Abstract
Shortened telomere length (TL) and oxidative stress have been described in several vascular disorders at both the tissue and circulating level. However, to our knowledge, there are no reports about TL associated with varicose vein (VV) disease. This paper aimed to evaluate, at the tissue and circulating level, TL and oxidative stress in VV disease, compared to the corresponding counterparts from abdominal aortic aneurysm (AAA) patients and control healthy subjects. TL was measured using quantitative fluorescence in situ hybridization (Q-FISH). Oxidative stress was evaluated by measuring the malondialdehyde (MDA) concentration by thiobarbituric acid reactive substance/s (TBARS) assay. At the vascular tissue level, VV patients had shortened TL and a high MDA concentration, similarly to AAA patients. Conversely, blood lymphocytes and epidermal cells from VV patients had a TL similar to healthy controls and significantly longer than the same cells from AAA patients. Moreover, the MDA concentration in plasma from VV patients was significantly lower than from the AAA group. Linear regression analysis showed a statistically significant inverse correlation between the blood lymphocyte TL and plasma MDA level. Our results suggest that, unlike AAA, telomere attrition in VV tissue is not a systemic phenomenon but it may be attributable to tissue microenvironment conditions and possibly to high local oxidative stress.
Introduction
Varicose disorder is characterized by excessive dilation and tortuosity of the superficial veins of lower limbs. Varicose veins (VVs) show histological alterations including abnormal and disorganized collagen deposition, lower elastin content and leucocyte infiltration.1,2 In the intima, endothelial cells (ECs) are dysfunctional and desquamation of cellular fragments frequently occurs, in accordance with biochemical studies showing a high level of endothelial damage markers in patients with VVs. 3 Oxidative stress has been suggested as a key contributing factor for VV formation because of its ability to promote oxidative modifications of cellular and matrix molecules, but the exact mechanism underlying the alterations observed in VV endothelium is not fully elucidated. Recently, we showed that abdominal aortic aneurysm (AAA) patients display shorter telomere length (TL) associated with increased oxidative stress not only in ECs and vascular smooth muscle cells (VSMCs) but also in circulating lymphocytes and keratinocytes. 4 The human telomere is a simple repeating sequence of TTAGGG bases, located at the ends of chromosomes.5,6 In human diploid cells, every replicative cycle is associated with a progressive reduction in TL due to an uncompensated loss by elongation enzymes. So, TL is an effective indicator of the number of cell divisions undergone up to a critical length inducing cell senescence.7–9 Natural shortening can be accelerated by iatrogenic or environmental factors including oxidative stress.10,11 Shorter TL is considered a good marker of cell aging and senescence, suggesting a key role in endothelial damage. Shortened telomeres have been described in several vascular disorders, including AAA, peripheral artery disease and carotid disease,4,12 but to our knowledge there are no reports about TL in VV disease.
In the present work, we investigated for the first time TL in vascular wall, circulating lymphocytes and epidermal cells from patients affected by VVs and compared the levels of these parameters and of oxidative stress with corresponding counterparts from AAA patients and control healthy subjects. Since VVs share many features at the tissue level with AAA, and given our previous data on aneurysmal disease, our aim was to compare TL and oxidative stress in two vascular dilative pathologies such as VV and AAA in order to further evaluate the role of oxidative stress and telomere shorting in EC damage and in the progression of these vascular diseases.
Materials and methods
Study design
The study included 30 male subjects: 10 AAA patients whose mean age was 71.3 years (range 61–78 years), 10 VV patients whose mean age was 67.9 years (range 64–72 years) and 10 apparently healthy control subjects whose mean age was 68.7 years (range 62–77 years). The patients and control subjects were selected by the outpatient clinic of the Vascular and Endovascular Surgery Division of IRCCS San Martino – IST, Genoa, Italy. Participation in the study of was based upon informed consent of patients or legal representatives. The main exclusion criteria included the presence of neoplasms, infections and chronic inflammation, chronic obstructive pulmonary disease, diabetes mellitus, nephropathy, liver disease, symptomatic obstructive coronary artery disease, cerebrovascular diseases, and current or past smoking habits. Blood lymphocytes were obtained from a peripheral venous blood sample of the superficial vein of the arm. AAA samples were obtained from the aneurysmal sac at the time of the surgical repair. VV samples were obtained during varicectomy. Skin biopsies were obtained from six AAA patients and six VV patients at the time of surgery. The study was approved by the Ethics Committee of the IRCCS San Martino – IST.
