Abstract
Adequate vitamin D levels may promote cardiovascular health by improving endothelial function and down-regulating inflammation. The objective of this pilot trial was to investigate the effects of vitamin D repletion on endothelial function and inflammation in patients with coronary artery disease (CAD). Using a double-blind placebo wait-list control design, 90 subjects with CAD and vitamin D deficiency (< 20 ng/ml) were randomized 1:1 to 50,000 IU of oral ergocalciferol or placebo weekly for 12 weeks. Endothelial function (reactive hyperemia peripheral arterial tonometry, RH-PAT), circulating adhesion molecules, and pro-inflammatory cytokines were measured at baseline and 12 weeks. The median increase in serum 25-vitamin D from baseline was 26 ± 17 ng/ml in the active group and 4 ± 8 ng/ml in the placebo group (between-group difference = 22 ng/ml, p < 0.001). The median within-subject change in RH-PAT score was 0.13 ± 0.73 with active treatment and −0.04 ± 0.63 with placebo (between-group difference = 0.17, p = 0.44). Within-group and between-group differences in intercellular adhesion molecule levels were greater with placebo (between-group difference = 6 ng/ml, p = 0.048). Vascular cell adhesion molecule levels decreased in both groups by a similar magnitude (median difference between groups = 8.5 ng/ml, p = 0.79). There was no difference between groups in magnitude of reduction in interleukin (IL)-12 (−8.6 ng/ml, p = 0.72) and interferon-gamma (0.52 ng/ml, p = 0.88). No significant differences in blood pressure, e-selectin, high-sensitivity c-reactive protein, IL-6 or the chemokine CXCL-10 were found with treatment. In conclusion, repleting vitamin D levels in subjects with CAD failed to demonstrate any benefits on surrogate markers of cardiovascular health. These results question the role of vitamin D supplementation in modifying cardiovascular disease.
Keywords
Introduction
Vitamin D deficiency remains highly prevalent amongst community-dwelling populations1–3 in urban and multi-ethnic low-income northern US cities, where cardiovascular disease (CVD) is also endemic.4–8 Adequate vitamin D levels may promote cardiovascular (CV) health by improving endothelial function and down-regulating systemic inflammation.9,10 Accumulating epidemiologic evidence linking low vitamin D status to increased CV morbidity and mortality11–14 has contributed to large increases in serum vitamin D testing and consumption of supplements in the United States.15,16
Nevertheless, critical questions about the role of vitamin D in CV health remain unanswered. Does vitamin D supplementation reduce the risk of developing coronary artery disease (CAD) and improve the outcomes of patients with established CAD? The Institute of Medicine (IOM) in a recent update noted the paucity of causal evidence supporting a benefit for vitamin D in CV health. 17 The issue of CVD prevention is the subject of two large, randomized, placebo-controlled trials (VITAL and ViDA) to be completed over the next 6 years.18,19 At the same time, the impact of vitamin D repletion on markers relevant to active CVD in subjects with CAD has not been investigated.
We have completed a randomized pilot trial to determine the effect of vitamin D repletion on endothelial function, serum adhesion molecules and pro-inflammatory cytokines in subjects with vitamin D deficiency and established CAD. We hypothesized that vitamin D repletion will improve endothelial function, and decrease the expression of adhesion molecules and pro-inflammatory cytokines relevant to the pathogenesis of CAD and its complications.
Methods
The study was a randomized, double-blind, placebo-controlled trial. A wait-list design was used to allow subjects on placebo to be treated with open-labeled vitamin D after the conclusion of the study. The study protocol was approved for all recruiting sites by the Institutional Review Board of the Albert Einstein College of Medicine (Bronx, NY, USA) and all subjects provided witnessed informed consent.
Subjects were recruited from October 2008 through December 2010 from the cardiac catheterization laboratories and outpatient clinics of the Jacobi Medical Center and Montefiore Medical Center in the northeastern section of the Bronx, NY. Additional recruitment occurred at Crystal Run Health in Orange County, NY. Subjects greater than 18 years of age with ≥ 50% angiographic stenosis of at least one coronary artery or documented previous revascularization were screened for vitamin D deficiency by measurement of serum 25-hydroxy vitamin D (25-vitamin D). Exclusion criteria included confinement to a nursing facility, institution or home, glomerular filtration rate (GFR) < 60 ml/min (by modification of diet in renal disease (MDRD) equation), presence of liver disease, hypercalcemia, New York Heart Association (NYHA) class III or IV heart failure, cardiogenic shock at time of presentation, current planned or emergent coronary artery bypass graft, prior gastric or small bowel surgery, pancreatitis, malabsorption, inflammatory bowel disease, autoimmune disease, active malignancy, current use of > 800 IU/day of vitamin D, Dilantin, phenobarbitol, immunosuppressant, or immunostimulant therapy.
Eligible subjects with a 25-vitamin D level < 20 ng/ml were randomly assigned 1:1, in a double-blind manner from computer-generated random number lists, to active treatment with 50,000 IU per week of ergocalciferol or placebo for 12 weeks. The randomization visit (baseline) was conducted 4 weeks after screening for subjects with acute coronary syndromes (ACS) or recent percutaneous coronary interventions to allow adequate clinical recovery time and to assure adjustment to appropriate guideline-based medications. Subjects were instructed not to use any form of dietary vitamin D supplementation during the study period and to continue all home medications with the exception of non-study vitamin D. Double-blinding was maintained until the conclusion of the 12-week study period, after which subjects on placebo with persistent vitamin D deficiency were prescribed open label vitamin D replacement. Calcium levels were drawn at week 4 as a safety measure. Queries on sun exposure and changes in dietary habits were made at baseline, week 4 and at the conclusion of the study period. At the week 4 and 12 visits, study medication compliance was assessed via pill count and confirmed by measurement of the vitamin D2 subfraction at the conclusion of the study.
