Abstract
Prospective studies supporting a relationship between elevated lipoprotein-associated phospholipase A2 (Lp-PLA2) and incident peripheral arterial disease (PAD) are limited. We evaluated the association of Lp-PLA2 with incident PAD in a multi-ethnic cohort without clinical cardiovascular disease. A total of 4622 participants with measurement of Lp-PLA2 mass and Lp-PLA2 activity and an ankle–brachial index (ABI) between 0.9 and 1.4 were followed for the development of PAD (median follow-up = 9.3 years), defined as an ABI ⩽0.9 and decline from baseline ⩾0.15. There were 158 incident PAD events during follow-up. In adjusted logistic regression models, each higher standard deviation of both Lp-PLA2 activity and mass did not confer an increased risk of developing PAD [odds ratios, (95% confidence intervals)]: 0.92 (0.66–1.27) for Lp-PLA2 activity and 1.06 (0.85–1.34) for mass. Additionally, no significant interaction was found according to ethnicity: p=0.43 for Lp-PLA2 activity and p=0.55 for Lp-PLA2 mass. We found no evidence of an association between Lp-PLA2 and incident PAD.
Introduction
Eight million people in the United States and over 200 million people worldwide are estimated to have peripheral arterial disease (PAD) and these individuals have higher mortality rates than those without PAD.1–3 Prevalence of PAD varies substantially according to age and ethnicity with significantly higher rates in older populations and African American individuals and lower rates in Asian and Hispanic individuals.4–7 Established risk factors alone do not fully explain ethnic-specific variations in PAD prevalence.6,8
Lipoprotein-associated phospholipase A2 (Lp-PLA2) is an enzyme highly expressed by macrophages in atherosclerotic lesions, particularly within the necrotic core and fibrotic cap of rupture-prone plaques.9–11 Lp-PLA2 is responsible for the hydrolysis of oxidized phospholipids on low-density lipoprotein (LDL) particles and is considered a marker specific for vascular inflammation.12–14
A consistent, direct association between Lp-PLA2 levels and both incident coronary heart disease and ischemic stroke has been reported in prospective cohorts with very different characteristics.15–20 Determining whether Lp-PLA2 is also associated with incident PAD is important considering the prevalence of PAD and related mortality. The association of Lp-PLA2 with incident PAD has only been reported in one prospective study that included predominantly white elderly participants. 21 In this analysis, we studied the association between baseline Lp-PLA2 mass and activity levels and the development of PAD, defined as the development of a low ankle–brachial index (ABI).
Methods
The Multi-Ethnic Study of Atherosclerosis (MESA) is a population-based study of 6814 adults aged 45–84 years free from cardiovascular disease who were recruited from six field centers (Baltimore, Maryland; Chicago, Illinois; Forsyth County, North Carolina; Los Angeles, California; New York, New York; and St Paul, Minnesota) and underwent baseline examination between July 2000 and September 2002. 22 The study participants were 53% female and consisted of four ethnic groups: non-Hispanic white (38%), black (28%), Hispanic (22%), and Chinese-Americans (12%). MESA conducted four subsequent examinations of the cohort between 2002 and 2012. Institutional review boards at each site approved the study, and all participants gave written informed consent.
Risk factor assessments
Standardized questionnaires were used at baseline to obtain demographic information, level of education, annual household income, physical activity, alcohol consumption, smoking history, and medication usage. Body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared. Three separate systolic and diastolic resting blood pressure measurements were taken in seated participants, with the last two measurements being averaged for analysis. 23 Physical activity was recorded as participant-reported number of intentional exercise metabolic equivalent (MET)-minutes per week. Alcohol consumption was determined by a self-reported number of drinks per week. Cigarette smoking was calculated in pack-years and also defined as current, former, or never. Aspirin use was defined as a self-reported use of at least 3 days per week. Hypertension was defined as a self-report of physician diagnosis and use of an anti-hypertensive medication, or systolic blood pressure ⩾140, or diastolic blood pressure ⩾90 mmHg.
