Abstract
Objective:
Under diabetic conditions, glucose is converted to sorbitol via aldose reductase, whose process could contribute to diabetic vascular complications. However, effects of aldose reductase inhibitors are modest in diabetic patients. This may be attributed to weak inhibitory activity of aldose reductase inhibitors. We compared effects of ranirestat on endothelial cell damage with those of epalrestat.
Materials and methods:
Intracellular formations of sorbitol and superoxide were measured by liquid chromatography–mass spectrometry–mass spectrometry and dihydroethidium staining, respectively. Vascular cell adhesion molecule-1 gene expression was analysed by reverse transcription polymerase chain reaction. THP-1 cell adhesion to human umbilical vein endothelial cells was evaluated using a fluorescent probe.
Results:
High glucose significantly increased sorbitol levels, superoxide generation and vascular cell adhesion molecule-1 mRNA levels in, and THP-1 cell adhesion to, human umbilical vein endothelial cells, all of which were prevented by 500 nM ranirestat, but not epalrestat except for superoxide production.
Conclusion:
Our present results suggest that ranirestat has a stronger inhibitory activity on aldose reductase than epalrestat and suppresses inflammatory reactions in high glucose–exposed human umbilical vein endothelial cells.
Introduction
Diabetes is highly prevalent all over the world and is a growing health problem. 1 Despite intensive management of hyperglycaemia, hypertension and dyslipidaemia, a substantial number of diabetic patients still have experienced vascular complications, which could account for disabilities and high mortality rate in patients with diabetes. 1 Although various hyperglycaemia-associated biochemical and hemodynamic changes contribute to the development and progression of vascular complications in diabetes, endothelial cell (EC) damage is considered to be an initial event of diabetic micro- and macroangiopathy. 2 Therefore, protection of ECs against hyperglycaemic injury may be a therapeutic target for vascular complications in diabetes.
Under hyperglycaemic conditions, glucose is catalysed to sorbitol via aldose reductase (AR), a rate-limiting enzyme of the polyol pathway, whose process has been shown to play a pathological role in diabetic neuropathy in addition to other vascular complications.2–4 Numerous structurally distinct AR inhibitors (ARIs) are developed, some of which had been advanced to clinical trials. 5 However, epalrestat is the sole ARI which is clinically available for the treatment of diabetic neuropathy until now. 5 Furthermore, although a few studies with small number of patients reported that epalrestat modestly prevented the progression of diabetic retinopathy and/or nephropathy, there is no clinical evidence to show the beneficial effects of epalrestat on EC injury or macrovascular complications in diabetes.6,7 In addition, high glucose–induced EC damage was only suppressed by high concentrations of epalrestat (⩾10 µM).8,9 Since recommended oral dose of epalrestat is 50 mg three times a day and its plasma concentration up to 8 h after an administration of 50 mg is about 0.5–10 µM, weak or incomplete AR inhibitory activity of epalrestat may account for its modest or ambiguous vascular protection. Therefore, in this study, we compared the protective effects of ranirestat, a well-tolerated front-line ARI against EC damage with those of a classical ARI, epalrestat. 10
Materials and methods
Human umbilical vein endothelial cells (HUVECs) obtained from Lonza Group Ltd (Basel, Switzerland) were cultured in endothelial basal medium-2 (glucose concentration; 5.5 mM) supplemented with 2% foetal bovine serum, human fibroblast growth factor, heparin, insulin-like growth factor-1, human epidermal growth factor, vascular endothelial growth factor, hydrocortisone and ascorbic acid according to the manufacturer’s recommendation. HUVECs were treated with or without 24.5 mM glucose (high glucose) in the presence or absence of 50 nM, 500 nM ranirestat or 500 nM epalrestat in basal medium-2 with 2% foetal bovine serum, human fibroblast growth factor and heparin for 24 h. Then, according to the method of Liang et al., 11 sorbitol contents were measured by liquid chromatography tandem mass spectrometry, which consisted of an SIL-HTC and LC-10A (Shimadzu Corp., Kyoto, Japan) and the API4000 tandem mass spectrometer (Applied Biosystems/MDX SCIEX, MA, USA) with atmospheric pressure chemical ionization. The column and autosampler temperatures were 35°C and 10°C, respectively. The separation was performed on an CAPCELL PAK NH2 UG80 (5 µm, 4.6 mm ID × 250 mm L.; Shiseido Co., Ltd, Tokyo, Japan) using a mixture of 0.1% dichloromethane-acetonitrile/water (90/10, v/v) at a flow rate of 1 mL/min. The analytical run time was 18 min. The monitored ion was used for 217 m/z → 181 m/z for sorbitol. Superoxide generation was evaluated by dihydroethidium staining (DHE); vascular cell adhesion molecule-1 (VCAM-1) and 18S gene expression was determined by quantitative real-time reverse transcription-polymerase chain reactions using Hs01003372_m1 and Hs99999901_s1 primers, respectively; and THP-1 cell adhesion to HUVECs was assayed with a fluorescent probe as described previously. 12
All values were presented as mean ± standard error. Dunnett’s test was performed for multiple statistical comparisons; p < 0.05 was considered significant.