Reactive hyperemia peripheral arterial tonometry (RH-PAT)
RH-PAT (EndoPAT 2000; Itamar Medical, Caesarea, Israel) was performed in all subjects as a primary endpoint, in a quiet temperature-controlled room in the supine position after an overnight fast, at baseline and after 12 weeks of treatment. The technique has been described in detail elsewhere. 20 Briefly, after a 15-minute rest period, RH-PAT pneumatic plethysmograph probes were placed on the index finger of each hand. A blood pressure cuff was placed on one arm and the opposite arm served as the control. Continuous recording of the pulse wave tracing was performed for 10 minutes until the signal equilibrated. The blood pressure cuff was then inflated above systolic blood pressure for 5 minutes. PAT recording was continued during cuff inflation, and for 10 minutes after cuff deflation. An RH-PAT score was calculated as the ratio of the average amplitude of the peripheral arterial tonometry signal over a 1-minute time interval starting 60 seconds after cuff deflation divided by the average amplitude of the PAT signal of a 3.5-minute time period before cuff inflation (baseline) and was adjusted for systemic changes as reflected in the recording in the contralateral control fingertip.
A Framingham study-derived modification of the RH-PAT score (Framingham RH index or FRHI) was also calculated using the natural logarithm of the PAT ratio during the 90–120-second time period post-cuff deflation as described previously, 21 without baseline correction; FRHI = ln (average amplitude occluded arm (90–120 s)/average amplitude control arm (90–120 s)). The 90–120-second time period has been shown to have the strongest relationship with CV risk factors.
Endothelial function was measured by RH-PAT in a group (n = 21) of healthy age-matched controls (AKG; Pennington Biomedical Research Center, Louisiana State University System, Baton Rouge, LA, USA). This group had no CV risk factors, was free of known CAD, and was not taking any prescription or non-prescription medications (Appendix Table 1).
Blood pressure was measured as a secondary endpoint, at baseline and at week 12 in the seated position after adequate rest. Three measurements were made 5 minutes apart and the final two measurements were averaged and used for analysis.
Laboratory testing
Serum 25-hydroxy vitamin D (mass-spectroscopy liquid chromatography; Quest Diagnostics, Teterboro, NJ, USA) was measured from screening and week 12 samples. The lower limit of detection was 4 ng/ml. Results are reported as total 25-hydroxy vitamin D and vitamin D2 and D3 fractions. The hs-CRP (high-sensitivity c-reactive protein) (high-sensitivity latex turbidimetric immunoassay; Olympus Diagnostica, Olympus America Inc; performed at the Clinical Research Center Core Laboratory, Albert Einstein College of Medicine (AECOM), Bronx, NY), serum calcium, intact parathyroid hormone (PTH), blood urea nitrogen (BUN) and creatinine (Jacobi Medical Center Clinical Laboratory, Bronx, NY) were measured at baseline and week 12. Circulating cytokine levels of interleukin-12 (IL-12), interferon-gamma (IFN-γ), the chemokine CXCL-10 (laboratory of GT, Yale University School of Medicine, New Haven, CT), and adhesion molecules s-VCAM (soluble vascular cell adhesion molecule), s-ICAM (soluble intercellular adhesion molecule), and soluble e-selectin (Immunology and Pathology Core Laboratory, AECOM) were measured by enzyme-linked immunosorbent assay (ELISA) (R&D Systems, Minneapolis, MN). Changes in inflammatory marker levels were considered as a primary endpoint.
Plasma levels of IL-12, IFN-γ and CXCL-10 from a referent group of 10 healthy volunteers (GT, Yale University School of Medicine) free of known CAD, or any chronic medical conditions, was used to demonstrate normal values of these markers (Appendix Table 1).
Statistical analysis
Standard deviations (σ) of RH-PAT scores and inflammatory variables measured at a single time point were obtained from our prior studies.22,23 Assuming a standard deviation of 0.6 in the change in RH-PAT score from baseline to 12 weeks, we estimated that the study would need a sample size of 45 patients per treatment group to have 80% power at a two-tailed alpha = 0.05 level to detect a minimum difference in change in RH-PAT score of 0.36 between treatment groups. To allow for an expected drop-out rate of 10%, we enrolled 50 subjects per treatment group. All analyses were based on the intent-to-treat approach. Continuous variables were summarized as mean ± standard deviation or median ± interquartile range if the data were skewed. Differences between treatment groups in baseline characteristics were evaluated with the two-sample t-test or Wilcoxon rank sum test depending on the distribution of the continuous variables. The chi-square test was used to compare categorical variables between groups. Differences in change from baseline in endothelial function, adhesion molecules, and inflammatory markers between treatment groups were evaluated by fitting analysis of covariance (ANCOVA) models to adjust for baseline levels in these variables. Rank transformations were applied prior to model fitting when the data were non-normally distributed. Spearman rank correlation coefficients were used to estimate correlations between different measures of endothelial function. A two-sided p-value < 0.05 was considered statistically significant. Analyses were performed with SAS software version 9.2 (Cary, NC, USA).
Results
A summary of the study design is provided in Figure 1. The prevalence of vitamin D deficiency was 46.3% in subjects with CAD. Of the subjects randomized, six withdrew for non-clinical reasons and 90 completed the trial. There were no significant differences in baseline characteristics between active and placebo groups (Table 1), including season of randomization. Compliance was 99% by pill count but two subjects in the active group did not have an increase in vitamin D2 subfraction, suggesting non-compliance or malabsorption. D2 increased substantially in one subject on placebo, suggesting unreported use of D2 supplementation. No significant side effects of therapy were reported, and calcium levels remained stable in all subjects.