Serum measurements
Fasting blood samples were drawn and promptly centrifuged at either 2000 g for 15 minutes or 3000 g for 10 minutes at 4ºC. Plasma or serum were separated, aliquotted, and stored at −70ºC or colder at a Central Blood Analysis Laboratory (University of Vermont). Total and high-density lipoprotein (HDL) cholesterol, triglycerides, and glucose were measured. LDL-cholesterol was calculated by the Friedewald equation in those with triglycerides <400 mg/dL. Diabetes was defined as a fasting glucose >125 mg/dL or use of anti-diabetic medications. Serum creatinine was measured with rate reflectance spectrophotometry using thin film adaptation of the creatine amidinohydrolase method on the Vitros analyzer (Johnson & Johnson Clinical Diagnostics, Inc., Rochester, NY, USA; inter-assay coefficient of variation: 2.2%). The estimated glomerular filtration rate (eGFR) was calculated using the Chronic Kidney Disease Epidemiology Collaboration equations. 24
Plasma Lp-PLA2 measurement
Lp-PLA2 mass and activity were measured in stored baseline plasma samples. Measurements were performed by diaDexus, Inc. (San Francisco, CA, USA). 25 Lp-PLA2 mass was measured using a sandwich enzyme immunoassay (PLAC™ Test; diaDexus). Lp-PLA2 activity was measured by an enzymatic assay using a tritium-labeled platelet activating factor (PAF) analog as the substrate. The inter-assay coefficients of variation were 6.0% for Lp-PLA2 mass and 5.0% for Lp-PLA2 activity. Lp-PLA2 values were not available in 1328 participants, mostly due to lack of consent for research involving a commercial entity.
ABI measurement
The ABI was performed at baseline examination as well as clinic exam 3 (between September 2002 and February 2004) and clinic exam 5 (between April 2010 and December 2011). To obtain the measurements used to calculate the ABI, participants rested supine for 5 minutes, and then systolic blood pressures were measured in both arms and legs with the appropriate-sized cuffs. For each leg, the systolic blood pressure in each posterior tibial and dorsalis pedis artery was measured using a continuous-wave Doppler ultrasound 5-mHz probe. The leg-specific ABI was calculated as the higher systolic blood pressure in the posterior tibial or dorsalis pedis divided by the average of the left and right brachial pressures. In the event that left and right brachial pressures differed by 10 mmHg or more, the higher of the brachial pressures was chosen, since subclavian stenosis could be present. 26 The lower of the two leg-specific ABIs was used for analysis.
Prevalent PAD was defined as an ABI ⩽0.90. Incident PAD was defined as a decline in ABI of at least 0.15 and to 0.9 or less in either leg. This approach was used to limit the impact of regression to the mean and measurement error, and avoids small clinically insignificant changes (e.g. 0.91 at baseline to 0.89 at follow-up) being included in the incident low ABI definition.27,28 If only one follow-up ABI was available, then that was used for the analysis. If both follow-up ABIs were available, then the lower of the two values from exam 3 and exam 5 was used for the analysis. Change in ABI was calculated as the difference between ABI at baseline and the lowest ABI at follow-up.
Statistical analysis
Individuals having baseline Lp-PLA2 mass or activity and ABI measurements with at least one follow-up ABI measurement were eligible. Additionally, participants with a baseline ABI <0.9 or non-compressible vessels (ABI >1.4) at baseline were excluded from the analysis. Comparison of baseline characteristics was initially performed between those included in the analysis and those excluded using χ2 tests for categorical measures and t-tests for continuous measures. Among participants included in the analysis a comparison of baseline characteristics was also performed between those who developed incident PAD and those who did not.
Associations of Lp-PLA2 mass and activity with the development of PAD were assessed separately using logistic regression modeling. Odds ratios (ORs) and 95% confidence intervals (CIs) for incident PAD were calculated per standard deviation increment for both Lp-PLA2 mass and activity. Models were initially adjusted for age, sex, and race (Model 1). Additional adjustments included alcohol use, aspirin use, baseline ABI, BMI, cigarette smoking, diabetes, eGFR, HDL-cholesterol and total cholesterol, hypertension, physical activity, and statin use (Model 2). The difference in time between the baseline and follow-up ABI measures was also adjusted for in multivariable analyses, depending on which exam was used for the participants. Age- and sex-adjusted analyses were performed stratified by ethnicity. A p-value for interaction was also performed to determine if associations for Lp-PLA2 variables and incident PAD significantly differed by race/ethnicity.
Additionally, we performed a multivariable linear regression sensitivity analysis adjusting for the same variables listed above to assess continuous change in ABI per standard deviation increment in Lp-PLA2 mass and activity for the entire cohort and stratified by ethnicity. Continuous change in ABI was annualized and defined as the total change in ABI divided by the time between the baseline exam and lowest follow-up ABI.
For each model we used multivariable regression splines to explore the linearity assumption for mass and activity. There was no evidence to suggest that non-linearity was present; hence, these variables were used continuously to maximize power and information.
Finally, to see if associations might differ by including both exam 3 and exam 5 ABI measurements, and accounting for time-varying confounding variables, we performed a repeated measures longitudinal analysis using generalized linear mixed modeling. These associations were performed for both the logistic and linear regression analyses described above to assess the relationship of Lp-PLA2 levels with incident PAD and continuous change in ABI, respectively. For the continuous model, the outcome was taken to be the change in ABI from baseline to either exam 3 or exam 5. For the incident PAD model, the outcome was binary, and a logistic link was used. These models include a random effect for subject to allow for the correlation between repeated observations on the same participant.