Sample preparation
Peripheral blood samples were collected in tubes with K3EDTA and centrifuged at 3000 rpm for 15 minutes to obtain plasma. Mononuclear cells were isolated by Histopaque-1077 (Sigma, St Louis, MO, USA) gradient centrifugation and washed three times in phosphate buffered saline (PBS). Cytospin preparations of peripheral blood lymphocytes from patients and controls were cytocentrifuged and fixed in 4% paraformaldehyde in PBS. In the operating room, AAA and VV tissues were dissected, blood was washed with saline and the surrounding connective tissue and fat were removed. The specimen was then divided into two sequential blocks: block A was used for TL study, while block B was submitted to oxidative stress evaluation. Block A was fixed in 4% phosphate-buffered formaldehyde, processed into paraffin blocks, and subjected to sectioning. Block B was subjected to homogenization and immediately frozen. Skin specimens were fixed in 4% phosphate-buffered formaldehyde and processed into paraffin blocks.
Telomere length measurement by Q-FISH in interphase nuclei
Quantitative fluorescence in situ hybridization (Q-FISH) of telomeres has been extensively used to obtain quantitative information on TL distributions.13,14 In addition, the low detection limit of Q-FISH (0.1 kb of telomere repeats) allows quantification of critically short telomeres that go undetected by Southern blot analysis. The interphase Q-FISH method, which builds on conventional Q-FISH, combines labeling of telomeres in interphase nuclei, using a fluorescent peptide nucleic acid (PNA) probe against telomeric repeats, with automated microscopy. The PNA probe for telomeric sequences is a ready-to-use probe included in the Telomere PNA FISH Kit/Cy3 (Dako Cytomation, Hamburg, Germany). The hybridization was performed according to the manufacturer’s instructions. Slide scanning, cell identification, intensity measurement, and quantification of TL were performed using the fluorescence-based microscopic scanning system Nikon E1000 (Nikon, Japan) and a high-resolution CCD camera. For TL quantification, we measured Cy3 intensity in single nuclei. The slide scanning and cell analysis procedures were performed using a 100× Nikon objective. The Cy3 pan-telomeric probe intensity was measured by an appropriate filter. Telomere fluorescence signals were quantified by using the Genikon program (Nikon). A minimum of 20 nuclei were scanned for every sample and the mean value of the fluorescence ratios of all cells analyzed was calculated.
Immunofluorescence studies
Indirect immunofluorescence was performed on 4-µm thick, formalin-fixed, paraffin-embedded vascular tissue sections. After antigen retrieval slides were incubated with anti-human CD31 (diluted 1/50; Dako Cytomation) overnight at 4°C, followed by Alexa Fluor 568-conjugated anti-mouse immunoglobulin G (1/200 dilution; Molecular Probes, Eugene, OR, USA).
Malondialdehyde assay
Plasma and tissue malondialdehyde (MDA) concentration, one of the final products of lipid peroxidation, was measured with thiobarbituric acid reactive substance/s (TBARS) assay kit (Abnova, Taiwan) according to the manufacturer’s instruction. This procedure is based on the formation of a pink-colored complex between MDA and thiobarbituric acid (TBA), with an absorbance maximum at 540 nm. Briefly, 0.5 mL of plasma or tissue homogenates was added to the reaction mixture formed by equal parts of acetic acid solution, sodium hydroxide and 0.53% TBA with the addition of sodium dodecyl sulfate (SDS). The mixture was heated for 1 hour at 95°C. The samples were then cooled on ice and centrifuged, and the absorbance was measured spectrophotometrically at 540 nm. A calibration curve prepared by serial dilutions of a MDA stock solution (0–12.5 nmol/mL) was used for quantitation.
Statistical analysis
Variables with a skewed distribution were presented as medians (including the first and third quartiles), and were log-transformed to normalize the distribution before performing statistical analysis. Comparisons of quantitative variables were made by the Mann–Whitney U-test and adjusted for age, body mass index, blood pressure level, and serum total cholesterol level. Data have also been adjusted for these two last parameters because they are involved in oxidative stress, and statistical analysis suggested a trend for a difference between the three groups. Correlations were evaluated by Spearman’s rank correlation coefficient (r); r-values from 0.40 to 0.59 were considered moderate, from 0.60 to 0.79 were considered strong, and from 0.80 to 1 were considered very strong. A p-value <0.05 was considered statistically significant.