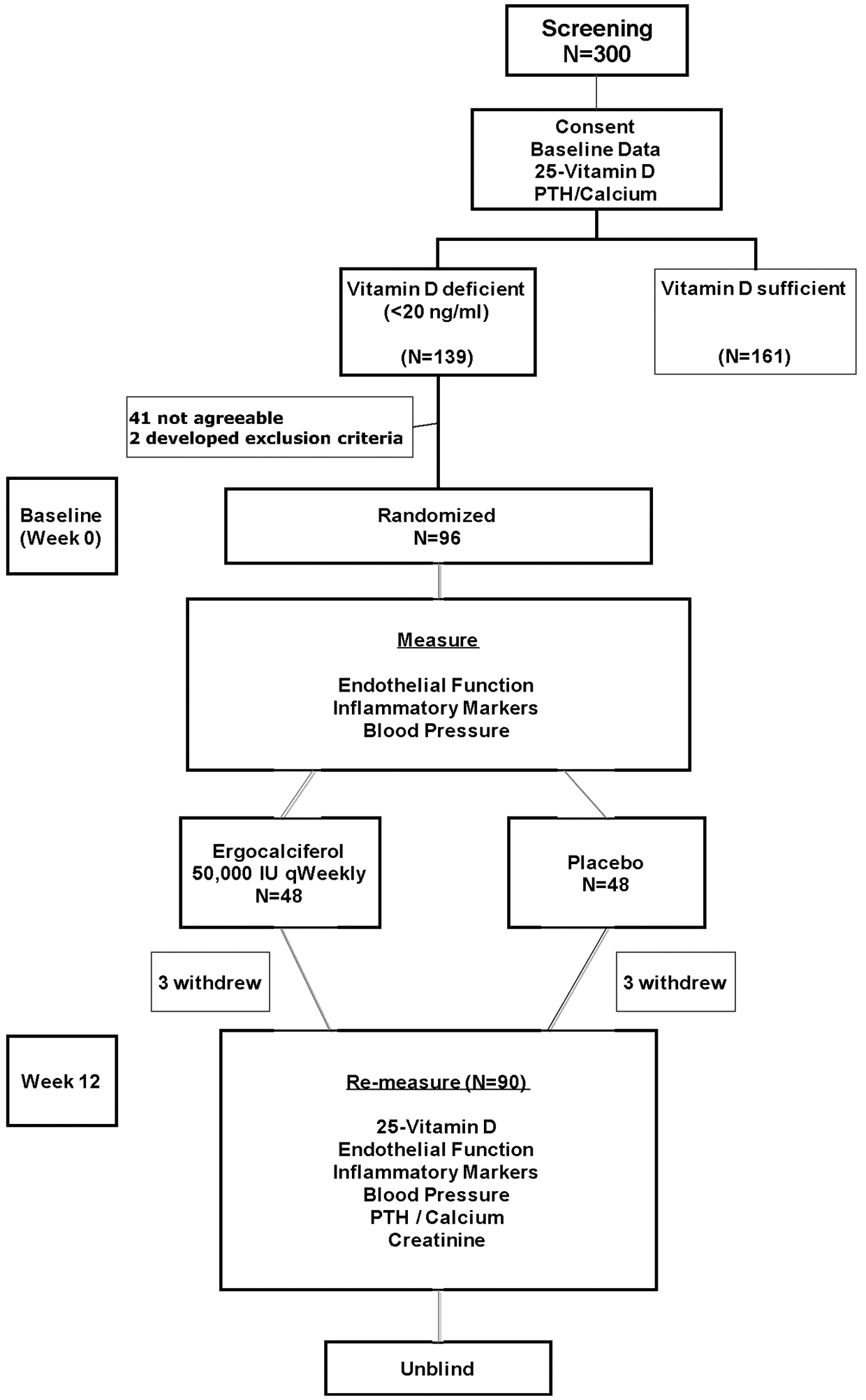
Study design. Of 300 subjects screened, 139 were vitamin D deficient, of which 96 were eventually randomized to active or placebo treatment. Of the 96 subjects randomized, six withdrew for non-clinical reasons leaving 45 subjects in each group who completed the trial.
Baseline characteristics of the 90 randomized subjects with vitamin D deficiency and CAD who completed the study
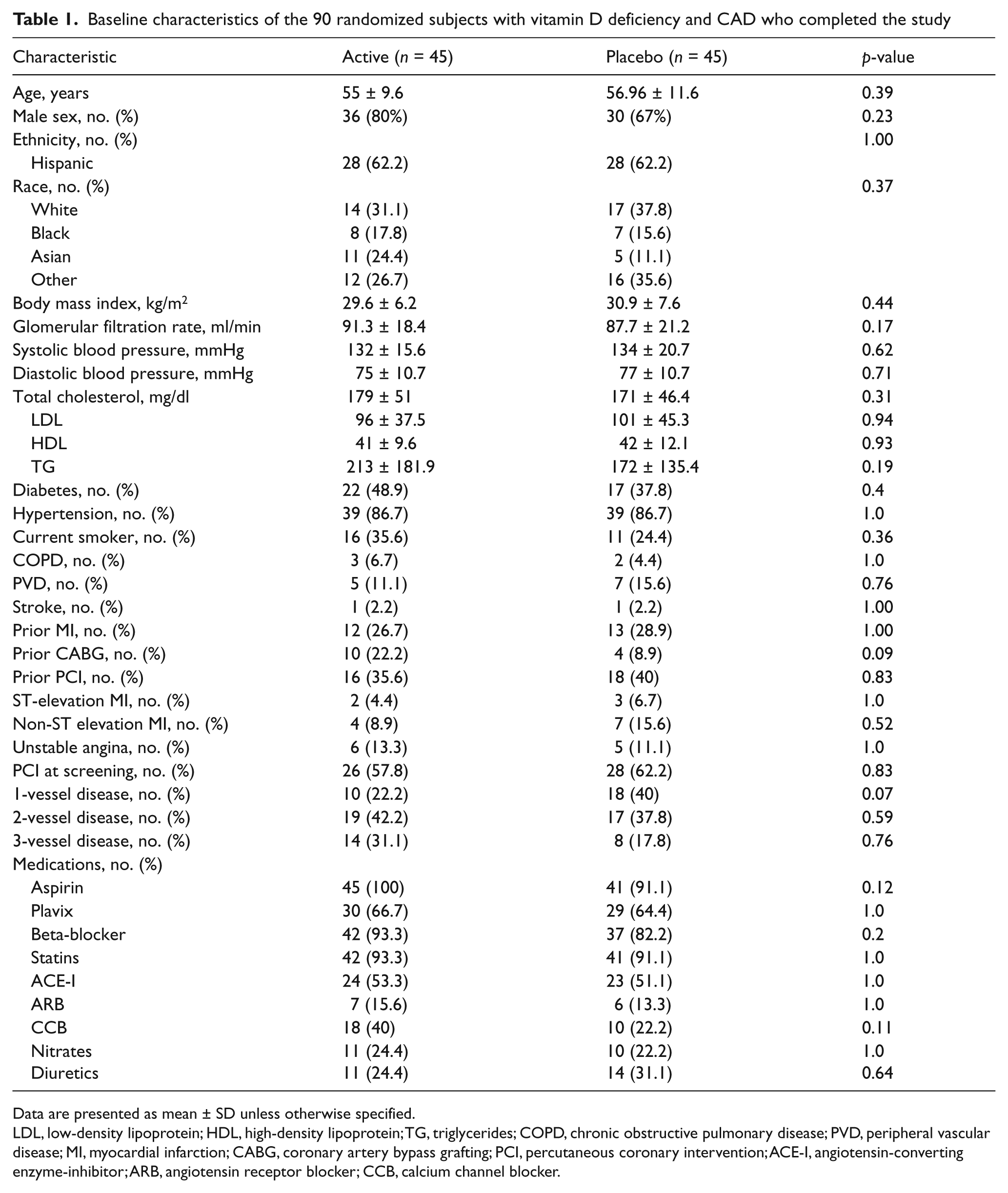
Data are presented as mean ± SD unless otherwise specified.