Results
As shown in Figure 1(a), high concentration of ranirestat (500 nM) or 500 nM epalrestat, but not low concentration of ranirestat (50 nM), significantly reduced sorbitol contents in normal glucose-exposed HUVECs. High glucose exposure for 24 h significantly increased sorbitol levels in HUVECs, which was dose-dependently suppressed by the treatment with ranirestat (Figure 1(b)). However, 500 nM epalrestat did not decrease sorbitol levels in high glucose–exposed HUVECs.
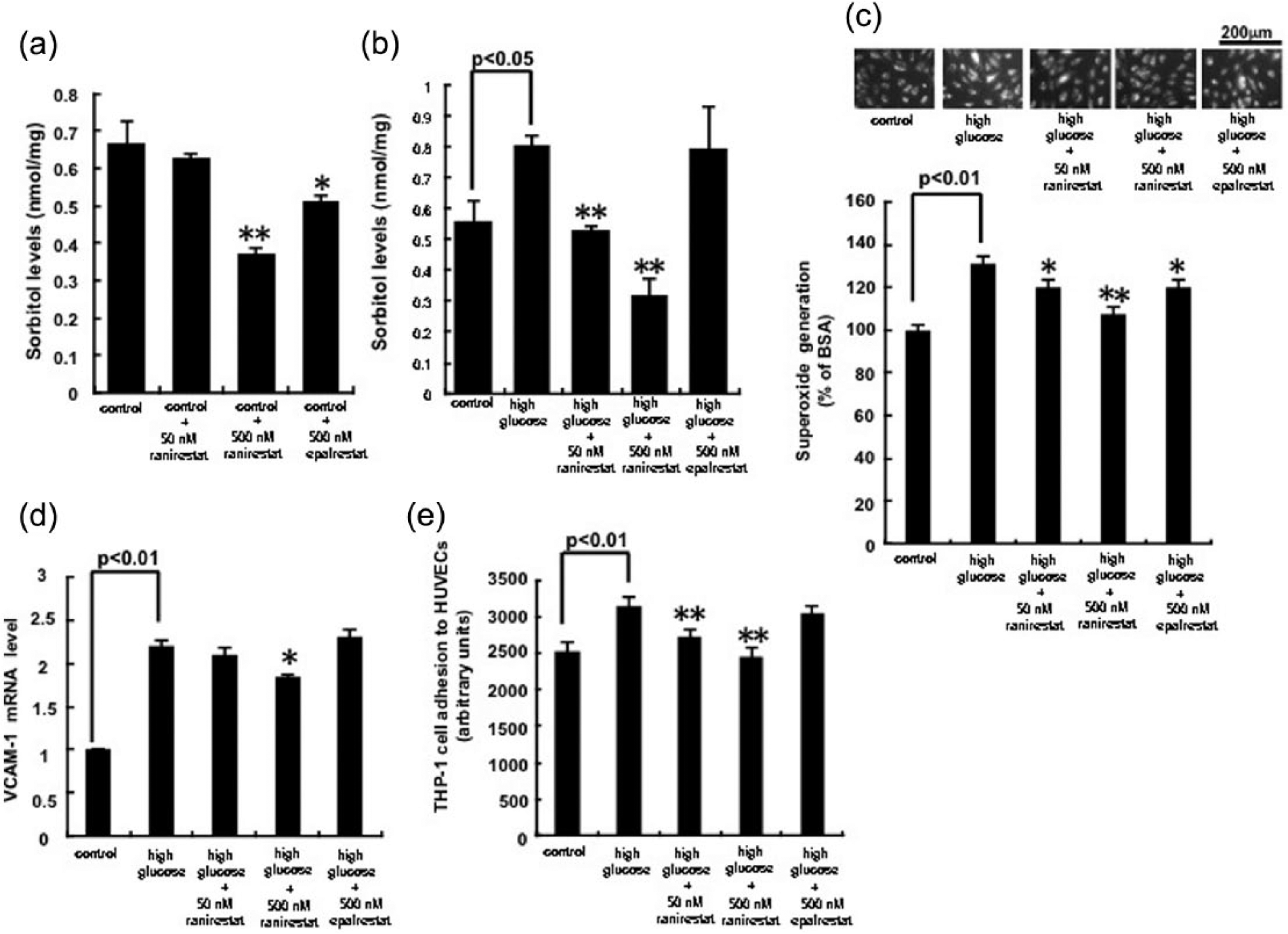
(a, b) Effects of ranirestat or epalrestat on intracellular sorbitol levels, (c) superoxide generation, (d) VCAM-1 gene expression in, and (e) THP-1 cell adhesion to, 5.5 mM glucose (control)- or high glucose–exposed HUVECs. HUVECs were treated with or without 24.5 mM glucose (high glucose) in the presence or absence of 50 nM, 500 nM ranirestat or 500 nM epalrestat in basal medium-2 with 2% foetal bovine serum, human fibroblast growth factor and heparin for 24 h. (a and b) Then sorbitol contents were measured N = 4–6 per group. (c) Each upper panel shows typical microphotographs of the cells. Each lower panel shows quantitative data of superoxide generation evaluated by fluorescent intensity. Intensity of DHE staining in five different fields of each sample was analysed by microcomputer-assisted image J. N = 3 per group. (d) Total RNAs were transcribed and amplified by real-time PCR. Data were normalized by the intensity of 18S mRNA–derived signals and then related to the control values. N = 3 per group. (e) THP-1 cell adhesion to HUVECs was evaluated. N = 3 per group. (a) *p < 0.05 and **p < 0.01 compared to the control values, respectively. (b to e) *p < 0.05 and **p < 0.01 compared to the values with high glucose, respectively.
