Abstract
Background:
Peripheral artery disease (PAD) affects 200 million people worldwide and is associated with impaired quality of life, increased morbidity, and mortality. Supervised exercise therapy (SET) and lower-extremity revascularization (LER) are both proven strategies to improve patient symptoms. Short and long-term functional outcomes after LER for symptomatic PAD in a large, international cohort have not previously been described.
Methods:
The VOYAGER PAD trial (ClinicalTrials.gov identifier: NCT02504216) enrolled subjects after LER for symptomatic PAD (Rutherford category 2–6). Participants completed the Walking Impairment Questionnaire (WIQ) at baseline, 1, 3 and 6 months, and every 6 months thereafter. The primary outcome analysis was degree of difficulty walking two blocks at each of the aforementioned time points. Difficulty walking three blocks and climbing one flight of stairs at these time points was also analyzed. Data about supervised and home exercise therapy before or after revascularization were not collected in the VOYAGER PAD trial.
Results:
Of the 5614 VOYAGER PAD participants completing the WIQ at baseline, three-quarters presented with claudication and one-quarter with critical limb ischemia. Of these, the majority (62% with claudication and 74% with CLI) reported inability or much difficulty walking two blocks prior to LER. Walking improved after LER regardless of revascularization strategy, but one-fifth with claudication and one-third with CLI reported continued inability or much difficulty walking two blocks 1 month after LER. Participants who reported improved walking ability 1 month after LER experienced a durable functional result out to 3 years. Although the proportion of participants reporting significant baseline difficulty climbing one flight of stairs or walking three blocks differed, the trend in immediate and sustained improvement after LER was similar to that observed for walking two blocks.
Conclusion:
In this large, international cohort undergoing LER for symptomatic PAD, nearly two-thirds reported inability or much difficulty walking two blocks at baseline. Although many participants reported improved walking ability after LER, a substantial proportion remained severely disabled. These observations may help motivate providers, patients, and medical systems to improve awareness and engagement in SET referral after LER.
Keywords
Introduction
Peripheral artery disease (PAD) affects more than 200 million individuals worldwide, 1 including 8.5 million Americans above the age of 40, 2 causes functional impairment, 3 and is associated with increased morbidity and mortality. 4 Previous reports have demonstrated improved walking function after vascular surgery, percutaneous revascularization, and supervised exercise therapy (SET), although many of these studies included a small number of participants and limited observations to 12 months or less.5–10 More recent research has shown that management with both structured exercise training and lower-extremity revascularization (LER) improves clinical outcomes to a greater extent than either strategy alone and may decrease or delay the need for subsequent revascularization. 11 Despite the strong evidence and guideline recommendations, 12 there is continued under-utilization of structured exercise training in the United States. 13
In this context, better characterizing the immediate effect and long-term durability of LER on walking ability in patients with PAD could help inform providers and patients about appropriate expectations for symptomatic improvement after these procedures. To address this gap, we assessed the impact of LER on walking ability in the VOYAGER PAD (Vascular Outcomes studY of ASA alonG with rivaroxaban in Endovascular or surgical limb Revascularization for Peripheral Artery Disease) study using the Walking Impairment Questionnaire (WIQ), a validated patient-reported functional assessment instrument developed by Dr Hiatt and colleagues. 14
Methods
The VOYAGER PAD trial (ClinicalTrials.gov identifier: NCT02504216) was an international, multicenter, randomized, double-blind, placebo-controlled, event-driven phase 3 study conducted at 542 study sites in 34 countries for which primary results have been previously reported. 15 The protocol was designed and overseen by a collaborative group that included physicians at the Colorado Prevention Center (CPC Clinical Research, an Academic Research Organization affiliated with the University of Colorado), an academic executive committee, and the sponsors – Bayer and Janssen Pharmaceuticals. The protocol was approved by local ethics committees and all participants provided informed consent. CPC Clinical Research independently conducted analyses for this publication.