Results
Demographics
Relevant characteristics of the two study groups are shown in Table 1. The main clinical characteristics were similar in patients and controls. A trend for a difference was present for levels of systolic blood pressure (p=0.10) between the AAA group and control subjects and for levels of serum total cholesterol between patient groups and control subjects (p=0.07).
Demographics of study groups.
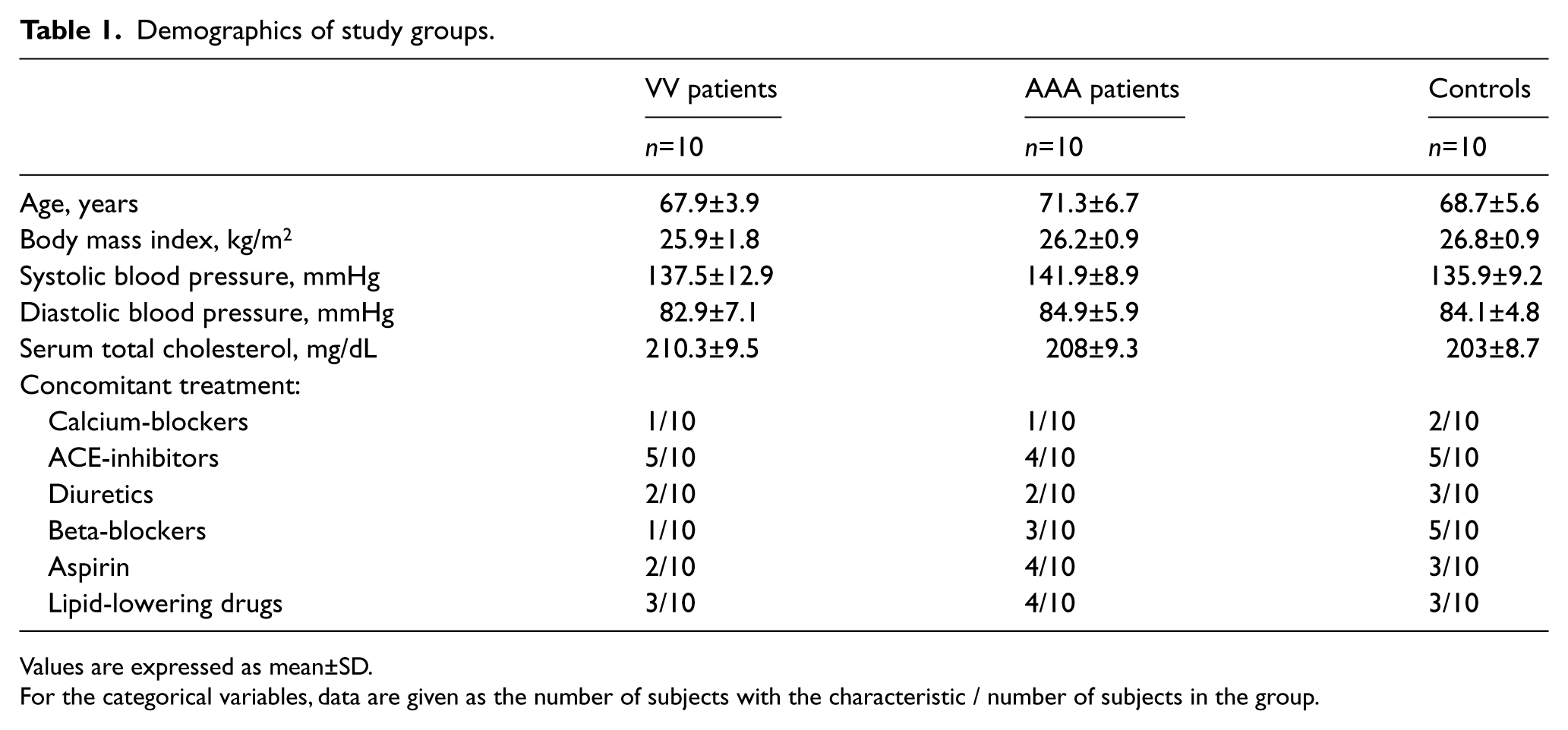
Values are expressed as mean±SD.
