Abstract

Peripheral artery disease (PAD), stroke, and coronary artery disease are the main clinical manifestations of atherothrombosis. 1 Recent reports indicate a high prevalence of PAD in patients with acute ischemic stroke, with prevalence estimates ranging from 31% to 51%.1,2 Additionally, PAD is also associated with an increased all-cause mortality and an increased risk of cardiac events in patients with ischemic stroke.3–5 This study aims to assess the prevalence of PAD among our patients hospitalized for ischemic stroke and to evaluate the common risk factors associated with recurrent stroke.
During the period January 2010 to December 2015, 2340 patients with ischemic stroke were consecutively admitted to the stroke unit in Ege University, Izmir, which is a center where tertiary care treatments are provided. A total of 2030 eligible patients fulfilling the inclusion criteria (age > 50 years, patient legally competent and able to cooperate appropriately, and written informed consent provided) were recruited. Primary stroke was diagnosed by two senior clinicians (EK and FEÇ) as any neurological deficit, persisting for more than 24 hours, and a lesion on computed tomography (CT) or magnetic resonance imaging (MRI). The study was approved by Ege University institutional review board and ethics review committee, and written informed consent was obtained from all participants.
Baseline examinations included a questionnaire, demographic characteristics, ankle–brachial index (ABI) measurement, and vascular risk factors. ABI was calculated according to the American Heart Association (AHA) recommendations. 6 Symptomatic PAD was defined as definitive or atypical claudication, ABI < 0.9, and/or a history of peripheral vascular revascularization, and amputation due to PAD, according to the Edinburgh questionnaire. 