Study population
Between August 2015 and January 2018, 6564 patients were randomized (1:1) to rivaroxaban 2.5 mg twice daily or placebo on top of aspirin 100 mg daily; median follow-up was 28 months. Enrolled subjects had symptomatic PAD (Rutherford category 2–6) and had undergone a technically successful LER procedure no more than 10 days prior to randomization. 16 Participants were required to have undergone a technically successful revascularization procedure to qualify for the study; the choice of revascularization procedure was at the discretion of each participant’s clinical team. VOYAGER PAD qualifying revascularization procedures included endovascular balloon angioplasty and/or stenting with or without drug-coated products, surgical endarterectomy, bypass with venous or prosthetic conduits, and hybrid procedures; anatomic targets ranged from the pelvis to foot. 16 Data regarding supervised and home exercise therapy either before or after LER were not collected in the trial.
Walking Impairment Questionnaire (WIQ)
Walking ability was assessed at baseline, 1, 3, and 6 months, and every 6 months thereafter using the WIQ, a validated tool to assess the improvement or deterioration in the daily walking ability of patients with PAD.13,17–19 Although patients may have completed the baseline questionnaire temporally after LER occurred, they were instructed to answer the questions based on pre-revascularization walking function and symptoms. Three questions from the WIQ were included in this analysis: Report the degree of physical difficulty that best describes how hard it is for you to walk on level ground without stopping to rest:
for 600 feet (two blocks) for 900 feet (three blocks) Report the degree of physical difficulty required for you to climb one flight of stairs without stopping to rest.
Responses included ‘unable’, ‘much difficulty’, ‘some difficulty’, ‘slight difficulty’, and ‘no difficulty’, which corresponded to scores of 0–4, respectively. WIQ data were collected prospectively as a prespecified exploratory efficacy endpoint.
Analysis
The WIQ analysis dataset included all randomized patients in VOYAGER PAD with the WIQ completed within 7 days of qualifying LER (baseline) and at least once during follow-up. Participants were categorized into one of two mutually exclusive symptomatic groups: claudication or critical limb ischemia (CLI). Claudication was defined as Rutherford stages 2–3; CLI as stages 4–6. Categorical variables are reported as count (percentage), and continuous variables reported as mean (SD). Comparisons of baseline characteristics were by Wilcoxon rank-sum tests for continuous variables and Fisher’s exact tests for categorical variables. WIQ responses were tabulated at longitudinal time points and were presented by revascularization strategy (surgical/hybrid vs endovascular). Although subjects were required to have undergone a revascularization procedure in order to qualify for the study, the choice of revascularization procedure was at the discretion of the subject’s clinical team. The statistical software used for the analysis was SAS 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Baseline and follow-up WIQ were recorded for 5614 VOYAGER PAD subjects, representing 86% of the randomized population. Of those, 2027 (36%) had qualifying surgical procedures and 3587 (64%) had endovascular procedures. All were to receive low-dose aspirin per protocol; approximately 80% received statins and two-thirds angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blockers (ARBs). At baseline, subjects with qualifying surgical procedures were more often male, White, and residents of Eastern Europe with lower prevalence of diabetes and hyperlipidemia compared to those with endovascular procedures (Table 1). Subjects with surgical revascularization had more severe PAD than those with endovascular procedures as evidenced by a lower ankle–brachial index (ABI), higher Rutherford stage, and a higher proportion with a history of CLI (Table 1).
Characteristics of VOYAGER PAD subjects completing the Walking Impairment Questionnaire targeting pre-lower-extremity revascularization symptoms.
