Abstract
Prior studies have assessed the prognostic value of a decrease, not an increase, of the post-exercise ankle–brachial index (ABI) among patients with normal resting results. Thus, we sought to evaluate the prognostic significance of an increase in post-exercise ABI among these patients. From a single center vascular laboratory database between September 2005 and January 2010, we retrospectively identified 1437 consecutive patients with a normal resting ABI (1.00–1.40) and available post-exercise results. We classified them into group 1 (normal subjects; post-exercise ABI drop ⩽ 20%, 58%) and group 2 (post-exercise ABI increase, 42%) after excluding those with an ABI drop > 20% (peripheral artery disease) as they had known disease (n=192). The primary outcome was to assess the risk of ischemic events, defined as a composite of unadjudicated death, stroke, or myocardial infraction (MACE). Associations between groups and outcomes were examined using multivariable Cox proportional hazard and propensity analyses. Both groups had similar prevalence of cardiovascular comorbidities. In unadjusted analysis, group 2 was more likely to have MACE (p = 0.001). After adjusting for all baseline characteristics, an increase in post-exercise ABI compared to normal subjects was associated with a higher MACE (adjusted HR: 1.70, 95% CI: 1.14–2.53; p=0.009). This association stayed statistically significant after propensity matching (adjusted HR: 1.80, 95% CI: 1.17–2.76; p=0.007). This hypothesis-generating analysis showed that an increase in post-exercise ABI appears to identify a population with a higher risk for MACE. A prospective study of this association and mechanisms of risk should be conducted.
Keywords
Introduction
In patients with a normal resting ankle–brachial index (ABI) and suspected intermittent claudication, a post-exercise ABI is recommended. 1 Healthy subjects may have no change to a mild drop in ABI with exercise, while a drop of > 20% is indicative of peripheral artery disease (PAD). These patients have a higher incidence of lower extremity revascularization and cardiovascular events.1–5 More recently, it has been reported that between 5% and 20% of patients undergoing exercise ABI testing may have an increase in post-exercise ABI.5–7 The clinical significance of this increase, however, has never been evaluated. We hypothesize that an increase in post-exercise ABI could serve as a useful marker for future cardiovascular events. In this retrospective analysis, we will investigate the prognostic implications of this increase and suggest plausible mechanisms.
Methods
Study population
From a tertiary medical center and its affiliated eight community hospitals between September 2005 and January 2010, we retrospectively identified 1437 consecutive subjects with a normal resting ABI (1.00–1.40) and available post-exercise measurements. We excluded those with PAD (n = 192), a post-exercise ABI drop > 20%, as per the current American College of Cardiology/American Heart Association (ACC/AHA) guidelines, 1 due to the well-known association with adverse cardiovascular events. The study population was composed of 1245 subjects. According to post-exercise ABI, patients were classified into two groups: group 1 = post-exercise drop ⩽ 20%, if both legs of a patient had a ⩽ 20% drop with exercise (normal response); group 2 = post-exercise increase, if either leg of a patient had an increase in ABI with exercise. This study was approved by the Institutional Review Board and informed consent was waived.
Data collection, ABI measurement, and outcomes
ABI data were acquired from the institutional Non-Invasive Vascular Laboratory Database. All other data including baseline characteristics and outcomes were obtained through electronic medical records review. This was supplemented by the Social Security Death Index (SSDI) with a closing date on 30 September 2011.
Registered vascular technologists performed all ABI studies in an accredited vascular laboratory (Intersocietal Accreditation Commission) using a standardized protocol. Systolic blood pressure was obtained at the level of the brachial arteries and the ankle arteries (dorsalis pedis and posterior tibial arteries) in all four extremities using a continuous wave Doppler device after patients had been resting in a supine position for at least 5 minutes. The resting ABI for each leg was calculated using the higher of the brachial systolic pressures and the higher of the two ankle systolic pressures in the corresponding leg. The total resting ABI was the lower of the two legs. For post-exercise ABI, patients were asked to walk on a motorized treadmill at a 12.5% grade at 2.0 mph (3.2 km/h) for up to 5 minutes or until patients had to stop due to symptoms. The speed or incline of the treadmill may have been adjusted for patient safety at the discretion of the technologist. Following completion of exercise, systolic blood pressures were immediately remeasured in the arm that had the higher resting brachial pressure and at the ankle vessel used for the resting ABI calculation, and the ABI was recalculated for each leg.
