Abstract
Endothelial dysfunction is an independent predictor of incident atherosclerotic cardiovascular disease (ACVD). Niacin possesses high-density lipoprotein (HDL)-elevating, antioxidant and anti-inflammatory properties, all potentially contributing to the amelioration of endothelial function. However, controversies exist among trials reporting the effects of niacin on endothelium-dependent flow-mediated dilation (FMD) as a reliable surrogate of endothelial function. The objective of this study was to assess the effect of niacin on brachial artery FMD using a meta-analysis of available evidence. MEDLINE and Scopus databases were searched for randomized controlled trials investigating the impact of niacin therapy on brachial artery FMD. Meta-analysis of eligible studies was conducted using a random-effects model. Pooled effects were measured by weighted mean difference (WMD) and 95% confidence intervals (CIs). Quality assessment, and subgroup, meta-regression and sensitivity analyses were conducted using standard methods. Among 596 citations, 19 full-text articles were read and seven were found to be eligible for inclusion. Eligible studies involved 441 subjects comprising 228 in the niacin and 213 in the control groups. Niacin therapy significantly improved FMD (WMD: 1.98%; 95% CI: 0.91–3.05%; p = 0.0003) and this effect was robust in the sensitivity analysis. The effect size was greater in the subgroup of studies administering higher doses of niacin (≥ 2000 mg/day) as well as those studies administering niacin for primary prevention of ACVD. Meta-regression indicated no association between niacin-induced changes in FMD and changes in plasma HDL-cholesterol, low-density lipoprotein-cholesterol or triglycerides. None of the included studies could find any significant effect of niacin on nitroglycerin-mediated dilation. In conclusion, treatment with niacin improves endothelial function.
Keywords
Introduction
As the largest body organ, endothelium plays a key role in maintaining the homeostasis of vasculature. 1 Endothelial dysfunction, characterized by impaired vascular reactivity and reduced bioavailability of vasodilators, is a pivotal step in the initiation of atherogenesis. 2 Several lines of evidence have confirmed the role of endothelial dysfunction as an independent predictor of the incidence and recurrence of atherosclerotic cardiovascular disease (ACVD). 3 A major contributor to endothelial dysfunction is oxidative stress and increased formation of reactive oxygen species which quench endothelium-derived nitric oxide (NO) and diminish its vasorelaxant effects. 4 Moreover, supplementation with antioxidants has been shown to restore normal endothelial function. 5 Aside from oxidative stress, dyslipidemia and inflammation are other risk factors which reduce NO bioavailability and impair endothelial function.6,7 Recent data indicate that measurement of flow-mediated dilation (FMD) in the brachial artery by non-invasive ultrasonography is a reliable surrogate marker of endothelial function and has a prognostic value in patients with ACVD. 8
Although statin therapy is the cornerstone of pharmacotherapy for lowering low-density lipoprotein cholesterol (LDL-C) concentrations and prevention of ACVD, compelling evidence indicates a substantial residual risk in patients with target LDL-C levels.9,10 Such a residual risk has been attributed, at least in part, to low levels of high-density lipoprotein cholesterol (HDL-C). 11 HDL is a lipoprotein with a multitude of biological effects contributing to cardiovascular health. 12 Some of the cardioprotective effects of HDL include promotion of reverse cholesterol transport, and antioxidant, anti-inflammatory and anti-thrombotic effects. 12 Moreover, there is epidemiological evidence indicating the association between low HDL-C concentration and endothelial dysfunction. 13
Niacin (nicotinic acid; vitamin B3) is the most potent HDL-C-elevating medication currently available in the market. In addition, niacin possesses pleiotropic non-lipid effects including antioxidant and anti-inflammatory properties. 14 All these effects can help in ameliorating endothelial function. However, randomized controlled trials investigating the effects of niacin therapy on the brachial artery FMD have yielded inconsistent results.15 –22 The present study aimed to systematically review the literature and perform a meta-analysis of individual findings on the impact of niacin on FMD.
Methods
Search strategy
This study was designed according to the guidelines of the 2009 preferred reporting items for systematic reviews and meta-analysis (PRISMA) statement. 23 An electronic search of English-language literature was performed in MEDLINE (www.ncbi.nlm.nih.gov/pubmed) and Scopus (www.scopus.com). The combination of following search keywords was used in titles and abstracts: (niacin OR “nicotinic acid” OR “vitamin B3” OR “vitamin PP” OR niaspan OR acipimox) AND (endothelium OR endothelial OR “flow-mediated” OR “flow mediated” OR FMD). The literature was searched from inception to 9 June 2013. Retrieved papers were manually screened for the identification of any other relevant publication.
Study selection
Original studies were included if they fulfilled all of the following criteria: (1) be a randomized clinical case–control or case –crossover trial; (2) investigated the impact of niacin on brachial artery FMD; (3) inclusion of an appropriate control group; (4) presentation of sufficient information on FMD values at baseline and at the end of study; and (5) published in a peer-reviewed journal. Exclusion criteria were: (1) non-clinical studies; (2) inappropriate controlling for niacin (e.g. comparing combination lipid-modifying therapy containing niacin versus placebo or ‘no treatment’); and (3) not reporting baseline and endpoint FMD values sufficient for quantitative data synthesis. Exclusion of an article for the latter reason was done if no feedback was received after contacting the author(s).
Data extraction
Eligible studies were reviewed and the following data were abstracted: (1) first author’s name; (2) year of publication; (3) study location; (4) number of participants in the case and control groups; (5) age, sex and body mass index (BMI) of study participants; (6) circulating concentrations of lipid profile parameters (comprising total cholesterol, LDL-C, HDL-C and triglycerides) and high-sensitivity C-reactive protein (hs-CRP); (7) systolic and diastolic blood pressures; (8) fasting glucose concentration; (9) homeostasis model assessment-estimated insulin resistance (HOMA-IR) index; (10) prevalence of smoking, type 2 diabetes, dyslipidemia, hypertension and coronary artery disease (CAD); and (11) baseline and post-trial percentage change values of brachial artery nitroglycerin-mediated dilation (NTGMD) and FMD.
Quality assessment
Eligible studies were systematically assessed for potential risk of bias using instructions described in the Cochrane Handbook for Systematic Reviews of Interventions. 24 The items used for the assessment of each study were adequacy of sequence generation, allocation concealment, blinding, addressing drop-outs (incomplete outcome data), selective outcome reporting, and other potential sources of bias. According to the recommendations of the Cochrane Handbook, a judgment of ‘Yes’ was indicative of low risk of bias, whilst ‘No’ indicated high risk of bias. Labeling as ‘Unclear’ indicated unclear or unknown risk of bias.
