Abstract
Carotid intima–media thickness (cIMT) has received interest as a predictor of cardiovascular events in recent years. Use of cIMT in a clinical setting is limited by the variability in measurement and the lack of evidence for its use in clinical risk prediction. This review examines the major studies that have assessed the relationship between cIMT and cardiovascular event risk and discusses the current role of IMT in cardiovascular risk prediction.
Keywords
Introduction
Despite considerable progress made in recent years in the management of known risk factors, cardiovascular disease (CVD) remains the leading cause of death worldwide. 1 Economic constraints placed upon modern health services mean that not only is it desirable to reduce vascular risk in the entire healthy general population but also to identify those at particularly high risk in order to appropriately allocate more intensive and expensive preventive regimes.
Contemporary research has given clinicians a range of tools to help identify asymptomatic individuals who are at higher risk of cardiovascular events. Multifactor risk scores based on traditional risk factors such as age, smoking, hypercholesterolaemia, hypertension and diabetes mellitus (such as the Framingham risk score) are the most commonly used, but interest is increasing in the use of less conventional markers of risk such as coronary artery calcium scoring, the ankle–brachial index (ABI) and C-reactive protein (CRP).2–4 Carotid ultrasound has also become a prominent focus of research and carotid intima–media thickness (cIMT) in particular has received attention as a possible non-invasive and relatively simple to perform predictor of future CVD risk. 5 However, controversy exists over the use of novel risk markers as predictors of CVD, including cIMT. A recent United States Preventive Services Task Force (USPSTF) statement was not in favour of the use of any of a range of non-traditional risk factors when it was published in 2009. 6 Although there was some evidence that cIMT could predict cardiovascular events in asymptomatic people with no history of coronary heart disease (CHD), the recommendation was to focus research on the predictive ability of cIMT over and above Framingham risk factors (the current gold standard) and that more evidence was required on its potential use in diabetic populations (a group at high risk for CHD).
Conversely, in 2010, the ACCF/AHA published their own guideline for the assessment of cardiovascular risk in asymptomatic individuals. 7 In the guideline, it is recommended that cIMT could be measured for the purposes of aiding cardiovascular risk classification in asymptomatic adults who are deemed to be at intermediate risk. They highlight that there is broad evidence in support of the use of cIMT for risk prediction, although they acknowledge that much of the evidence is drawn from a research setting. Thus, in clinical practice, there must be a high degree of adherence to published guidelines regarding measurement of cIMT to allow the collection of high-quality data. 7
Given the potentially conflicting messages on the use of cIMT in clinical practice, the current article aimed to review critically the evidence for cIMT as a predictor of future vascular events, and to discuss its possible place in current cardiovascular risk assessment in light of this evidence. A Medline search was performed including the search terms ‘carotid intima media thickness’ or ‘carotid intima media thickening’ and ‘cardiovascular disease’ or ‘myocardial infarction’ or ‘stroke’. Large epidemiological studies were identified and the references of review articles were manually searched for additional sources.
Development and prediction of atherosclerotic CVD
Atherosclerotic CVD begins as asymptomatic lesions of the artery walls, including the coronary arteries. These lesions manifest as fatty streaks present in the endothelial surface of the artery, which can appear as early in life as childhood. Fatty streaks tend to be common at sites of turbulent blood flow, but can be present in nearly all areas of the vascular tree. Following continued exposure to common cardiovascular risk factors, including hypertension, smoking and dyslipidaemia, early atherosclerotic change can progress to plaque formation and calcification. Narrowing of the arterial lumen by these growing plaques leads to ischaemia of target tissues and, once complete occlusion occurs, tissue infarction is inevitable.
Since CVD only becomes clinically apparent at a point where atherosclerosis is well established in the arteries and often at the point of an acute event, there is little potential to use clinical signs and symptoms to identify those at high risk for CVD early enough to prevent events. 8 Clinicians therefore look to techniques which can identify disease at an early stage in disease development, before symptoms manifest.
