Abstract

What is superficial venous disease?
Veins are blood vessels that carry blood back to the heart. Leg veins consist of a network of superficial (near the skin surface) and deep veins, which refers to their location relative to the major muscle groups in the legs. Both superficial and deep veins run the entire length of the leg and are connected at multiple locations to help drain blood from the leg. When these veins become too diseased to adequately return blood to the heart, patients experience symptoms such as pain, fatigue, heaviness, itching, or swelling in the legs. Diseased veins in the superficial system may appear as spider veins, varicose veins, leg discoloration, leg swelling, or nonhealing wounds (Figure 1).
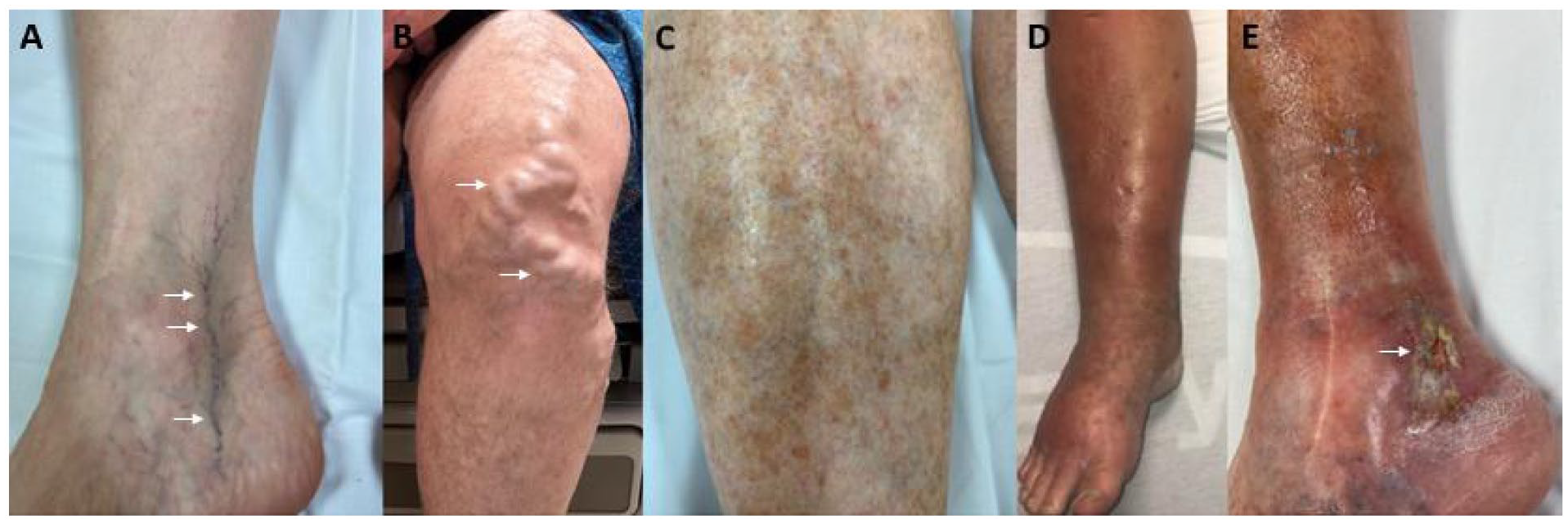
Photos of patients with various signs of superficial venous disease in the leg:
How is superficial venous disease diagnosed?
The first step in determining the presence of superficial venous disease is a thorough history and physical examination to discuss leg symptoms that could be due to vein problems and to examine the feet and legs for signs of venous disease. Some patients will have an ultrasound of the legs to look at both the superficial and deep venous systems. For more information, read the Vascular Disease Patient Information Page on venous reflux testing. 1 This test takes place in a vascular lab and is performed by a trained vascular ultrasonographer. The ultrasound determines which superficial vessels are having problems returning blood to the heart by having backflow of blood (known as venous reflux). A treatment plan is created based on the location of the vessels that are causing the problem, as well as the severity of the problem.
How is superficial venous disease managed?
Superficial venous disease is often managed conservatively with compression garments (socks or wraps), daily exercise, and leg elevation. 2 For those with wounds related to their venous disease, wound care is also recommended. 3 Patients who continue to have leg symptoms or nonhealing wounds despite conservative therapy may also benefit from different minimally invasive treatments for venous disease.