The primary endpoint was major cardiovascular adverse events (MACE), a composite of death, stroke, or myocardial infarction obtained from a review of the medical record and query of the SSDI. Criteria for stroke involved documented new or worsening focal neurological symptoms for > 24 hours along with neuroimaging confirmation. Criteria for myocardial infarction consisted of documented cardiac ischemic symptoms or electrocardiographic changes along with positive cardiac biomarkers.
Statistical analysis
Continuous variables were displayed as mean ± standard deviation, and categorical variables were displayed as number (%). Continuous data comparisons were performed using the t-test (for parametric variables) or Wilcoxon rank-sum test (for non-parametric variables). Categorical data comparisons were performed using the chi-squared test or Fisher’s exact test when variable frequency was less than 10. Only four variables (high-density lipoprotein, low-density lipoprotein, body mass index, and creatinine clearance) had missing values in 33%, 30%, 21%, and 19% of the study population, respectively. The R package ‘mice 2.22’ was used to impute missing laboratory values under fully conditional specification; 8 10 datasets were imputed and one randomly chosen dataset was used for all analyses.
The primary outcome was studied using Kaplan–Meier plots (date of ABI testing represented time zero) in addition to unadjusted and multivariable adjusted (for all variables in Table 1) Cox proportional hazard regression analyses. A secondary analysis was performed utilizing a greedy matching algorithm in a 1:1 fashion for all the variables in Table 1 to create propensity score-matched groups. 9 The primary outcome was then re-analyzed using Cox multivariable analyses. A series of sensitivity analyses were conducted to support the level of confidence in the association between the increased post-exercise ABI group and MACE. Each of the following variables – resting brachial systolic blood pressure, post-exercise brachial systolic blood pressure, change in brachial systolic