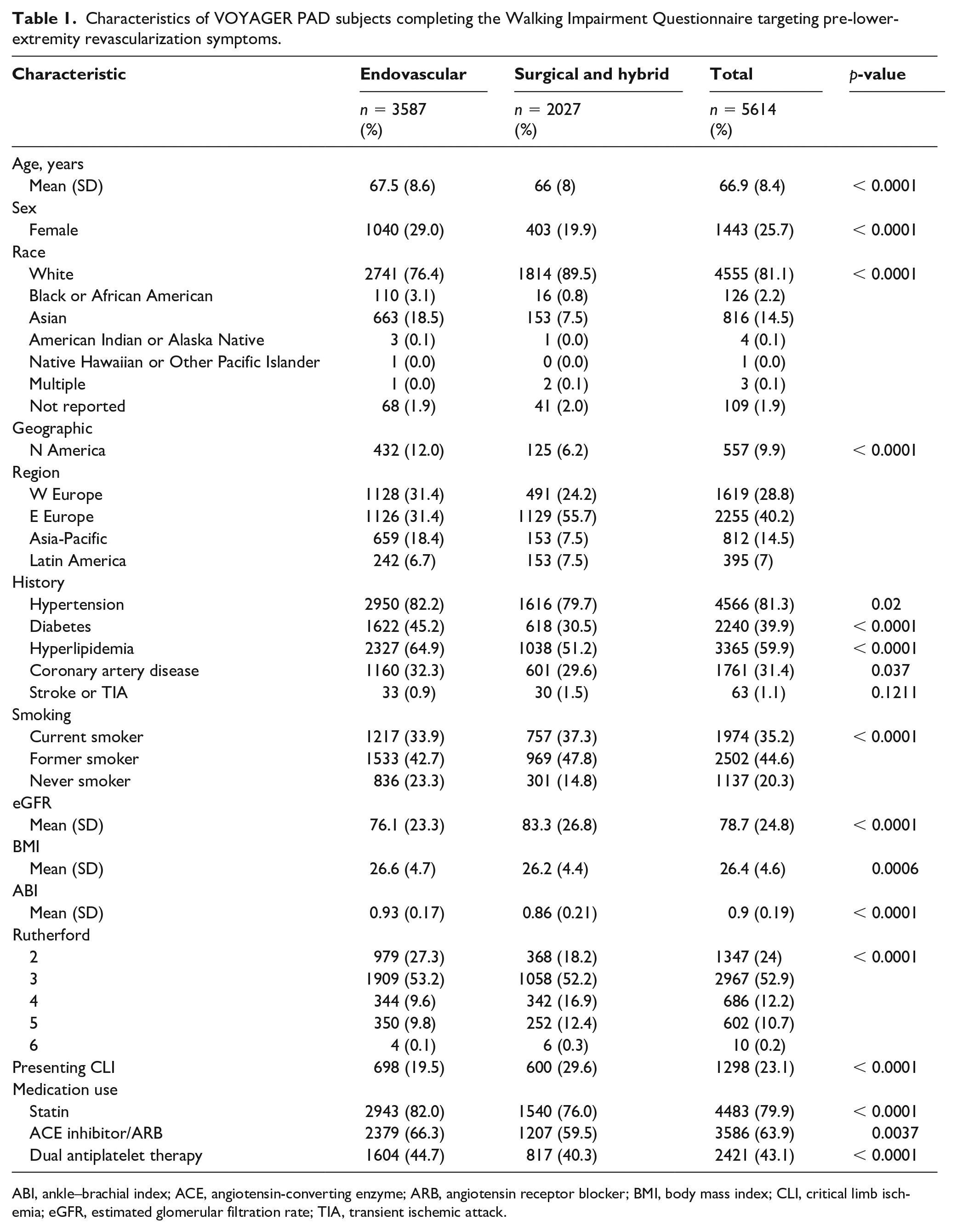
ABI, ankle–brachial index; ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; BMI, body mass index; CLI, critical limb ischemia; eGFR, estimated glomerular filtration rate; TIA, transient ischemic attack.
Approximately half of the participants presented with Rutherford category 3 symptoms, and approximately one in five had CLI (Table 1). Patients with surgical index LER reported greater functional disability walking two or three blocks or climbing one flight of stairs at baseline (Figure 1). As expected, the degree of disability was greater in those presenting with CLI – prior to revascularization 74% reported inability or much difficulty walking two blocks, compared to 62% in the claudication group (Figure 2).