For the categorical variables, data are given as the number of subjects with the characteristic / number of subjects in the group.
Telomere length evaluation in plasma, ECs and epidermal cells from patients affected by VV and AAA
TL was measured using Q-FISH. We chose to use Q-FISH because this technique is suitable to perform TL analysis in individual cells. The telomere hybridization fluorescence intensities were expressed in arbitrary telomere fluorescence units (TFU).
We first investigated TL in VV tissue and AAA tissue. Serial tissue sections were stained with CD31 mAbs by immunofluorescence in order to identify ECs, and tested for TL in these cell types by Q-FISH. As shown in Figure 1A, TL in ECs from VV patients (median: 0.1027 TFU; 1st–3rd q: 0.1021–0.1029) was similar to TL measured in ECs from AAA subjects (median: 0.1013 TFU; 1st–3rd q: 0.0988–0.102) (p<0.0001).
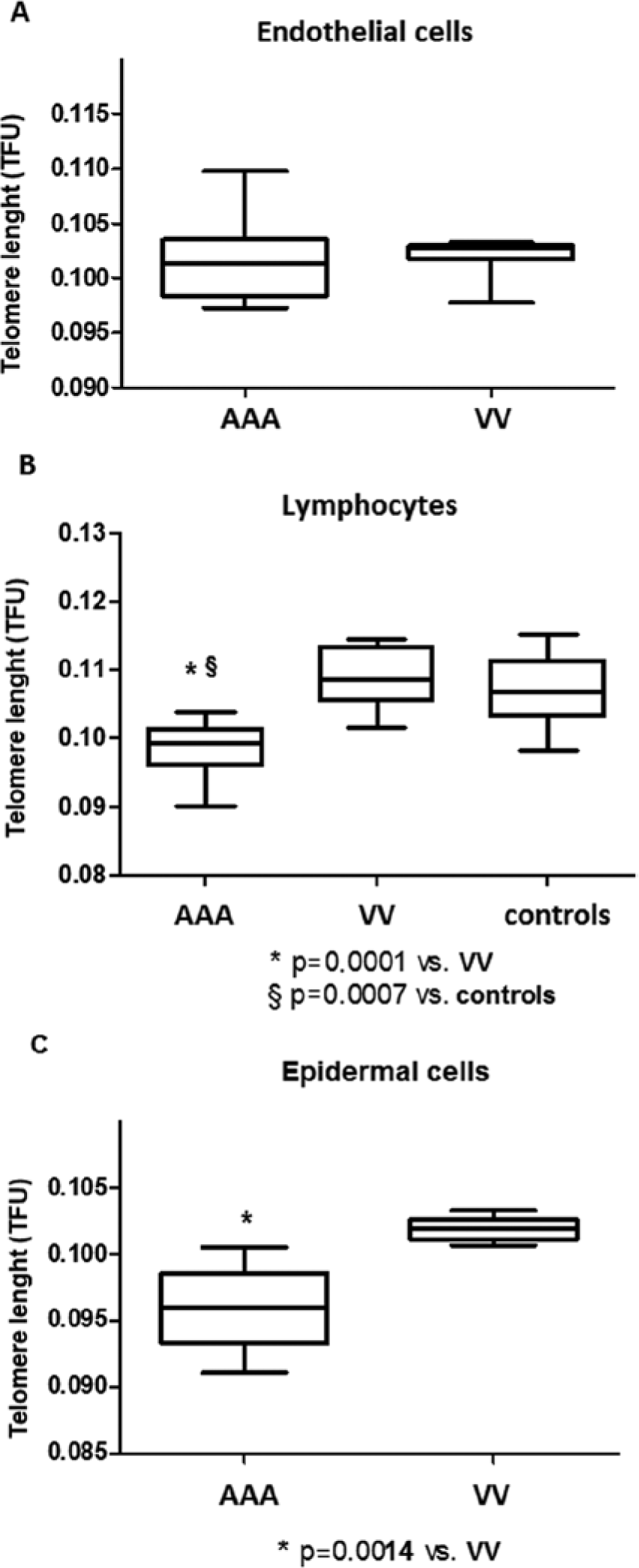
The telomere length of endothelial cells (ECs), blood lymphocytes and epidermal cells from VV and AAA patients and controls. TL was measured using quantitative fluorescence in situ hybridization (Q-FISH) and fluorescence intensities were expressed in arbitrary telomere fluorescence units (TFU). (A) TL in ECs from 10 VV patients and 10 AAA patients. (B) TL in blood lymphocytes from 10 VV patients, 10 AAA patients and 10 healthy controls. (C) TL in epidermal cells from six VV patients and six AAA patients.