Results
Figure 1 shows the flow of participants that were eligible for the analysis. Among the 4622 participants, 1105 had an ABI value at exam 3 only, and 79 had an ABI value at exam 5 only. The ABI used for analysis was taken from 2735 participants at exam 3 and 1887 participants at exam 5. In Table 1, compared to participants included in the analysis, excluded participants were older and were more likely to be black, smokers, diabetics, and be on anti-hypertensive and statin medications. BMI, systolic blood pressure, C-reactive protein (CRP), and HDL-cholesterol were higher while exercise levels were lower in those who were excluded.
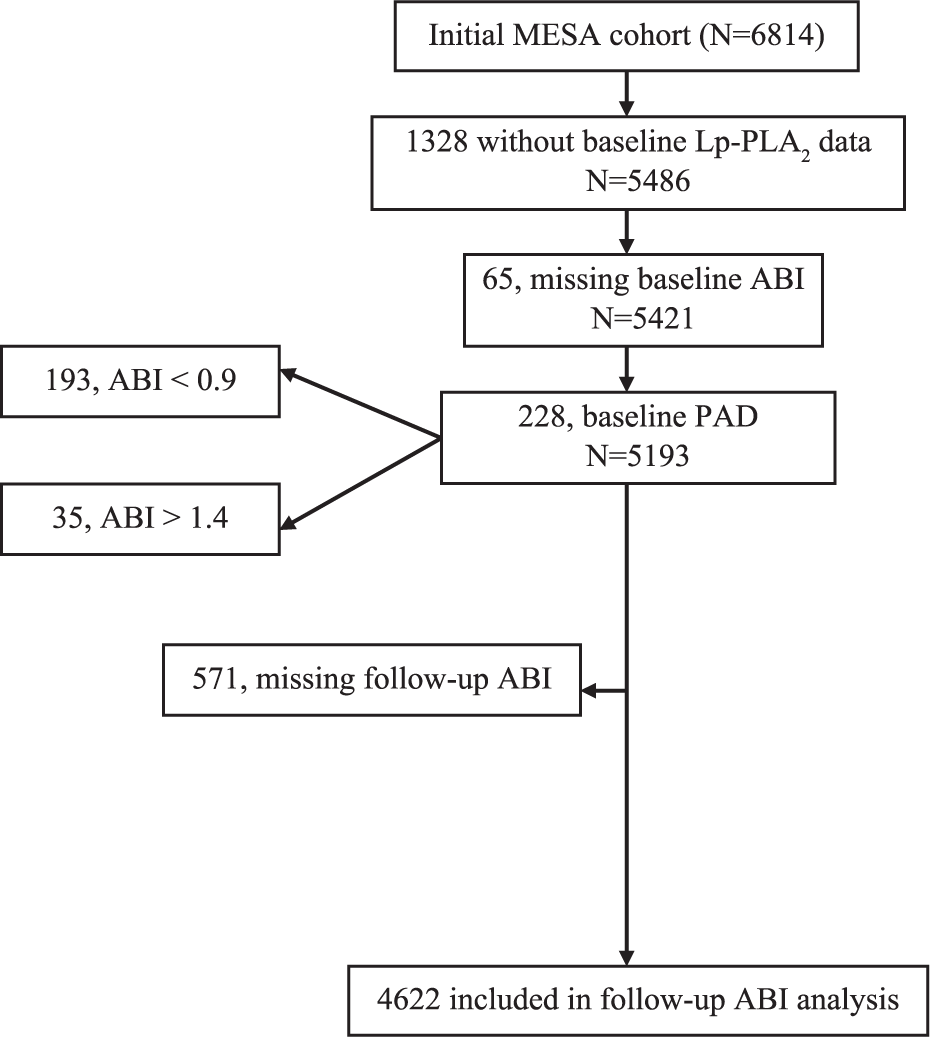
Flowchart of participants included in the analysis for developing a low ankle–brachial index (ABI). Lp-PLA2, lipoprotein-associated phospholipase A2; PAD, peripheral arterial disease.
Baseline characteristics of the Multi-Ethnic Study of Atherosclerosis (MESA) participants based on study inclusion.
