Abstract
Venous thromboembolism is a common complication of malignancy. Lung cancer is considered one of the most thrombogenic cancer types. Primary thromboprophylaxis is not currently recommended for all ambulatory patients with active cancer. In the present narrative review we aim to summarize recent data on the safety and efficacy of primary thromboprophylaxis as well as on venous thromboembolism risk assessment, focusing on ambulatory patients with lung cancer. A potential benefit from prophylactic anticoagulation with low molecular weight heparins in terms of venous thromboembolism risk reduction and increased overall survival in patients with lung cancer, without a significant increase in bleeding risk, has been reported in several studies. Recent studies also reveal promising results of direct oral anticoagulants regarding their efficacy as primary thromboprophylaxis in patients with cancer, including those with lung cancer. However, the use of different study methodologies and the heterogeneity of study populations among the trials limit the extraction of definite results. More randomized, controlled trials, restricted to a well-characterized population of patients with lung cancer, are greatly anticipated. The use of risk assessment tools for stratification of venous thromboembolic risk is warranted. The development of an accurate and practical risk assessment model for patients with lung cancer represents an unmet need.
Keywords
Introduction
Venous thromboembolism (VTE), including deep vein thrombosis (DVT) and pulmonary embolism (PE), represents a typical complication of malignancy.1,2 VTE risk is elevated four- to sevenfold in patients with cancer, as active malignancy has been delineated as an independent risk factor for thrombosis. 1 Occult malignancy coexists in 20% of individuals suffering from VTE. 3 Well established, superimposed risk factors, such as major cancer surgery, placement of central venous catheters, hospitalization, or anticancer treatment administration, further increase thrombotic risk in these patients.1,4 Thrombosis represents the second leading cause of death in patients with cancer. Long-term complications, such as chronic thromboembolic pulmonary hypertension and post-thrombotic syndrome, affect the quality of life in VTE survivors.3,5
Lung cancer is regarded as one of the most thrombogenic cancer subtypes, as individuals with non-small cell lung cancer (NSCLC) are at a 20-fold increased risk of suffering from VTE in comparison to the general population.4,6,7 Pneumonectomy, chemotherapy, and anti-angiogenic therapy have also been demonstrated to increase thromboembolic risk.4,8 VTE often appears early in the course of lung cancer since half (50%) of the patients are diagnosed with cancer less than 3 months before VTE 9 and early occurrence of VTE is associated with a worse survival regardless of stage, comorbidities, and performance status of the patients. 10
VTE is a potentially preventable disease with the use of several classes of drugs that act as anticoagulants, including parenteral anticoagulants, and also direct oral anticoagulants (DOACs), the role of which is currently being explored in the population of patients with cancer.11–13 Moreover, it has been hypothesized that anticoagulants may improve survival in patients with cancer through an anti-tumor effect beyond their well-described antithrombotic effect.14,15 Currently, international guidelines recommend thromboprophylaxis with parenteral anticoagulants in hospitalized patients with cancer and those who are undergoing cancer surgery.16,17 However, primary thromboprophylaxis for VTE in ambulatory patients with solid tumors remains uncertain because the potential benefit in VTE reduction or overall survival should be well established and must outweigh the risk of bleeding. It is recommended though that VTE risk should be assessed with the use of risk stratification models, and prophylactic administration of anticoagulants should be considered in high-risk ambulatory patients on a case-by-case basis.17,18 Indeed, among patients with cancer, there is a considerable variation of VTE risk depending on patient-, cancer-, and treatment-related factors 19 and, therefore, the stratification of the patients could improve the clinical benefit of primary thromboprophylaxis by reducing the number needed to treat to avoid an episode of VTE. 19 In fact, various risk assessment models have been developed to identify patients with cancer at high risk for VTE events. 20
The purpose of this narrative review is to summarize recent evidence from randomized controlled trials about the safety and efficacy of prophylactic anticoagulation in ambulatory patients with lung cancer, focusing on the use of low molecular weight heparins (LMWHs) or DOACs. In addition, we aim to provide a summary of currently available risk stratification models for assessment of VTE risk, focusing on patients with lung cancer.