blood pressure with exercise, resting ankle systolic blood pressure, post-exercise ankle systolic blood pressure, change in ankle systolic blood pressure with exercise, and the ratio of the change in ankle systolic blood pressure to the change in the brachial systolic blood pressure – was individually included in the propensity model to predict MACE, after excluding group assignment. For those variables which were predictive of MACE, sensitivity analyses were repeated after adding group assignment back into the model. Statistical significance for all comparisons was defined as p < 0.05. All statistical analyses were performed using R Statistical Software, version 3.1 (R Foundation for Statistical Computing, Vienna, Austria).
Baseline characteristics according to post-exercise ABI status among patients with a normal resting ABI.
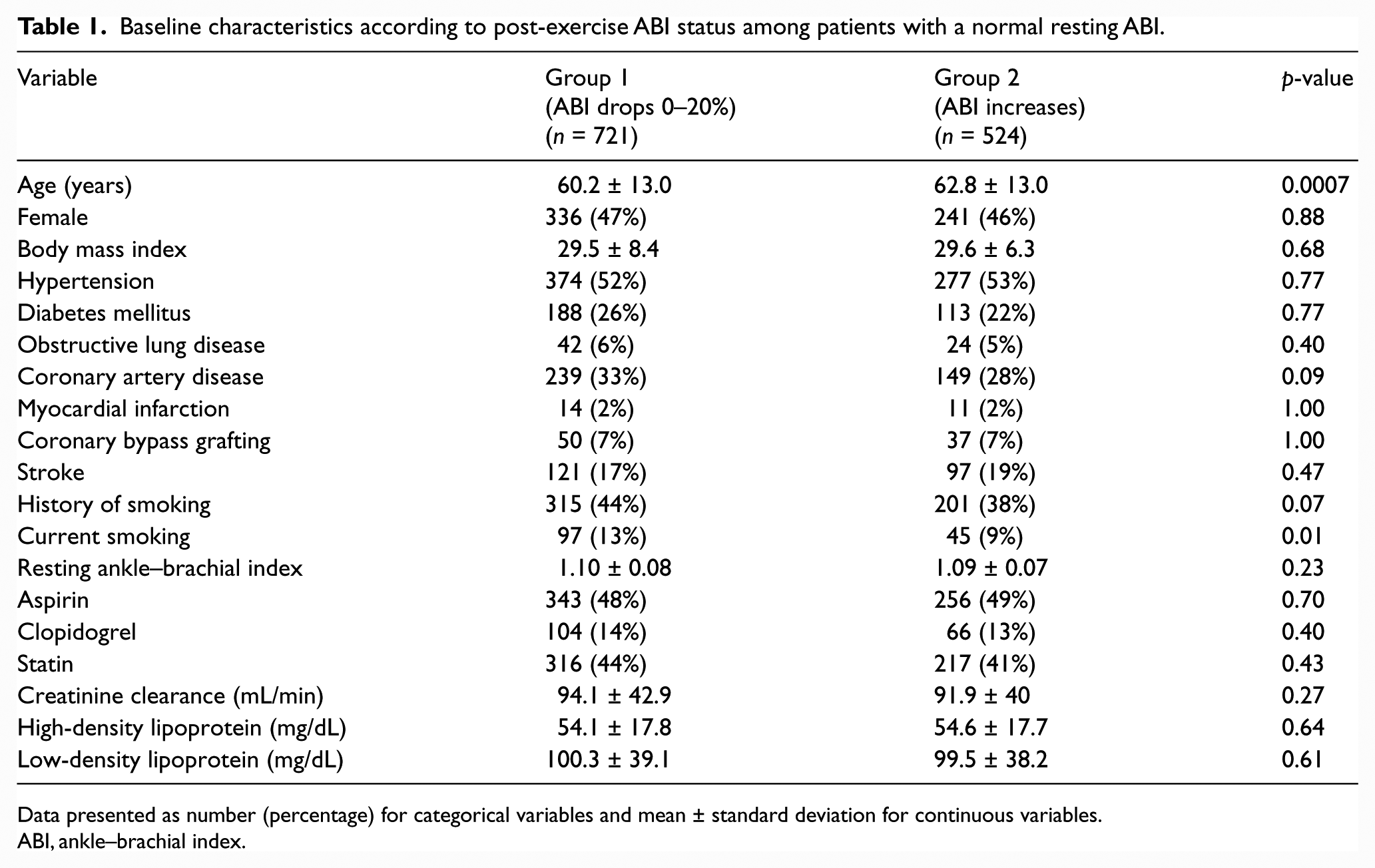
Data presented as number (percentage) for categorical variables and mean ± standard deviation for continuous variables.
ABI, ankle–brachial index.
Results
Baseline characteristics
A total of 1245 patients had a normal resting ABI (1.00–1.40), of which 721 (58%) had no change or a ⩽ 20% drop in post-exercise ABI, and 524 (42%) had an increase in post-exercise ABI. Fifty-nine percent of the patients had atypical symptoms (the most common presentation in groups 1 and 2), while 24% had classic symptoms of intermittent claudication, and 17% were asymptomatic. All subjects had a median follow-up of 3.8 years, and referral for ABI testing was made by vascular medicine (24%), primary care (24%), cardiology (20%), vascular surgery (9%), and other specialties (23%).
There were significant differences in few baseline characteristics (Table 1) and ABI testing data (Table 2) between the two groups. Patients with increased post-exercise ABI (group 2), compared with those who dropped ⩽ 20% (group 1), were older, less likely to be current smokers, and had higher resting brachial and post-exercise ankle pressures.
ABI testing data according to post-exercise ABI status among patients with a normal resting ABI.
