with clopidogrel and aspirin versus aspirin alone in this setting. CAMPER began enrolling in November 2002 and planned to enroll 2000 patients from 100 US sites. Only approximately 300 patients were enrolled by February 2004 and the trial was stopped due to slow recruitment (William R Hiatt, personal communication). Previously, a single, small, randomized trial has compared contemporary peri-procedural or post-procedural antiplatelet agents in patients receiving percutaneous treatment of PAD.21,22 In this study, 80 patients with PAD were randomized to DAPT with aspirin and clopidogrel or to aspirin and placebo following a successful endovascular procedure. 22 The DAPT group experienced less target vessel revascularization at 6 months (2 vs 8, p=0.04), but no change in ABIs or Rutherford category. In the absence of more abundant randomized data, the Steering Committee felt that any use of DAPT in the setting of peripheral revascularization was largely an extrapolation from the coronary percutaneous coronary intervention data. Therefore, given an overall lack of evidence, it was decided to compare antiplatelet monotherapies in TI-PAD, rather than including an arm of DAPT.
Reluctance of providers to commit patients to antiplatelet monotherapy (with a preference for dual therapy) proved to be a major obstacle to enrollment in TI-PAD, with implications for future trials in this setting. However, the overall evidence to guide clinicians regarding the benefit of DAPT in lower extremity endovascular revascularization is limited. 23 Likewise, in stable PAD not undergoing revascularization, the CHARISMA (Clopidogrel for High Atherothrombotic Risk and Ischemic Stabilization, Management, and Avoidance) trial compared DAPT using clopidogrel and aspirin to aspirin alone. 24 The overall trial results were negative as was the PAD subgroup for the reduction of myocardial infarction, stroke or death, although the small sample size of the PAD subgroup limited the power to detect a significant difference in therapies. Finally, TI-PAD was designed prior to approval of most contemporary drug-eluting devices approved for use in PVI. Extrapolating from the evidence base for coronary intervention, providers may have favored use of DAPT for these new PVI devices. Clearly, clinical trials are needed to further evaluate the role of DAPT and other antithrombotic strategies after lower extremity revascularization while including investigators who have equipoise regarding competing antithrombotic regimens.
A key factor in the success of a clinical trial is the predictive value of the feasibility assessment during site and investigator selection. As soon as slow enrollment was recognized, the Steering Committee and academic clinical trial organization (ACRO, CPC Clinical Research) collected information to determine enrollment barriers and worked to overcome these. A study protocol amendment addressing these challenges was finalized within a few months after recognizing this issue, and site IRBs approved the amended protocol in an expeditious manner. After sites began operating under the amended protocol, there was a slight uptick in enrollment but unfortunately even after 6 months of intensive efforts by the Steering Committee, ACRO and investigators to impact enrollment, the trajectory of enrollment did not change significantly. All of the study sites were active clinically, but the proportion of patients who met all inclusion and none of the exclusion criteria turned out to be much lower than anticipated. There were no regional, clinic or health system differences detected. A strategy that may inform future successful trial operations may be to ask potential sites to perform simulated prescreening for a specified period of time with study-specific inclusion and exclusion criteria in order to obtain a more accurate estimate of predicted enrollment rate.
Despite efforts to optimize identification of potential subjects, prescreening, and streamlining the enrollment process, sites that did not screen or randomize subjects reported that they could not find any patients that met all of the inclusion and none of the exclusion criteria, and there was no other identifiable factor that could be found for this difference. Sites that were successful had an efficient process for identifying subjects ahead of time through prescreening of potential subjects scheduled for peripheral vascular procedures, frequent contact between the study staff and the investigator to discuss study progress, and enough volume to be able to enroll eligible patients.
The use of antiplatelet therapy in patients after revascularization for lower extremity PAD appears to vary geographically. For example, data from the Swedish National Registry for Vascular Surgery (Swedvasc) and the Prescribed Drug Register reveal that among patients with intermittent claudication who underwent lower extremity revascularization in Sweden, 73.5% were on low-dose aspirin alone, 7.6% were on clopidogrel alone, and 5.4% were on DAPT consisting of low-dose aspirin with clopidogrel, prasugrel or ticagrelor. 25 After revascularization, the percentage of patients treated with aspirin was 80% and with clopidogrel was 21%; data were not available for use of DAPT after revascularization. In contrast, in a US study of 85,830 Medicare beneficiaries, 81% were prescribed a P2Y12 inhibitor around the time of revascularization (P2Y12 prescription being a rough surrogate of DAPT use). 26 Enthusiasm for DAPT in the US may stem in large part from evidence in patients undergoing percutaneous coronary intervention. In this setting, decreased rates of stent thrombosis are seen with DAPT compared to aspirin alone.27–30 More recently, newer antiplatelet agents (ticagrelor and prasugrel) have been shown to reduce myocardial infarction, stroke, and death compared to clopidogrel on background aspirin therapy in patients with myocardial infarction.31,32
Based on the available evidence of antiplatelet therapy in patients with PAD undergoing endovascular procedures and based on a signal of different treatment effects of antiplatelet therapy in patients with PAD versus CAD, the rationale of TI-PAD to compare ticagrelor to aspirin, rather than a comparison involving DAPT, was justified. There is significant variation in practice patterns for PAD, and current practice that tends to favor DAPT in the US is not based on existing evidence, and makes it more difficult to recruit for this type of trial and thus to accumulate evidence for antiplatelet therapy in this setting. One potential solution for future randomized trials of antiplatelet therapy in patients with PAD undergoing endovascular treatment might be to enroll into a mono-therapy trial after completion of a period of DAPT.
