Abstract
Ankle–brachial indices (ABIs) are important for the assessment of disease burden among patients with peripheral artery disease. Although low values have been associated with adverse clinical outcomes, the association between non-compressible ABI (ncABI) and clinical outcome has not been evaluated among patients with critical limb ischemia (CLI). The present study sought to compare the clinical characteristics, angiographic findings and clinical outcomes of those with compressible (cABI) and ncABI among patients with CLI. Consecutive patients undergoing endovascular evaluation for CLI between 2006 and 2013 were included in a single center cohort. Major adverse cardiovascular events (MACE) were then compared between the two groups. Among 284 patients with CLI, 68 (24%) had ncABIs. These patients were more likely to have coronary artery disease (p=0.003), diabetes (p<0.001), end-stage renal disease (p<0.001) and tissue loss (p=0.01) when compared to patients with cABI. Rates of infrapopliteal disease were similar between the two groups (p=0.10), though patients with ncABI had lower rates of iliac (p=0.004) or femoropopliteal stenosis (p=0.003). Infrapopliteal vessels had smaller diameters (p=0.01) with longer lesions (p=0.05) among patients with ncABIs. After 3 years of follow-up, ncABIs were associated with increased rates of mortality (HR 1.75, 95% CI: 1.12–2.78), MACE (HR 2.04, 95% CI: 1.35–3.03) and major amputation (HR 1.96, 95% CI: 1.11–3.45) when compared to patients with cABIs. In conclusion, ncABIs are associated with higher rates of mortality and adverse events among those undergoing endovascular therapy for CLI.
Keywords
Introduction
Peripheral artery disease (PAD) results from obstructive atherosclerotic narrowing of the lower extremity arteries. PAD prevalence is estimated to vary from 4% in the general population to 30% among primary care practices.1,2 Ankle–brachial index (ABI) screening is recommended for the detection of unrecognized PAD in at-risk populations. 3 An ABI <0.9 is sensitive and specific for the diagnosis of PAD and is associated with increased cardiovascular morbidity and mortality, independent of traditional risk factors.4–6 However, the ABI may be unreliable and termed ‘non-compressible’ when the infrapopliteal arteries cannot be compressed by a blood pressure cuff. A non-compressible ABI (ncABI) is defined by an ABI of >1.4 and is histologically associated with medial arterial calcification (Mönckeberg’s calcific sclerosis), which is classically observed in diabetics and those with advanced renal disease. 7 When used as a screening tool, the presence of ncABIs can mask or confound the detection of PAD.8,9 Important differences in cardiovascular disease risk factors also exist between those with low and high ABI.10–13 While it has long been known that very low ABI is associated with a poor prognosis, recent studies have also suggested that a non-compressible (>1.4) ABI is associated with increased cardiovascular events and mortality 14 when compared to the overall population of patients with PAD.
Little has been published regarding the presentation and outcomes of patients with ncABI and critical limb ischemia (CLI). We therefore sought to evaluate the association between ncABI with the angiographic patterns of disease and clinical outcomes among patients with CLI. We hypothesized that ncABIs among patients with CLI would be associated with increased rates of mortality, major adverse cardiovascular events (MACE), and increased amputation rates.
Methods
The PAD-UCD (University of California, Davis) Registry consists of all patients with a clinical diagnosis of PAD who underwent diagnostic peripheral angiography and therapeutic endovascular intervention at the University of California, Davis Medical Center between 1 June 2006 and 31 December 2013. During this timeframe, four vascular surgeons and one interventional cardiologist performed the procedures. At the time of data extraction, 1400 patients and 2000 procedures were included in the overall registry.
Data collection and definitions
After approval by the Institutional Review Board at University of California, Davis Medical Center, baseline data were collected from review of the electronic medical record and procedure notes. Pre- and post-procedure clinical notes and the admission history and physical documentation were used to identify clinical presentation as well as post-procedure outcomes and medical management. This information was entered into a pre-specified case report form with standardized data entry, as previously described. 15
CLI was defined as Rutherford category 4–6 disease (defined as ischemic rest pain, minor tissue loss, or major tissue loss, respectively) based on review of clinic notes, history and physical examination, and hospital discharge summaries. In each case, the Rutherford classification and indication for the procedure were recorded by the treating cardiologist or vascular surgeon at the time of the procedure. Rutherford category 5 disease was classified as a non-healing lower extremity ulcer with minor tissue loss, while Rutherford category 6 disease was defined as major tissue loss extending beyond the transmetatarsal level. 16
ABI and toe–brachial index (TBI) measurements and duplex ultrasonography (DUS) were obtained in patients before endovascular treatment and at follow-up. A ncABI was defined as an ABI value >1.4, as measured at the posterior tibial and anterior tibial arteries; patients with an ABI ≤1.4 were considered to have a compressible ABI (cABI). Among patients with cABIs, 15/216 (7%) had ‘partially compressive’ ABI values of 0.9–1.4. A target lesion was defined as any continuous vascular segment of the lower extremity arterial tree (e.g. common iliac artery, common femoral artery, anterior tibial artery, etc) treated or attempted to be treated during the procedure. The target lesion was also defined as the treated segment, including 10 mm proximal and ending 10 mm distal to the index device or therapy (stent, balloon, or atherectomy catheter). Quantitative angiography was performed on all target lesions to evaluate the pre- and post-procedure percent diameter stenosis, lesion length, and reference vessel diameter. Chronic occlusions were defined as complete arterial occlusion with absence of antegrade flow. Acute technical success was defined as the achievement of a final residual diameter stenosis <30% for stent and <50% for angioplasty or atherectomy by angiography at the end of the procedure (and without a flow-limiting arterial dissection or hemodynamically significant translesional pressure gradient <10 mmHg) for endovascular revascularization. Acute procedural success was defined as both acute technical success and absence of major adverse events (e.g. death, stroke, myocardial infarction, acute onset of limb ischemia, index bypass graft or treated segment thrombosis, and/or need for urgent/emergent vascular surgery) within 72 hours of the index procedure.