7 Patients without available ABI were not included in the analysis of symptomatic PAD. Verbal information was obtained from patients without aphasia, but information about complaints and pain from aphasic patients was obtained from their relatives. The primary outcome measure was stroke recurrence and the secondary outcome measure was cardiac events and vascular death at 5 years. Recurrent stroke was diagnosed as any new clinical neurological deficit after the index stroke, persisting for more than 24 hours, and a new lesion on CT or MRI. Cardiac events were recorded over the study period if they had at least one of the following: myocardial infarction, unstable angina, percutaneous coronary intervention, or coronary artery bypass graft surgery.
Univariate and multivariate Cox regression analyses were performed, and the corresponding hazard ratios (HR, and their 95% CI) were calculated to assess associations between PAD (and other risk factors) and 5-year stroke recurrence. A secondary outcome including all cardiac events and death was also analyzed.
A total of 2030 patients aged 50 years or older were included in the analyses. Table 1 shows the baseline characteristics of 1712 patients without PAD (84.3%) and 318 patients with symptomatic PAD (15.7%).
Baseline clinical characteristics in patients with and without PAD.
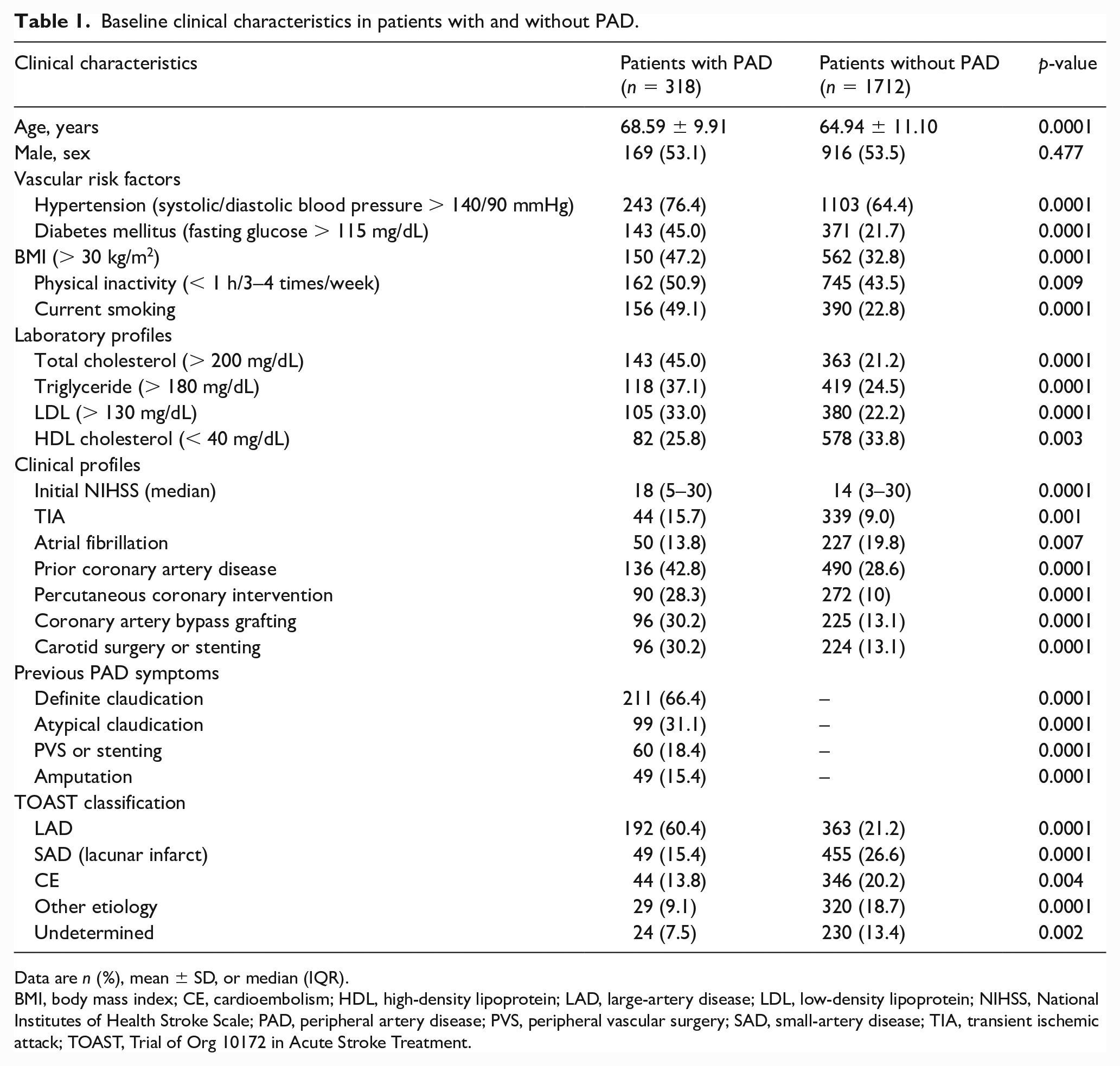
Data are n (%), mean ± SD, or median (IQR).
BMI, body mass index; CE, cardioembolism; HDL, high-density lipoprotein; LAD, large-artery disease; LDL, low-density lipoprotein; NIHSS, National Institutes of Health Stroke Scale; PAD, peripheral artery disease; PVS, peripheral vascular surgery; SAD, small-artery disease; TIA, transient ischemic attack; TOAST, Trial of Org 10172 in Acute Stroke Treatment.
During the 5-year follow-up, 102 patients had a stroke in patients with PAD (32.1%), and 348 without PAD (20.3%) had a recurrent stroke (HR, 1.85; 95% CI 1.42–2.41; p = 0.001). Hemorrhagic stroke was found in 31 patients (6.4%). There was significant difference in rate of hemorrhagic stroke between PAD (4.1%) and non-PAD (1.1%) groups in follow up (HR, 4.02; 95% CI 1.95–8.29; p = 0.001). In patients with PAD compared to those without PAD, the incidence of all cardiac events was excessively increased (HR, 11.24; 95% CI 8.46–14.93; p = 0.001).