LDL, low-density lipoprotein; HDL, high-density lipoprotein; TG, triglycerides; COPD, chronic obstructive pulmonary disease; PVD, peripheral vascular disease; MI, myocardial infarction; CABG, coronary artery bypass grafting; PCI, percutaneous coronary intervention; ACE-I, angiotensin-converting enzyme-inhibitor; ARB, angiotensin receptor blocker; CCB, calcium channel blocker.
In the randomized cohort, median (± interquartile range) levels of 25-vitamin D increased from baseline by 26 ± 17 ng/ml (p < 0.001) in the active group and 4 ± 8 ng/ml (p < 0.001) in the placebo group (between-group difference = 22 ng/ml, p < 0.001) (Figure 2). Increases in the active group were mostly explained by ergocalciferol treatment (median change in vitamin D2 = 35 ± 19, p < 0.001), and by increases in vitamin D3 in the placebo group (median change = 3.0 ± 7 ng/ml, p = 0.01) (Table 2).
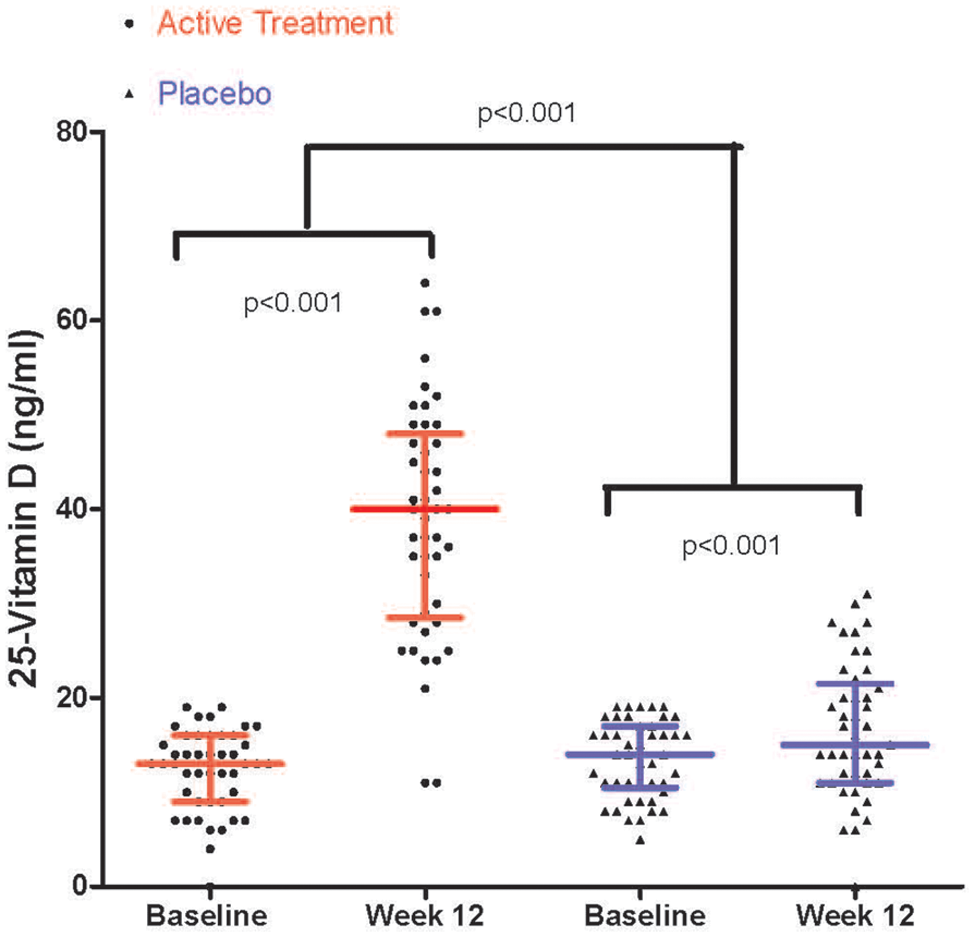
25-Vitamin D levels pre- and post-treatment – active vs placebo. Median (± interquartile range) levels of 25-vitamin D at each time point increased from 13 (± 7) ng/ml to 40 (± 18) ng/ml in the active group and from 14 (± 6) ng/ml to 15 (± 10) ng/ml in the placebo group. The difference in median change between the two groups was significant (p < 0.001).
