Abstract
Peripheral artery disease (PAD) is a manifestation of generalized atherosclerosis which results in hemodynamic compromise of oxygen and substrate delivery to the lower extremity skeletal muscles. Hemodynamic assessments are vital in PAD diagnosis and in the evaluation of strategies aimed at treating claudication (i.e. exercise training, revascularization, and pharmacological agents). Venous occlusion plethysmography (VOP) is a century-old, non-invasive technique used to quantify limb blood flow and has been used to evaluate hemodynamic compromise in patients with PAD. However, the literature suggests a wide array of methodological variability in the measurement and analysis of limb blood flow using VOP. In this manuscript, we overview the clinical application of VOP measurement, and secondly we review the methodological variation that occurs during the measurement and analysis of VOP in healthy individuals and in patients with claudication.
Keywords
Introduction
Lower extremity, non-invasive physiologic vascular studies play an important role in the diagnosis and characterization of peripheral artery disease (PAD). Such studies can include ankle–brachial index (ABI), toe–brachial index (TBI), Doppler ultrasound, transcutaneous oxygen pressures (TcpO2), segmental pressure analysis, pulse volume recordings (PVR), magnetic resonance angiography (MRA), and venous occlusion plethysmography (VOP). In addition, newer techniques such as flow mediated dilation (FMD) and peripheral arterial tonometry (PAT) are commonly utilized measurements that reflect systemic endothelial function.1,2 Collectively, vascular studies can comprise a powerful toolset for defining the functionality of the arterial system, localizing the site of disease, providing prognostic data, and evaluating the effectiveness of therapies. 3 One method in particular, VOP, which has been in practice for over 100 years, 4 is a technique that has been used for the diagnosis and evaluation of PAD. However, despite its longevity, VOP measurement has noted variation that is most commonly attributed to differing protocols utilized and the interpretation of the generated curves. The purpose of this article is to: (1) provide an overview of the application of VOP in the clinical assessment of PAD; (2) review the current literature pertaining to sources of variability within VOP measurement and analysis (Table 1); and (3) discuss the variability in VOP methodologies for patients with PAD.
Summary of variability in VOP methodology and evidence-based recommendations.
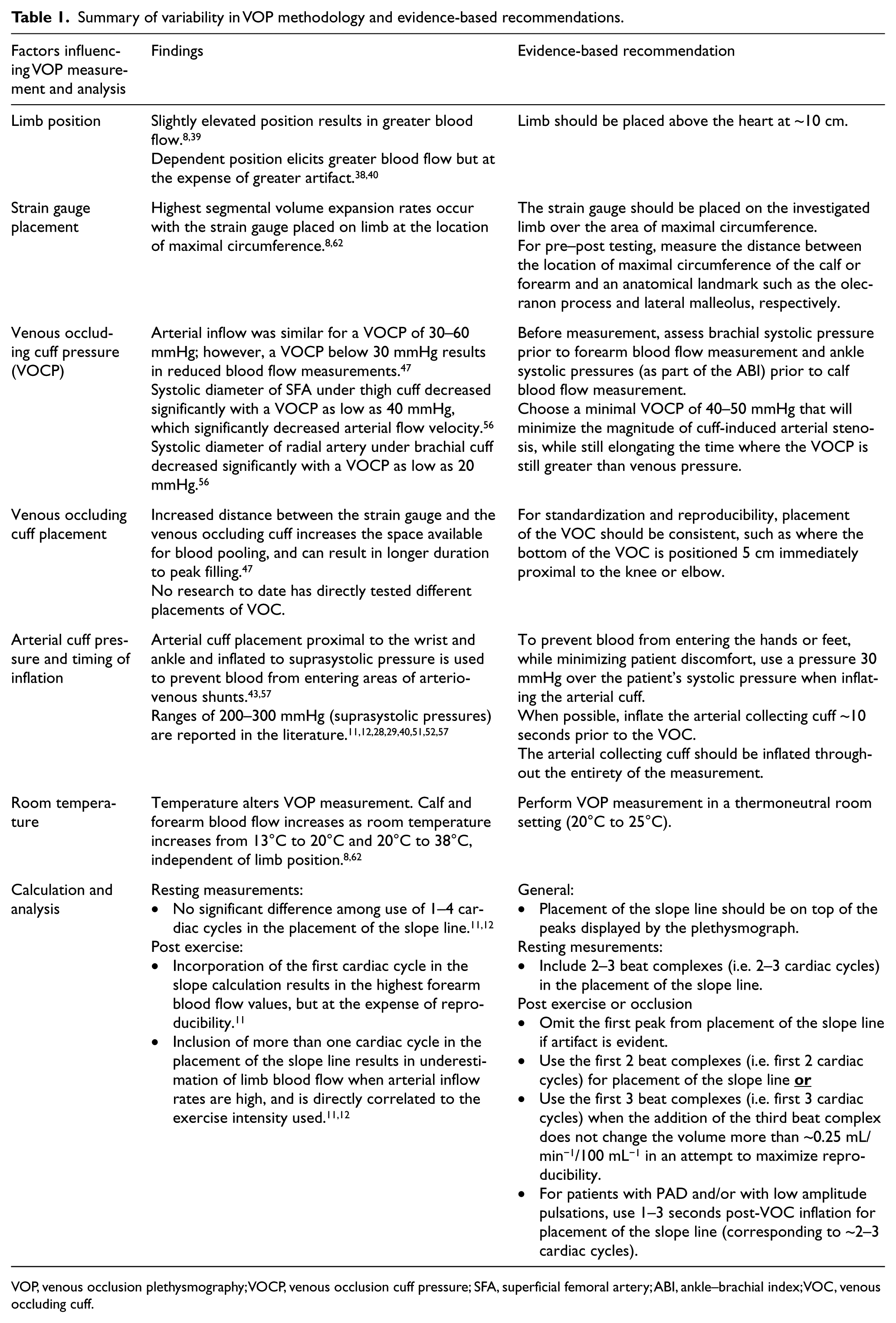
VOP, venous occlusion plethysmography; VOCP, venous occlusion cuff pressure; SFA, superficial femoral artery; ABI, ankle–brachial index; VOC, venous occluding cuff.