Two cardiologists who were unaware of the ABI status of each patient reviewed all angiographic images to verify lesion morphology, calcification, and status of the distal run-off vessels. Patients were routinely seen 30 days after the revascularization procedure. The follow-up visit included an assessment of the patient’s clinical improvement as well as interval ABI, TBI, and DUS examinations. Further post-procedure follow-up was then conducted every 3 months during the first year post-procedure and every 6–12 months thereafter. Target lesion patency was evaluated by DUS at 0–30 days, 4–6 months, 9–12 months, and every 6 months thereafter. Major amputation was defined as any amputation above the level of the ankle joint. 17 A major adverse cardiovascular event was defined as documented death, stroke or myocardial infarction. These outcomes were verified based on follow-up clinic visits within the healthcare system. Death was verified based on follow-up clinic visits as well as review of the Social Security Death Index.
Statistical analysis
Mean values with standard deviation were used to describe continuous variables, and numerical values (percentages) were used for categorical variables. Continuous variables were compared using the Mann–Whitney test and categorical values were compared by the Fisher’s exact test. Freedom from major adverse cardiovascular events, amputation, or overall mortality was analyzed with the use of Kaplan–Meier survival estimates and the log-rank test. Univariate analysis was used to identify predictors of survival in patients with non-compressible ABIs. A multivariable Cox regression model was developed to assess the relationship between ncABIs and MACE. Patient variables known to be associated with mortality including coronary artery disease (CAD), chronic kidney disease, and dialysis were automatically forced into the model. Lesion-specific characteristics that increase the risk of MACE, including chronic occlusion, were also automatically included. Generalized estimating equations were used to account for patient clustering in situations of multiple target lesions per procedure.