Methods currently used by clinicians to predict vascular risk are dominated by the combination of known cardiovascular risk factors into risk score prediction models or algorithms. A variety of such risk scores have been developed and proposed for use in the general population, including the Framingham risk score, 9 SCORE, 10 ASSIGN, 11 and QRISK212,13 among others. These algorithms are based upon known cardiovascular risk factors including age, sex, smoking status, diabetes status, total and high-density lipoprotein (HDL) cholesterol, and systolic blood pressure, and differ from each other primarily in the exact combination of risk factors included in the model. Although their non-invasive nature means they are often used in clinical practice and have been robustly validated in general populations, cardiovascular risk algorithms have been shown to be inaccurate in both high-risk populations and low-risk populations. 14 Other methods of risk evaluation involving direct measurement of disease, for example coronary angiography, are often highly invasive or risk prone, and are not suitable methods of investigation in those who may be at low risk of CVD. Interest has therefore increased in the use of non-invasive measures of atherosclerosis, including those measurable on carotid ultrasound such as cIMT and plaque. A review by Wyman et al. on the use of carotid plaque as a predictor of cardiovascular events suggested that carotid plaque could be useful in the assessment of future vascular risk and that patients could be referred for cIMT screening if carotid plaque screening was negative. 15 The current review looks at the ability of cIMT to predict future vascular events in its own right.
Carotid intima–media thickness
cIMT is defined as the distance between the lumen–intima interface and the media–adventitia interface. 16 Traditionally, atherosclerotic disease of the carotid arteries was determined by observing luminal narrowing secondary to plaque development. However, because atherosclerosis is a process which directly affects the arterial wall, it was suggested by Pignoli et al. that measuring the thickness of this complex may be a more accurate representation of atherosclerosis than simply measuring luminal diameter, especially as there is often luminal expansion in compensation for a thickened arterial wall. 17 The process underlying intimal thickening is not fully understood but is thought to be similar, though not identical, to that underlying atherosclerosis. The hypothesis that IMT represents subclinical vascular disease may be supported by the finding of graded associations between IMT and concurrent atherosclerotic change visualized in the coronary arteries during angiography.18,19 It is important to note, however, that whilst in many cases thickening of the intima–media does represent atherosclerotic change, in other cases it may represent non-atherosclerotic lesions such as hypertrophy in response to shear stress on the artery wall. 20
In 1986, Pignoli et al. demonstrated that B-mode ultrasound was a useful method for the measurement of cIMT, 17 where it appears as a ‘double echo’ in the artery wall. Since then, further studies have shown that the measurement of IMT by B-mode ultrasound is reliable and reproducible.21–23 Because of its ease of measurement and its status as a marker of atherosclerosis, IMT has been used in observational epidemiological studies and clinical trials as a surrogate marker of CVD. More recently, investigators have measured changes in IMT in response to cardiovascular drugs or interventions as a method of reducing follow-up time and sample sizes needed for individual studies. 24
Measurement of IMT
There is considerable variation between studies regarding measurement of IMT and how to summarize these measures. Subjects are assessed supine, with the neck extended and head turned to the side that is contralateral to the side on which the measurement is being taken, allowing maximal access to the great arteries of the neck. When the carotid artery wall is visualized by B-mode ultrasound, the characteristic ‘double echo’ of the intima–media complex can be seen on both the near and far wall of the carotid artery. 17 Early opinion differed on which of these two sites should be used to measure cIMT. However, near wall measurement of IMT has been shown to be heavily gain dependant and not representative of anatomical IMT, 16 whereas far wall measurements have been demonstrated to be representative of the true thickness17,25 such that it is now widely accepted that IMT should be measured on the far wall of the artery. 16 It has also been recommended to measure IMT at the end of diastole, as measurement during systole is affected by stretching of the arterial wall, and subsequent reduction in IMT. 26
There are several segments of the carotid artery in which IMT can potentially be measured – the common carotid, the carotid bifurcation or the internal carotid artery. Practically, visualization of the internal carotid or carotid bifurcation is more difficult than visualization of the common carotid artery (CCA), as these segments are more anatomically obscure. Several epidemiological studies have demonstrated a higher proportion of missing values and variability in measurements when measurements are taken from the internal carotid artery or carotid bifurcation. 27 Howard et al. found a moderate correlation between IMT measurements taken from different sites on the carotid artery but IMT at one site could not accurately predict IMT at another. 28 Overall, IMT measurement in the CCA is often favoured due to ease of access and reliability of measurements.