We next analyzed TL in peripheral blood lymphocytes from VV patients, AAA patients and age-matched healthy donors (Figure 1B). Telomeres were significantly shorter in AAA patients (median: 0.0993 TFU; 1st–3rd q: 0.0975–0.101) than in VV patients (median: 0.1087 TFU; 1st–3rd q: 0.1056–0.1128) (p<0.0001) and controls (median: 0.1067 TFU; 1st–3rd q: 0.104–0.111) (p<0.0007). Lymphocytes from VV patients and from controls showed a similar TL.
In order to test cells not directly involved in vascular lesions, we next measured TL in epidermal cells from skin biopsies. Q-FISH revealed that telomeres in epidermal cells from VV patients (median: 0.1019 TFU; 1st–3rd q: 0.1013–0.1022) (p<0.0001) were significantly longer than in epidermal cells from AAA subjects (median: 0.0959 TFU; 1st–3rd q: 0.0944–0.0974) (p<0.0001) (Figure 1C).
These results suggest that in varicose disease, telomere shortening occurs in VV ECs not in circulating blood lymphocytes and epidermal cells, unlike AAA where telomere shortening was shown in all domains. These data underline once again the systemic nature of AAA disease, while they suggest a key role of local microenvironment in telomere attrition in varicose tissue.
Malondialdehyde level in plasma and tissue homogenates from patients affected by VV and AAA