Comparison of baseline and post-treatment levels of vitamin D, endothelial function, adhesion molecules and pro-inflammatory markers
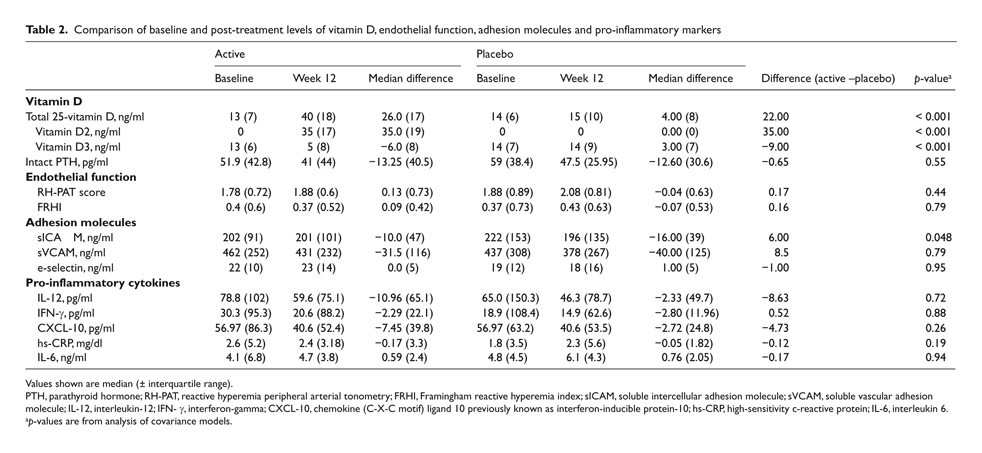
Values shown are median (± interquartile range).
PTH, parathyroid hormone; RH-PAT, reactive hyperemia peripheral arterial tonometry; FRHI, Framingham reactive hyperemia index; sICAM, soluble intercellular adhesion molecule; sVCAM, soluble vascular adhesion molecule; IL-12, interleukin-12; IFN- γ, interferon-gamma; CXCL-10, chemokine (C-X-C motif) ligand 10 previously known as interferon-inducible protein-10; hs-CRP, high-sensitivity c-reactive protein; IL-6, interleukin 6.
p-values are from analysis of covariance models.
Endothelial function
Mean RH-PAT scores at baseline were significantly lower in our cohort as compared to healthy aged-matched controls (1.62 ± 0.3 vs 2.18 ± 0.6, respectively; p = 0.0004) (Appendix Table 1).
In our cohort, the RH-PAT score and FRHI correlated directly with each other (r = 0.9; p < 0.0001). Endothelial function was similar at baseline across active and placebo groups as shown in Table 2. The median within-subject change in the RH-PAT score in the active group was 0.13 (± 0.73, p = 0.85) and −0.04 (± 0.63, p = 0.43) within placebo, without significant differences between groups (0.17, p = 0.44). Similar results were found using the FRHI (median change: 0.09 (± 0.42), p = 0.59 for active; −0.07 (± 0.53), p = 0.94 for placebo; between-group = 0.16, p = 0.79) (Table 2, Figure 3).
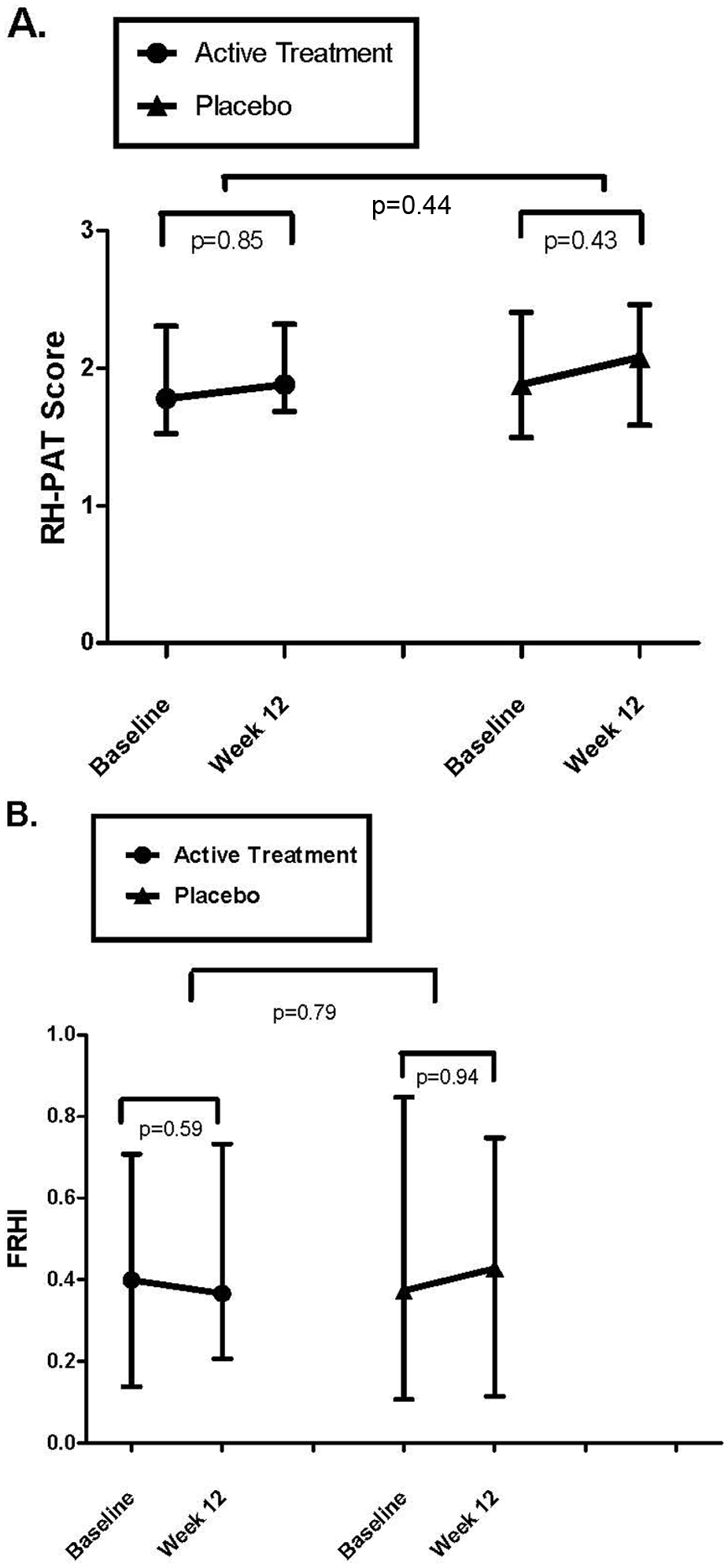
RH-PAT scores and FRHI pre- and post-treatment – active vs placebo. (A) Median (± interquartile range) RH-PAT score at each time point increased from 1.78 (± 0.72) to 1.88 (± 0.6) with active treatment (p = 0.85) and from 1.88 (± 0.89) to 2.08 (± 0.81) with placebo (p = 0.43). The difference in median change between groups was not significant (p = 0.44). (B) Median FRHI at each time point decreased from 0.4 (± 0.6) to 0.37 (± 0.52) with active treatment (p = 0.59) and increased from 0.37 (± 0.73) to 0.43 (± 0.63) with placebo (p = 0.94). The difference in median change between groups was not significant (p = 0.79). (RH-PAT, reactive hyperemia peripheral arterial tonometry; FRHI, Framingham reactive hyperemia index.).