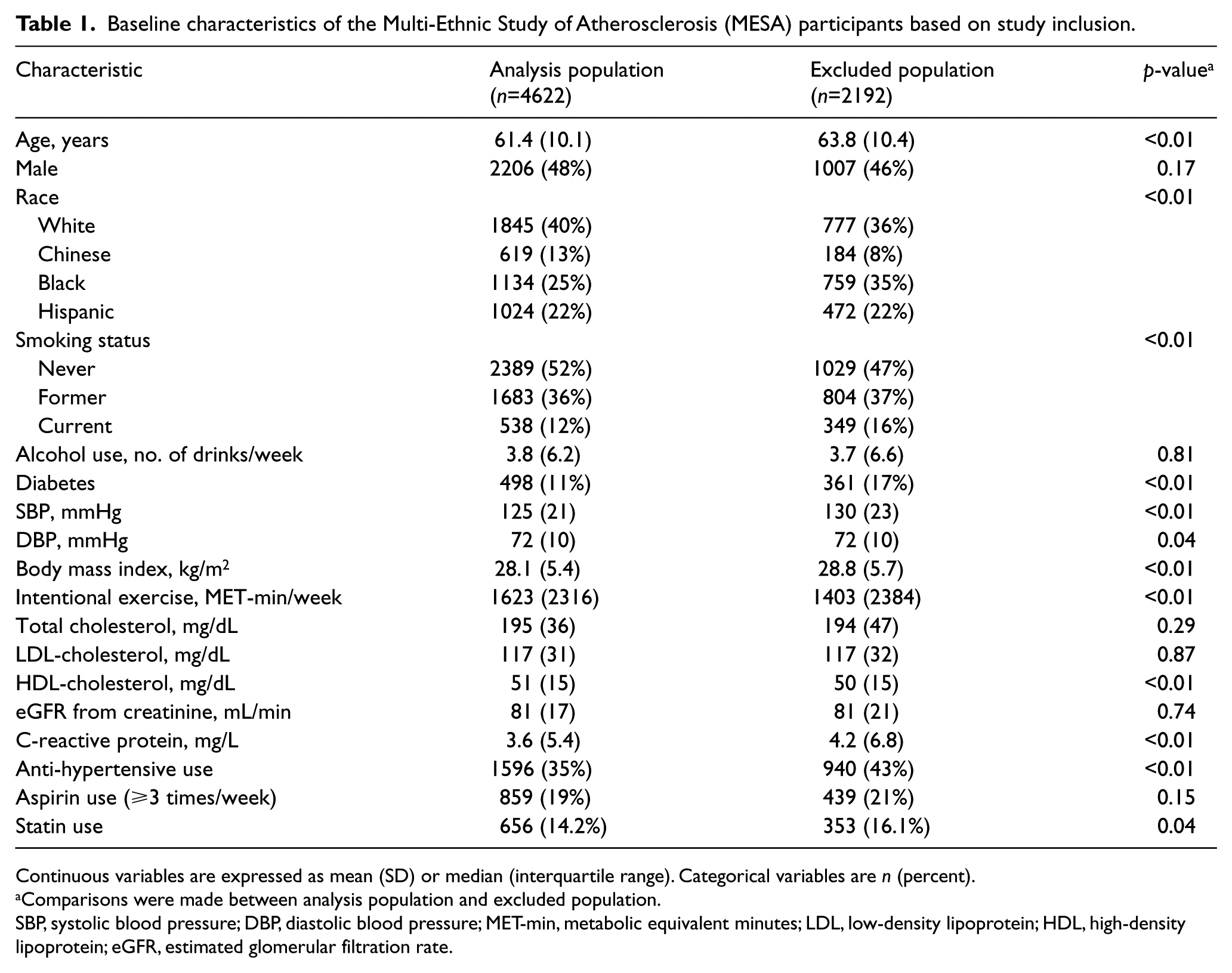
Continuous variables are expressed as mean (SD) or median (interquartile range). Categorical variables are n (percent).
Comparisons were made between analysis population and excluded population.
SBP, systolic blood pressure; DBP, diastolic blood pressure; MET-min, metabolic equivalent minutes; LDL, low-density lipoprotein; HDL, high-density lipoprotein; eGFR, estimated glomerular filtration rate.
There were 158 (3.4%) incident cases of PAD over a median follow-up period of 9.3 years. Table 2 compares baseline characteristics in participants who developed incident PAD to those who did not. Participants who developed PAD were older and more likely to be black, smokers, diabetics, and be on anti-hypertensive medication. Baseline BMI, CRP, and systolic blood pressure were higher, while ABI, exercise levels and HDL-cholesterol were lower in those who developed PAD. Lp-PLA2 mass and activity levels did not differ between the two groups.
Baseline characteristics of the Multi-Ethnic Study of Atherosclerosis (MESA) participants according to presence or absence of incident peripheral arterial disease (PAD).