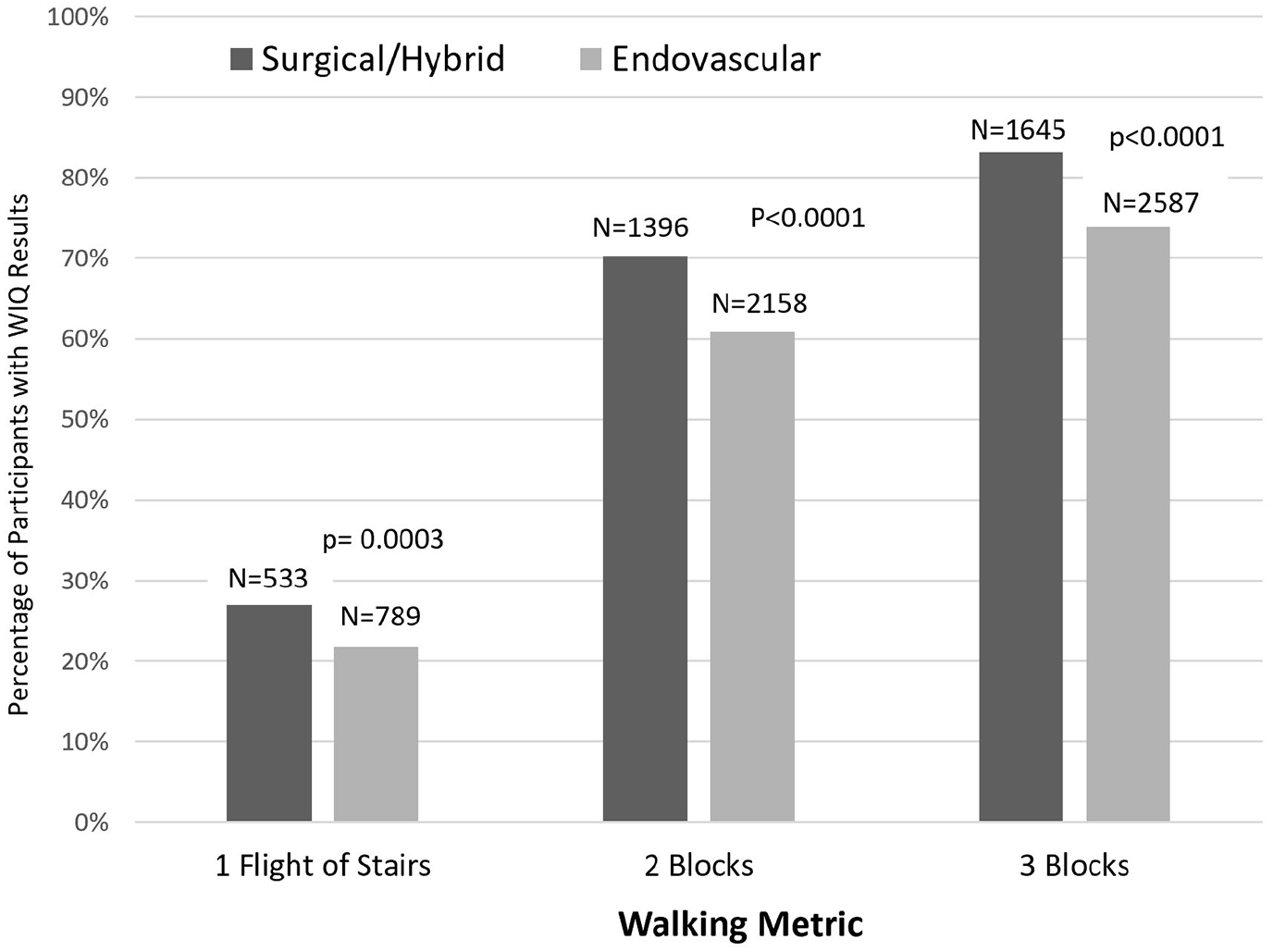
Percentage of participants who were ‘Unable’ or ‘Able with much difficulty’ to walk the designated metric before lower-extremity revascularization, presented by revascularization strategy.