Literature review
For the first part of the review a comprehensive search of PubMed-MEDLINE, Embase, and Scopus databases was performed, using combinations of the keywords ‘lung cancer’, ‘prophylactic anticoagulation’, ‘thromboprophylaxis’, ‘LMWHs’, and ‘DOACs’. We only included reports from randomized controlled trials and only if they involved adult patients diagnosed with lung cancer who received primary thromboprophylaxis with LMWHs or DOACs. Non-English literature was excluded. This search was followed by a manual search of the reference list of the included articles to identify additional studies.
For the second part of the review, we searched PubMed-MEDLINE, Embase, and Scopus databases with combinations of the following keywords: ‘risk assessment model’, ‘VTE risk’, ‘lung cancer’, and ‘cancer-associated thrombosis’. Among the retrieved abstracts, we included only original articles that described the development of risk stratification models for assessment of VTE risk in patients with cancer, as well as original articles that examined the validation of risk assessment models in patients with lung cancer.
Safety and efficacy of primary thromboprophylaxis – review of clinical trials
Primary thromboprophylaxis with the use of LMWHs
Twelve clinical trials have focused on the use of LMWHs as prophylactic anticoagulation in ambulatory patients with cancer, including lung cancer (Table 1). Among the trials, five were restricted to patients with lung cancer only21–25; the remaining seven included a mixed population of patients with cancer of various primary sites.26–32 In the study of Altinbas et al., in which 84 patients with small-cell lung cancer (SCLC) were randomized to receive either chemotherapy with LMWH (dalteparin) or chemotherapy with placebo, the results revealed a statistically significant increase in overall survival as well as progression-free survival in the dalteparin arm. 21 Similarly, the ABEL study, which involved 38 patients with limited-stage SCLC, who were randomized to receive standard chemoradiotherapy or the same therapy plus the LMWH bemiparin, demonstrated a statistically significant increase in overall survival for patients in the bemiparin arm. 22 However, the recently published RASTEN trial, with 377 patients with SCLC, concluded that the addition of LMWH enoxaparin to standard therapy did not improve overall survival in patients with SCLC despite being administered at a supraprophylactic dose. 23 This was true for patients with extended disease as well as limited disease. In the same study, the addition of enoxaparin resulted in a statistically significant reduction of VTE occurrence. A total of 547 patients with NSCLC were included in the TOPIC-2 study, which examined the prophylactic administration of certoparin. This study’s primary efficacy endpoint was the occurrence of objectively confirmed VTE events. The results did not exhibit a decrease in the frequency of thromboembolic events in stage III disease, but a significant decrease was proven in patients with stage IV disease (3.5% in the certoparin vs 10.2% in the control group) without an increase in bleeding events. 24 Finally, FRAGMATIC was a large, open-label, phase III clinical trial restricted to patients with lung cancer only (2202 patients with either NSCLC or SCLC) that examined 1-year survival as a primary efficacy endpoint. The results did not show a survival benefit when prophylactic anticoagulation therapy with dalteparin was administered. However, a statistically significant decrease in the frequency of thromboembolic events was documented. 25
Trials examining prophylactic LMWH administration in patients with lung cancer.
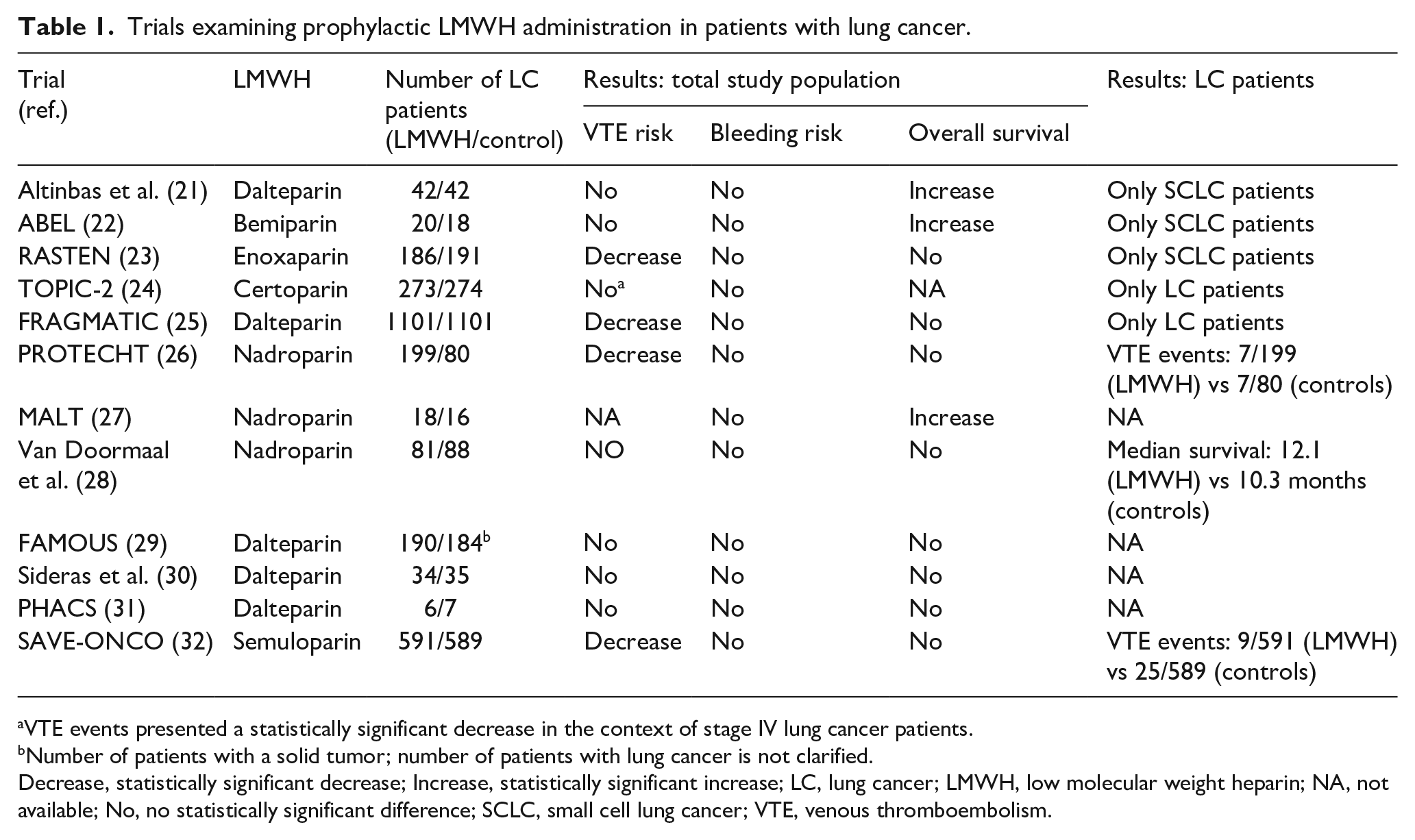
VTE events presented a statistically significant decrease in the context of stage IV lung cancer patients.
Number of patients with a solid tumor; number of patients with lung cancer is not clarified.
Decrease, statistically significant decrease; Increase, statistically significant increase; LC, lung cancer; LMWH, low molecular weight heparin; NA, not available; No, no statistically significant difference; SCLC, small cell lung cancer; VTE, venous thromboembolism.