What are minimally invasive treatment options?
Minimally invasive treatment options include either permanently sealing off the problematic veins or removing them completely. Both of these options divert blood from diseased veins to healthy veins that can carry blood back to the heart. The two primary ways in which diseased superficial veins can be permanently sealed are through heat-based techniques and nonheat-based techniques. Endovenous ablation is a heat-based technique that typically targets the larger superficial veins in the legs, such as the great saphenous vein (GSV) and small saphenous vein (SSV) (Figure 2). Sometimes endovenous ablation may be used on perforator veins, which connect the superficial and deep venous systems.
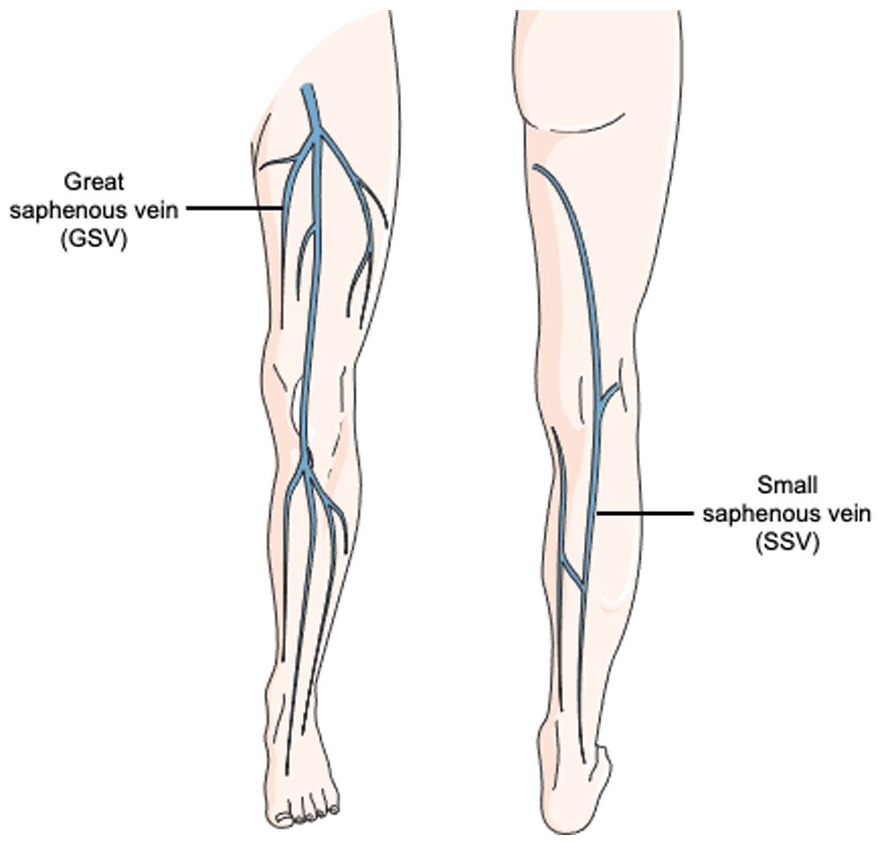
Superficial veins of the leg that are often treated with endovenous ablation include the great saphenous vein (GSV) and the small saphenous vein (SSV).