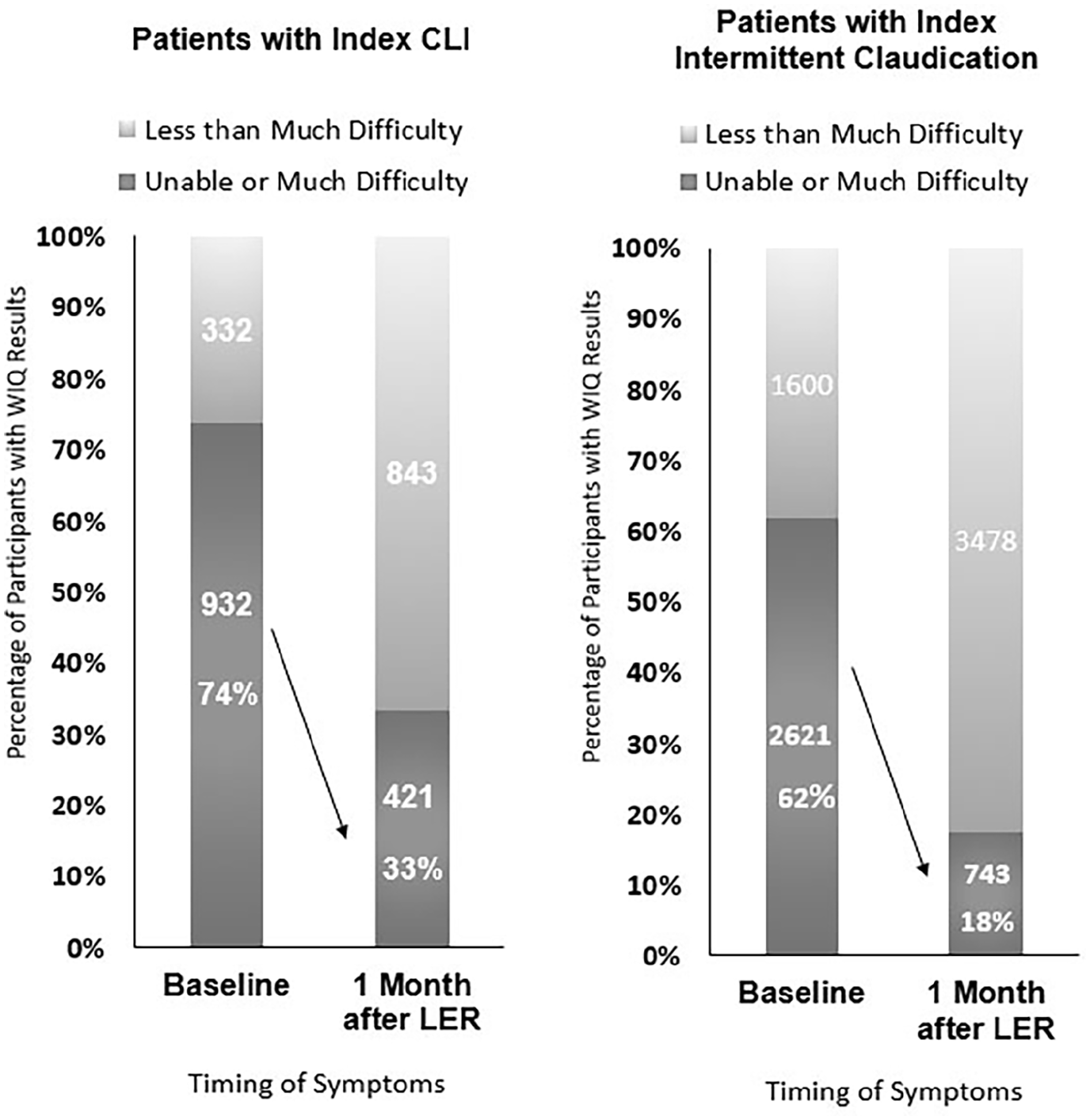
Proportion of patients with inability or much difficulty walking two blocks on the Walking Impairment Questionnaire before and 1 month after revascularization.