The value for statistical significance was <0.05. Analyses were conducted using Stata statistical software, Release 11.2 (StataCorp LLC, College Station, TX, USA).
Results
During the study period, 284 patients with CLI underwent peripheral angiography with intervention to 329 limbs. Of these patients, 68 (24%) had ncABIs prior to angiography. Baseline demographics for the two groups are summarized in Table 1. Patients with ncABIs were more likely to have a history of CAD (65% vs 44%, p=0.003), diabetes (87% vs 57%, p<0.001), or end-stage renal disease (46% vs 12%, p<0.001). Patients with CLI and ncABI were also more likely to have tissue loss (Rutherford 5 and 6: 91% vs 80%, p=0.01). Baseline TBIs (0.20 ± 0.19 vs 0.20 ± 0.18, p=0.9) and absolute toe pressures (28 ± 26 vs 33 ± 31 mmHg, p=0.2) were similar between the two groups.
Demographic characteristics of patients presenting with critical limb ischemia.
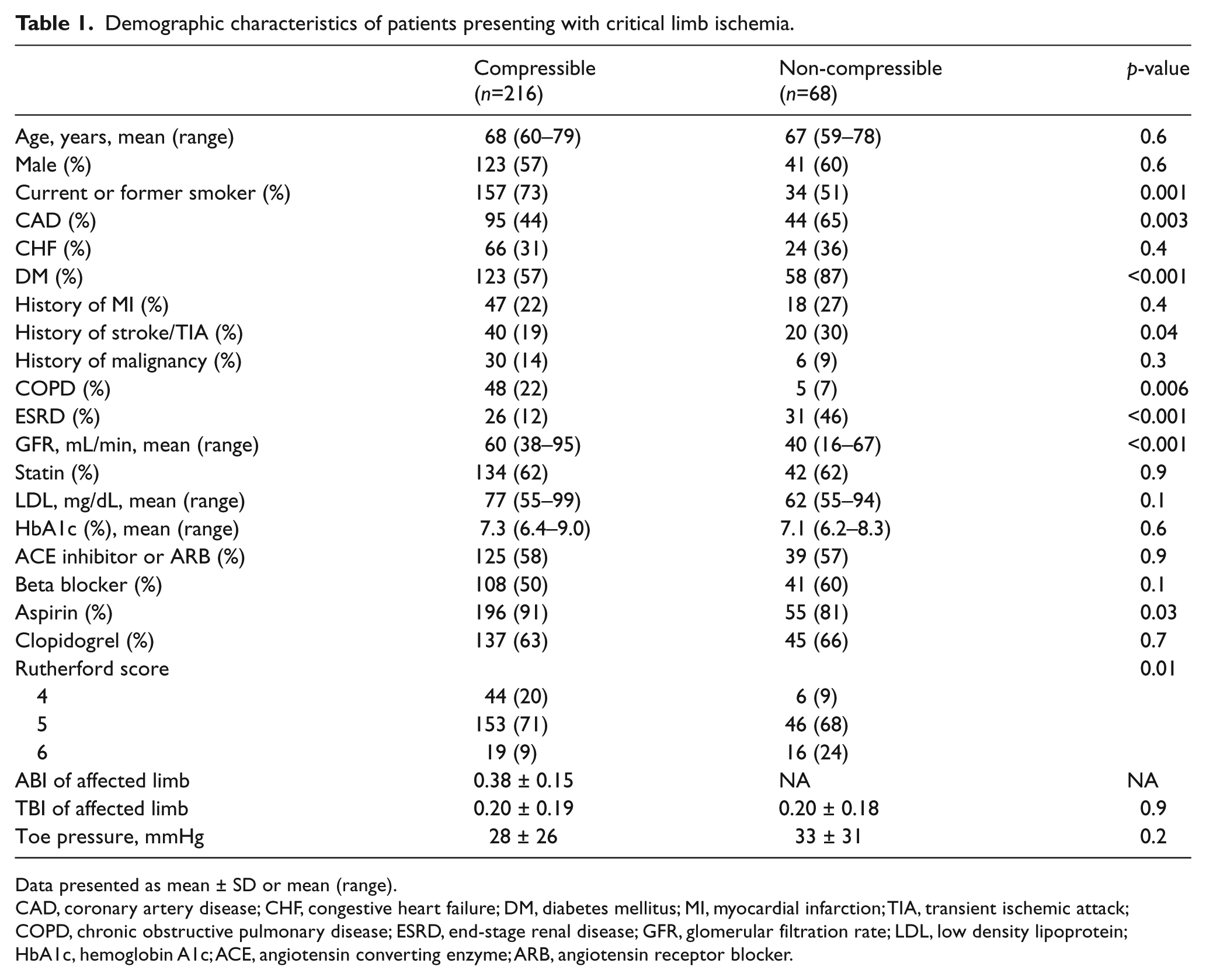
Data presented as mean ± SD or mean (range).
CAD, coronary artery disease; CHF, congestive heart failure; DM, diabetes mellitus; MI, myocardial infarction; TIA, transient ischemic attack; COPD, chronic obstructive pulmonary disease; ESRD, end-stage renal disease; GFR, glomerular filtration rate; LDL, low density lipoprotein; HbA1c, hemoglobin A1c; ACE, angiotensin converting enzyme; ARB, angiotensin receptor blocker.
Baseline angiographic characteristics of the two groups are depicted in Figure 1. Overall, the presence of multilevel disease (Figure 1A) was similar between the two groups (56% ncABI vs 65% cABI, p=0.2). However, patients with ncABIs were less likely to have significant aortoiliac (8% vs 28% cABI, p=0.004) or SFA/popliteal disease (62% vs 80% cABI, p=0.003). The frequency of infrapopliteal lesions was similar (87% ncABI vs 79% cABI, p=0.1). Chronic total occlusions (Figure 1B) were less common in ncABI patients within the iliac (2% vs 12% cABI, p=0.04) and SFA/popliteal segments (22% vs 52% cABI, p<0.001) but were noted with the same frequency in the infrapopliteal segments (87% vs 79% cABI, p=0.1). Finally, there was no significant difference in the lower extremity run-off (Figure 1C) between the two groups (p=0.3).
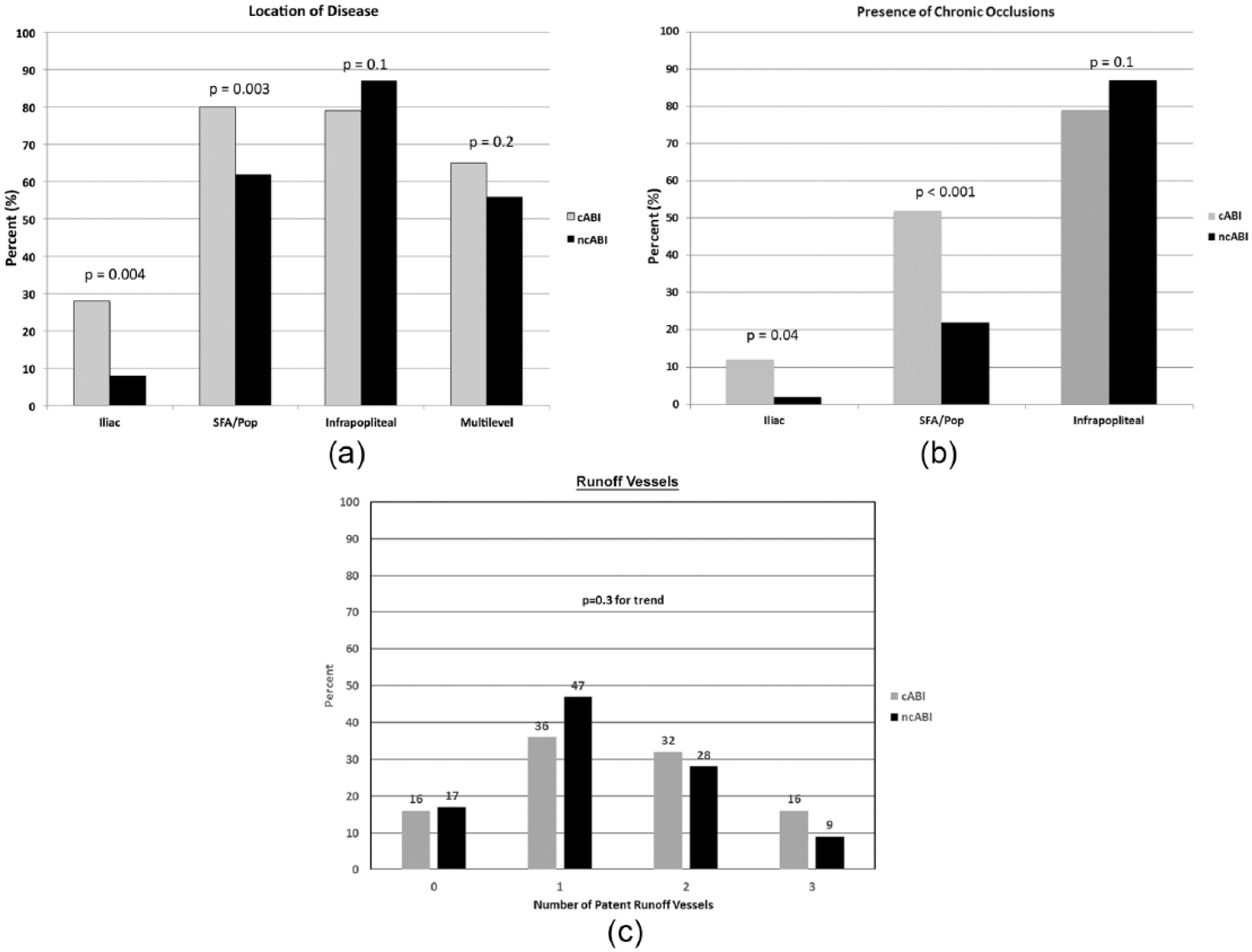
Location and severity of peripheral artery disease among patients with compressible and non-compressible ankle–brachial indices (ABIs). (A) Patients with compressible ABIs were more likely to have significant iliac stenosis and superficial femoral artery (SFA)/popliteal disease. The prevalence of infrapopliteal or multilevel disease was similar between groups. (B) Patients with compressible ABIs were more likely to have iliac or SFA/popliteal chronic total occlusions (CTOs). (C) Patients with compressible versus non-compressible ABIs had a similar number of runoff vessels.
Infrapopliteal target lesion-specific characteristics are provided in Table 2. Patients with ncABIs had a similar prevalence of infrapopliteal chronic occlusions, but longer target lesions (104 vs 92 mm, p=0.05), as well as smaller diameter target vessels (2.5 vs 3.1 mm, p=0.01). There was a significantly higher rate of intraprocedural flow-limiting dissection among patients with ncABIs (6% vs 0%, p=0.02), and a trend towards lower overall procedural success (82% vs 91%, p=0.08). These findings are likely consistent with the presence of dense calcification in the target lesions of patients with ncABIs. The overall patency at 6 months was not significantly different between the groups (60% vs 64%, p=0.3); the number of patients with dedicated duplex follow-up to 1 year was low, limiting analysis of long-term patency.
Angiographic and interventional characteristics of infrapopliteal lesions.
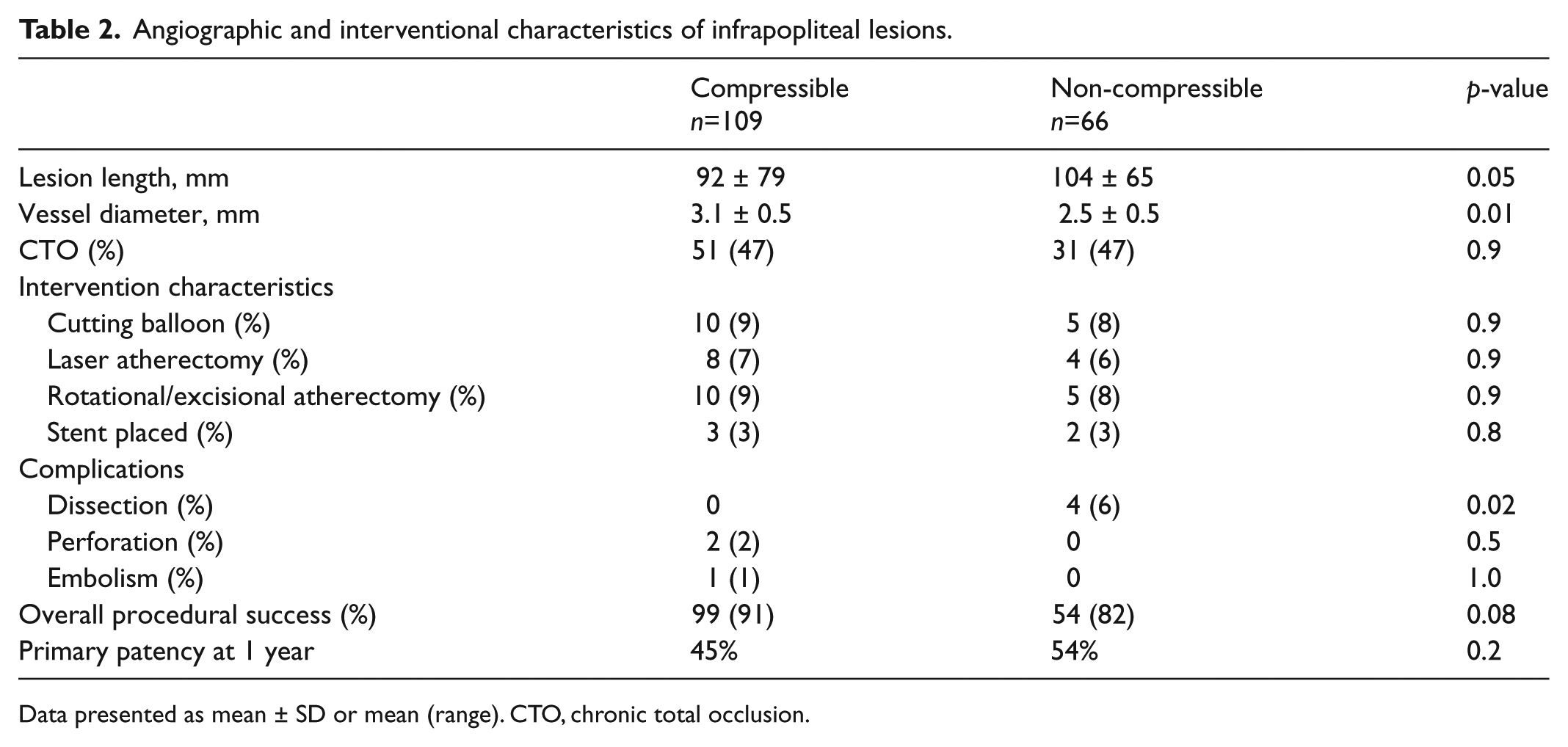
Data presented as mean ± SD or mean (range). CTO, chronic total occlusion.