Adhesion molecules
Baseline levels of adhesion molecules were similar between groups except for e-selectin (p = 0.03) (Table 2). The median within-subject change in ICAM levels from baseline to post-treatment was −10 (± 47) ng/ml in the active group and −16 (± 39) ng/ml in the placebo group. The between-group difference in change in ICAM levels from baseline was significant (6 ng/ml, p = 0.048). Median VCAM levels decreased in both groups by a similar magnitude (−31.5 ± 115.7 ng/ml active vs −40 ± 125 ng/ml placebo; median between-group difference = 8.5 ng/ml, p = 0.79).
Inflammatory markers
Baseline IL-12, IFN-γ and CXCL-10 were similar between groups (Table 2). The median within-subject change in IL-12 levels from baseline to post-treatment was −11.0 ± 65 ng/ml (p = 0.04) with active treatment and −2.3 ± 50 ng/ml (p = 0.03) with placebo, but the between-group difference was not significant (−8.6 ng/ml, p = 0.72). Similarly, IFN-γ decreased in the active (−2.3 ± 22 ng/ml, p = 0.12) and placebo (−2.8 ± 12 ng/ml, p = 0.03) groups, but the between-groups difference was not significant (0.52 ng/ml, p = 0.88). There were no significant changes in CXCL-10, hs-CRP or IL-6 with active or placebo treatment.
Effect of severity of vitamin D deficiency
The analysis was repeated with the active and placebo groups stratified by severity of vitamin D deficiency (mild–moderate deficiency: ≥ 10 to < 20 ng/ml vs severe deficiency: < 10 ng/ml). In the severely deficient category the magnitude of decrease in VCAM levels was significantly greater in the active group as compared to placebo. However, in those with mild–moderate deficiency, VCAM levels also declined but by a greater magnitude in the placebo group. Analysis of RH-PAT scores, FRHI, and all other inflammatory markers failed to show any significant differences based on baseline severity of deficiency, despite adequate repletion of vitamin D levels in the active group.
Effect of ACS on baseline measurements
At baseline, median levels of IL-12 and IFN-γ trended higher in subjects with ACS (measured 4 weeks after event) as compared to those with chronic stable CAD. There were no other notable differences in any measured pro- inflammatory cytokine, adhesion molecule or endothelial function at baseline after stratification by presence or absence of ACS (Appendix Table 2).
Blood pressure
The mean systolic blood pressure (SBP) and diastolic blood pressure (DBP) were similar in active and placebo groups (130/76 mmHg vs 133/77 mmHg, respectively; p for SBP = 0.38, p for DBP = 0.6) at baseline. There were no significant changes in blood pressure after 12 weeks of treatment in either group.
Discussion
In subjects with vitamin D deficiency and documented CAD, repletion of vitamin D over a 12-week period did not improve endothelial function (as measured by RH-PAT) or blood pressure. Decreased expression of the adhesion molecule VCAM and the cytokine IL-12 were observed with active treatment; however, changes of a similar direction and magnitude were also seen with placebo treatment. ICAM expression decreased significantly but only with placebo.
Our results are surprising given the available biologic evidence supporting a role for vitamin D in modulating vascular function. This evidentiary trail includes in vitro findings of vitamin D receptors on human endothelial cells, the ability of active vitamin D to inhibit the expression of adhesion molecules on these cells,24,25 and in vivo studies in animals demonstrating reductions of blood pressure and endothelial-dependent contractions in spontaneously hypertensive rats treated chronically with vitamin D.26,27 Additionally, recent observational and small interventional trials have linked low vitamin D status to poor vascular health, 28 and vitamin D sufficiency or repletion to improvement in brachial artery flow-mediated vasodilatation (FMD), reactive hyperemia index, aortic stiffness, and blood pressure.28–30 These studies, however, were cross-sectional/observational, 28 non-randomized, and included only healthy asymptomatic subjects 29 and adolescents. 30
We are unaware of any prior studies evaluating the effects of vitamin D repletion on endothelial function in subjects with established CAD and documented vitamin D deficiency at baseline. Given that changes in endothelial function were detectable with repletion in small trials of healthy subjects over relatively short periods of time, we expected to detect changes in our subjects who likely had advanced or ongoing vasomotor dysfunction. Additionally, it was important to investigate this cohort as the persistence of endothelial vasomotor dysfunction despite optimized conventional therapy for CAD is associated with worse outcomes. 31 At baseline the median RH-PAT score in our cohort was above threshold scores correlated with coronary endothelial dysfunction (≤ 1.67). 32 However, when we age-matched our subjects to a group of 21 healthy adults, the mean RH-PAT score in our cohort was significantly lower, confirming that our subjects had poorer endothelial function at baseline (as measured by RH-PAT). We also reanalyzed the data in subsets with a baseline RH-PAT score of 1.67 or less. Mean RH-PAT scores increased significantly in the active and placebo groups but the magnitude of change was the same between groups (data not shown). Given the high use of ACE inhibitors, the further improvement of angiotensin receptor blockers (ARBs) and statins in endothelial function beyond that attributed to these medications may not have been achievable or measurable in our subjects. This was suggested in two recent randomized trials of vitamin D repletion in patients with type 2 diabetes 33 and prior stroke 34 and high use of statins and ACE-I, where no measurable improvement in endothelial function, evaluated using flow-mediated dilatation (FMD) of the brachial artery, was found at the conclusion of the trials.