The surgical and endovascular groups both reported improved walking function for all metrics 1 month after LER, and these results were durable out to 3 years (Figure 3). Most subjects with significant difficulty walking two blocks at baseline reported functional improvement 1 month after LER, although this was less frequently observed among those with CLI (Figure 2). Functional impairment persisted in a notable proportion of subjects: one in five claudicants and one in three with CLI continued to have much difficulty or inability walking two blocks after LER (Figure 2). Although the proportion of participants reporting significant baseline difficulty climbing one flight of stairs or walking three blocks differed, the trend in immediate and sustained improvement after LER was similar to that observed for walking two blocks (Figure 3).
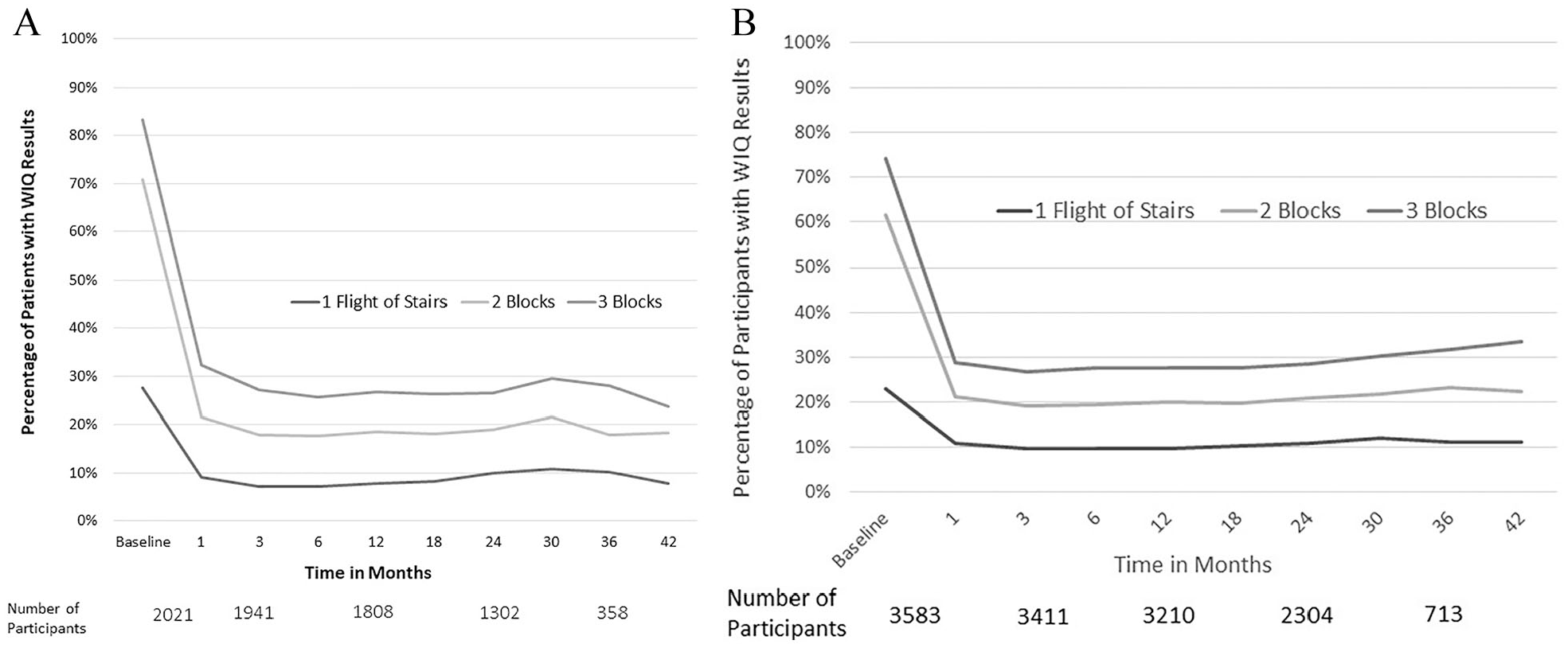
Percentage of participants ‘Unable’ or ‘Able with much difficulty’ to climb one flight of stairs (bottom line), or walk two blocks (middle line) or three blocks (top line) before and at indicated time points after index revascularization.