Telomere shortening has been associated with oxidative stress. In order to evaluate the role of oxidative stress in telomere attrition in varicose tissue, we measured the MDA concentration, one of the final products of lipid peroxidation, in plasma and tissue from VV patients, AAA patients and control healthy subjects. At the vascular tissue level, the MDA concentration in VV patients (median: 325.5 TFU; 1st–3rd q: 288.25–371.25) (p<0.0001) was similar to the AAA group (median: 321 TFU; 1st–3rd q: 298.25–365.75) (p<0.0001) (Figure 2A).
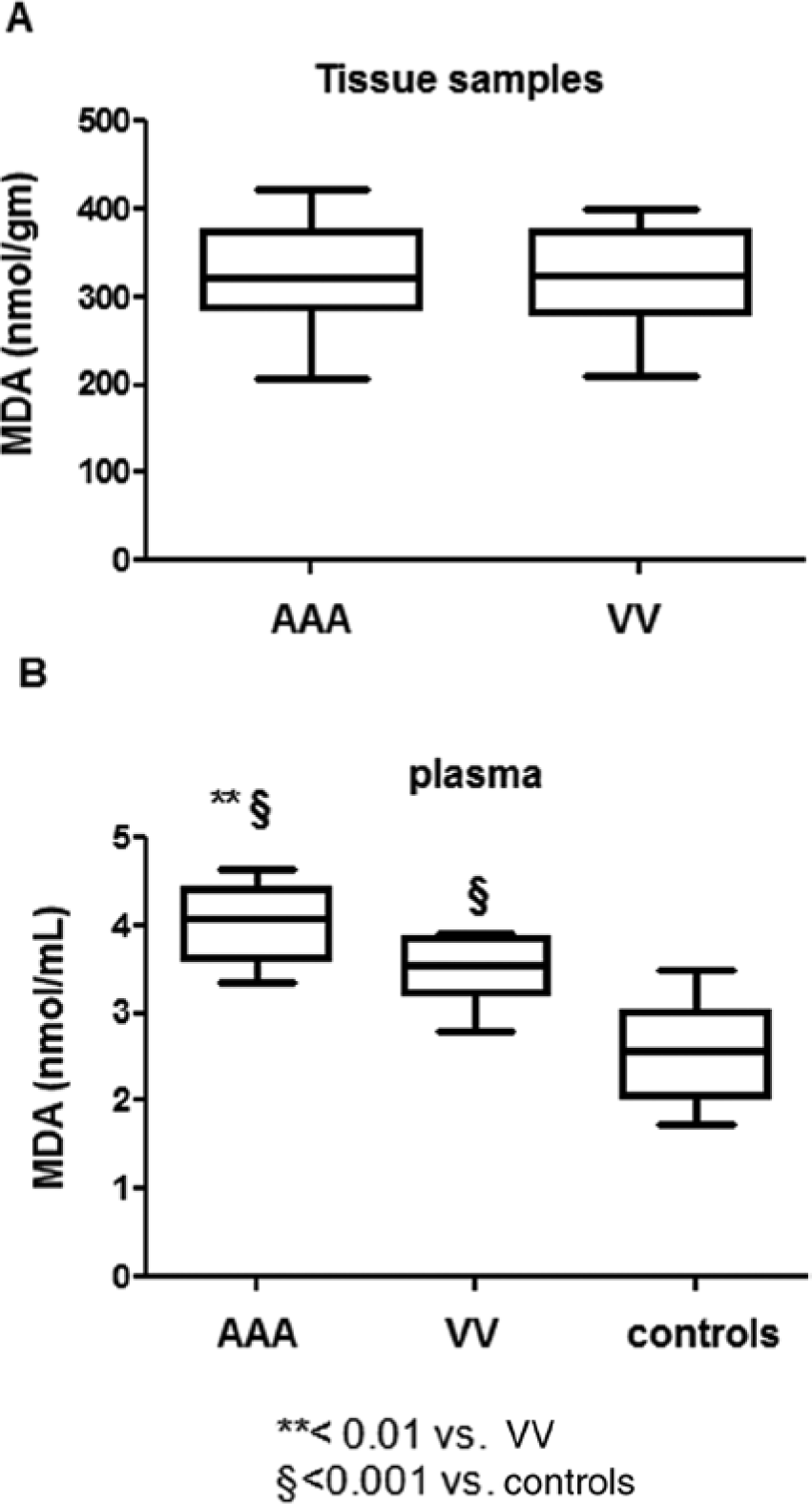
Oxidative stress in plasma and vascular tissue from VV and AAA patients and controls. Oxidative stress was evaluated by measuring the MDA concentration by thiobarbituric acid reactive substance/s (TBARS) assay. (A) MDA concentration in tissue homogenate from 10 VV patients and 10 AAA patients. (B) MDA concentration in plasma from 10 VV patients, 10 AAA patients and 10 healthy controls.
Conversely, the mean concentration of MDA in plasma from patients with VV (median: 3.545 TFU; 1st–3rd q: 3.272–3.782) was significantly lower than from the AAA group (median: 4.07 TFU; 1st–3rd q: 3.715–4.367) (p<0.0001). Moreover, compared to control subjects, VV samples also showed a slight increase of the MDA level (Figure 2B).
Taken together, these data show that oxidative stress measured in the vascular lesion was similar between VV and AAA patients. Conversely, at the circulating level, oxidative stress was lower in VV subjects than in the AAA group.
Oxidative stress and telomere shortening correlation in blood lymphocytes