Quantitative data synthesis
Meta-analysis was conducted using the Cochrane Program Review Manager, version 5.1 (Cochrane Collaboration, Oxford, UK). Blood lipid and glucose levels were collated in mmol/L. A multiplication by 0.0259, 0.0113 or 0.0555 was used to convert cholesterol (total cholesterol, HDL-C or LDL-C), triglyceride and glucose levels expressed in mg/dL into mmol/L, respectively. Standard deviations (SDs) of the mean difference were calculated using the following formula: SD = √[(SDpre-treatment)2 + (SDpost-treatment)2 – (2R × SDpre-treatment × SDpost-treatment)], assuming a correlation coefficient (R) = 0.5. In case of reporting FMD values in median and interquartile range, the mean and SD were estimated using the recommendations of Hozo et al. 25 In case of reporting 95% confidence intervals (CIs), SD was estimated using the following formula: SD = (CIupper – CIlower) × √(n)/3.29, where n is the number of subjects. 24
Net changes in measurements (change scores) were calculated for parallel and crossover trials, as follows: (measure at end of follow-up in the treatment group − measure at baseline in the treatment group) − (measure at end of follow-up in the control group − measure at baseline in the control group). A random-effects model and the generic inverse variance method were used to accommodate for the heterogeneity of studies in terms of design (parallel or crossover), duration of niacin treatment, demographic characteristics of included populations (such as underlying disease, age, sex), and niacin dosages that were administered. If a study provided both intention-to-treat and per-protocol analysis, only the results of the former analysis were used in the quantitative data synthesis. In order to evaluate the influence of each study on the overall effect size, sensitivity analysis was conducted using the one-study remove approach. 26 Heterogeneity analysis was performed using Cochran’s Q test and I2 index. 26
Meta-regression
Random-effects meta-regression was performed using the unrestricted maximum likelihood method to evaluate the association between changes in lipoprotein (HDL-C, LDL-C and triglycerides) concentrations and standardized mean difference in FMD values.
Publication bias
Potential publication bias was explored using visual inspection of Begg’s funnel plot asymmetry, and Begg’s rank correlation and Egger’s weighted regression tests. The Duval and Tweedie ‘trim and fill’ method was used to adjust the analysis for the effects of publication bias. 27 Comprehensive Meta-Analysis (CMA) V2 software (Biostat, NJ, USA) 28 was used for performing meta-regression and publication bias analyses.
Results
Flow of included studies
The initial literature search yielded 595 articles (after removing duplicates), to which one article was added following a manual search of eligible studies. The title and abstract of these 596 articles were carefully checked, leading to the exclusion of 577 articles for not meeting the inclusion criteria. Nineteen articles were initially selected and their full-texts were evaluated. After careful assessment of full-texts, seven articles were found to fulfill the inclusion criteria and were selected for meta-analysis.15 –21 Reasons for rejecting the other 12 articles were: not measuring FMD,29–34 not having a randomized design, 35 not including an appropriate control group, 36 FMD not being measured in the brachial artery, 37 insufficient data on baseline and/or endpoint FMD values,22,38 and not being in the English language. 39 A summary of the study selection process is illustrated in Figure 1.
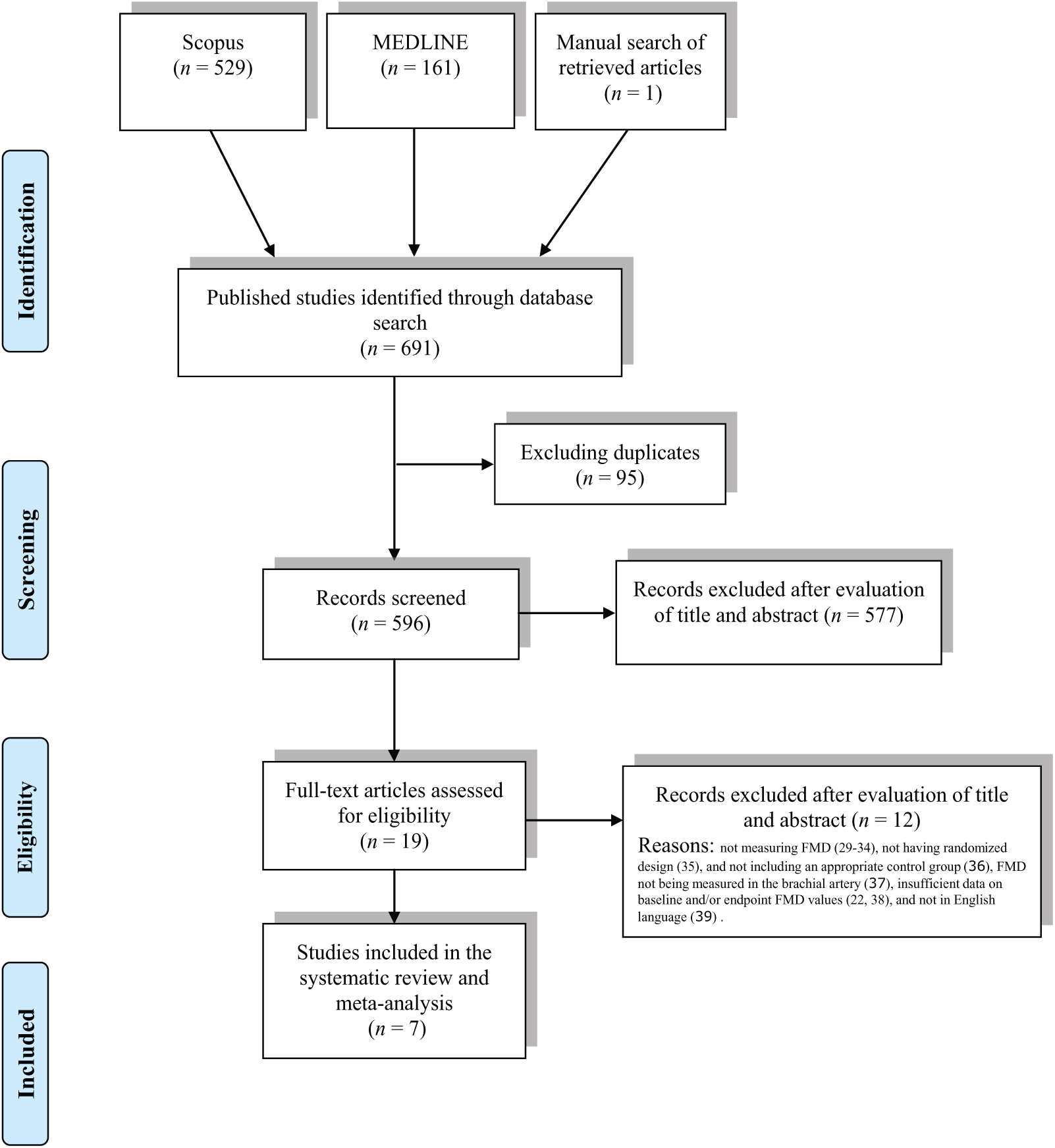
Flow diagram of the study selection procedure showing the number of eligible randomized controlled trials for the meta-analysis of the impact of niacin therapy on brachial artery flow-mediated dilation (FMD).