Concept
VOP works by creating a system where arterial blood is trapped in a limb segment by inflating occluding cuffs above and below the site under investigation. Proximal (venous occluding) cuffs, placed on the thigh or upper arm, prevent venous blood from leaving the system, and a distal (arterial occluding) cuff, placed above the ankle or wrist, prevents arterial blood from entering the branching vasculature of the foot or hand. Arterial blood flows into the system, while venous flow is halted by the venous occluding cuff, resulting in a condition where blood can enter the extremity but cannot escape. This enables measurement of the accumulating arterial inflow of the investigated limb.5–8
Mercury-in-silastic strain gauges are used to determine change in limb circumference following venous occlusion. The silastic tubing of the strain gauge is stretched upon segmental volume increase, sending an electrical current through the mercury. Changes in the electrical current signaled by the silastic tubing are subsequently graphed by the plethysmograph. 5 Blood flow is quantified using the graph of each cardiac cycle by calculating the rate of change between cycles. Additionally, the strain gauge can register arterial pulse waves traveling through the arterial tree at high velocities, providing additional insight regarding arterial wall integrity. 7
Change in limb blood flow from resting conditions can be assessed following transient limb ischemia (reactive hyperemia (RH)),9,10 exercise (active hyperemia),11,12 administration of oral or nasal nitroglycerin, or intra-arterial infusions of acetylcholine and sodium nitroprusside.13,14 The latter will be omitted from discussion in this article as it represents an invasive measurement of endothelial-dependent vasodilation. Protocols that assess RH, as a surrogate measurement of endothelial function, promote a transient state of ischemia in the examined limb by inflating the cuff on the upper arm (or thigh) to a suprasystolic pressure for a variable period of time. Blood flow is then measured immediately after the cessation of limb ischemia.9,10,15,16 Active hyperemia, as a surrogate indicator of endothelial function, is assessed immediately following metronome paced, isotonic plantar flexion or finger flexion exercise that is performed to maximal fatigue using either a foot ergometer or grip strength dynamometer, respectively.11,12
Clinical application
VOP has been repeatedly proven to be both a reliable and valid measurement for resting blood flow17,18 and conditions that produce high rates of arterial inflow, 11 including RH 19 and active hyperemia following exercise.20,21 Therefore, VOP has been commonly used as the reference to compare other popular peripheral blood flow measurement techniques such as Doppler ultrasound 22 and FMD. 23 Measurement of limb blood flow using VOP has clinical relevance, including for the prediction of cardiovascular events and the evaluation of lifestyle and pharmaceutical therapies. In particular, endothelial dysfunction, measured by RH, has been used to evaluate long-term cardiovascular prognosis in patients with PAD.24,25 The acute responses to therapy, such as passive heating in patients with PAD, are commonly evaluated using VOP. 26 Furthermore, VOP assessment of endothelial function, measured during states of both RH and active hyperemia, has been used to successfully evaluate the effects of exercise,27–29 pharmacological,30,31 surgical, and stem cell32,33 therapies in patients with PAD.
Newer technologies such as FMD and Doppler ultrasound are highly utilized techniques; however, both have limitations when measuring blood flow and endothelial function in patients with PAD. FMD has been utilized in patients with PAD to examine changes in brachial artery diameter during RH and active hyperemic states, which is believed to reflect systemic endothelial function. 34 However, FMD has not been validated for use in the lower extremities in individuals with PAD, particularly for use over time following intervention.
Doppler ultrasound, another non-invasive technique, allows blood flow calculation in conduit arteries during rest and exercise, but does not take into account blood flow in collateral arteries or irregular vessel cross sections; therefore, it fails to measure total blood flow in a leg segment, particularly one affected by PAD. 35 Despite an appearance of being tedious and somewhat dated, the well-tolerated, non-invasive and cost-effective nature of VOP makes it an appropriate choice for the measurement of blood flow in the periphery, particularly in patients with PAD.