Overall, our results suggest that vitamin D repletion adds little to improving endothelial function above and beyond that already achieved by conventional treatment.
Inflammation
Vitamin D affects both innate and adaptive immune responses. In vitro, vitamin D decreases the production of the cytokines IL-12 and IFN-γ. 35 IL-12 can elicit the secretion of IFN-γ (and its associated chemokine CXCL-10) in human atherosclerotic coronary arteries with T cells being the primary source of IFN-γ in the arterial wall. Detection of this cytokine axis is associated with poorer outcomes in patients with CAD. 22 An innate pathway causing vascular pathology via NFκβ and downstream release of the pro-inflammatory cytokine IL-6, has recently been associated with vitamin D deficiency in healthy middle-aged and older adults. 36 Increased levels of IL-6, CRP and the cellular adhesion molecules ICAM and VCAM have been associated with lower vitamin D status. 37 In addition, CRP levels were reduced with injections of cholecalciferol in healthy British Bangladeshi men 38 and with high-dose oral supplementation in the critically ill. 39 We found no significant correlations between vitamin D and any measured adhesion or inflammatory markers, nor any significant decreases in markers solely due to active treatment. Indeed, similar reductions were seen with placebo in IL-12 and VCAM. Moreover, ICAM levels decreased significantly only with placebo. These results are similar to several recently reported findings. In the Framingham Offspring Study, no associations with vitamin D status were found for CRP, IL-6 and ICAM. 12 In healthy postmenopausal woman, short-term treatment (12 weeks) with calcium and 800 IU of vitamin D did not reduce circulating levels of IL-6 and CRP. 40 In otherwise healthy overweight and obese adults, no correlation between vitamin D status and serum cytokines could be demonstrated. Moreover, supplementation for 1 year with once-weekly doses of vitamin D failed to reduce the levels of IL-12, IFN-γ, CRP or ICAM in overweight or obese individuals. 41
Study strengths
Several strengths of our study are noteworthy, including the randomized, double-blind, placebo-control design, inclusion of subjects with angiographically documented CAD, exclusion of subjects with renal insufficiency, measurement of baseline and post-treatment vitamin D levels, and excellent compliance with treatment assignment confirmed by pill counts and measurement of vitamin D subfractions. Additionally, our study is generalizable to patients with CAD who are on guideline-based therapies for secondary prevention of disease.
Study limitations
As RH-PAT is a measure of microvascular function at the level of the digits, it is possible that an alternative method measuring macrovascular function in conduit arteries or after infusion of endothelial agents may have been more sensitive to detect changes in our cohort. RH-PAT is a validated 32 method for non-invasively measuring endothelial function and has been used in trials of patients with advanced CAD to show changes in endothelial function with interventions such as enhanced external counterpulsation 42 and the administration of ranolazine. 43 It also provides its own distinct information regarding risk, and a low reactive hyperemia signal detected by RH-PAT is independently associated with higher adverse cardiac event rates. 44
The relationship between endothelial function measured in large conduit arteries versus small resistance vessels is complex and incompletely understood, and in some studies did not correlate well with each other. 45 Different physiological mechanisms may be involved in the changes that are observed with different measures of reactive hyperemia in conduit and resistance vessels. 45 However, each technique appears to provide distinct information regarding risk, 46 but it is unclear which method or vascular bed is best for predicting outcomes. For example, endothelium-dependent vasodilation in resistance arteries but not in the brachial conduit artery, was recently shown to be associated with a 5-year risk of adverse events in an elderly population. 47
Small increases in vitamin D in the placebo group may have confounded our results. Most of the increase was attributable to rises in the vitamin D3 subfraction, suggesting under-reported sun exposure or dietary supplementation. Only one subject in the placebo group had a substantial increase in D2, which can only be obtained via dietary supplementation. Subjects were advised not to use any form of dietary vitamin D supplementation throughout the study period and were asked at all follow-up visits about dietary changes, use of vitamin D supplements and sun exposure. Inherent to any study of vitamin D in free-dwelling communities is the inability to control for sun exposure or involuntary dietary intake given the addition of vitamin D to the food supply. It is unlikely though that the small increases in vitamin D levels in the placebo group had any significant effect on our results. To support this assumption we performed an exploratory analysis within the placebo group which failed to demonstrate a threshold effect for small increases in vitamin D on changes in endothelial function (Appendix Table 3). In fact, the RH-PAT score and FRHI improved in Group 1 despite a further decline in vitamin D levels, which is contrary to our original hypothesis.
The inclusion of subjects with ACS only 4 weeks after their event may not have provided sufficient time for heightened levels of inflammation to resolve. Our findings may simply have reflected a delayed but predictable regression back to baseline levels over time. Our subgroup analysis supports this explanation in relation to IL-12 and IFN-γ, as they tended to be higher in those with ACS versus those with stable CAD at randomization, but this pattern was not evident for ICAM and VCAM. The 4-week waiting period was adequate for the other markers (including hs-CRP) and adhesion molecules to decline to levels seen in stable disease (Appendix Table 2).
Finally, as with any pilot trial, it is possible that our study was underpowered and resulted in a type II error. We did, however, calculate our sample size using the best available data for RH-PAT score estimates from vitamin D-deficient and sufficient subgroups with high CV risk, 23 and inflammatory marker estimates from subjects with and without angiographically proven CAD, 22 cohorts we have previously reported on.
In summary, in subjects with CAD and vitamin D deficiency, vitamin D repletion using 50,000 units per week of ergocalciferol for 12 weeks failed to improve multiple surrogate markers of cardiovascular health. These results question the role of vitamin D supplementation for cardiovascular disease modification in subjects with already established CAD. Whether vitamin D repletion is effective over longer periods of time, in higher doses or in the preclinical stages of CAD pathogenesis remains to be tested.