Characteristics of included studies
A total of 441 subjects were included in the seven eligible studies, comprising 228 subjects receiving niacin and 213 receiving control treatment (subjects in the only included crossover trial were counted in both groups). Included studies were published between 2006 and 2013, and were reported from the USA,17,20 UK, 18 Germany, 19 Slovenia, 15 Canada 16 and Brazil. 21 All seven studies used either extended-release niacin or no-flush niacin (as inositol hexasuccinate) at a dose range of 1000–2000 mg/day. Four studies used a titrating schedule to reach the predefined dosing of niacin.16 –19 Duration of treatment with niacin ranged between 1216,17,19,21 and 52 20 weeks. The largest study had a population size of 106 subjects, 19 whilst the smallest study recruited 22 subjects. 21
All studies were conducted in parallel case–control groups apart from the study by Philpott et al., 16 which was designed as a crossover trial. Four studies were conducted in patients with a history of ACVD or acute coronary syndrome,15,16,18,19 whilst the other three studies recruited subjects with metabolic syndrome, 20 HIV infection 17 or isolated low HDL-C. 21 Males were the predominant sex in all of the included studies and two studies were exclusively conducted among male subjects.15,21 Case and control groups were matched regarding age and sex in all of the studies. Demographic and baseline biochemical parameters of included studies are illustrated in Table 1.
Demographic characteristics of the included studies.
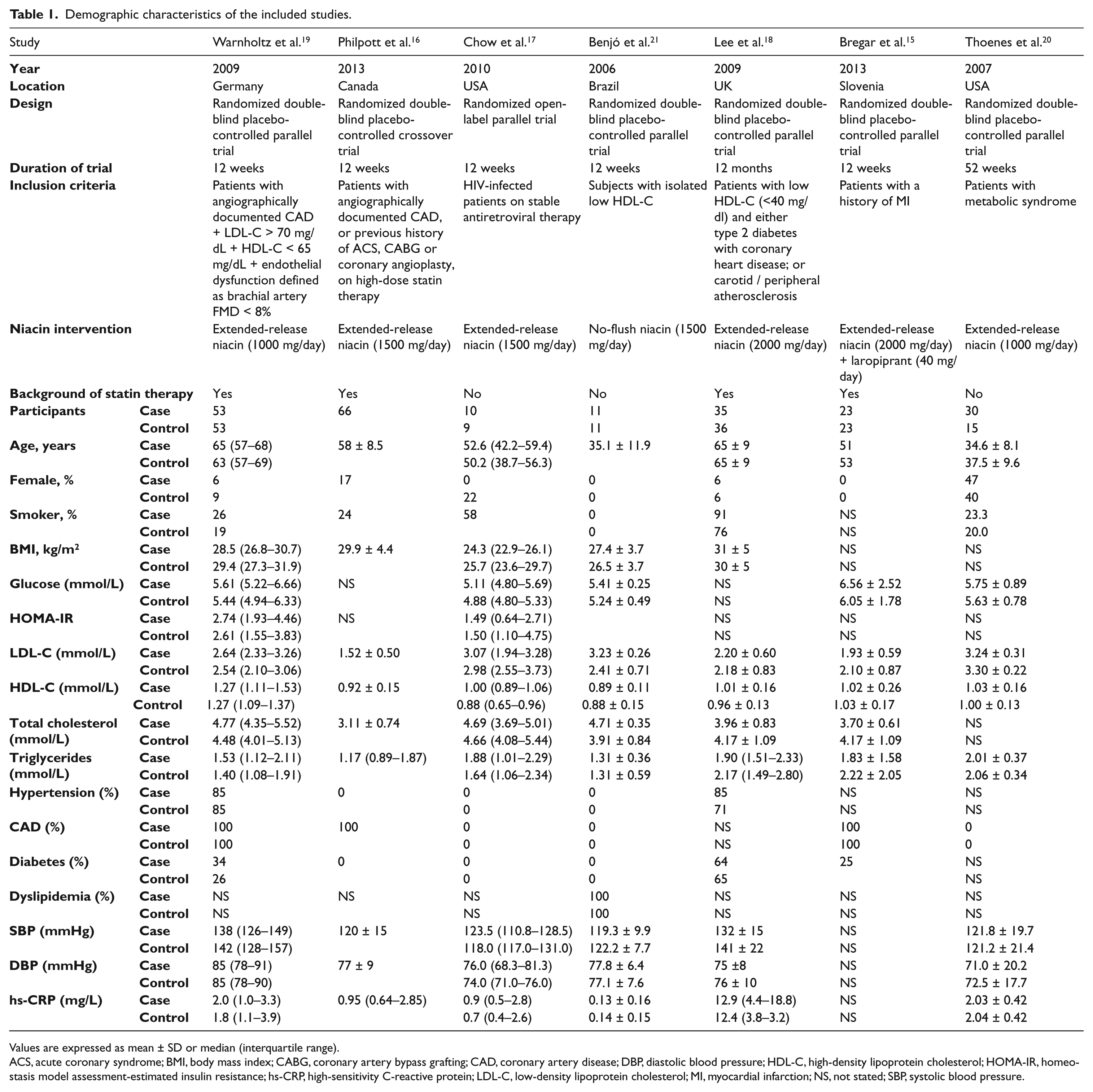
Values are expressed as mean ± SD or median (interquartile range).
ACS, acute coronary syndrome; BMI, body mass index; CABG, coronary artery bypass grafting; CAD, coronary artery disease; DBP, diastolic blood pressure; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, homeostasis model assessment-estimated insulin resistance; hs-CRP, high-sensitivity C-reactive protein; LDL-C, low-density lipoprotein cholesterol; MI, myocardial infarction; NS, not stated; SBP, systolic blood pressure.
Quality assessment
Overall, the risk of bias was found to be low in the studies included in this meta-analysis, though most of studies did not report sufficient information regarding random sequence generation and allocation concealment methodology. All of the studies were double-blind apart from the study by Chow et al., which was open-label. 17 Intention-to-treat analysis was carried out in two of the studies,18,19 whilst one study had no dropout. 21 Although all studies used extended-release or no-flush niacin, the major reason for dropouts was flushing. Two studies did not report the source of funding20,21 and three studies received financial support from commercial sources.16,18,19 Details on the risk of bias among included trials are summarized in Table 2.
Quality assessment of studies selected for analysis.