The importance of taking IMT measurements in areas of the artery wall that are free of plaque is increasingly recognized. Measuring in areas free of plaque is not only easier but also improves reproducibility and accuracy.29,30 The Mannheim consensus has defined plaque as a focal thickening that encroaches into the lumen by 0.5 mm or by 50% of the surrounding IMT or where intima–media thickening is > 1.5 mm.29,30 It is increasingly recognized that in terms of assessing disease burden and risk of subsequent vascular events, both IMT and plaque should be considered separately. Carotid plaque itself has been shown to be associated with concomitant CVD, and indeed is considered to be an indicator of high vascular risk. 31
The method used to summarize multiple IMT measurements from a given individual varies widely between studies. A systematic review and meta-analysis conducted by Lorenz et al. in 2007 found that in the eight key studies used for the meta-analysis, measures of IMT could be divided into two main groups: mean IMT and maximal IMT. Within each group, studies also differed in their use of observations from the far wall alone or from both the near and far wall. 5 Such variation in both the measurement of IMT and summary of multiple measurements affects our ability to make meaningful comparisons of findings across different studies, including those in which IMT has been assessed as a possible predictor of CVD.
As with any physiological measurement, measurement of IMT is prone to variability from a number of different sources, including the type of ultrasound machine used, the sonographer taking the images and the reader making the measurement. Thus, within research settings, the capture of images for the measurement of IMT, using B-mode ultrasound, is often done by a single radiographer, with measurement of IMT performed off line by several different readers. Salonen et al. found that when measured in this way, most variability in IMT measurements was due to inter-observer variation, with only 4% attributable to intra-observer variation. 32 In the Rotterdam Study, 87% of variability in cIMT after repeated scanning was due to difference between subjects and the remaining 13% was due to differences between sonographers and readers. 21 Methods used to reduce variability include standardized protocols for performing IMT measurement, and the use of edge detection programs. The latter may benefit from the development of automatic edge detection systems, although high-quality images are required for these systems.
Edge detection programs
The use of only one highly trained sonographer to reduce measurement variability can be achieved with reasonable ease within a research setting. In contrast, applying this method to clinical practice is almost impossible. Several groups have attempted to tackle this by employing computer-based measurement programmes to reduce inter-reader differences (Figure 1). Such programmes have evolved from those which require a significant level of user input, to systems which use edge detection algorithms to allow user-independent identification of the lumen–intima interface and the media–adventitia interface of the cIMT. Both the American Society of Echocardiography (ASE) and Mannheim consensuses suggest the measurement of cIMT with edge detection programs,30,33 although other methods have been developed, several of which are discussed in a 2010 review of computer-aided cIMT measurement by Molinari et al. 34
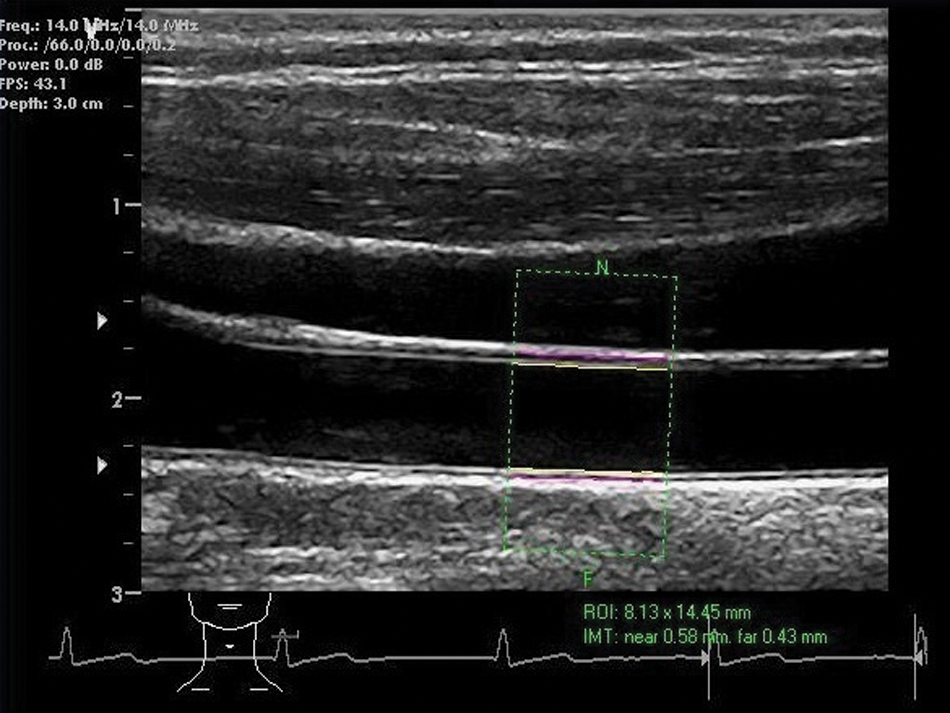
Measurement of cIMT using edge detection software.