Literature review of venous occlusion plethysmography (VOP) methodologies
Although VOP is considered, by some, as a gold standard for the analysis of peripheral blood flow,22,36,37 particularly in lower extremity vascular disease, there are subtle differences in current methodologies, which suggests a need for standardization. Common methodological differences found in the literature in regard to data acquisition, data analysis, and biological variability are discussed below. The online electronic database, MEDLINE, was queried for the following key terms: peripheral artery disease, peripheral vascular disease, strain gauge plethysmography methods, and venous occlusion plethysmography methods. The query included e-publications ahead of print, in-press, and other non-indexed citations. Reference lists were cross-examined. No date limits were set. Guidelines, editorials, comments, and letters were excluded. Only studies in English, examining methodologies of VOP or investigations using VOP in patients with PAD were included in the literature review.
Limb position
Limb position influences the transmural pressure, vascular compliance, and distention and therefore affects the quality and reproducibility of blood flow measurements. The effects of varying limb position on VOP blood flow values have been evaluated by several researchers.8,38–40 Findings regarding which limb position, including the elevated (i.e. just above the level of the heart), horizontal, and dependent positions, results in the greatest blood flow measurement have been inconsistent. Researchers have reported a wide range of variability in blood flow measurements using the dependent position, which may be explained by the simultaneous opposing physiological forces at work on the blood vessels. When the limb is in the dependent position, gravity increases the vascular hydrostatic pressure, which increases the blood flow values. However, there is evidence that the dependent position produces more artifact on the plethysmograph compared to an elevated position, 40 making the measurements difficult to read and reproduce. In contrast, elevating the limb slightly above the level of the heart promotes venous drainage, reduces vasoconstriction,41,42 and the resulting drop in transmural pressure promotes the collapse of the veins. Lower transmural pressure affords greater filling potential, which allows for greater blood accumulation, 38 assuming normal arterial inflow.
Strain gauge placement
The strain gauge measures only the blood flow in the area encircled by the band, therefore controlling for its placement is vital. VOP measurements at the calf or forearm give information on muscle blood flow, while measurements at the ankle, wrist, foot, or hand determine cutaneous circulation, each with unique and differing physiologies. 43 Therefore, if the goal is to measure calf or forearm blood flow, the strain gauge should be placed at the area of highest density of limb or segment of skeletal muscle, which is at the maximal circumference.12,44–50 For optimal reproducibility in pre–post testing, measure the distance between the location of maximal circumference and an anatomical landmark such as the olecranon process40,51 or antecubital crease in the forearm or the lateral malleolus in the leg. 52 This ensures that the same location is used in post testing.
Venous occluding cuff pressure (VOCP) and inflation
The purpose of the venous occluding cuff is to produce a state of venous tamponade, preventing venous return. VOCP must be sufficient to induce venous tamponade but low enough to ensure regular arterial inflow. However, using a VOCP low enough to guarantee arterial inflow may result in venous leakage. 47 This compromises the integrity of the closed system, as the accumulation of blood is reduced. Therefore, assessment of ankle and brachial systolic pressures should be assessed prior to calf and forearm VOP measurement, respectively. The state of venous tamponade exists until the venous pressure rises above that of the venous occluding cuff; 53 thus, there is a short window of time to capture valid blood flow measurements. This time frame is reduced during RH or following exercise, as venous pressure will equalize and surpass VOCP more quickly. Hellige et al. 54 suggest that during conditions that increase rates of blood flow, the venous pressure may equal the VOCP within two cardiac cycles. Using the optimal VOCP is particularly important in populations with decreased lower limb arterial pressure, as is seen in the PAD population, where the difference between VOCP and ankle systolic pressure may be small. The amount of pressure that should be applied to collapse the veins of the investigated limb during VOP has been a major source of controversy among researchers.17,41,42,54–56
The possibility that VOP may decrease arterial inflow has been reported.17,54,56 Therefore, VOP is a reliable and valid technique to assess change in blood flow over time; however, it may not be an optimal measurement technique for the absolute quantification of limb blood flow. This limitation in VOP was demonstrated by Hiatt and colleagues 56 who found that VOCP as little as 40 mmHg induced a 27–44% stenosis in the superficial femoral artery. This is close to the reported minimal VOCP (30 mmHg 47 ) where the venous pressure will exceed the VOCP resulting in venous leakage and lowering of the blood flow measurement.