Overall mortality, shown in Figure 2, was higher in the ncABI cohort at 3-year follow-up (49% vs 32%, hazard ratio (HR) for death 1.75, 95% CI 1.12–2.78, p=0.01). Similar findings were also noted for MACE (60% vs 36%, HR for MACE 2.04, 95% CI 1.35–3.03, p=0.001) (Figure 3). Major amputation rates (Figure 4) were also significantly elevated in patients with ncABIs (32% vs 17%, HR 1.96, 95% CI 1.11–3.45, p=0.02). The majority of the major amputation events occurred within the first year in both groups. Amputation-free survival (Figure 5) followed a similar pattern, with only 38% of ncABI patients surviving without amputation at 3 years versus 60% of patients with cABIs (HR 1.85, 95% CI 1.25–2.70, p=0.002).
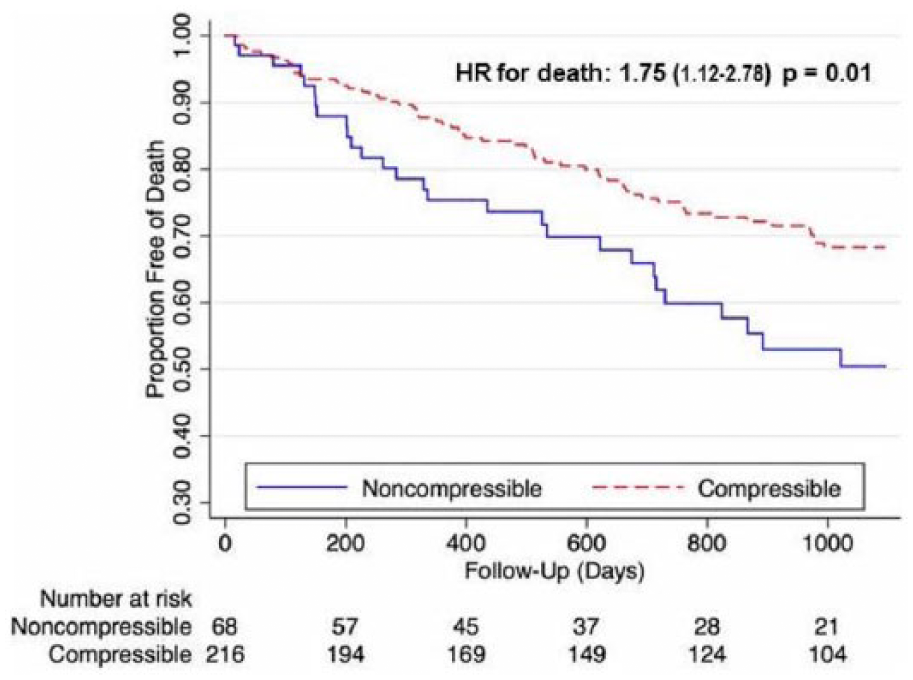
Overall mortality.
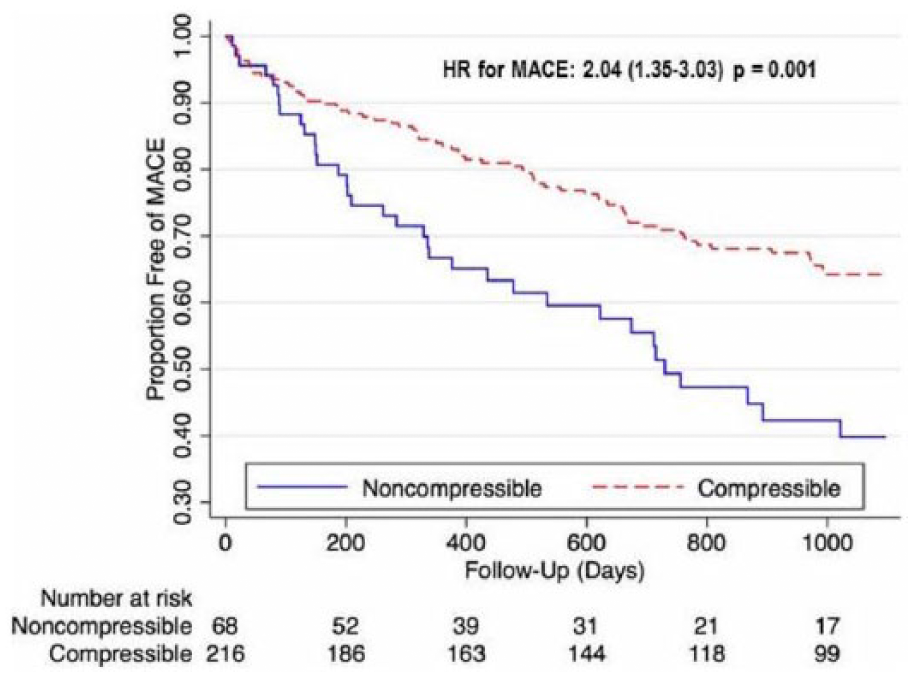
Freedom from major adverse cardiovascular events (MACE).