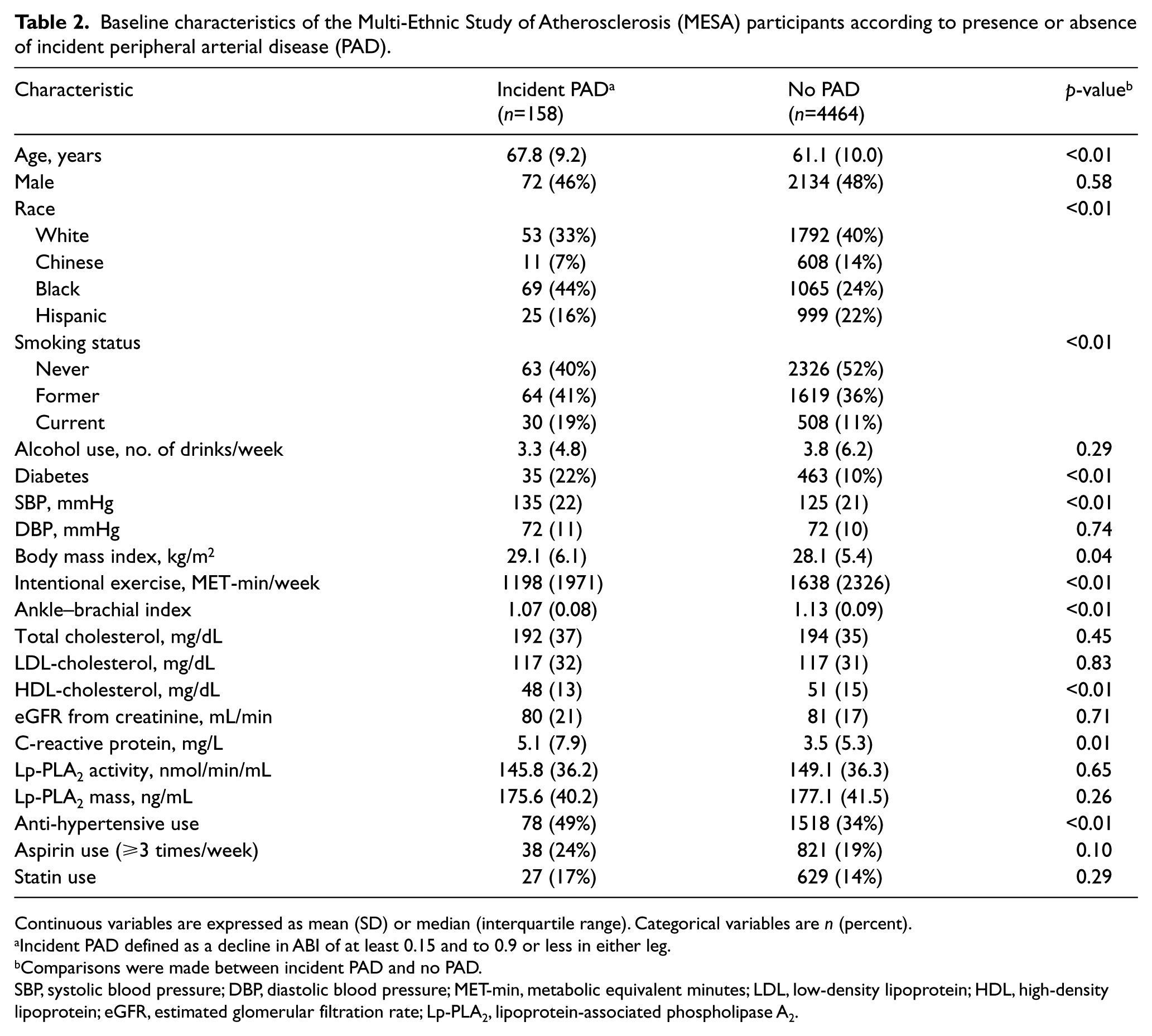
Continuous variables are expressed as mean (SD) or median (interquartile range). Categorical variables are n (percent).
Incident PAD defined as a decline in ABI of at least 0.15 and to 0.9 or less in either leg.
Comparisons were made between incident PAD and no PAD.
SBP, systolic blood pressure; DBP, diastolic blood pressure; MET-min, metabolic equivalent minutes; LDL, low-density lipoprotein; HDL, high-density lipoprotein; eGFR, estimated glomerular filtration rate; Lp-PLA2, lipoprotein-associated phospholipase A2.
Table 3 shows the odds ratios for developing PAD for both Lp-PLA2 activity and Lp-PLA2 mass. Adjusting for age, sex, race, and risk factors, neither Lp-PLA2 activity nor Lp-PLA2 mass were associated with an increased odds of incident PAD. The adjusted odds ratios were 0.92 (95% CI: 0.66, 1.27) and 1.06 (95% CI: 0.85, 1.34) per standard deviation (SD) increment of Lp-PLA2 activity (36 nmol/min/mL) and Lp-PLA2 mass (42 ng/ml), respectively. Lp-PLA2 activity and Lp-PLA2 mass were also not associated with a continuous decline in ABI in age- and sex-adjusted analyses; average annual changes in ABI were −0.0008 (95% CI: −0.0016, 0.0001) and −0.0008 (95% CI: −0.0018, 0.0002), respectively.
Associations between Lp-PLA2 and incident peripheral arterial disease (PAD).