Venous occluding cuff placement
The specific placement of the venous occlusion cuff on the thigh or upper arm is a critical factor in the accuracy of the blood flow measurement and therefore must be standardized. 5 The further away the venous occluding cuff is from the strain gauge, the longer it will take for venous filling to occur due to increased filling space. At rest, when the rate of inflow is constant, this effect is negligible; however, this can be a source of error in measurements taken immediately post exercise. The additional segmental volume between the strain gauge and the venous occluding cuff increases the space available for blood pooling, causing prolonged upsloping traced by the plethysmograph, reflecting a delay in peak filling. 47 This source of error can be exacerbated when there is a minimal amount of time before venous pressure overcomes the induced state of venous tamponade, as seen in PAD, and therefore can result in failure to capture peak blood flow values.
Arterial cuff pressure and inflation
The purpose of the arterial occlusion cuff, placed immediately proximal to the wrist or ankle, is to prevent arterial blood from entering the hand or foot. 57 As discussed earlier, because hands and feet have a high amount of arteriovenous shunting, 43 it is common practice to omit each from the measurement of forearm or calf blood flow, respectively. This ensures that the measured arterial inflow reflects what is trapped in the calf or forearm. An arterial occluding cuff pressure of 200 mmHg was originally used as a standard presssure to prevent blood flow to the hands or feet. 58 However, it is likely that any suprasystolic pressure is sufficient for this purpose and the use of extreme suprasystolic pressures is unnecessary.59,60 Previous studies have demonstrated that the arterial occlusion cuff may be inflated safely for up to 13 minutes; 16 however, high cuff pressure and prolonged inflation may cause unnecessary pain to the patient.
Timing of arterial cuff inflation can also affect the accuracy of the measurement. As the arterial collecting cuff causes a surge in retrograde venous blood flow when inflated, it should be inflated prior to the venous cuff. 57 This order of inflation allows blood flow to stabilize, which in turn reduces artifact and enhances measurement reproducibility.
Temperature
Ambient temperature is another variable that affects regional and limb blood flow. Sympathetic nervous system (SNS) activity is directly influenced by body temperature and controls blood distribution throughout the body. At high ambient temperatures, blood flow to the periphery increases due to a concurrent decrease in SNS activity causing a reduction in catecholamine binding to alpha-receptors in the smooth muscle. 61 Low body temperatures increase SNS activity in an attempt to localize blood flow to the central regions of the body. Cooler conditions, which increase vasoconstriction, will reduce peripheral blood flow while high temperatures promote venous capacitance and likely produce the highest VOP measurements. Therefore, room temperature should be used for optimal flow8,62 and tests should be performed in a temperature-controlled environment, particularly for pre–post testing.
Calculation and analysis
During blood flow measurements, the plethysmograph creates a graph showing ‘beat complexes’ (Figure 1A), where one beat complex (Figure 1B) corresponds with one cardiac cycle when VOP is simultaneously measured with electrocardiography. This represents the rate and/or rate of change of volume expansion for the area under the strain gauge, which is collectively referred to in the literature as blood flow values. 62 Blood flow values are determined by calculating the slope between beat complexes (Figure 1B). Assuming proper measurement, the predominant source of error during analysis is the number of beat complexes selected to calculate slope. There is no consensus in the literature regarding the time to onset of venous leakage (venous pressure > VOCP), which is influenced by the VOCP used, stimuli for hyperemia, and patient population. Graphically, venous leakage appears as a progressive plateau across the beat complex peaks (Figure 1B).
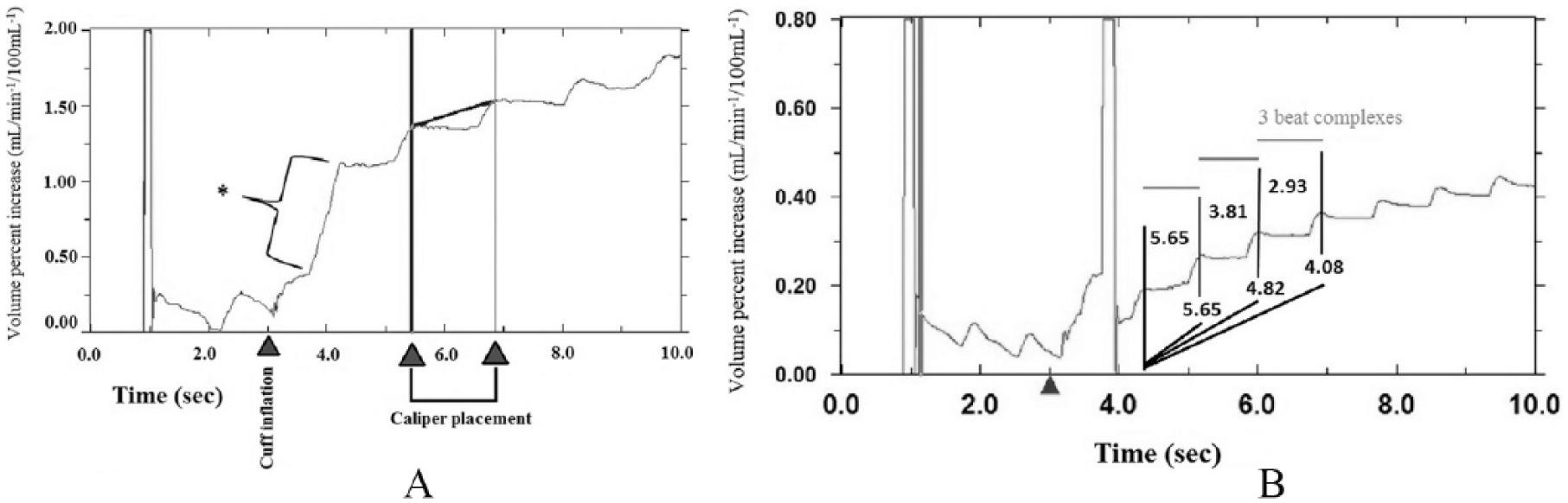
Morphology of slope lines: (A) *cuff artifact upon cuff inflation and illustration of caliper placement; (B) illustration of venous pressure exceeding venous occlusion collection cuff pressure and its quantification and definition of a beat complex.