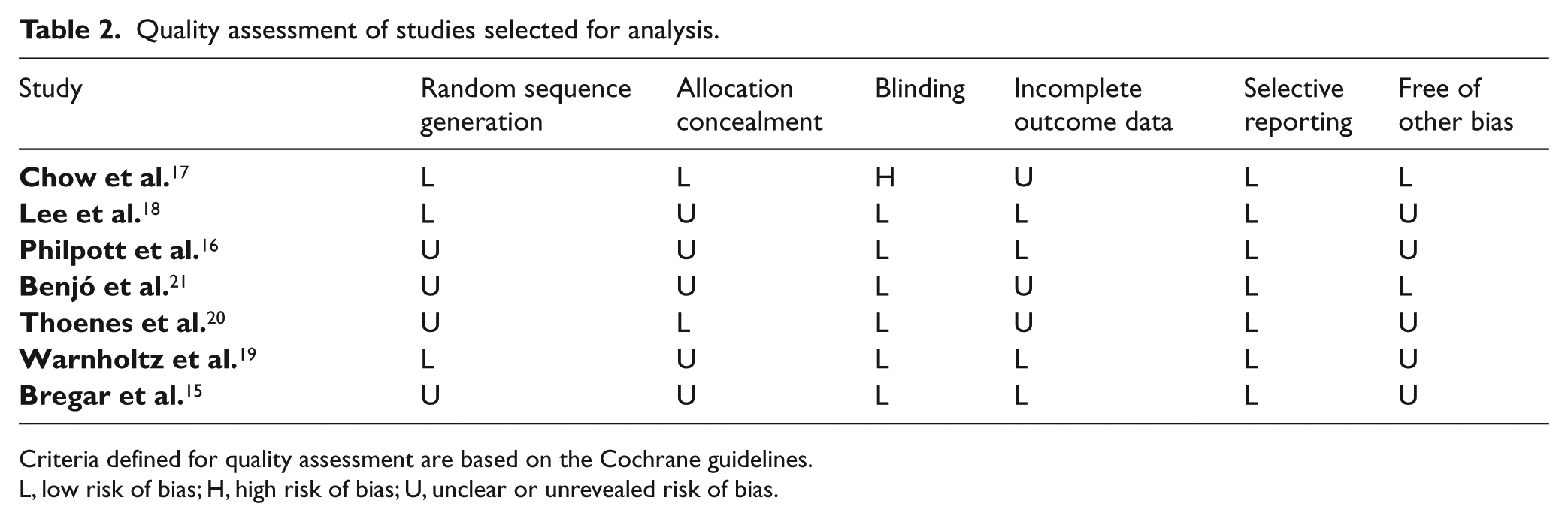
Criteria defined for quality assessment are based on the Cochrane guidelines.
L, low risk of bias; H, high risk of bias; U, unclear or unrevealed risk of bias.
FMD measurement
Non-invasive ultrasound was used for the measurement of brachial artery FMD in the included studies. Only one study used magnetic resonance imaging (MRI) for measurement of FMD. 18
Effect of niacin on FMD and NTGMD in individual studies
FMD in the niacin group ranged between +0.24% 16 and +5.90%, 15 whilst the corresponding changes in the control group were between −0.70% 17 and +1.50%. 15 Niacin was found to significantly improve FMD in three studies.15,20,21 In one study, the effect of niacin therapy on FMD was significant in the per protocol but not ITT analysis. 19 The mean NTGMD change ranged between −5.4% 18 and +4.2% 15 in the niacin group, and between −3.6% 21 and +1.7% 15 in the control group. None of the studies could find a significant effect of niacin on NTGMD values. The percentage changes in FMD and NTGMD reported by each trial are summarized in Table 3.
Effect of niacin therapy on brachial artery FMD and NTGMD in selected studies.
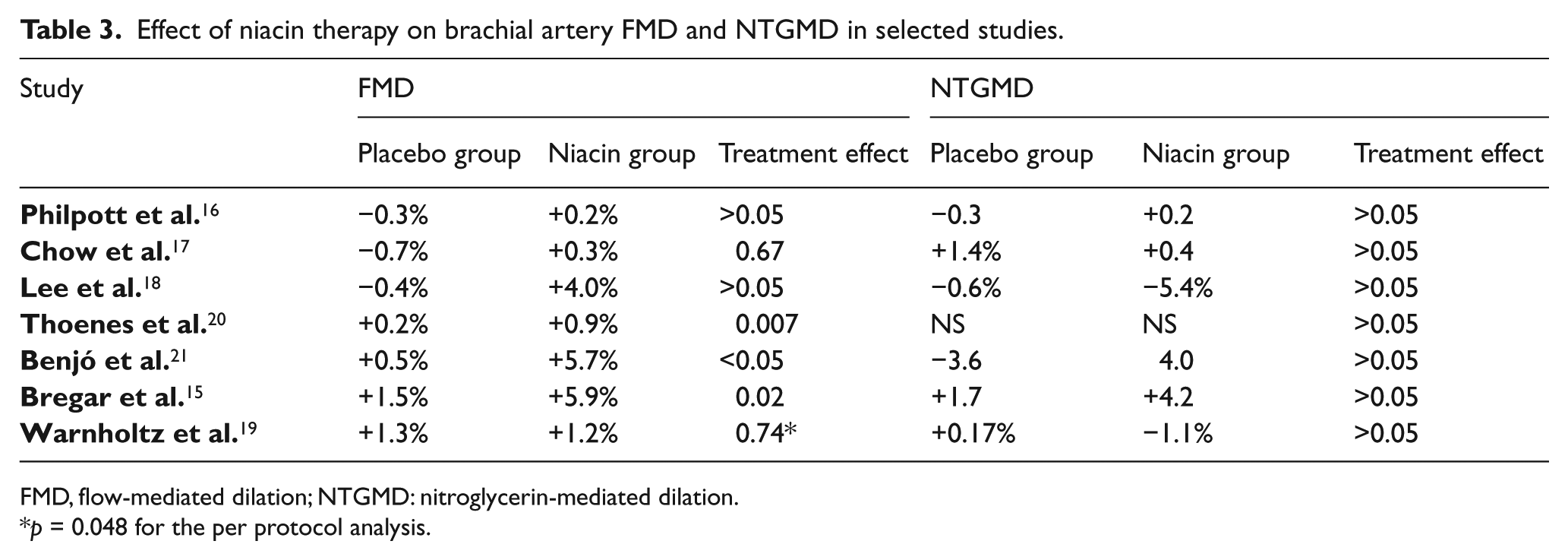
FMD, flow-mediated dilation; NTGMD: nitroglycerin-mediated dilation.
p = 0.048 for the per protocol analysis.