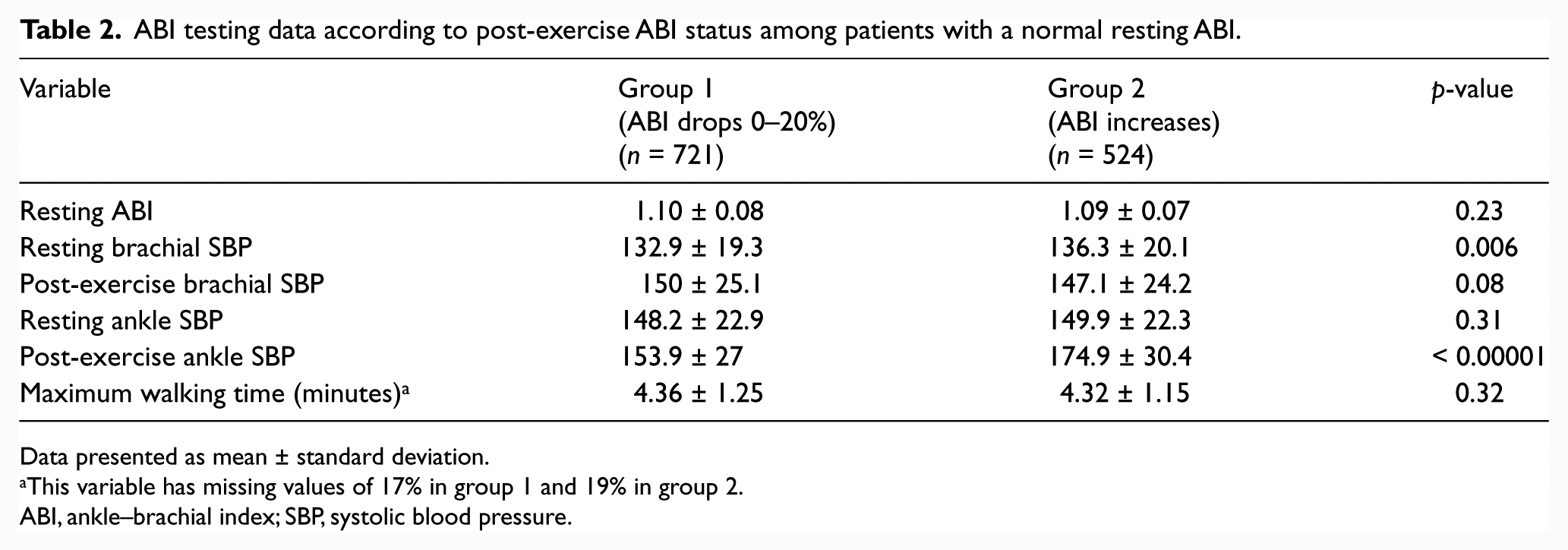
Data presented as mean ± standard deviation.
This variable has missing values of 17% in group 1 and 19% in group 2.
ABI, ankle–brachial index; SBP, systolic blood pressure.
Unadjusted and multivariable adjusted analyses
Percentages of the primary outcome and its components across the two ABI groups are displayed in Table 3. In unadjusted analysis, there was a significant difference in the outcomes between the ABI groups; risk of MACE was higher among those with increased post-exercise ABI (Table 4, Figure 1). After multivariable Cox proportional hazard adjustment (for all variables in Table 1) and when comparing Group 2 (post-exercise increase) to Group 1 (post-exercise drop ⩽ 20%), an increase in post-exercise ABI was associated with a higher MACE (adjusted hazard ratio (HR): 1.7, 95% confidence interval (CI): 1.14–2.53;p = 0.009) (Table 4). In order to exclude the possibility that the association between MACE and the post-exercise increase of ABI is driven by an underlying existing association between MACE and increased arterial stiffness, we excluded individuals with an ABI between 1.3 and 1.4 (n=120). Multivariable Cox proportional hazard regression analysis of the remaining 1125 showed that an increase in post-exercise ABI remained significantly associated with MACE (adjusted HR: 1.72, 95% CI: 1.14–2.60; p = 0.01).
Incidence of MACE among patients with a normal resting ABI according to their post-exercise ABI status.