Early practice was to omit the first and last beats from measurement as it was hypothesized that venous occlusion starts and ends at arbitrary moments in the cardiac cycle. 38 The first beat was also commonly omitted due to the possibility of artifact caused by inflation of the collecting cuffs. Additionally, common practice was to place the slope line through the lowest points (valleys) of the beat complex(es). 38
The current evidence suggests that for resting measurements, blood flow to the limb under investigation is constant and venous pressure should not exceed the VOCP; therefore there is no significant difference in the use of one to three beat complexes for the placement of the slope line. 12 However, following a stimulus that decreases the time where venous pressure is less than VOCP, incorporating more than one beat complex for the placement of the slope line results in an underestimation of blood flow, although the reproducibility is improved with the inclusion of additional beat complexes.11,12 Unfortunately, there is currently no standardized method for analysis in regard to placement of the slope line, and the variability in methodologies demonstrates the need for more research to show consistency in the analysis of VOP.
Special considerations for VOP measurement in patients with PAD
Individuals with PAD have anatomical and physiological factors including atherosclerotic occlusion(s) and endothelial dysfunction that can alter VOP measurement and analysis as compared to healthy persons. A major difference is that the waveforms in persons with PAD may have low amplitude pulsations 19 (Figures 2A and 2B). The magnitude of resting pulsation is not correlated to resting blood flow values, but is correlated to peak flow response following plantar flexion exercise and during RH, suggesting that the pulsation magnitude seen on the plethysmograph may be an indicator of disease severity. 19
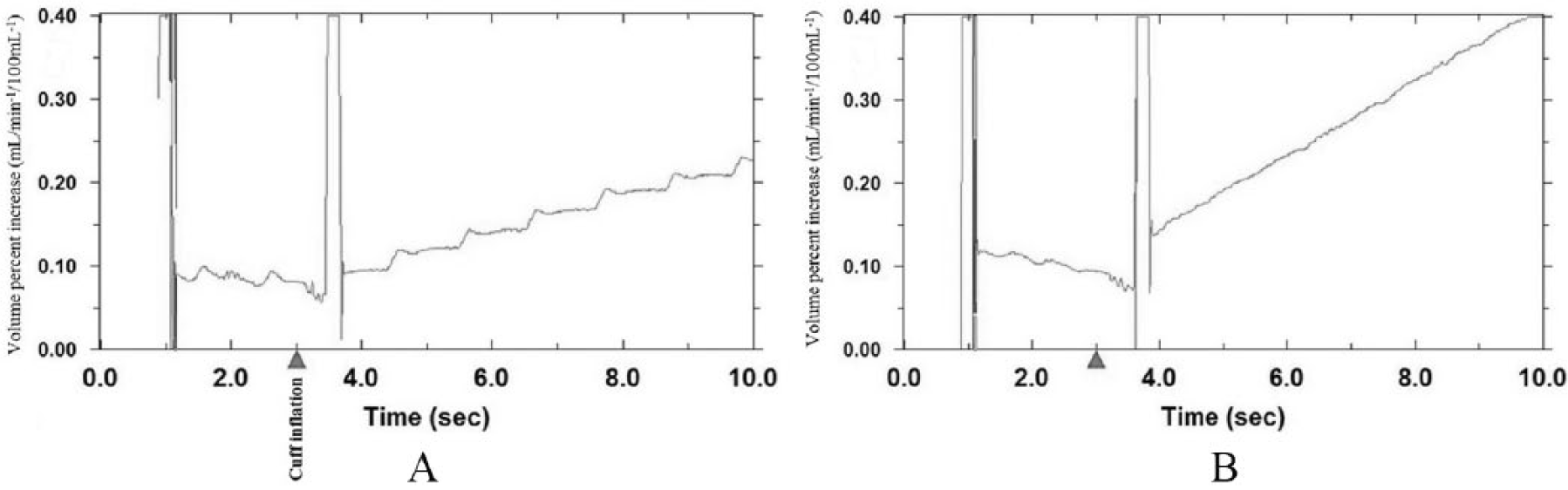
Morphology of slope lines: (A) visible pulsations at rest in a healthy participant during calf blood flow assessment; (B) poorly defined pulsations at rest in a participant with PAD during calf blood flow assessment. Triangle indicates cuff inflation.