Three studies have examined the prophylactic use of LMWH nadroparin in patients with cancer of various primary sites, including lung cancer. In the PROTECH study, 279 patients with lung cancer were enrolled: 199 patients received nadroparin thromboprophylaxis, whereas 80 constituted the control group. VTE events occurred in 7/199 (3.5%) and 7/80 (8.8%), respectively, resulting in a twofold decrease in thromboembolic risk, without a significant difference in bleeding risk. 26 The MALT study included a total of 302 patients with cancer of various primary sites (34 patients with lung cancer). One group (148 patients) received nadroparin in a therapeutic dose for 2 weeks followed by a prophylactic dose for 4 more weeks, and the other group received placebo. The primary efficacy endpoint of this study was overall survival; results indicated an increase in overall survival for individuals receiving nadroparin without an apparent increase in bleeding events. 27 However, Van Doormaal et al., in a multicenter, randomized, controlled clinical trial involving 503 patients with lung, prostate or pancreatic cancer, did not demonstrate a statistically significant benefit in overall survival or the incidence of thromboembolic events for patients receiving prophylactic nadroparin over placebo neither for the total cancer study population, nor for the lung cancer subgroup. 28
As far as prophylactic administration of dalteparin is concerned, three studies were analyzed. In the FAMOUS trial, a total of 374 patients were included, but the number of those with lung cancer was not clarified. Dalteparin was prescribed in a prophylactic dose for 12 months in the study arm, whereas the control group received placebo. The results did not report a statistically significant difference in overall survival; however, a post hoc analysis of the subset of patients with a better prognosis revealed a statistically significant survival benefit. 29 Nevertheless, Sideras et al. did not report a statistically significant survival benefit from the prophylactic use of dalteparin in 138 patients with advanced cancer. 30 More recently, the PHACS trial’s investigators examined the prophylactic use of dalteparin in a mixed cancer population who were stratified as high risk for VTE according to the Khorana score. The results demonstrated a nonsignificant reduction of VTE events in the dalteparin arm at the expense of nonmajor clinically relevant bleeding. 31
The efficacy of the ultra-LMWH semuloparin was assessed in the SAVE-ONCO study. In this trial, 3212 patients with various solid tumors were included, 1180 of whom had lung cancer. Although semuloparin administration (20 mg daily for 3.5 months) did not exhibit a substantial survival benefit, a statistically significant decrease in the frequency of thromboembolic events was documented with no difference in the risk of major or clinically relevant nonmajor bleeding. It should be noted that semuloparin maintained its efficacy in VTE reduction among all different subgroups of patients according to the primary cancer site. 32
Primary thromboprophylaxis with the use of DOACs
Regarding administration of prophylactic DOACs in patients with lung cancer, three studies were identified (Table 2). Levine et al. examined the prophylactic administration of apixaban in patients with metastatic malignancies, including lung cancer, undergoing first or second-line chemotherapy in a randomized, phase II study. Apixaban appeared to be well tolerated and results indicated a reduction in thromboembolic episodes in all subgroups that received study treatment. 33 Very recently, two trials that examined primary thromboprophylaxis with DOACs among patients with cancer were published. The AVERT trial, which included 574 patients in total (59 with lung cancer), showed that thromboprophylaxis with apixaban led to a significantly lower rate of venous thromboembolic complications compared to placebo among ambulatory patients with cancer who were commencing chemotherapy and had a Khorana score of 2 or higher, but at the cost of a higher risk of major bleeding episodes. 34 In the CASSINI trial, the addition of rivaroxaban in ambulatory patients with cancer who had a Khorana score of 2 or higher led to a substantially lower incidence of VTE events in the intervention period, but not in the 180-day trial period, without a significant difference in bleeding events between the groups. 35