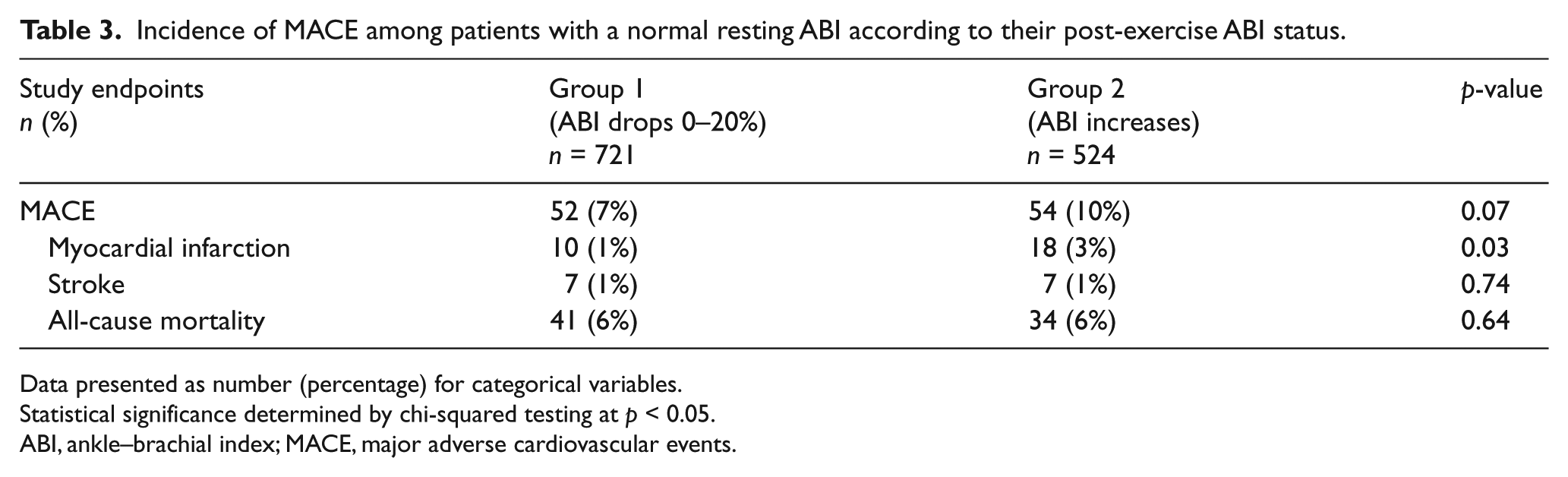
Data presented as number (percentage) for categorical variables.
Statistical significance determined by chi-squared testing at p < 0.05.
ABI, ankle–brachial index; MACE, major adverse cardiovascular events.
Prognostic value of an increase in post-exercise ABI compared to normal subjects among patients with a normal resting ABI.
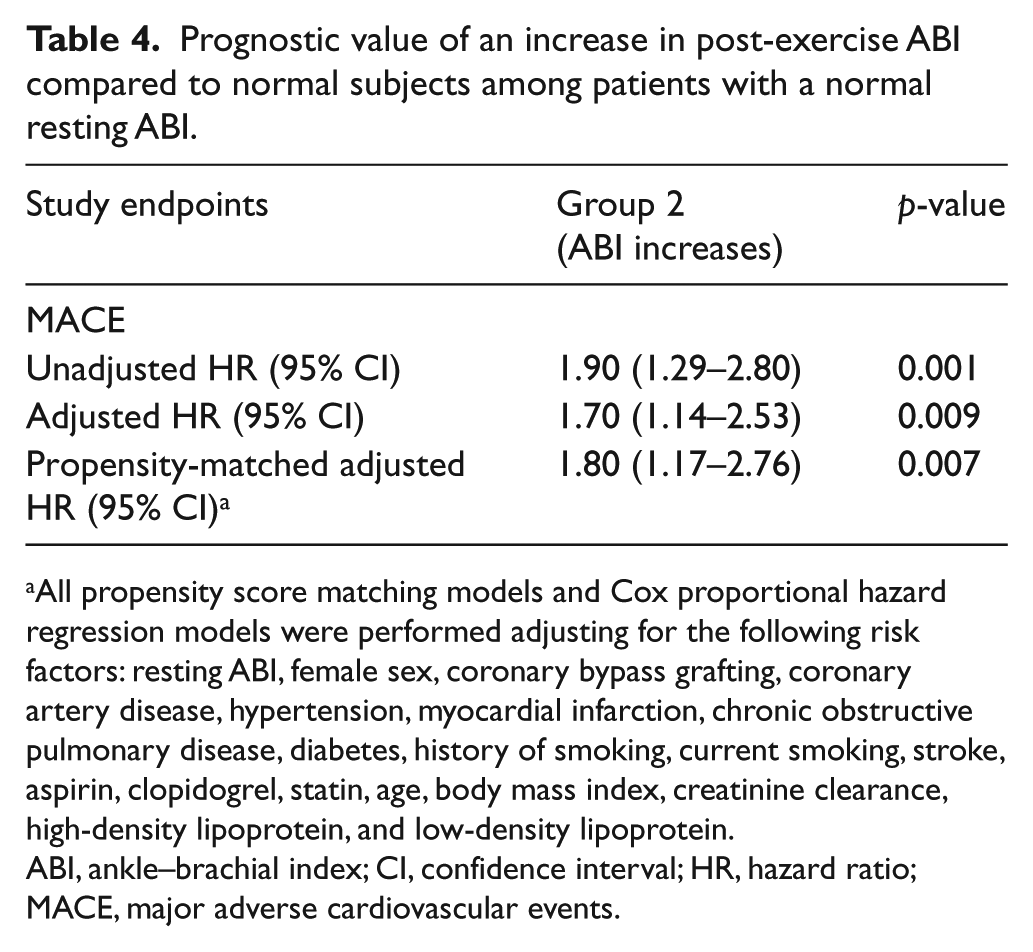
All propensity score matching models and Cox proportional hazard regression models were performed adjusting for the following risk factors: resting ABI, female sex, coronary bypass grafting, coronary artery disease, hypertension, myocardial infarction, chronic obstructive pulmonary disease, diabetes, history of smoking, current smoking, stroke, aspirin, clopidogrel, statin, age, body mass index, creatinine clearance, high-density lipoprotein, and low-density lipoprotein.