Quantitative data synthesis
Weighted mean difference (WMD) in FMD following treatment with niacin was 1.98% (95% CI: 0.91–3.05%). This effect size turned out to be statistically significant (p = 0.0003) (Figure 2), and was robust in the leave-one-out sensitivity analysis (Table 4). In subgroup analyses, improvement of FMD was greater in studies administering niacin at a daily dose of ≥ 2000 mg (WMD: 4.40%; 95% CI: 2.75–6.05%; p < 0.0001) versus studies that used lower doses (WMD: 1.19%; 95% CI: 0.17–2.20%; p = 0.02). In the same manner, the effect of niacin therapy was greater in the subgroup of studies recruiting patients with CAD or acute coronary syndrome (WMD: 3.09; 95% CI: 0.50–5.68; p = 0.02) compared to studies conducted on patients free from these conditions (WMD: 1.22; 95% CI: −0.83–3.26%; p = 0.24). There was no improved efficacy of niacin on FMD in trials with > 12 weeks duration (WMD: 2.42; 95% CI: −1.13–5.98%; p = 0.18) versus trials with shorter duration (WMD: 2.30; 95% CI: 0.09–4.51; p = 0.04) (Figure 3). When the studies were stratified based on the baseline LDL-C levels, a significant improvement of FMD was observed in both subsets of studies with baseline LDL-C < 100 mg/dL (WMD: 3.00; 95% CI: 0.21–5.79; p = 0.03) and LDL-C > 100 mg/dL (WMD: 1.36; 95% CI: 0.20–2.52; p = 0.02).
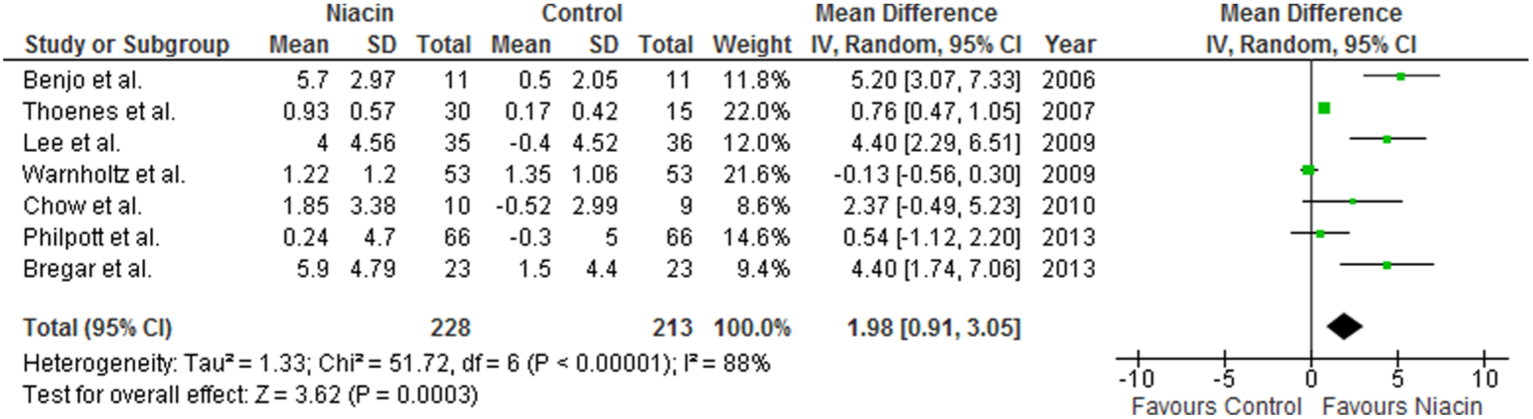
Forest plot detailing weighted mean difference and 95% confidence intervals for the impact of niacin therapy on brachial artery flow-mediated dilation.
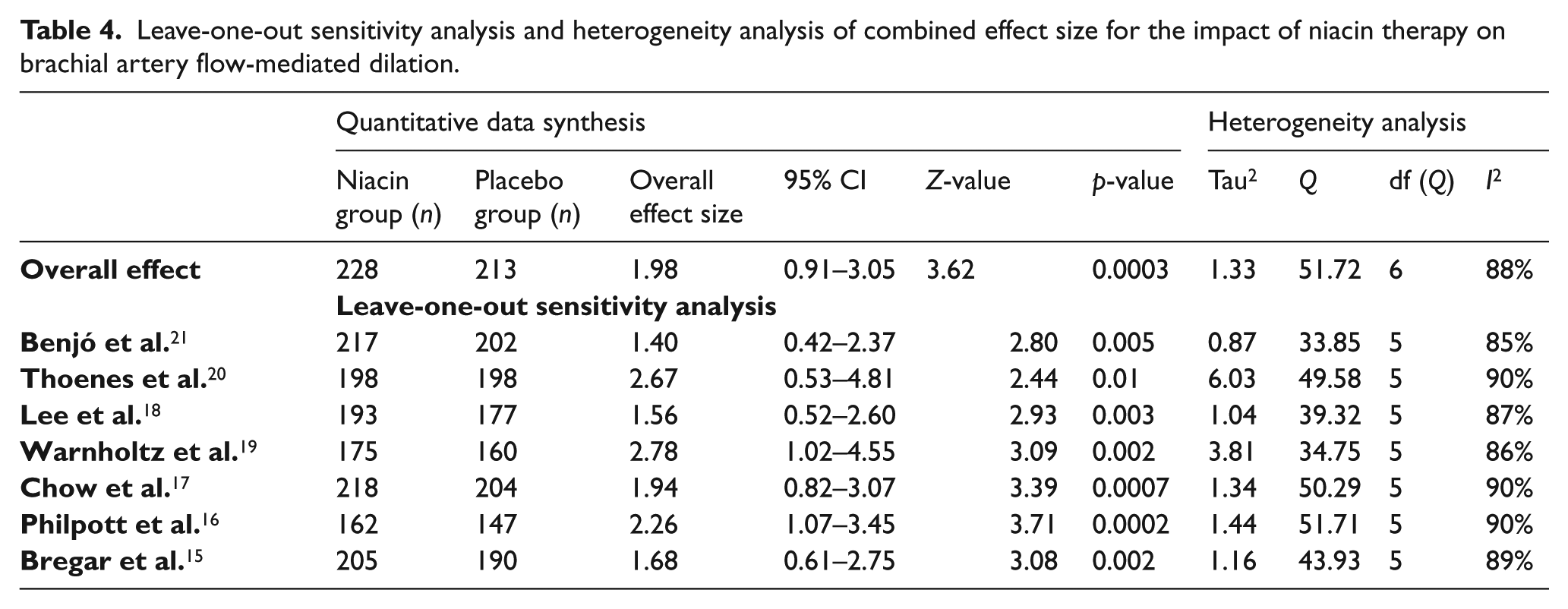
Leave-one-out sensitivity analysis and heterogeneity analysis of combined effect size for the impact of niacin therapy on brachial artery flow-mediated dilation.