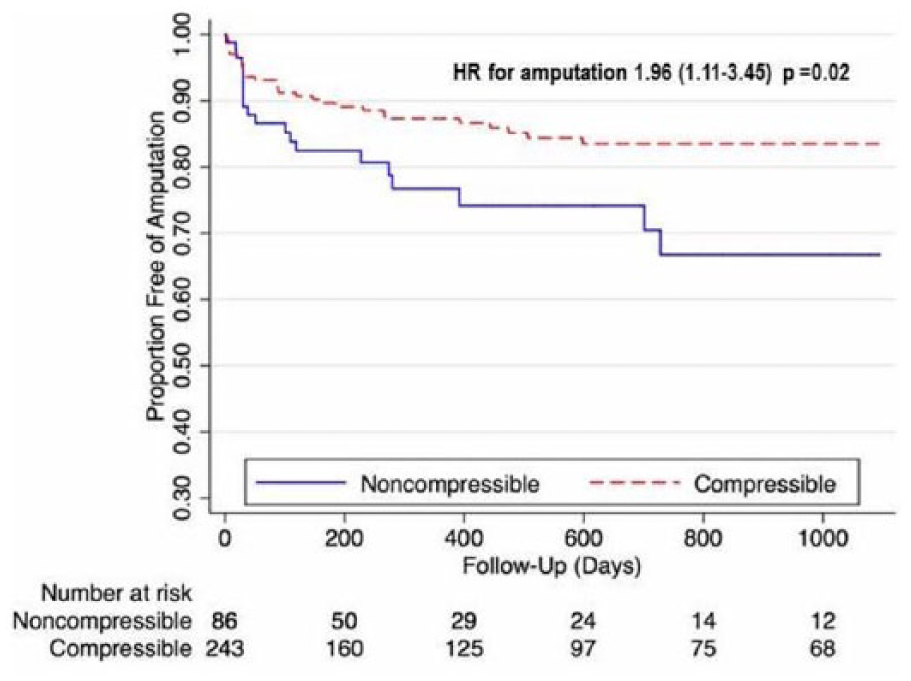
Freedom from major amputation.
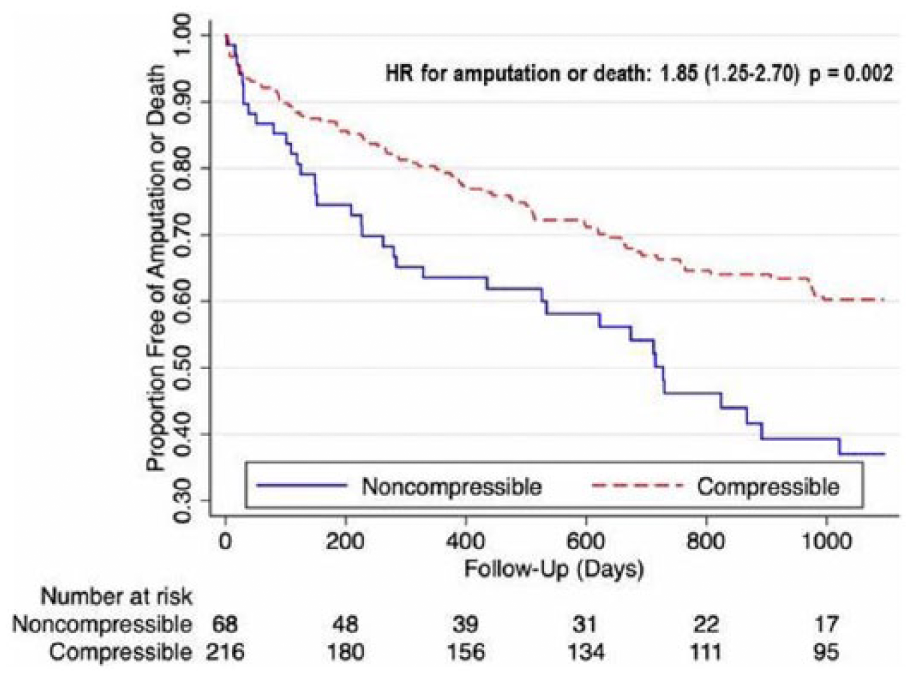
Freedom from major amputation or death.
Multivariable predictors of MACE were identified using a Cox proportional hazards model (Table 3). Unadjusted predictors of MACE included ncABI (HR 2.04; 95% CI: 1.35–3.03), history of CAD (HR 2.47; 95% CI: 1.7–3.6), glomerular filtration rate (HR 1.25; 95% CI: 1.1–1.3), and prior myocardial infarction (HR 1.56; 95% CI: 1.06–2.29). After multivariable adjustment, independent predictors of MACE included ncABI (HR 1.65; 95% CI: 1.02–2.70), history of CAD (HR 2.45; 95% CI: 1.52–3.95), glomerular filtration rate (HR 1.26; 95% CI: 1.11–1.31), and history of COPD (HR 2.17; 95% CI: 1.29–3.64).