In addition, the atherosclerotic burden and endothelial dysfunction are both believed to be mechanisms by which individuals with PAD may present with lower peak blood flow values,19,44,50,63,64 and a delay in peak blood flow response (Table 2).44,50,63,64 In extreme cases, the delay in peak blood flow response has been documented to be upwards of 6 minutes following exercise stimuli. 44 These findings suggest inadequate oxygen delivery to support the metabolically active skeletal muscle. 65 Another major difference is that persons with PAD often require a longer time to re-establish baseline blood flow values. Reports in excess of 15 minutes for recovery have been published. 44 The delay in recovery time suggests that the tissues are failing to recover in a hypoxic condition and thereby not clearing metabolites, or repaying the oxygen debt, so flow would remain elevated for longer durations following the stimulus. 65
Delay in peak blood flow measurements following isotonic plantar flexion exercise in patients with PAD.
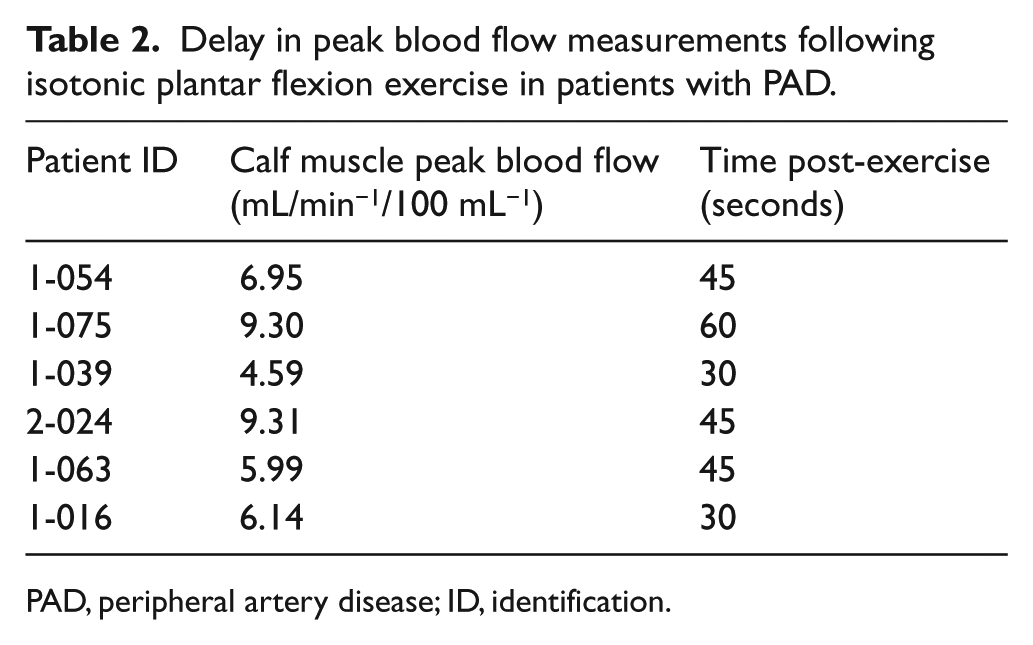
PAD, peripheral artery disease; ID, identification.
Not all patients with PAD show these VOP characteristics, as19,50,63,66 atherosclerosis-related resistance in the large arteries can be compensated by collateral circulation. This can make it difficult to distinguish a healthy artery from a diseased artery when analyzing the classic VOP characteristics discussed above. Therefore, when using VOP for assessing peripheral blood flow in persons with PAD, the following must be considered and/or applied: (1) the first post-exercise and RH measurement may not be indicative of the peak flow response; (2) recovery measurements, in addition to the five post-exercise or RH measurements, should be taken until baseline blood flow values have been re-established; and (3) due to low amplitude oscillometric pulsations, care must be taken to ensure that the slope line is placed in the time frame where two cardiac cycles would normally be found.
Implementation of VOP measurement in a clinical PAD trial
These methods and considerations, along with those outlined in Table 1, were successfully used to guide the National Heart, Lung, and Blood Institute (NHLBI) Exercise Training to Reduce Claudication (EXERT): Arm Ergometry Versus Treadmill Walking study protocol. The EXERT study (NCT00895635) was a large randomized controlled trial investigating the effects of different modes of supervised aerobic exercise training on walking capacity in patients with PAD with a secondary outcome of elucidating the underlying mechanisms of exercise-induced improvements in walking distance using VOP. The Hokanson EC6 plethysmograph, E20 rapid cuff inflator system, and NIVP3 software (Hokanson, Bellevue, WA, USA), Baseline Smedley hand dynamometer (Smedley Fabrication Enterprises, White Plains, NY, USA), and pedal foot apparatus (University of Minnesota School of Nursing and School of Engineering, Minneapolis, MN, USA) were used for VOP blood flow measurement (Figures 3A–F and 4A–C).