Trials examining prophylactic DOAC administration in patients with lung cancer.

The incidence of VTE was lower with rivaroxaban than with placebo in the per-protocol analysis but not in the primary intention-to-treat analysis.
Decrease, statistically significant decrease; DOAC, direct oral anticoagulant; Increase, statistically significant increase; LC, lung cancer; NA, not available; No, no statistically significant difference; VTE, venous thromboembolism.
Assessment of VTE risk in patients with lung cancer
The Khorana score (range: 0–6, with higher scores signifying a higher risk of VTE) has been developed and validated to identify patients with an elevated risk of VTE and is the most widely used risk assessment model. It takes into account five variables: primary site of cancer, pre-chemotherapy platelet count, hemoglobin level, pre-chemotherapy leukocyte count, and body mass index. A low-risk category according to the Khorana score is associated with a rate of 0.3–0.8% of VTE events at 2.5 months of follow-up, while intermediate- and high-risk categories are associated with 1.8–2.0% and 6.7–7.1%, respectively. All patients with lung cancer are assigned 1 point, which leads to an at least intermediate risk of VTE. 36 The PROTECHT score has been suggested as a modification of the Khorana score, with the addition of platinum or gemcitabine-based chemotherapy to the variables already taken into account in the Khorana score, 37 and the CONKO score is a modified Khorana score in which body mass index is replaced by WHO performance status. 38 The Vienna CATS score has combined the variables of the Khorana score with two other biomarkers: D-dimers and soluble P-selectin. 39 In the ONKOTEV risk assessment model, the combination of three clinical parameters (metastatic disease, malignancy-related macroscopic vascular or lymphatic compression, and a history of VTE) with a Khorana score > 2 has been shown to improve the prediction of VTE in ambulatory patients with cancer. 40
The COMPASS-CAT model, which has been developed in a cohort of patients with breast, colorectal, lung or ovarian cancer, includes the following categories of variables: (1) cancer-related risk factors including anthracycline or hormonal treatment, time since cancer diagnosis ⩽ 6 months, central venous catheter, and advanced stage of cancer; (2) predisposing risk factors including cardiovascular risk factors (at least two of the following: personal history of peripheral artery disease, ischemic stroke, coronary artery disease, hypertension, hyperlipidemia, diabetes, obesity), recent hospitalization for acute medical illness, and personal history of VTE; and (3) biomarkers consisting of platelet count ⩾ 350 × 109/L. It leads to the stratification of patients into a high-risk category for VTE, where the rate of thrombotic events is 13%, and low-/intermediate-risk category for VTE, where the rate is 1.7%. An advantage of the COMPASS-CAT model is that it can be implemented to outpatients at any time after treatment initiation during the patient’s anticancer therapy. 41 Recently, the CATS-MICA prediction model was developed in a large prospective cohort of ambulatory patients with a variety of solid cancers, and then it was externally validated in an independent prospective cohort. In this model, only two variables are incorporated: one clinical factor (tumor-site category) and one biomarker (D-dimer), a very commonly tested biomarker in most centers worldwide. 42
For patients with lung cancer, in particular, a retrospective study of 719 patients demonstrated that a high risk defined by the Khorana score did not accurately predict VTE events. 43 Those findings are in line with the results from a subgroup analysis of patients receiving chemotherapy in the FRAGMATIC study. In this study, the rate of VTE was not significantly different between the high-risk (Khorana score ⩾ 3) and intermediate-risk groups of patients.25,44 In another recent retrospective study of 118 patients with lung cancer, the following risk assessment models were compared in terms of accuracy in prediction of VTE events: Khorana score, PROTECHT, CONKO, and COMPASS-CAT. The results demonstrated that the COMPASS-CAT score was the most accurate in the prediction of VTE events. 45