Predictors of major adverse cardiovascular events (MACE).
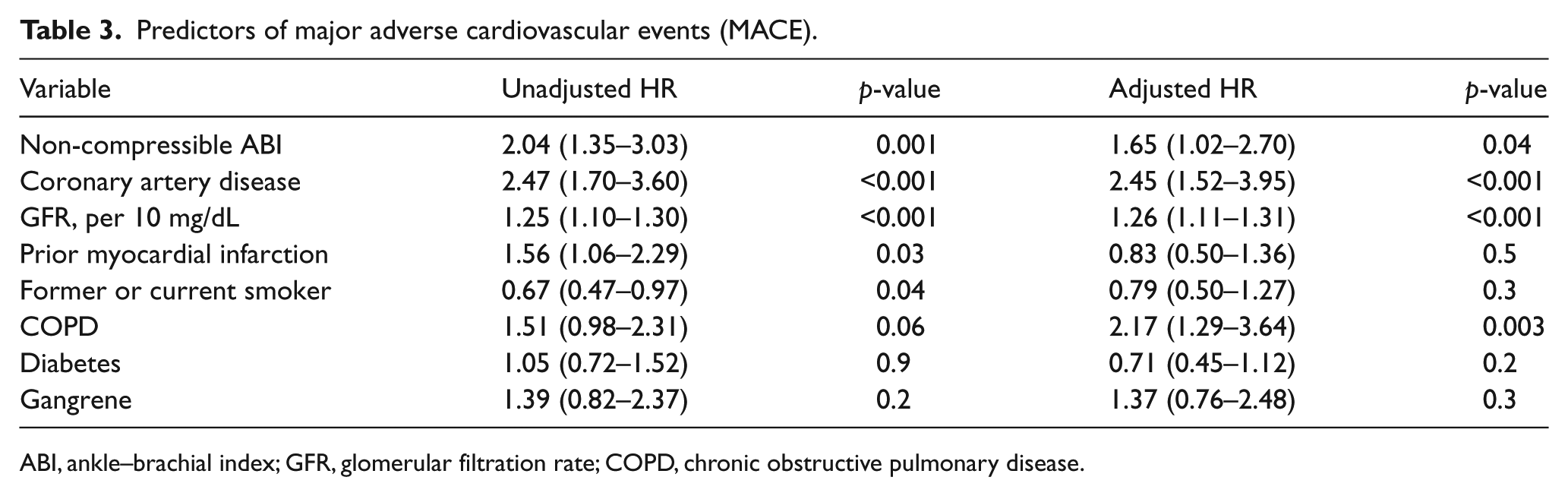
ABI, ankle–brachial index; GFR, glomerular filtration rate; COPD, chronic obstructive pulmonary disease.
Discussion
Non-compressible ABI are a frequent finding among patients with CLI, but little has been published on the presentation and outcomes of this important subgroup of patients. In this study, we identified many clinically significant findings among patients with ncABI and CLI. First, the presence of ncABI was associated with less inflow disease but a similar prevalence of infrapopliteal lesions that were on average longer and with smaller vessel diameters. Second, the presence of ncABI was associated with an increased rate of mortality and major amputation when compared to patients with CLI and cABIs. Finally, even after adjusting for known cardiovascular risk factors, the presence of ncABI remained a significant predictor for MACE and major amputation.
Non-compressible lower extremity arteries are pathologically related to the presence of medial artery calcification. 7 The presence of ncABI has been associated with accelerated atherosclerosis, diabetes, and chronic renal insufficiency 18 in varying populations. Our series focused solely on patients with CLI, with 87% of the ncABI patients having diabetes and 46% having ESRD. This demographic finding is notable, as ncABIs are typically associated with chronic kidney disease, but in our cohort >50% of the patients with ncABI did not have ESRD. The overall association with a higher prevalence of CAD likely reflects more advanced systemic atherosclerosis, and that patients with ncABI have a higher prevalence of polyvascular disease.
Despite the literature implicating these risk factors with ncABI, the angiographic pattern of atherosclerotic disease and relationship to adverse cardiac and vascular events is largely unknown. 19 Angiographically, patients with CLI and ncABI have a similar prevalence of infrapopliteal disease, but a lower prevalence of inflow (aortoiliac and femoropopliteal) disease when compared to those with cABI and CLI (Figure 1A). Patients with ncABI and CLI also had longer target lesions and smaller vessel diameters on average in the infrapopliteal segments. These findings have important technical and clinical implications. In the most recent guidelines, 6 the optimal revascularization strategy for patients with ncABI is not addressed. Owing to the relative lack of inflow disease in our series, endovascular intervention with a goal for limb salvage should be attempted in this high-risk population, especially given that these patients also have a higher prevalence of comorbidities that may make patients a higher risk for surgical bypass. 20 The increased rates of major amputation among patients with ncABI did not appear to be due to worse primary patency, although the rate of dedicated duplex follow-up in our cohort was low. Instead, the increased rates of major amputation may be related to a higher prevalence of Rutherford 5/6 disease, treatment of longer diseased segments, smaller vessel diameters, and a greater proportion of patients with diabetes and ESRD.