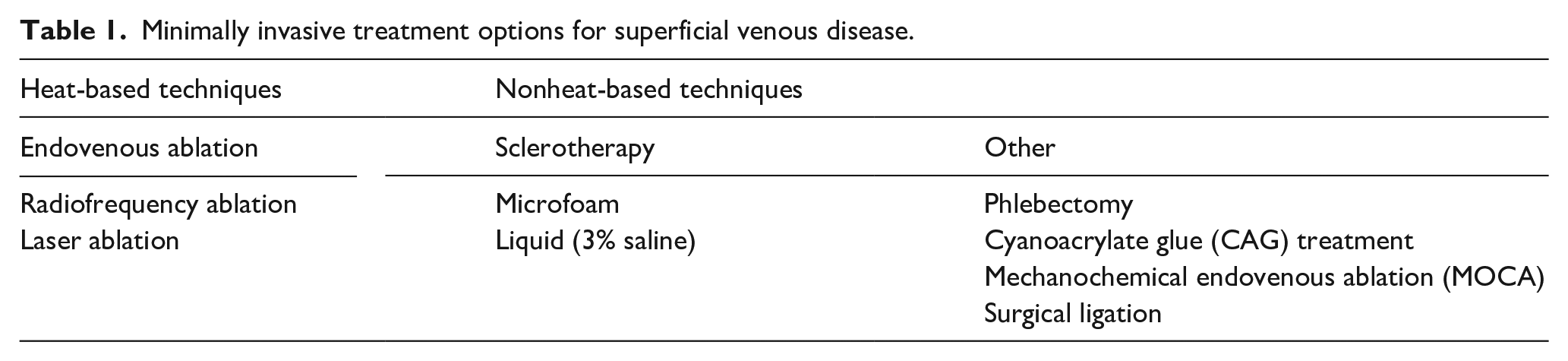
Nonheat-based techniques include sclerotherapy, phlebectomy, cyanoacrylate glue (CAG), and mechanochemical endovenous ablation (MOCA). Some of these techniques can be utilized in the GSV and SSV but are primarily reserved for varicose veins and spider veins. Most of these procedures are performed in an outpatient setting with local anesthesia. In more complex cases, they may be performed in an operating room. The treatment method can consist of one or multiple procedures based on the location of venous disease (Table 1). 4
Minimally invasive treatment options for superficial venous disease.
For large veins, surgical ligation may be recommended. This procedure involves making a small incision at the site of the vein to tie a stitch around it and permanently close it.
Endovenous ablation
There are two types of endovenous ablation: radiofrequency ablation and laser ablation. Both procedures heat the vein from the inside to create a scar and seal off (or close) the diseased vein, thus redirecting blood flow to healthy veins. Numbing medication (anesthesia) is injected just under the skin along the length of the vein to be treated. Under ultrasound guidance, a small intravenous catheter (IV) is placed in the vein. The ablation catheter is then inserted through the IV and into the vein. The ablation catheter can then be moved inside the vein along the portion that needs to be treated. The ablation process takes less than 10 minutes. The catheter is removed, and a small bandage is placed at the site of the IV. Patients are typically asked to wear compression garments for 12–24 hours after the procedure.
Endovenous ablation treatment seals off the vein immediately after the procedure is complete. Occasionally, the scarring in the treated vein might not be enough to prevent the vein from filling with blood once again. These veins can be re-treated if necessary.
Overall, endovenous ablation is extremely safe and complications are rare. The most common short-term side effects are bruising and mild swelling. In rare cases, patients can have skin burns, deep vein thrombosis (DVT; a clot that forms inside a nearby deep vein), or numbness in the area of the ablated vein due to damage to superficial nerves. Patients who have endovenous ablation will need an ultrasound several days after the procedure to look for blood clots.
Sclerotherapy
Sclerotherapy is the injection of a liquid solution directly into the vein, causing it to scar and either shrink or disappear. Sclerotherapy solutions come in microfoam and liquid preparations. Sclerotherapy can be used on portions of the main superficial veins (particularly those below the knee), varicose veins, or spider veins. 5 Varithena (Boston Scientific, Marlborough, MA) is the only United States Food and Drug Administration (FDA)-approved microfoam for treatment of the GSV. The microfoam sclerotherapy solution is injected directly into the vessel using a small syringe. For larger vessels, ultrasound guidance can be used for sclerotherapy. Sclerotherapy can take a few weeks to months for the vein to fully fade.
There are very few side effects of sclerotherapy. Sclerosing medication that is not directly injected into the vein can cause breakdown of tissue around the vein. Short-term side effects include pain, bruising, and mild bleeding at the injection site. For more superficial veins, the skin may darken, which will fade over time. If skin darkening does not fade, a topical bleaching cream may be prescribed.
Phlebectomy
Phlebectomy is the removal of veins, which can be performed in the office through small cuts in the skin. Phlebectomy is typically used for large and more superficial varicose veins. 5 For these veins, there tends to be a more durable and better cosmetic result with phlebectomy than with sclerotherapy.
For this procedure, the area around the veins to be removed is numbed. Small incisions (2–3 mm) are made above the veins, which are then removed using a small hook. No stitches are necessary; the incisions are closed with surgical glue or adhesive bandages. Rare side effects of phlebectomy include skin infection, soreness, bruising, and mild bleeding.