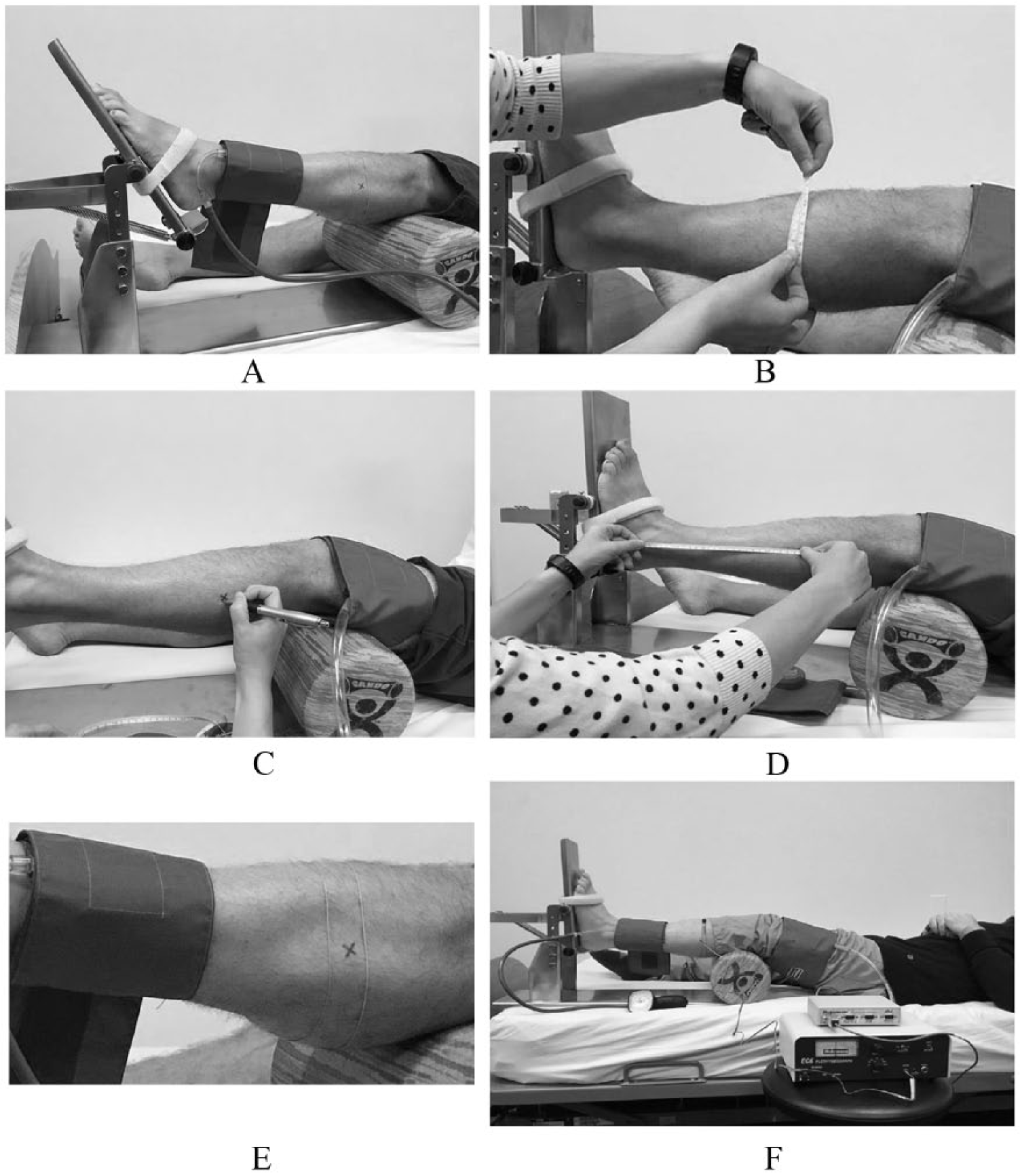
Set up of VOP for calf blood flow assessment: (A) calf ergometer for plantar flexion exercise; (B) measurement of maximal calf circumference for strain gauge placement; (C) mark maximal calf circumference; (D) measurement of the distance between the lateral malleolus and area of maximal calf circumference; (E) strain gauge placement and placement of arterial occlusion cuff; and (F) complete calf apparatus set up.