ABI, ankle–brachial index; CI, confidence interval; HR, hazard ratio; MACE, major adverse cardiovascular events.
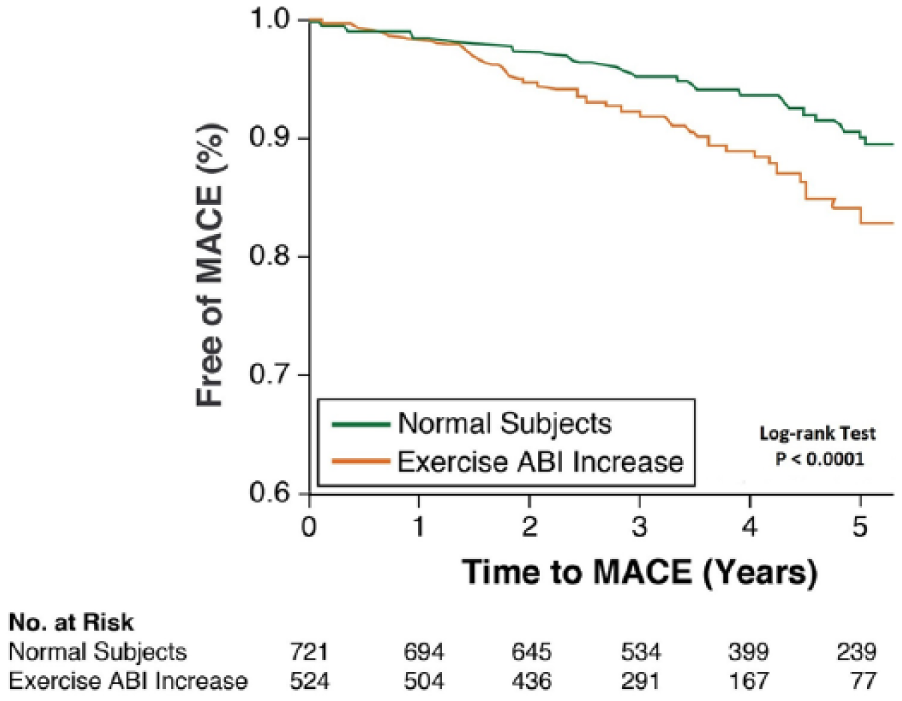
Unadjusted Kaplan–Meier curves of MACE for the two ABI groups. The log-rank test revealed a statistically significant difference in MACE across the two ABI groups over time (p < 0.0001). (ABI, ankle–brachial index; MACE, major adverse cardiovascular events.)
Propensity adjusted analysis
Propensity matching was performed between the two groups. Each of group 1 (post-exercise ABI drop ⩽ 20%) and group 2 (post-exercise increase) had 524 matched patients (1048 patients total) with similar baseline characteristics and maximal walking time (Supplementary Tables 1 and 2). An increase in post-exercise ABI, compared to a drop ⩽ 20%, stayed significantly associated with higher MACE (adjusted HR: 1.80, 95% CI: 1.17–2.76; p=0.007) (Table 4).