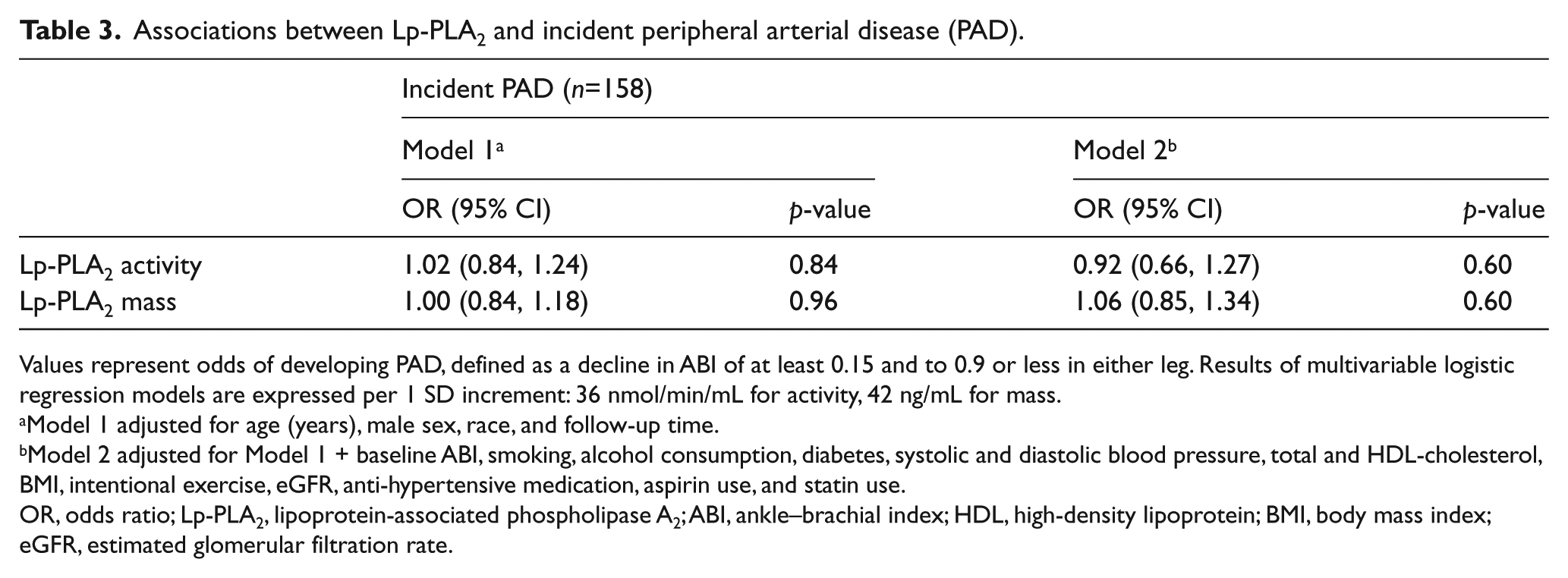
Values represent odds of developing PAD, defined as a decline in ABI of at least 0.15 and to 0.9 or less in either leg. Results of multivariable logistic regression models are expressed per 1 SD increment: 36 nmol/min/mL for activity, 42 ng/mL for mass.
Model 1 adjusted for age (years), male sex, race, and follow-up time.
Model 2 adjusted for Model 1 + baseline ABI, smoking, alcohol consumption, diabetes, systolic and diastolic blood pressure, total and HDL-cholesterol, BMI, intentional exercise, eGFR, anti-hypertensive medication, aspirin use, and statin use.
OR, odds ratio; Lp-PLA2, lipoprotein-associated phospholipase A2; ABI, ankle–brachial index; HDL, high-density lipoprotein; BMI, body mass index; eGFR, estimated glomerular filtration rate.
Analysis of incident PAD for both Lp-PLA2 activity and Lp-PLA2 mass stratified by race/ethnicity is shown in Table 4. Despite an apparent difference in findings between white individuals and the other groups, the difference was not statistically significant: p-values for interaction of race with Lp-PLA2 activity and Lp-PLA2 mass were 0.43 and 0.55, respectively, for incident PAD, and 0.45 and 0.59 for annualized continuous change in ABI. Among white participants, in age- and sex-adjusted analysis, each SD increment (36 nmol/min/mL) of Lp-PLA2 activity was associated with an increased odds of incident PAD (OR 1.45, 95% CI: 1.06, 2.00) and an annualized continuous decline in ABI (OR −0.0018, 95% CI: −0.0035, −0.0001). No significant associations were observed for either Lp-PLA2 activity or Lp-PLA2 mass with incident PAD in Chinese, black, or Hispanic participants.
Associations between Lp-PLA2 and peripheral arterial disease (PAD) stratified by race/ethnicity.