Discussion
The current analysis provides several insights with respect to walking function in patients undergoing LER for symptomatic PAD, with long-term follow-up in a multinational cohort receiving contemporary medical therapy and using a validated patient-reported outcome measurement. First, patients undergoing LER for claudication and CLI have important walking functional impairment. Second, LER is associated with real functional benefit after both surgical and endovascular procedures. Third, a large proportion of patients continue to have significant disability after LER; in these patients, SET may provide even more important adjunctive benefit.
Current professional guidelines endorse LER to prevent tissue loss in CLI as a Class I indication, but LER for claudication receives a lesser recommendation level and is primarily performed for symptomatic functional improvement.2,7 However, the threshold of walking dysfunction claudicants should have to justify LER remains a topic of debate. In this international cohort of symptomatic patients with PAD undergoing LER, three-quarters underwent revascularization for claudication, and patients were significantly disabled – approximately two-thirds reported inability or much difficulty walking two blocks. This observation underscores the severe individual and collective disability associated with symptomatic PAD.
The current analysis observed meaningfully improved walking ability following LER in patients regardless of indication (CLI or claudication) or revascularization approach. Furthermore, this benefit appeared to be durable – beyond 3 years for some participants. These data are in line with the plethora of previous evidence showing improved short-term functional outcomes after LER,5,7–9 and markedly add to the limited body of evidence regarding functional outcomes beyond 12–18 months.
These observations, however, must be tempered by the substantial proportion of patients who remained disabled after LER, most of whom were on appropriate medical therapy: all patients received aspirin, nearly 80% of patients received statin therapy, more than 40% received dual anti-platelet therapy, and approximately two-thirds received ACE inhibitor or angiotensin II receptor blocker (ARB) therapy. The 20–30% of participants who reported continued, severe limitations walking two blocks after LER remain a population in need of other effective management strategies to improve function.
There are notable limitations of this analysis. First, because the data were obtained as an exploratory endpoint – participants were not randomized to study walking outcomes— the results are hypothesis-generating only. Second, a functional capacity test was not performed, which would have provided more accurate data on claudication distance and peak walking ability. Third, information on supervised and home exercise therapy (before and after LER) was not collected and may have been an important confounding factor. Fourth, we could not control for walking limitations that may have been the result of other (nonvascular claudication) physical ailments (e.g., musculoskeletal problems, heart failure, chronic obstructive pulmonary disease, angina, etc.).
SET remains the most effective nonprocedural intervention for improving function in patients with PAD. Despite numerous confirmatory trials and a grade 1A recommendation as first-line therapy for PAD claudication by the major US vascular surgery and cardiology societies,2,20,21 and recent changes in reimbursement in the United States, SET remains grossly underutilized. 22 Factors contributing to poor utilization exist at the provider, patient, and system levels. 22 Specific patient-cited issues for quitting supervised exercise programs include lack of motivation, health problems, and perceived lack of claudication improvement during program participation. 23 Providers have cited the lack of SET center availability and concerns about costs and travel incurred by patients as major reasons for poor SET referral. 13 From a system level, the Centers for Medicare and Medicaid only reimburse facility fees and not physician fees or relative value units (RVUs), resulting in less sustainability for systems to build and invest in SET facilities and programs. 13 Recognition of the residual disability as described in the current paper serves as a call to action for broader and systematic use of SET overall in PAD and also as an adjunct to LER, as there are strong data that combined SET and LER results in the greatest functional benefit. 13 Programs to systematize use of cardiac rehabilitation in coronary populations may serve as a model.