As shown in Figure 1(c) to (e), high glucose treatment significantly increased superoxide generation and VCAM-1 mRNA levels in, and THP-1 cell adhesion to, HUVECs, all of which were prevented by ranirestat in a dose-dependent manner. In contrast to the case with 500 nM ranirestat, 500 nM epalrestat did not affect these parameters except for superoxide production.
Conclusion
In this study, we found for the first time that 50–500 nM ranirestat, but not 500 nM epalrestat, completely inhibited the high glucose–induced sorbitol accumulation in HUVECs. Plasma concentration of ranirestat after administration of 40 mg once a day, a recommended oral dose of ranirestat, is about 1–10 µM, whose level is nearly equal to that of epalrestat. Therefore, our present findings suggest that ranirestat has a stronger inhibitory activity on AR than epalrestat. In support of this speculation, we have recently found that inhibitor constant (Ki) of ranirestat against AR is about 250-fold lower than that of epalrestat (0.38 vs 89 nM).
Activation of the polyol pathway under hyperglycaemic conditions has been shown to contribute to the pathogenesis of diabetic neuropathy.2–4 However, its causal role in the development and progression of other vascular complications in diabetes remains ambiguous. In this study, we found that 500 nM ranirestat, a comparable level that is achieved in the treatment for diabetic patients with neuropathy, inhibited oxidative stress generation and VCAM-1 gene expression in high glucose–exposed HUVECs. AR pathway has been known to stimulate oxidative stress generation in ECs via various mechanisms. 2 VCAM-1 gene expression is regulated by redox-sensitive transcriptional factor, nuclear factor-κB. 2 Therefore, it is conceivable that ranirestat may block the high glucose–induced oxidative stress and inflammatory reactions in HUVECs partly via the suppression of AR pathway. The recruitment and firm adhesion of inflammatory cells to ECs are one of the earliest characteristic changes in diabetic retinopathy, nephropathy and atherosclerosis, whose process is mainly mediated by adhesion molecules, such as VCAM-1. 2 Furthermore, we have previously shown that fidarestat, another type of ARI with Ki of 1.9 nM, inhibits high glucose–induced pericyte damage and prevents retinal hyperpermeability in diabetic rats, 13 thus suggesting that complete inhibition of AR may also be a therapeutic target for diabetic retinopathy. These observations indicate that ranirestat is a newly developed, more potent ARI and may protect against vascular complications in diabetes. Further clinical studies are needed to examine whether ranirestat treatment efficiently blocks the development and progression of vascular complications, including neuropathy and retinopathy in patients with diabetes.
Footnotes
Acknowledgements
S. Yamagishi conceptualized and designed the study; acquired, analysed and interpreted data; and drafted the manuscript; and he takes responsibility for the integrity of the data and the accuracy of the data analysis. Y. Ishibashi, T. Matsui and T. Matsumoto acquired, analysed and interpreted data. H. Kato acquired, analysed and interpreted data and critically revised the manuscript for important intellectual content. We thank Chie Kohayakawa, Drug Research Division, Sumitomo Dainippon Pharma Co., Ltd, Osaka, Japan, for her excellent technical assistance.
Declaration of conflicting interests
Dr Yamagishi has received honoraria such as lecture fees from Sumitomo Dainippon Pharma Co., Ltd. T. Matsumoto and H. Kato are employees of Sumitomo Dainippon Pharma Co., Ltd, Japan.
Funding
This project was financially supported in part by Sumitomo Dainippon Pharma Co., Ltd, Japan. This work was supported in part by Grants-in-Aid for Scientific Research B (grant number 22390111) (S.Y.) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