This study also identified two important contributors to the higher risk of overall mortality in patients with CLI and ncABI: increased major amputation and cardiovascular event rates. CLI patients represent the highest risk PAD patients and in this population, overall mortality, cardiovascular mortality and amputation rates are interrelated. It is well known that presence of symptomatic PAD carries an increased risk for death, cardiovascular events, and limb loss. 21 In a recent large series of approximately 40,000 patients with PAD, 3-year mortality and amputation rates in Rutherford 5/6 patients ranged from 40% to 60% and 20% to 60%, respectively. 22 The authors also reported 1-year amputation rates of 20% and 50% in Rutherford 5 and 6 patients, respectively. In the present series, the majority of the amputations occurred within the first year, underscoring the need to aggressively treat patients with CLI. These findings are not entirely dissimilar from the TASC II data, which report a 30% amputation rate in patients with CLI at 1 year. 6 The current investigation reports an overall 1-year amputation rate of 17% and 3-year amputation rates of 25%. The present series represents contemporary endovascular approaches, which may partly explain the improved limb salvage rates. While the overall amputation rates in our cohort are lower compared to historical controls for the aforementioned reasons, the presence of ncABI nonetheless carries a substantial increased risk for major amputation (HR of 1.96 relative to cABIs). Whether aggressively treating risk factors or approaching lesions for successful revascularization changes, these outcomes in patients with ncABIs are not known and should be a future direction for research.
One of the more striking findings in our study was the 50% observed 3-year mortality in patients with ncABI, versus 30% in those with cABI. Several prior population-based studies have evaluated the association between ncABIs and adverse outcomes in the overall population of patients with PAD, and have reported mortality rates ranging from 30% to 60%.23–25 However, the applicability of these broad data sets to the patients with CLI is uncertain. In a large study of 16,000 patients referred for outpatient lower extremity vascular duplex, the prevalence of ncABIs was noted to be 17%. 25 Only 37% of these patients carried a diagnosis of CLI, with an overall survival of approximately 70% at 3 years and 55% at the median follow-up period of 6 years. 25 The authors noted a HR for death of 1.3 when compared to patients with PAD and cABIs. In the remaining three published series,26–28 the mortality HR for patients with ncABIs was similar to those with cABIs. However, all of those studies used different criteria to define PAD, cABI, and ncABI, and had a heterogeneous population of patients with asymptomatic PAD, PAD with cABI, and PAD with ncABI. The current study reports findings exclusively in CLI patients.
In unadjusted analysis, the presence of ncABI was associated with a twofold increased risk for MACE. Even when adjusting for confounding variables, such as presence of CAD, renal dysfunction, and history of smoking, the presence of ncABI carried a 1.5-fold increased risk for MACE. As noted, cardiovascular outcomes in ncABI patients in prior investigations have comprised of a mixed population, with not all having symptomatic PAD, making direct comparison difficult. Amini et al. 29 reported on a series of PAD patients with ncABIs and noted high rates of MACE (33%) at a mean follow-up of 2 years. That series noted higher MACE rates in the ncABI cohort when compared to those with cABIs. The higher MACE rates in our and their series is likely driven by the higher presence of comorbidities including presence of CAD, diabetes, history of stroke or TIA, and renal dysfunction. In additional to poor angiographic targets in patients with ncABIs (long diffuse lesions with small vessel diameters), these additional comorbidities (e.g. diabetes, end-stage renal disease) may make patients a higher risk for surgical revascularization. To date, no large series has systematically evaluated outcomes in ncABI patients undergoing surgical revascularization. Hence, implementation of medical therapy and consideration for endovascular revascularization is of utmost importance for patients with ncABIs.
Limitations
There are several limitations to our study. This is a single center, retrospective series with the inherent bias of incomplete data sets and reporting bias. Second, this study includes results from CLI patients who underwent diagnostic angiography and revascularization. Hence, the findings of the present study may not be applicable to the larger population with ncABI, and the cohort is biased towards patients fit to undergo lower extremity angiography. However, the focus on CLI exclusively may help with patient risk stratification and optimal treatment. Third, the multivariable adjustment did not include lesion-related characteristics; it is possible that some of the independent association of ncABIs is associated with specific lesion characteristics. Fourth, our population was dichotomized between cABI and ncABI, whereas the actual risk likely represents a spectrum. Recent studies have suggested that a significant percentage of patients may have minimally decreased or ‘normal’ ABIs and have an ischemic ulcer. This is especially true in the presence of an incomplete pedal arch, where the angiosome of the wound may be the major determinant of wound healing. Fifth, we do not have information on functional status or patient-reported quality of life, which are potentially additional predictors of patient outcomes.
Conclusion
The presence of ncABI represents a subset of patients with CLI at elevated risk for mortality, major amputation, and adverse cardiovascular event rates. Whether the presence of ncABI is a surrogate to the elevated risk or a direct contributor is still not known. Additionally, whether targeted medical therapy towards associated medical comorbidities or specific endovascular intervention has any effect on outcomes should be the focus of ongoing research.
Footnotes
Declaration of conflicting interests
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: John R Laird is a consultant or advisory board member for: Bard Peripheral Vascular, Boston Scientific, Medtronic, and Abbott Vascular. He receives research support from WL Gore. Ehrin J Armstrong is a consultant or advisory board member for Abbott Vascular, Boston Scientific, Cardiovascular Systems, Medtronic, and Spectranetics.
All other authors declare no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