The sensitivity analyses evaluated components of the ABI at rest and post exercise. These analyses identified that both resting brachial and ankle systolic blood pressures were predictive of MACE (higher resting pressure indicative of increased risk of MACE). In contrast, only the post-exercise ankle pressure (not brachial) was predictive of MACE (Supplementary Table 3).
Discussion
Among patients with a normal resting ABI, a post-exercise ABI is usually performed and interpreted as either a drop ⩽ 20% (no PAD) or drop > 20% (PAD). Our study highlights an additional possible response of the ABI to exercise, which is an increase post exercise. The clinical significance of the latter is unknown and has not been investigated.5–7 In this study, we have shown that patients with an increase in their post-exercise ABIs compared to those with a mild drop (normal subjects) are at higher risk for major adverse cardiovascular events after adjusting for many variables including cardiac comorbidities, medications use, and resting ABI.
At rest, the resistance of the distal arterioles creates retrograde pressure waves that meet the anterograde waves and form the actual magnitude of the systolic pressure wave. This results in systolic blood pressure magnification as the blood streams distally in the arterial tree 10 – the central aortic pressure < brachial pressure < ankle pressure, if all are measured simultaneously at a given time. During walking, the brachial artery systolic pressure increases due to an increase in left ventricle systolic pressure along with peripheral vasoconstriction (forming retrograde pressure waves), whereas the ankle systolic pressure increases proportionally to the brachial pressure increase but to a lesser extent due to the vasodilation effect (reducing retrograde pressure waves) in the exercising limbs. These hemodynamic changes result in a mild drop in post-exercise ABI in healthy subjects3,4 and a higher drop (> 20%) when lower extremity atherosclerosis is present. 1 However, we were able to identify a separate group of patients who did not have either of the above responses, but rather their ABIs increased post exercise. This group appears to have worse outcomes compared to healthy subjects. While the exact mechanism of this phenomenon is unknown, a disproportionate increase in ankle pressure, relative to the brachial pressure increase, is responsible for the increase in post-exercise ABI in these patients (Figure 2). For example, a group 1 patient (normal subject) with an increase in brachial blood pressure with walking of 50 mmHg had an increase in ankle blood pressure of 20–40 mmHg, while a group 2 patient with a similar increase in brachial blood pressure with walking, had a higher increase in his/her ankle pressure: 50–75 mmHg (Figure 2). Impaired vasodilation (endothelial dysfunction), maintaining retrograde pressure waves, in the exercising lower limbs is a possible explanation. Both Table 1 (many similar baseline characteristics) and Figure 1 (late separation of the MACE curves) support this theory; endothelial dysfunction usually precedes other typical atherosclerotic risk factors and allows early detection of cardiovascular disease. Arterial stiffness is another possibility; however, subjects with a resting ABI > 1.4 were excluded. Measurement error is a possibility but less likely 11 given the large number of patients who had this phenomenon and the preformed sensitivity analyses. A drop in brachial systolic blood pressure after exercise, indicating possible upper extremity PAD, was observed in 54 (10%) of the 524 subjects who had increase in their post-exercise ABI (Figure 2); however, only two (4%) of those 54 patients had MACE compared to a MACE of 10% in the whole group 2 (increase in post-exercise ABI) population (Table 3). In addition, the change in brachial systolic blood pressure with exercise was not predictive of MACE in the performed sensitivity analyses (Supplementary Table 3).