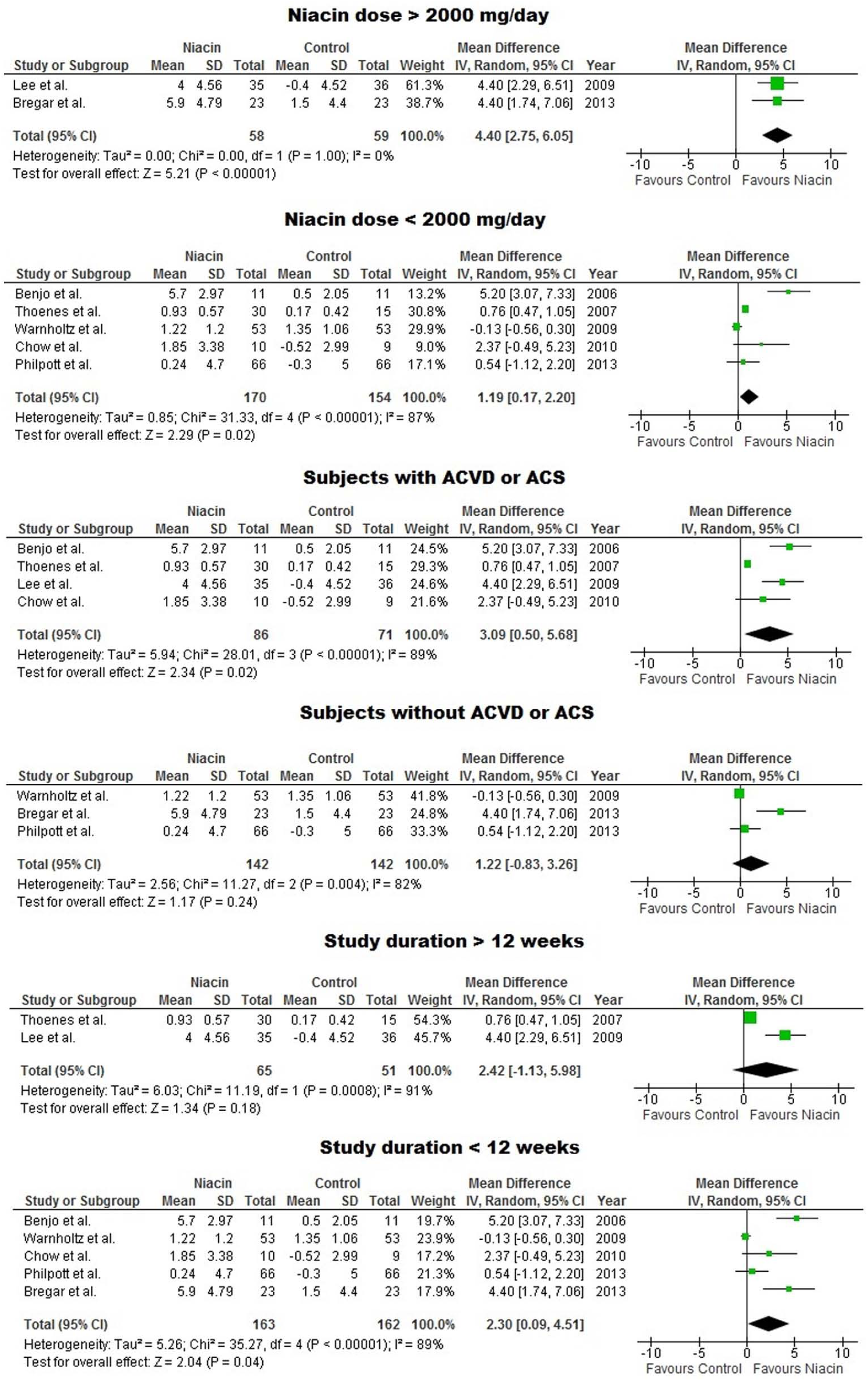
Forest plot detailing weighted mean difference and 95% confidence intervals for the impact of niacin therapy on brachial artery flow-mediated dilation in different subgroups of included studies.
Meta-regression
Meta-regression analysis was conducted to evaluate the association between niacin-induced changes in FMD and changes in lipid parameters during the study. Meta-regression did not reveal a significant association of FMD changes with changes in plasma HDL-C (point estimate for slope = −0.13, SE = 1.36, 95% CI = −2.79–2.53, z = −0.10, p = 0.92), LDL-C (point estimate for slope = −0.92, SE = 1.22, 95% CI = −3.31–1.47, z = −0.76, p = 0.45) or triglycerides (point estimate for slope = 1.25, SE = 1.19, 95% CI = −1.07–3.57, z = 1.05, p = 0.29) (Figure 4). Likewise, baseline LDL-C concentrations did not determine the calculated effect size (point estimate for slope = 0.65, SE = 0.37, 95% CI = 0.07 to −1.38, z = 1.77, p = 0.08) (Figure 4).