Three decades have passed since Dr Will Hiatt and colleagues developed the WIQ, reported the correlation of WIQ scores with graded treadmill exercise performance, and demonstrated that structured exercise training significantly improved walking performance and claudication symptoms in patients with symptomatic PAD.6,13,17 This call to action serves in the legacy of Dr Hiatt’s and many others’ work in the field of vascular disease to improve PAD care through integrated care and intensive use of lifestyle, exercise, medication, and appropriate revascularization. It also serves as a reminder that strategies such as SET and revascularization are not competing but are complementary and that many patients with PAD will need both to optimize their functional status.
Footnotes
Acknowledgements
This work would not have been possible without Dr William Hiatt’s pioneering contributions to the medical community’s knowledge of PAD pathophysiology and management. Thank you, Dr Hiatt—you are remembered and greatly missed.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The following authors receive salary support from CPC through funds transferred to the University of Colorado: Dr Hogan, Dr Jackson, Dr Nehler, Dr Hess, Dr Berkowitz, Dr Hsia, and Dr Bonaca. CPC, a nonprofit academic research organization affiliated with the University of Colorado, receives research funding from: Abbott, Agios, Alexion Pharma, Alnylam, Amgen, Angionetics, ARCA Biopharma, Array, AstraZeneca, Atentiv, Audentes, Bayer, Better Therapeutics, Brigham and Women’s Hospital, Bristol Myers Squibb, Cardiol Therapeutics, CellResearch, Cook Medical, Cook, CSL Behring, Eidos Therapeutics, EP Trading Co, Esperion Therapeutics, EverlyWell, Faraday, Fortress Biotech, DHL Therapeutics, HeartFlow, Hummingbird Bioscience, Insmed, Janssen, Kowa Research, Lexicon, Merk, Medtronic, Moderna, Novate Medical, Novo Nordisk, Pfizer, PhasBio, PPD Development, Prarie Education and Research, Prothena Ciosciences, Regeneron, Regio Biosciences, Sanafit Therapeutics, Sanofi, Smith and Nephew, Stealth Bio-Therapeutics, University of Colorado, Worldwide Clinical Trials, Wraser, Yale Cardiovascular Research Group. Dr Buchanan reports no disclosures. Dr King receives support from the American Heart Association Strategically Focused Research Network in Vascular Disease under award number 18SFRN3390085 (BWH-DH SFRN Center). Dr Anand reports receiving lecture fees from Bayer and Janssen. Dr Patel reports receiving grant support, advisory board fees, and consulting fees from AstraZeneca, Bayer, and Janssen, grant support from Medtronic and Philips Healthcare, and grant support and advisory board fees from HeartFlow. Dr Debus reports receiving grant support from Cook and Terumo Aortic. Dr Muehlhofer reports being employed by Bayer. Dr Haskell reports being employed full time by Janssen Pharmaceuticals, LLC. Dr Bauersachs reports receiving consulting fees and lecture fees from Bayer, Bristol Myers Squibb, and Pfizer. Dr Berkowitz was employed as a clinical research physician by Bayer at the time VOYAGER PAD was performed. Dr Bonaca is the Executive Director of CPC (see above) and receives grant support from Amgen, AstraZeneca, Merck, Novo Nordisk, Pfizer, and Sanofi and support from the AHA SFRN under award numbers 18SFRN3390085 (BWH-DH SFRN Center) and 18SFRN33960262 (BWH-DH Clinical Project). Dr Bonaca also reports stock in Medtronic and Pfizer and consulting fees from Audentes.
Funding
The VOYAGER PAD trial was supported by a grant from Bayer AG and Janssen Research & Development, LLC. Dr Anand receives lecture fees from Bayer. Dr Patel receives grant support, advisory board fees, and consulting fees from Bayer. Dr Muehlhofer is employed by Bayer. Dr Bauersachs receives consulting fees and lecture fees from Bayer. Dr Berkowitz was employed as a clinical research physician by Bayer at the time VOYAGER PAD was performed.