Discussion
Current evidence and guidelines
Patients with active cancer are at high risk of VTE, which results in substantial morbidity and mortality as well as in a considerable increase in health care cost. 46 Moreover, the diagnosis of VTE in a patient with cancer often leads to prolongation of anticancer treatment. Lung cancer, one of the most common cancer types worldwide, is strongly associated with VTE.8,47 In this review, we summarize the safety and efficacy outcomes of primary thromboprophylaxis from clinical trials that included patients with lung cancer. Different thromboprophylaxis options have been tested in the context of various clinical trials in patients suffering from lung cancer. The design of the studies is widely heterogeneous, which limits the extraction of definite conclusions.
Regarding LMWHs, efficacy results seem contradictory. Three studies (MALT, ABEL, and Altinbas et al.) reported a statistically significant increase in overall survival, while the FAMOUS study provided a survival benefit in the subgroup of patients who had a better prognosis. It should be noted that both the ABEL and the Altinbas et al. studies have included patients with SCLC only, suggesting a potential effect of LMWHs in this histologic type. Nevertheless, the RASTEN trial, which involved 377 patients with SCLC, did not report a survival benefit in patients with SCLC receiving enoxaparin in a supraprophylactic dose, either for patients with limited or extended disease. Interestingly, adherence to the treatment did not correlate with better survival, excluding the nonadherence as a potential cause of bias. 48 An increase in overall survival was not proven in the FRAGMATIC trial, which is the largest one involving patients with lung cancer. Regarding the incidence of VTE events, a statistically significant reduction was reported in the PROTECHT, FRAGMATIC, SAVE-ONCO, and RASTEN trials, as well as in the TOPIC-2 study in the subgroup of patients with lung cancer stage IV. Concerning safety endpoints, results seem more straightforward. The use of LMWHs as primary thromboprophylaxis appeared relatively safe; no trial reported a statistically significant increase in major bleeding events, while some trials reported an increase in nonmajor clinically relevant bleeding events.25,31
A meta-analysis of trials involving ambulatory patients with lung cancer receiving primary thromboprophylaxis indicated that LMWHs lead to a statistically significant reduction of VTE risk (4.0% vs 7.9% in LMWH and control groups) with an estimated number to treat of 25 to prevent one VTE event. The risk of bleeding events with the use of LMWH was not significantly increased. 49 Another meta-analysis reported that primary VTE prophylaxis with parenteral anticoagulation reduced the rate of VTE among ambulatory patients with lung cancer, without a significant increase in bleeding risk. Additionally, it appears that anticoagulants offer a benefit on mortality, but the effect remains unclear when the analysis is restricted to a single agent. 50
In recent years, DOACs are increasingly used as treatment of VTE in the general population. Regarding their use as primary thromboprophylaxis in ambulatory patients with cancer, Levine et al. showed that apixaban is well tolerated and safe in this specific population. 33 Additionally, both the AVERT and CASSINI trials provided encouraging efficacy results: the AVERT trial demonstrated a statistically significant reduction of VTE events with the administration of apixaban and the CASSINI trial also revealed a lower rate of VTE episodes, but only in per-protocol analysis.34,35 The risk of major bleeding reported in the AVERT study requires attention and directs additional studies to be conducted. DOACs are an emerging option of treatment and prevention of VTE in the general population since they represent a simple oral treatment regimen without the need for laboratory monitoring.12,51 It should be noted that recently, rivaroxaban and edoxaban have been added as options for treatment of VTE in patients with cancer. 18 More trials regarding the use of DOACs as treatment and as primary thromboprophylaxis in patients with cancer are greatly anticipated.