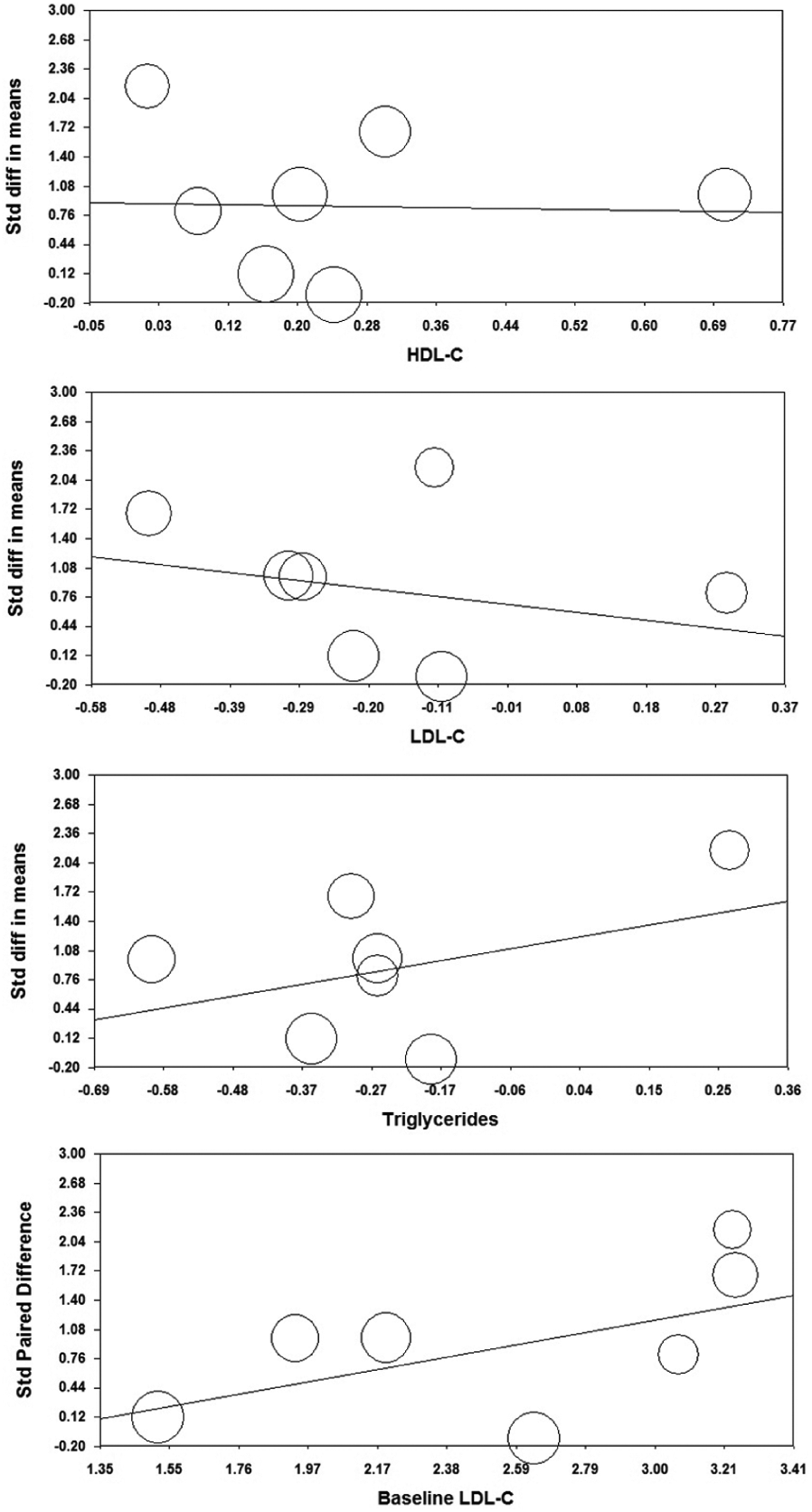
Meta-regression plots of standardized change in brachial artery flow-mediated dilation (FMD) according to changes in plasma lipid concentrations and baseline low-density lipoprotein cholesterol levels. The size of each circle is inversely proportional to the variance of change. (LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol.)
Publication bias
Visual inspection of the funnel plot of the study standard error by respective mean difference indicated an asymmetry and a potential presence of publication bias. However, there was no evidence of bias based on the Begg’s rank correlation test (tau with continuity correction = 0.10, z-value = 0.30, one-tailed p-value = 0.38) and Egger’s weighted regression test (intercept = 2.68, SE = 1.33, 95% CI = −0.75–6.11, t-value = 2.01, df = 5.00, two-tailed p = 0.10). Adjustment for publication bias using the Duval and Tweedie trim and fill procedure led to the imputation of two missing studies and yielded an overall effect size of 1.19%; 95% CI: 0.15–2.24% (Figure 5).
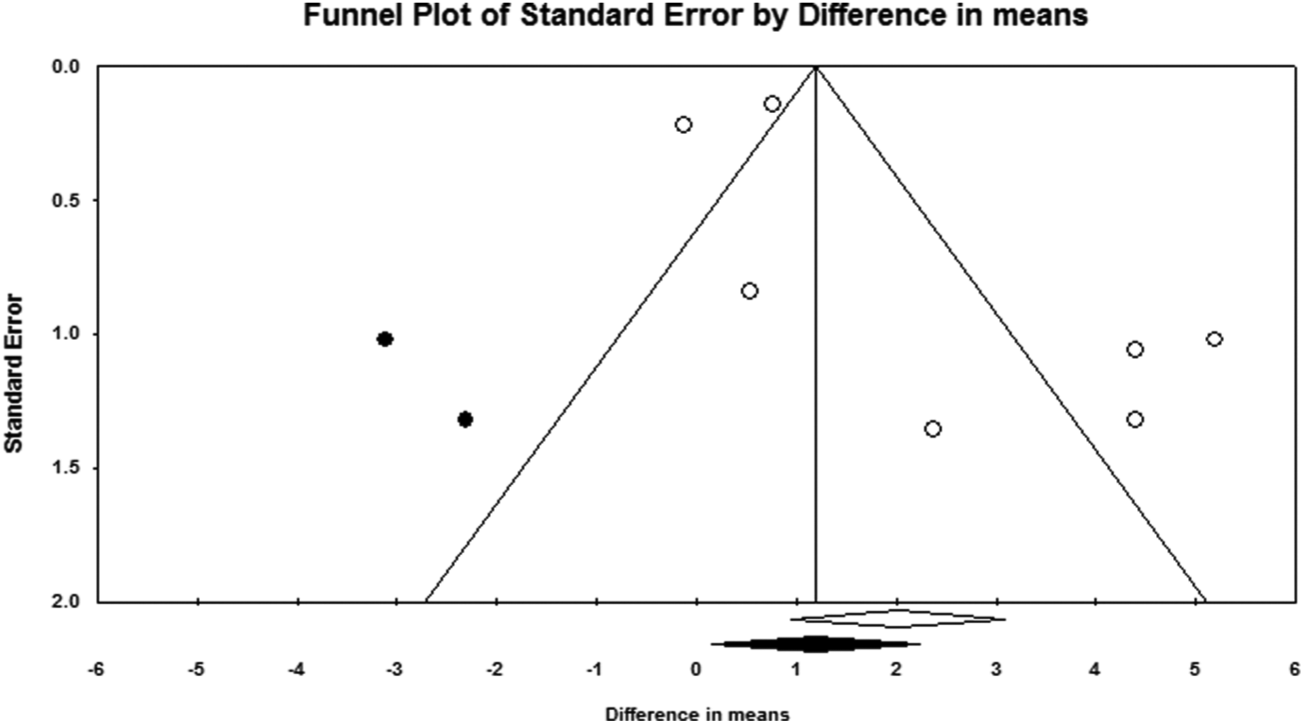
Funnel plot of trim and fill analysis. (Open circles represent observed published studies; closed circles represent imputed unpublished studies.)
Discussion
The present study represents the first meta-analysis on the impact of niacin therapy on brachial artery endothelial responsiveness measured by FMD. The results indicated that niacin can significantly improve endothelium-dependent vasodilation compared to placebo. The placebo-adjusted increase in FMD by niacin was estimated to be 1.98%. This is a clinically relevant effect size and is numerically larger than that reported for statins (0.94%) by a recent meta-analysis, 40 although direct head-to-head comparative trials are required to assess the FMD-improving effect of niacin versus that of statins. Although this remarkable effect size for niacin was robust and not mainly influenced by a single study, it must be noted that an FMD improvement by around 2% is unlikely to be observed following treatment with a single intervention. Furthermore, although the combined analysis favored the significant FMD-improving effect of niacin therapy, no such an effect was observed in larger studies by Philpott et al., 16 Lee et al. 18 and Warnholtz et al. 19 However, the mentioned studies were all secondary prevention trials and performed in populations with a background of statin therapy. Hence, a floor effect on FMD in these statin-treated populations might be a possible explanation for lack of further improvement by niacin to a significant level. In a similar way, the overall effect size is very likely to be influenced by statin therapy rather than a single effect of niacin.