Edge detection systems rely on the differing pixel intensities of the lumen and adventitia on ultrasound imaging of the artery wall. The adventitia is bright in intensity, in contrast to the lumen. These unique intensity profiles make both the lumen and adventitia easily identifiable when analysing pixel gradients down the artery wall, with the IMT being the distance between the two. Several studies have proposed techniques based on such image gradients. Liguori et al. describe such a system which had a measurement uncertainty of 0.02 mm, although noise interference provided an obstacle and the system was not fully automated. 35 Later systems have been based on similar gradient-based algorithms and have shown improved measurement error.36,37 However, some user input beyond scan acquisition is still required in these methods, with the user needed to select the region of interest for measurement. 34 The progression to completely automated edge detection systems is the next step and is a continued area of research. 38
In 2011, Peters et al. made a comparison between semi-automated and manually acquired cIMT measurements. They found both techniques to be highly reproducible and to have the same associations with cardiovascular risk factors. 39 The Multi-Ethnic Study of Atherosclerosis (MESA) study also found edge detected readings to be comparable to manually traced readings and they maintained associations with cardiovascular risk factors. 40 Importantly, the use of edge detection reduced inter-reader bias, making it more applicable to clinical practice.
Consensus recommendations for the measurement and application of IMT
cIMT is not commonly used in clinical practice, at least in part because of its variability and the need to have trained and dedicated staff to perform the measurements. However, cIMT is increasingly used as a research tool, including as a surrogate cardiovascular endpoint in clinical trials.41–47 In an attempt to standardize cIMT measurements and to provide recommendations for the use of cIMT in clinical, epidemiological and trial-based practice, a number of guidelines and consensus statements have been produced.26,29,30
Originally drafted in 2004, and updated in 2006, the Mannheim consensus aimed to standardize the measurement of cIMT in order to facilitate the analysis and meta-analysis of data on cIMT.29,30 It also aimed to clarify the classification of early and late atherosclerotic change. The consensus defines IMT and plaque separately and also notes that there are several intermediate stages between early and late atherosclerosis that are not easily identified on ultrasound imaging of the carotid artery and even on histological assessment. 29 The consensus also highlights that IMT and plaque do not share the same associations with atherosclerosis risk factors and recognizes that IMT and plaque have different predictive values for vascular disease. Recommendations for measurement of cIMT include taking readings in an area free of plaque, and separate documentation of readings from different sites (CCA, carotid bulb and internal carotid artery).
The ASE consensus has also provided recommendations for the use of cIMT in clinical practice.26,33 It is suggested that people categorized as intermediate risk by traditional scoring systems may benefit most from IMT scanning. This is often a heterogeneous group, consisting of those at a true low risk, those at true intermediate risk and those at true high risk. IMT may therefore be useful in identifying those at true high risk, enabling them to receive appropriate management. Conversely, those at true low risk could be correctly identified, avoiding unnecessary treatment. They also recommended that scanning be avoided in those whom the result would not change management. The ASE statement also provides recommendations for scanning procedures, including taking cIMT measurements from the far wall of the common carotid only, followed by assessment of plaque in the common carotid, bifurcation and internal carotid. 33
cIMT as a predictor of cardiovascular events
Early data from multiple large epidemiological studies showed that IMT is correlated with the extent of atherosclerosis in the coronary arteries18,19 and with other risk factors for CVD.48–52 More recently, several studies (Tables 1 and 2) have assessed the association of cIMT with incident CVD, including both CHD and stroke, to determine whether this measure can also predict future cardiovascular events.