It has been suggested that the stratification of the patients could improve the clinical benefit of primary thromboprophylaxis by reducing the number needed to treat to avoid an episode of VTE. 19 Indeed, the three recently published clinical trials (PHACHS, AVERT, and CASSINI) have applied a risk-stratification approach. The PHACS trial included patients with a Khorana score of 3 or higher and the AVERT and CASSINI trials involved patients with a Khorana score of 2 or higher. Although recent studies question the performance of the Khorana score in predicting VTE events, it is remarkable that a substantial percentage of patients enrolled in the PHACS and CASSINI trials presented VTE in baseline screening.31,34,35
The recent results of the aforementioned trials justify the implementation of risk assessment models before initiation of chemotherapy, as being suggested in the recent update of the clinical practice guidelines of the American Society of Clinical Oncology. 18 It is suggested that patients should be assessed with the Khorana score before starting a new chemotherapy regimen and for patients with a score of 2 or higher, prophylactic anticoagulation may be considered unless contraindicated. Apart from LMWHs, apixaban and rivaroxaban have also been added as options of prophylactic anticoagulation in patients with cancer. Given that lung cancer is associated with an at least intermediate risk of VTE (1 point), patients with any additional risk factor from the Khorana score model may be offered prophylactic anticoagulation. The benefits and risks of prophylactic anticoagulation should be discussed with the patient (Figure 1).
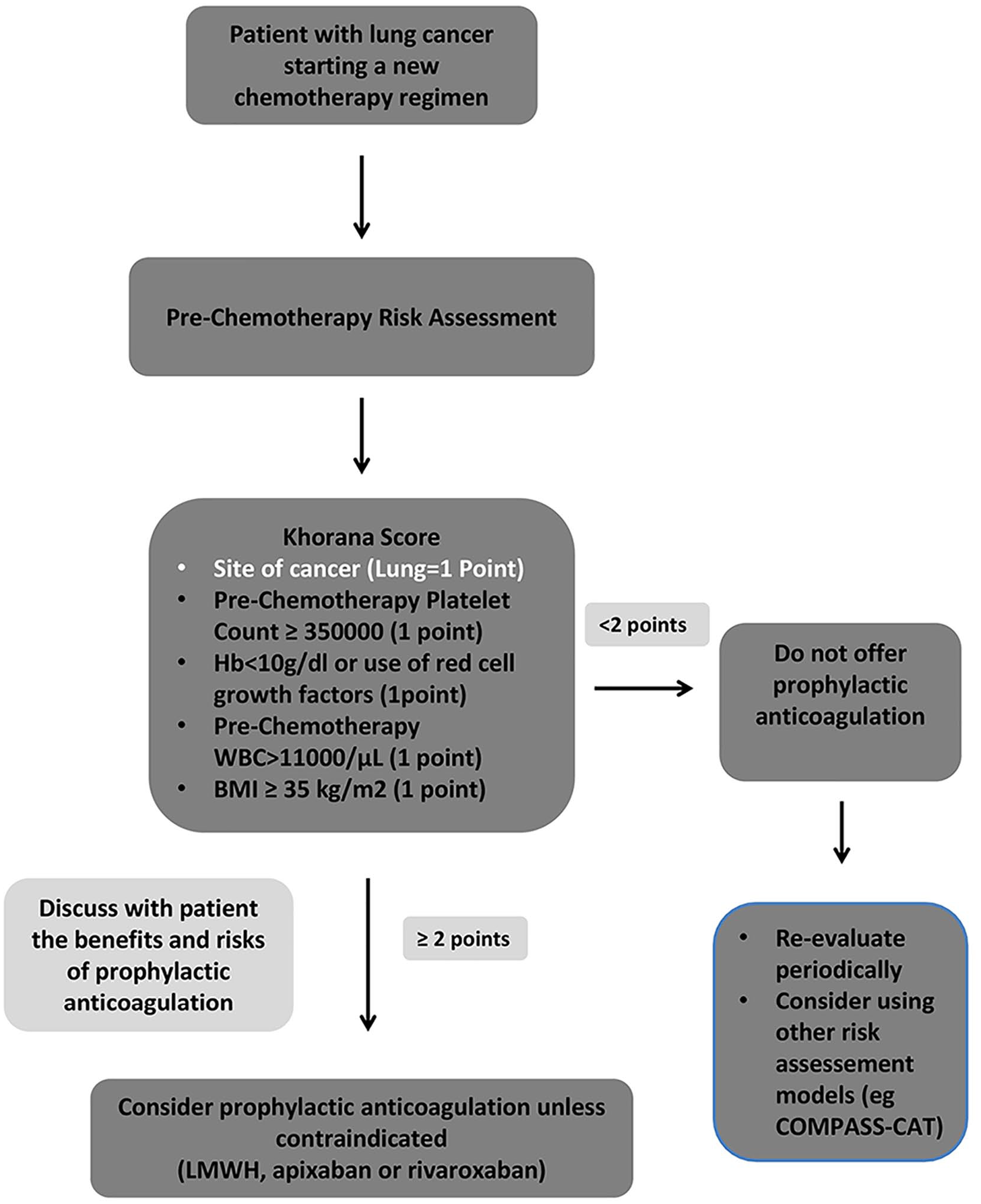
Algorithm of prophylactic anticoagulation in patients with lung cancer starting a new chemotherapy regimen.
Future points and perspectives
The development of an accurate and practical risk assessment model for prediction of VTE events in patients with lung cancer represents an unmet need. Several risk assessment models have been developed; however, their accuracy is questioned in patients with lung cancer, and for the most recent models there is not adequate external validation yet. Interestingly, recent studies have been performed to find novel biomarkers that could be used in VTE risk assessment for lung cancer that could guide clinicians in tailoring individualized treatment. In a post hoc analysis of the RASTEN trial, four assays were examined for potential association with VTE events – total tissue factor (TF); extracellular vesicle-associated TF (EV-TF); procoagulant phospholipids (PPL); and thrombin generation (TG) – but none of them did significantly associate with VTE incidence. 