Cyanoacrylate glue (CAG) treatment
CAG is a medical adhesive that can be used to seal diseased veins from the inside. CAG is used to treat the GSV. This allows for redirection of blood flow to healthy veins.
Under ultrasound guidance, an IV is inserted into the vein, then the CAG catheter is inserted inside the length of vein to be treated. The adhesive is released in small doses and pressure is applied on the skin, using ultrasound to ensure that the vessel is sealed off. Once the entire vessel is treated, the catheter is removed. The treatment works immediately after the adhesive is delivered and compression is completed.
Short-term side effects include phlebitis (inflammation of the vein), bleeding, and bruising. Patients can develop a superficial blood clot; a clot of the deep veins (DVT) is rare. Patients who have CAG will need an ultrasound several days after the procedure to look for blood clots.
CAG might be considered in patients at risk for bleeding or easy bruising, as no numbing injections are needed with CAG. CAG decreases the risk of skin burns, as the technique is not heat-based. Unlike endovenous ablation, patients do not need to wear compression garments after the procedure. Patients are able to return to daily activities immediately after CAG.
Mechanochemical endovenous ablation (MOCA)
MOCA is performed using a system that consists of two parts: a rotating catheter that creates scarring using mechanical abrasions within the vein and sclerotherapy. MOCA can be used on the GSV and SSV.
Under ultrasound guidance, a small IV is placed in the vein. The MOCA catheter is then inserted through the IV and into the vein to be treated. A sclerotherapy solution is attached to the MOCA system. The wire tip of the catheter is moved to the region of the vein to be treated closest to the pelvis. The device is turned on, delivering the sclerotherapy solution and causing abrasions to promote scarring of the vein. Once the entire vein is treated, the device and wire are removed. Compression garments are needed for 12–24 hours after the procedure to promote vein scarring and closure.
Rare short-term side effects of MOCA include phlebitis, infection, bleeding, and bruising. Patients can develop a superficial blood clot; DVT is rare. Patients who have MOCA will need an ultrasound several days after the procedure to look for blood clots.
MOCA and CAG are the only two methods for vein treatment that are not heat-based and do not require significant numbing medication. Though phlebitis is associated with both MOCA and CAG, the inflammation may last longer with CAG treatment. Both MOCA and endovenous ablation require compression garments for 12–24 hours after the procedure to ensure scarring of the treated vein.
Recovery after venous procedures
Recovery from all superficial venous procedures is typically brief, with the majority of patients able to return to work or daily activities the next day (Table 2). If any oral sedatives are used during the procedure, patients will need someone else to drive them home. After the procedure, adhesive bandages will be used to close any small incisions in the skin. These will fall off on their own. With the exception of CAG, the area of treated veins will be wrapped in an elastic bandage to decrease swelling and promote closure of the vein. Patients should elevate their legs and wear the elastic bandages for as long as tolerated for 12–24 hours after the procedure. Patients may shower, but prolonged submersion in water should be avoided to decrease the risk of infection. After showering, the compression bandage can be re-applied for comfort.
Recovery after superficial venous procedures.

For pain control and minor discomfort, over-the-counter analgesics such as ibuprofen and acetaminophen can be used. During the first 48 hours, a cold compress can be directly applied to treated areas for 15–20 minutes at a time to improve discomfort.
For patients who undergo treatment of the GSV or SSV, an ultrasound is performed a few days after therapy. This ultrasound is to ensure that the vein has been treated appropriately and to detect any evidence of blood clots that might require treatment. Patients who develop blood clots may need a blood thinner (especially if the clot involves the deep veins, a DVT), or they may need a follow-up ultrasound to monitor the size and location of the clot.
Conclusion
Therapies for superficial venous disease are safe, effective, and can typically be performed in an office setting. Treatment options are based on which veins are involved. With the majority of these procedures, patients can safely return to their daily tasks the next day.
The ‘Vascular Disease Patient Information Page’ is a regular feature of Vascular Medicine. All articles in the collection are available for free online at http://journals.sagepub.com/vmjpatientpage.
The Vascular Disease Patient Information Page is provided for educational purposes only and is not a substitute for medical advice.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