We correlated blood lymphocyte TL and serum MDA concentration from VV patients, AAA patients and the healthy control group and found a significant moderate inverse correlation (r = −0.4034; p=0.027). Interestingly, the correlation was strong when the statistical analysis was restricted to samples with a MDA level higher than 3.5 nmol/mL, corresponding to the MDA median value of the VV group (Figure 3)
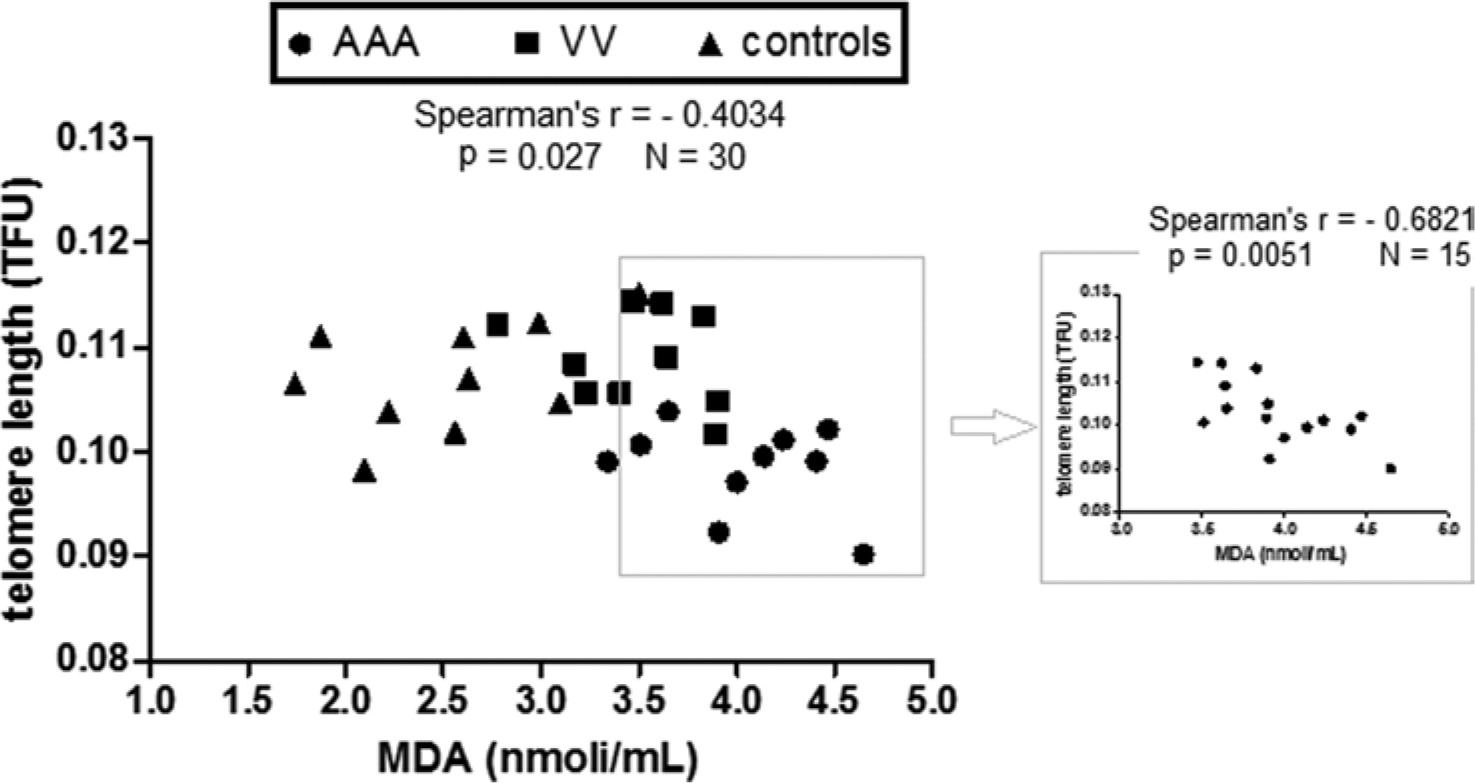
Inverse relationship between blood lymphocyte TL and plasma oxidative stress in VV patients, AAA patients and controls. Linear regression analysis and Spearman’s correlation test were performed between the blood lymphocyte TL and MDA plasma concentration in 10 VV patients, 10 AAA patients and 10 healthy controls. Spearman’s rank correlation coefficient was = −0.4034. On the right, linear regression analysis was performed including only subjects with plasma MDA concentrations higher than 3.5 nmol/mL (corresponding to the median value of the VV group). Spearman’s rank correlation coefficient was = −0.6821.
Discussion
Recent works have focused on the role of TL as a surrogate marker of vascular aging and oxidative stress-related diseases. Shortened tissue TL has been described in several vascular disorders including AAA, peripheral artery disease and carotid disease.4,12 However, to our knowledge, there are no reports about TL associated with VV disease. This paper aimed to evaluate, at circulating and tissue level, TL and oxidative stress in VV disease, compared to the corresponding counterparts from AAA patients and control healthy subjects.