52 Interestingly, a recent study in patients with NSCLC suggested that dynamic changes of coagulation biomarkers might be predictive for VTE events in opposition to the current models that are designed for baseline assessment. This study performed repeated measurements of a more extensive panel of investigational thrombogenic biomarkers in order to capture the dynamic VTE risk profile and optimize test sensitivity. Although the study proved that the overall biomarker profile did change over time, consistent with the proposed concept that VTE risk is dynamic, there was no characteristic longitudinal pattern, nor absolute or relative change in biomarker value that reliably predicted for VTE events. 53 Moreover, in the ROADMAP-CAT study, two coagulation biomarkers were identified as clinically relevant for the classification of ambulatory patients with lung adenocarcinoma into high and intermediate/low risk for VTE: the procoagulant phospholipid-dependent clotting time (Procoag-PPL) and the mean rate index (MRI) of the propagation phase of thrombin generation assessed with the Calibrated Automated Thrombinoscope. 54 Finally, perhaps future risk assessment models will incorporate ‘-omics’ data that is being gathered for patients with cancer in order to improve on existing sensitivity and specificity benchmarks. 19
Conclusion
Thromboembolic disease represents a common burden both for patients suffering from lung cancer and for their physicians. Several studies have implicated potential benefit from LMWH prophylaxis in terms of VTE risk reduction and increased overall survival. Most studies agree that prophylactic LMWH administration does not significantly elevate bleeding risk in treated individuals. DOACs could evolve into an effective and more patient-friendly alternative for VTE thromboprophylaxis. Current evidence suggests the implementation of a risk-stratification approach and the administration of prophylactic anticoagulation in selected high-risk patients. However, more randomized, controlled clinical trials are required to establish profit in patients with lung cancer. Risk assessment models should be developed and validated in order to help clinicians stratify patients based on their risk profile and apply prophylaxis that will maximize therapeutic benefits and reduce bleeding risk.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