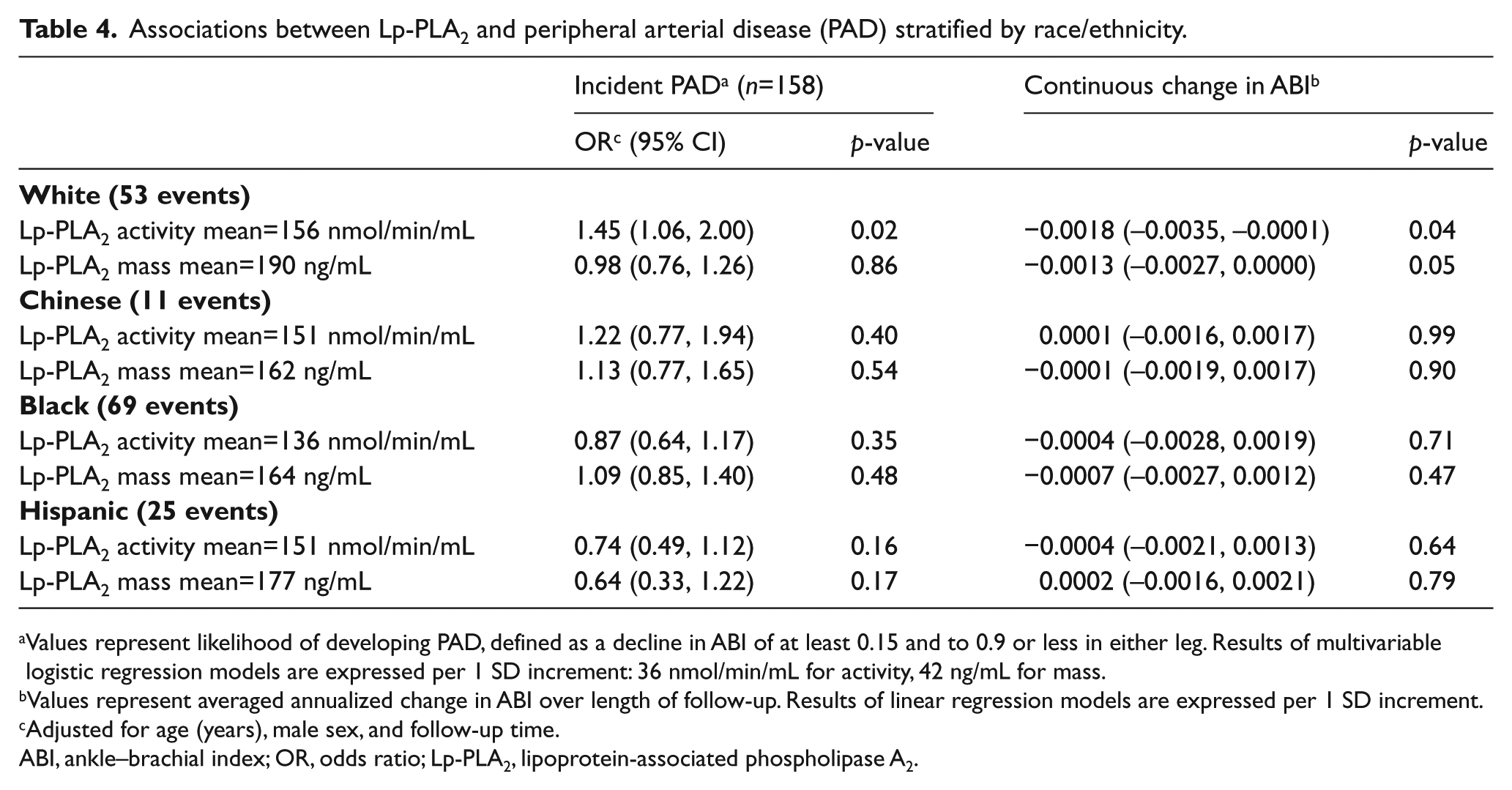
Values represent likelihood of developing PAD, defined as a decline in ABI of at least 0.15 and to 0.9 or less in either leg. Results of multivariable logistic regression models are expressed per 1 SD increment: 36 nmol/min/mL for activity, 42 ng/mL for mass.
Values represent averaged annualized change in ABI over length of follow-up. Results of linear regression models are expressed per 1 SD increment.
Adjusted for age (years), male sex, and follow-up time.
ABI, ankle–brachial index; OR, odds ratio; Lp-PLA2, lipoprotein-associated phospholipase A2.
Longitudinal associations using generalized linear mixed modeling of Lp-PLA2 mass and activity levels with either incident PAD or continuous change in the ABI for both the entire cohort and when stratified by race/ethnicity were similarly non-significant (Supplemental Tables 1 and 2). The p-values for interaction of race with Lp-PLA2 activity and Lp-PLA2 mass were 0.76 and 0.60, respectively, for incident PAD, and 0.27 and 0.98 for annualized continuous change in ABI.