We show here for the first time that ECs from VVs have shortened telomeres, displaying a pattern similar to ECs from AAA tissue. Our previous results showed a shorter telomere in ECs from AAA patients compared to healthy subjects. 4 Therefore, the present data suggest that telomere shortening occurs also in ECs from VV tissue. Since telomere attrition plays an important role in vascular aging and endothelial dysfunction, it is likely that it is involved in cell damage and endothelial alterations described in VVs. Conversely, telomeres in epidermal cells and blood lymphocytes from VV patients were significantly longer than in the same cells from AAA subjects and similar to healthy subjects. Our results are in agreement with Zee et al. who have related TL to venous disease and, in particular, to the incidence of venous thromboembolism (VTE). VV is an independent risk factor for VTE and Zee and co-workers showed that leucocyte TL from patients with VTE was similar to lymphocyte TL from healthy subjects, suggesting no association between TL and risk of VTE. 15 Taken together, these results show that telomere shortening in VV patients occurs only at the venous level, suggesting that it is not a systemic phenomenon but rather a response to local conditions. To further investigate this hypothesis, we quantified oxidative stress at vascular and circulating level in VV patients, AAA patients and control subjects. Oxidative stress plays a key role on vascular cell aging and senescence, partly related to its ability to react with DNA and to promote telomere shortening.10,11 Matthews and co-workers have demonstrated a potential relation between telomere attrition and oxidative stress in VSMC from human arterial atherosclerotic plaque. 16 In addition, we recently showed a strong relationship between shortened telomeres and augmented oxidative stress in EC and VSMC from patients with AAA. 4 In this context, we investigated the level of oxidative stress in plasma and in venous tissue homogenates from VV subjects by measuring MDA concentration, a marker of lipid peroxidation, in order to evaluate the possible role of oxidative stress in telomere shortening in VV tissue. The MDA level in VV tissue was high and comparable to that in AAA tissue. Conversely, the mean concentration of MDA in plasma from VV patients was lower than from AAA subjects. These results confirm the presence of high oxidative stress in the varicose tissue, in agreement with previous studies. 17 The presence of local oxidative stress in VV tissue was also described by Krzysciak et al., who detected an increased concentration of MDA in blood samples taken from the lower limbs compared with the samples of peripheral blood from the same subjects. 18
Moreover, statistical analysis revealed a moderate significant inverse correlation between lymphocyte TL and serum MDA level, suggesting that oxidative stress is involved in telomere shortening together with additional factors (e.g. genetic predisposition). Interestingly, when statistical analysis was performed including only subjects with plasma MDA concentrations higher than 3.5 nmol/mL (corresponding to the median value of the VV group), the correlation between TL and MDA level is strong, suggesting that over a certain threshold oxidative stress has a predominant role in telomere shortening compared to other factors that may be involved in telomere attrition.
Combining the data at tissue and circulating level, we can therefore postulate that oxidative stress is involved in telomere shortening in VV tissue. Local high oxidative stress may promote telomere shortening and consequent senescence and dysfunction in venous ECs as well, as it may induce the expression of proteolyic enzymes and adhesive molecules involved in matrix degradation and inflammatory cell infiltration responsible for the functional and structural alteration observed in VV lesions. Reactive oxygen species (ROS) and consequent oxidative stress in VVs may be due to local hypoxia and inflammation.19,20 Leucocytes are among the major producers of ROS and their presence in VV tissue has been described. 2
In accordance with other studies, we show here a slight increase in MDA concentration in the plasma of VV patients compared to healthy subjects, which is not associated with telomere shortening.17,21 Supported by the correlation data between TL and MDA concentration, we speculate that the slight increase of oxidative stress observed in VV patients at circulating level is not sufficient to act on the TL of circulating lymphocytes. However, we cannot exclude that antioxidant systems and telomerase reverse transcriptase (TERT) could be active in VV patients to counteract telomere attrition.21,22 The reduction of TERT expression, the main compensating factor for telomere loss, is associated with telomere shortening in AAA patients. To our knowledge, there are no data on TERT activity in VV patients to explain the difference observed in EC or blood lymphocyte TL. This is a very intriguing aspect and requires further investigation.
In conclusion, the study shows shortened telomeres and a high MDA level in VV tissue, not at the circulating level, suggesting a key role for oxidative stress in mediating the alteration observed in VV wall. In particular, our results suggest that, unlike AAA, telomere attrition in VV tissue is not a systemic phenomenon but it may be attributable to local microenvironment conditions strictly associated with high oxidative stress.
Footnotes
Declaration of conflicting interest
The authors have no conflicts of interest to declare.
Funding
This work was supported by ‘Compagnia di San Paolo’ (grant number 2528/SD-2010.0114).