Footnotes
Appendix
Subgroup analysis of endothelial function in placebo group stratified by change in vitamin D level
|
|
|||
|---|---|---|---|
| Group | Baseline | Week 12 | |
| Group 1 (vitamin D 16–11 ng/ml) | RH-PAT score | 1.84 (0.85) | 2.10 (0.95) |
| FRHI | 0.353 (0.62) | 0.545 (0.48) | |
| Group 2 (vitamin D 11–14 ng/ml) | RH-PAT score | 2.32 (1.21) | 2.08 (0.74) |
| FRHI | 0.628 (0.42) | 0.525 (0.58) | |
| Group 3 (vitamin D 14–25 ng /ml) | RH-PAT score | 1.67 (0.94) | 1.82 (0.94) |
| FRHI | 0.185 (0.72) | 0.358 (0.78) | |
|
|
|||
| Variable (week 12 to week 0) | Group 1 vs 2 | Group 1 vs 3 | Group 2 vs 3 |
| Difference in RH-PAT score | 0.06 | 0.93 | 0.04 |
| Difference in FRHI | 0.02 | 0.59 | 0.12 |
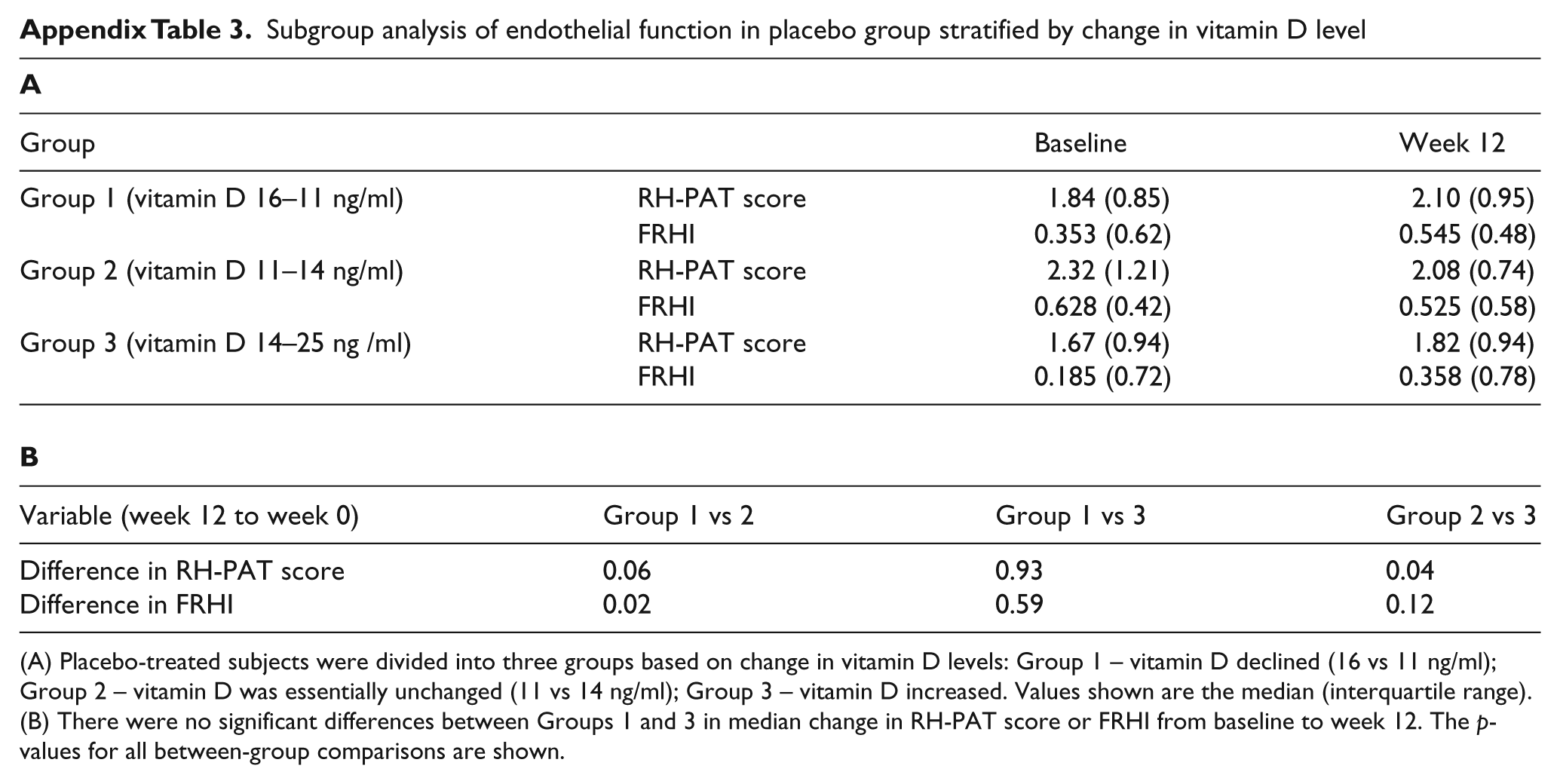
(A) Placebo-treated subjects were divided into three groups based on change in vitamin D levels: Group 1 – vitamin D declined (16 vs 11 ng/ml); Group 2 – vitamin D was essentially unchanged (11 vs 14 ng/ml); Group 3 – vitamin D increased. Values shown are the median (interquartile range).
(B) There were no significant differences between Groups 1 and 3 in median change in RH-PAT score or FRHI from baseline to week 12. The p-values for all between-group comparisons are shown.
Acknowledgements
We thank William Gotsis, MD of Crystal Run Health, Cinthi Pillai, MD, Claudia Calderon, BSN, and Heesun Huh, PharmD, at the Albert Einstein College of Medicine (CP) and the Jacobi Medical Research Center (CC, HH) for all their invaluable contributions in the conduct of this clinical trial; and the laboratories of Joan Berman, PhD and George Tellides, MD for inflammatory marker analysis.
Funding
Supported by a National Clinical Research Program Award (#0885041N) from the American Heart Association.
Conflict of interest
The authors have no relevant disclosures to make.