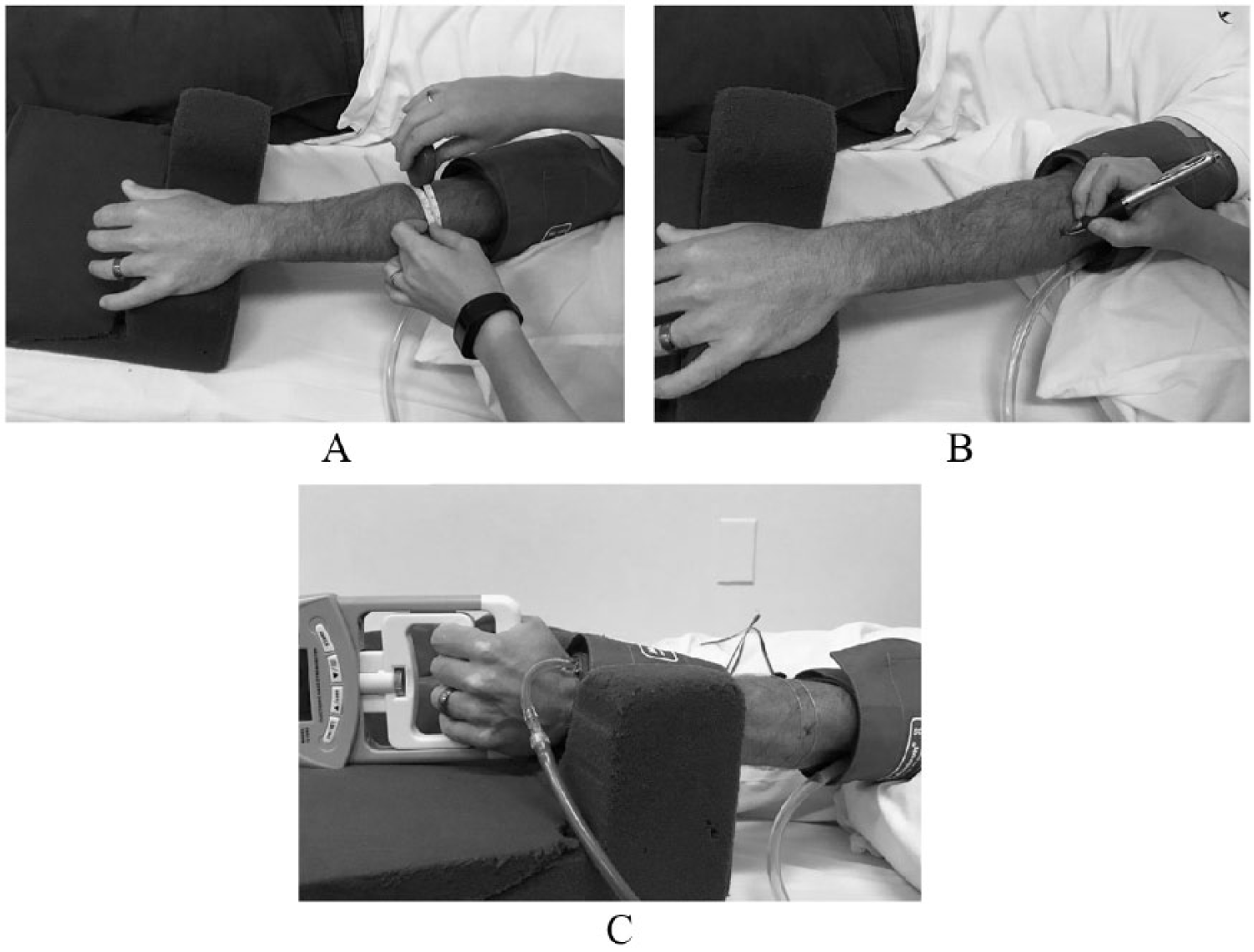
Set up of VOP for forearm blood flow assessment: (A) measurement of maximal forearm circumference for strain gauge placement; (B) mark maximal forearm circumference; and (C) complete forearm apparatus set up.
Conclusion
VOP has been highly utilized since its inception in the late 1800s and has been subjected to years of validity and reliability testing. Accurate measurement is dependent upon precise equipment set up and limb positioning. Careful examination of plethysmograph output is required to prevent the over or underestimation of peak blood flow while maximizing reliability. Additionally, extreme diligence must be taken when performing VOP measurement in patients with PAD to ensure valid blood flow values due to reduced lower extremity arterial pressures and low amplitude pulsations seen in this population. Lower extremity arterial pressures that approach venous occluding cuff pressures may result in incomplete venous occlusion and the underestimation of blood flow. Therefore, patients with lower extremity arterial pressures under 50 mmHg are not good candidates for VOP measurement. This should be considered when attempting to measure lower limb blood flow in patients with severe PAD. Furthermore, the potential for lower peak blood flow values, a delay in time to peak flow, and longer recovery time is expected in patients with PAD.
VOP can be used in the prognostic evaluation and effectiveness of therapeutic interventions in patients with PAD. Furthermore, VOP testing can provide insight into the underlying physiologic mechanism of change in physical functioning in patients with PAD following interventions such as supervised exercise programs. Therefore, VOP offers an analytic technique to evaluate the effectiveness of disease management strategies designed to enhance limb blood flow in patients with PAD.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by National Heart, Lung, and Blood Institute R01HL 090854-01A1.