The risk of patients with PAD for all types of stroke recurrence and cardiac events was significantly higher after adjustment for age, sex, tobacco use, hypertension, hyperlipidemia, prior coronary artery disease, history of coronary revascularization (bypass or stenting), carotid surgery or stenting, and amputation. HR of fatal stroke (11.73; 95% CI 7.48–18.42; p = 0.001) and cardiac events was higher after adjustment (8.31; 95% CI 6.01–11.48; p = 0.001).
In the Cox regression analysis, statistically significant predictors for recurrent stroke were higher age (defined as above the median of 60 years: HR, 1.99), PAD (HR, 1.67), hypertension (HR, 2.06), diabetes mellitus (HR, 2.39), and smoking (HR, 2.03). When patients with PAD were compared with those without PAD, HR was excessively higher in patients with cardiac events than in those with stroke recurrence (Table; online supplementary material).
Patients without PAD used aspirin (1360 [79.4%]) and clopidogrel (352 [20.5%]) whereas those with PAD used clopidogrel (160 [50.3%]), aspirin (80 [25.2%], and cilostazol (78 [24.5%]) during follow up. In the regression analysis of treatment effect on stroke recurrence, statistically significant preventive medication and intervention were antiplatelet treatment (HR, 0.71; 95% CI, 0.57–0.88), carotid surgery or stenting (HR, 0.67; 95% CI, 0.50–0.90), and coronary surgery or stenting (HR, 0.64; 95% CI, 0.51–0.81).
The current study showed that PAD is a strong predictor of recurrent stroke (HR, 1.84) and cardiac events (HR, 11.24) in patients with first-ever ischemic stroke. It seems that there is a more close association between cardiovascular diseases and PAD. Limited data are available regarding the predictability of PAD for recurrent stroke among first-ever ischemic stroke patients.8–10 With data from our population, we determined that PAD is a crucial predictor of total recurrent stroke, fatal stroke, and cardiac events and for nonfatal stroke and cardiac events.
In our cohort, significant vascular risk predictors for recurrent stroke in the regression analysis were age, PAD, hypertension, diabetes mellitus, and smoking. We noticed that in cardiac events, hyperlipidemia is among the other traditional vascular risk factors. Another interesting finding is that coronary intervention leads to a reduction in stroke. This can be explained by the reduction of rhythm disturbances, prevention of congestive heart failure, and reduction of emboli due to myocardial infarction. Some limitations must be considered. First, diagnoses of recurrent stroke and cardiac events from hospital or general practitioner records were centrally verified as best as possible, but misclassification of events cannot be completely excluded. Second, before the enrollment period, ABI measurement was not performed in a considerable number of potential subjects (11.5%) for several reasons – excluding severe disabling stroke, discharge prior to measurement, or refusal to enroll.
Third, the rate of PAD in the study’s patient population is a lower than expected rate of PAD, as quoted earlier in the study. The reason for this discrepancy might be related to ethnic and regional differences. 10 Fourth, cilostazol is preferred predominantly in patients with peripheral vascular diseases in our country. The differences in antiaggregant use between the two groups were related to this fact.
PAD has a substantially increased risk of recurrent stroke and cardiac events. In the context of other studies, our findings confirm the value of PAD for predicting secondary vascular events. PAD should be considered in the effective management of modifiable cardiovascular risk factors to reduce the risk of first-ever stroke patients and other vascular events.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231171610 – Supplemental material for Association between recurrence of stroke and peripheral artery disease after first-ever stroke
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231171610 for Association between recurrence of stroke and peripheral artery disease after first-ever stroke by Emre Kumral, Fatma Ece Çetin, Hüseyin Nezih Özdemir and Mehmet Orman in Vascular Medicine
Footnotes
Acknowledgements
We would like to express our gratitude to the stroke nurses for their great help in the care and follow-up of the patients.
Data availability
Individual deidentified participant data and study documents are available upon request to the corresponding author.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