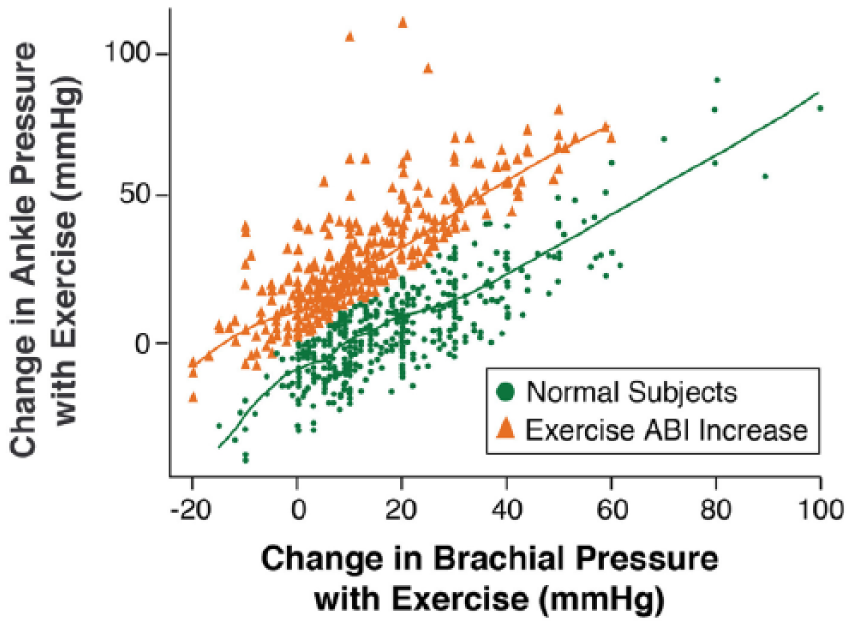
The change in brachial and ankle systolic blood pressures with exercise in the two ABI groups. During exercise and for a given increase in brachial pressure, a group 2 (increase in post-exercise ABI) patient has a higher increase in his/her ankle pressure compared to a normal subject (group 1). (ABI, ankle–brachial index.)
In addition to our previously published paper, 5 which shared the same database with this current study, only two other studies have reported similar findings, but none have investigated their implications.5–7 De Liefde et al. 6 reported 93 patients who had an increase in their ABI post exercise without specification of their resting ABI status. Diehm et al. 7 had 357 patients who had a resting ABI < 0.9 but a post-exercise ABI ⩾ 0.825.
When a post-exercise ABI is performed among patients with normal resting measurements, exercise results are traditionally reported as either ⩽ or > 20% drop. Based on this analysis, we encourage all reports to additionally highlight any increase in post-exercise ABI, when present, due to the associated increased risk for MACE. No specific or targeted management can be determined currently to reduce subsequent cardiovascular events in this group of patients due to the lack of understanding of this phenomenon or the etiology of its implications. However, general measures such as smoking cessation, diabetes and blood pressure controls if present, and following a healthy diet should all be more emphasized among this population.
Limitations
This is a large study but it has certain limitations. The prevalence of an increase in post-exercise ABI may be higher in our population given that our institution is a tertiary referral center and this prevalence may not be generalizable. No angiographic information was available for correlation with a post-exercise ABI increase; however, this would be hard to obtain even in future studies due to the absence of legitimate indications. Systematic bias due to lack of formal adjudication of the primary outcome is possible, and future studies evaluating this phenomenon should prospectively adjudicate MACE events. Unrecorded outcomes might have occurred at other facilities, but only 53 (3%) patients were lost to follow-up with no captured outcomes. Homogeneity in exercise ABI measurement is always a challenge; however, we follow a fixed exercise protocol in all patients with no exceptions unless for safety reasons. Lastly, missing variables such as individual heart rate responses to the exercise, cardiac output, left ventricle ejection fraction, and the use of vasoactive medications could have affected the blood pressure amplification process and thus our finding.
Conclusion
The current ACC/AHA PAD guidelines recommend post-exercise ABI measurement when the resting ABI is normal and intermittent claudication is suspected. However, those guidelines only comment on the diagnostic and prognostic value of a post-exercise drop > 20%. An increase in post-exercise ABI is usually overlooked and under-reported. In addition, its prognostic value has never been investigated. In this analysis, we have showed that an increased post-exercise ABI identified a population that is at higher risk for major cardiovascular events. Future prospective studies are needed to confirm this prognostic finding and determine possible mechanisms.
Footnotes
Acknowledgements
We thank Naomi Hamburg, MD, MS, Evans Department of Medicine and Whitaker Cardiovascular Institute, Boston University School of Medicine, Boston, MA; Suzanne Turner, Department of Graphics, Heart and Vascular Institute, Cleveland Clinic, Cleveland, OH; and Kathryn Brock, BA, Editorial Services Manager, Heart and Vascular Institute, Cleveland Clinic, Cleveland, OH.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