Discussion
In a multi-ethnic cohort without baseline clinical cardiovascular disease, higher baseline Lp-PLA2 mass and activity were not associated with incident PAD, defined by an ABI less than 0.90 or continuous decline in ABI. There was also no significant interaction by race observed for the relationship of either Lp-PLA2 activity or mass and incident PAD.
Associations for higher Lp-PLA2 levels and increased risk of incident cardiovascular disease have been established across multiple prospective cohorts.15–20 However, research regarding the association of Lp-PLA2 mass and activity with incident PAD is limited to one study. 21 In the Cardiovascular Health Study (CHS), both higher Lp-PLA2 mass and higher Lp-PLA2 activity were associated with incident PAD, defined either by development of clinical symptoms or a low ABI. 21 A difference in results was noted for the association of Lp-PLA2 mass and incident clinical PAD by race, with no association found in black individuals, while associations of Lp-PLA2 mass or activity and incident low ABI did not differ by race.
In contrast to the CHS, we found no significant association of either Lp-PLA2 mass or activity with incident PAD and no evidence of effect modification by race. Differences in cohort characteristics may help explain the discrepant findings between these studies. MESA participants, in comparison to CHS participants, are a healthier, younger group without baseline cardiovascular disease who experienced a much lower rate of incident PAD. The CHS cohort was recruited primarily in 1989–90 prior to the statin era. Additionally, the CHS included predominantly white individuals with black participants making up only 15% of the study population, while MESA is a more diverse cohort including ethnicities not represented in the CHS.
Prior research has already demonstrated that Lp-PLA2 mass and activity levels significantly vary across ethnicity, as do their associations with the development of cardiovascular disease. In a study of over 700 individuals, both Lp-PLA2 mass and activity were lower in black participants, intermediate in Asians and Hispanics, and highest in white individuals. 29 Similar differences were reported in the Dallas Heart Study, which included over 3000 black, Hispanic, and white participants. 30 In the Northern Manhattan Study that included nearly 2000 participants, Lp-PLA2 mass was associated with incident stroke in non-Hispanic white individuals but not in non-Hispanic black or Hispanic populations. 31 For MESA, Lp-PLA2 activity levels were not associated with incident cardiovascular disease in Chinese participants, but were in the other three ethnic groups. 20
Atherosclerosis is a complex process that can vary across different vascular beds and significant differences in the strength of the association of traditional cardiovascular risk factors such as diabetes or cigarette smoking with PAD versus other forms of atherosclerotic disease have already been established.32,33 It is possible the relationship of Lp-PLA2 with PAD may differ from its relationship with other forms of atherosclerotic disease. Lp-PLA2 is bound to lipoproteins in the circulation and most polymorphisms associated with Lp-PLA2 mass and activity are located in regions associated with levels of other lipoproteins. 34 Prospective studies of lipoprotein(a) and the risk of incident PAD have been mixed, suggesting a diminished role of lipoproteins in the progression of lower extremity arterial disease.35–37
Limitations
MESA participants did not have baseline clinical cardiovascular disease and findings may not apply to dissimilar populations. Hemoglobin A1c measurements were not available at study baseline and could not be used to identify any additional participants with prevalent diabetes. The significant amount of missing data for baseline Lp-PLA2 measurements and follow-up ABI values excluded a significant proportion of participants and may have introduced bias. Our assessment of incident PAD was restricted to the development of a low ABI due to the low number of symptomatic PAD events occurring during follow-up, precluding study of this endpoint. Progression of PAD may have been inadequately assessed, particularly in diabetics, due to exclusion of participants with an ABI >1.4 at baseline. Finally, ABI measurements did not include a post-exercise value and, therefore, may not have detected PAD in some individuals.
Conclusion
Our results demonstrate that, in a multi-ethnic cohort without baseline cardiovascular disease, higher Lp-PLA2 mass and activity were not significantly associated with an increased risk for incident PAD as defined by the ABI. Additional prospective studies in other observational cohorts will be helpful to better define the relationship of Lp-PLA2 levels and the development of PAD.
Footnotes
Acknowledgements
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this research was supported by contracts HHSN268201500003I, N01-HC-95159, N01-HC-95160, N01-HC-95161, N01-HC-95162, N01-HC-95163, N01-HC-95164, N01-HC-95165, N01-HC-95166, N01-HC-95167, N01-HC-95168, and N01-HC-95169 from the National Heart, Lung, and Blood Institute, and by grants UL1-TR-000040 and UL1-TR-001079 from the National Center for Research Resources. Additional support was from an investigator-initiated grant from GlaxoSmithKline. Dr Cushman and Dr Jenny have had other funding from Diadexus. The funding sources did not have a role in the design, analysis or approval of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