Augmentation of HDL-C status is generally regarded as the major mechanism for the cardiovascular effects of niacin. 41 HDL has a plethora of pleiotropic effects that affect endothelial NO production. Owing to its established paraoxonase and platelet-activating factor hydrolase activities, HDL has antioxidant properties that help mitigating vascular oxidative stress and prevent subsequent impairment of NO release. 42 Other mechanisms for the increased NO bioavailability by HDL include direct activation of endothelial NO synthase (eNOS) via interaction with scavenger receptor class B type I (SR-BI) receptor, 43 enhancement of intracellular ceramide levels, 43 and inhibition of oxidized LDL-induced disruption of plasma membrane caveola, thereby increasing the subcellular localization and activation of eNOS. 44 Additionally, HDL has been shown to inhibit apoptosis and enhance migration and re-endothelialization of endothelial cells. 45
In spite of the above-mentioned strong HDL-associated benefits on endothelial function, no significant association was found in the meta-regression analysis of FMD changes with HDL-C changes. There are several explanations for this finding. It is conceivable that an HDL-C-raising effect of niacin is more sizable in subgroups of patients with low HDL-C at baseline. However, only three of the included studies in this meta-analysis defined low HDL-C as inclusion criteria.18,19,21 Apart from one study, 19 the remaining studies did not provide post hoc analysis in subgroups of patients with low baseline HDL-C. Interestingly, the study by Warnholtz et al., which conducted such a post hoc analysis, reported a significant improvement of FMD in patients with low baseline HDL-C, whilst there was no effect in the total study population. 19 Another possible explanation is that improvement of FMD by niacin is the result of enhanced HDL functionality rather than increased HDL-C concentrations. It has been shown that measurement of HDL function, in particular reverse cholesterol transport, is a better indicator of HDL status than cholesterol content of HDL. 46 Finally, aside from HDL elevation, niacin has several lipid-independent actions that could account for its endothelial protective effects. 47 The most important lipid-independent effects of niacin are anti-inflammatory and antioxidant effects. 12 Several lines of in vitro and in vivo evidence have indicated the beneficial impact of niacin in mitigating vascular inflammation and reducing the infiltration of immune cells into the vessel wall. 48 Niacin down-regulates several key mediators of vascular inflammation including VCAM-1, MCP-1 and P-selectin 48 whilst increasing the expression and release of adiponectin, 49 an adipokine with documented protective effects against vascular inflammation and endothelial dysfunction. 50 Niacin also reduces endothelial oxidative stress via increasing the cellular content of nicotinamide adenine dinucleotide phosphate (NAD(P)H) and reduced glutathione (GSH) whilst inhibiting reactive oxygen species (ROS) generation in endothelial cells. 51
Based on the subgroup analyses of this study, higher doses of niacin may be associated with a greater effect on endothelial function. This is in line with previous findings on the dose–response pattern of niacin’s effect on HDL-C. 52 However, unlike the HDL-C-boosting effect, 53 improvement of FMD by niacin may not be time-dependent, as shown by lack of increase in the effect size in the subgroup of trials with longer durations of follow-up (> 12 weeks). Furthermore, when the studies were categorized into primary and secondary prevention trials, the FMD-improving effect of niacin reached statistical significance only in the primary prevention. Another important implication from the subgroup analysis is the meaningful reduction in the efficacy of niacin when used as adjunct to statins. Since statins have been shown to protect against endothelial dysfunction and improve FMD, 54 the efficacy of add-on therapy with niacin might have been masked by intensive statin therapy in this subset of trials. This latter effect may also explain the lack of significant effect of niacin in the secondary prevention trials as high-dose statin therapy is a constant component of drug regimens used for secondary prevention.
Although the finding from the current meta-analysis is promising, any beneficial effect of niacin should be interpreted in the context of its ability to reduce CVD endpoints. A large-scale clinical endpoint trial (AIM-HIGH) failed to find any benefit of niacin as an adjunct to intensive statin therapy in reducing ACVD. 55 Besides, unpublished reports from a recently completed clinical endpoint trial (HPS2-THRIVE) also imply on the lack of any significant benefit with extended-release niacin in reducing vascular events. Such discordance between the endothelial function-improving effects of an agent and its effect on hard clinical endpoints has been previously reported for folic acid, 56 hormone replacement therapy 57 and antioxidant vitamins. 58 Despite the negative findings of recent large-scale trials, several earlier studies – though at smaller scales – have reported an opposite finding.18,33,59–64 A recent meta-analysis also favored the benefits of niacin either as monotherapy or as an adjunct to statins in reducing CVD events. 64 It is also worth noting that both AIM-HIGH and HPS2-THRIVE studies were secondary prevention trials in subjects receiving optimal statin therapy. Moreover, impaired endothelial function was not among the inclusion criteria of either of these trials.
A number of limitations for this study need to be acknowledged. First, the number of included studies and total number of subjects was relatively small. Whilst this limitation did not preclude the significant effect of niacin on FMD, it might have influenced the meta-regression findings. Hence, negative findings on the association between changes in FMD values and lipid concentrations might be due to the lack of sufficient statistical power. Second, there was a significant heterogeneity among the included studies in terms of inclusion criteria, demographic characteristics of studied population and technical specifications employed for FMD measurement. Whilst the random-effects approach selected for quantitative data synthesis can compensate part of this inter-study heterogeneity, the comparison is still tender to bias. Third, in all studies included in this meta-analysis, males constituted the predominant sex. There have been a number of previous reports, though inconsistent, on sex difference in vascular function and FMD. 65 Finally, two relevant trials were excluded from the analysis because the FMD values were incomplete or unavailable.
Conclusions
In summary, the present meta-analysis revealed significant endothelial function-improving effects of niacin, as reflected by elevation of brachial artery FMD. This effect is more prominent in primary prevention of ACVD, and appears to be independent of the niacin-induced changes in conventional lipid parameters (HDL-C, LDL-C and triglycerides). Therefore, endothelial function-improving effects should be considered in parallel with the HDL-C-boosting properties to explain the effects of niacin on the cardiovascular system. Since the efficacy of niacin therapy in reducing ACVD events is argued, future randomized controlled trials are encouraged to assess the impact of niacin as monotherapy in patient populations with endothelial dysfunction and impaired FMD.
Footnotes
Declaration of conflicting interest
The author has no competing interests to declare.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