Conclusion
An objective of TI-PAD was to address the lack of data regarding the optimal antiplatelet therapy surrounding endovascular treatment of PAD. Focusing on a study population undergoing endovascular treatment for PAD was thought to help overcome barriers to enrollment that have plagued other PAD trials. 33 Furthermore, evaluating a newer antiplatelet agent (ticagrelor) in TI-PAD was an attractive addition to existing evidence which predominantly focuses on older antithrombotic medications. 21
A potential insight gained from TI-PAD was the mindset in the US that DAPT is important for patients undergoing endovascular revascularization. This practice pattern was a challenge in trial recruitment and must be considered when designing future studies. A strategy that could be used to increase the success of future clinical trials would involve asking potential sites to perform simulated prescreening for a specified period of time with study-specific inclusion and exclusion criteria in order to obtain a more accurate estimate of predicted enrollment rate. Factors shared by successful sites included active engagement by the investigator(s) and study staff, development of an efficient process for identification of potential subjects with adequate and timely prescreening of potential subjects scheduled for peripheral vascular procedures, streamlining of the enrollment process, frequent contact between the study staff and the investigator to discuss study progress, and a patient population that allows for enrollment of eligible patients.
Furthermore, future trial designs that are more pragmatic and allow for variations in clinical practice may more successfully enroll patients and come to fruition. Given the costs of carrying out contemporary randomized clinical trials, such data may be more economically gathered through registry randomized trials and other real world data sources. Such datasets can also be linked to mobile health platforms allowing measurements of not only clinical outcomes, but also more patient-centered and individualized outcomes. Results from such trials would not only guide clinicians toward the optimal medical regimen but also toward the optimal management of our patients with PAD.
Footnotes
Dr Joshua Beckman served as Guest Editor for this manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R Kevin Rogers: Bayer – Independent Clinical Endpoint Adjudication Committee. William R Hiatt: Bayer – grant funding; Janssen – grant funding. Manesh R Patel: AstraZeneca – research grant, advisory board; Bayer – research grant, advisory board; Janssen – research grant, advisory board. Mehdi H Shishehbor: no relevant conflicts. Robin White: no disclosures to report. Naeem D Khan: an employee of AstraZeneca. Narinder P Bhalla: an employee of AstraZeneca. W Schuyler Jones: research grants from the Agency for Healthcare Research and Quality, AstraZeneca, American Heart Association, Bristol-Myers Squibb, Doris Duke Charitable Foundation, Merck, and Patient-Centered Outcomes Research Institute; honoraria/other from the American College of Physicians, Bayer, Bristol-Myers Squibb, Daiichi Sankyo, and Janssen Pharmaceuticals. Cecilia C Low Wang: no disclosures to report.
Funding / Role of Sponsor
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AstraZeneca provided the funding and served as the sponsor for this study. The sponsor reviewed this manuscript but had no role in the data analysis, initial interpretation of results, or writing of this manuscript.
Trial committees
The Steering Committee consisted of William R Hiatt (University of Colorado School of Medicine and CPC Clinical Research), James Ferguson (AstraZeneca), Manesh R Patel (Duke University Health System and Duke Clinical Research Institute), R Kevin Rogers (University of Colorado School of Medicine), and Mehdi H Shishehbor (Cleveland Clinic).
The Clinical Endpoints Committee consisted of Christopher Fordyce, Jacob Kelly, Tiffany Randolph, Emily Zeitler, W Schuyler Jones, Manesh R Patel, Robert Mentz, Thomas Povsic, Sreekanth Vemulapalli, Robert Harrison, Keith Dombrowski, Bradley Kolls, Dedrick Jordan (all in the Duke University Health System); Warren Capell, Natalia Glebova, Connie N Hess, Mori Krantz (all at CPC Clinical Research).