Summary of major epidemiological studies of predictive ability of cIMT
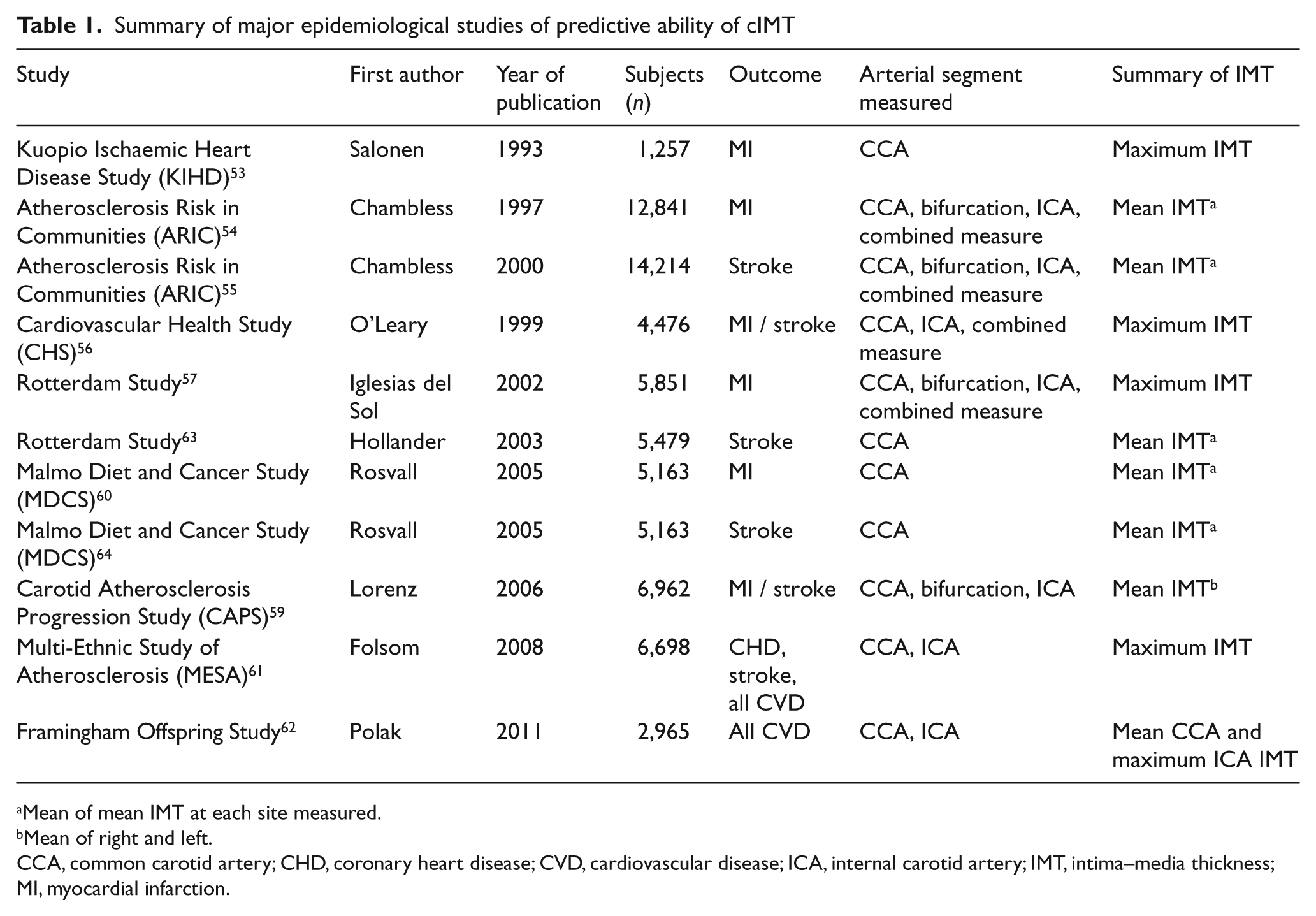
Mean of mean IMT at each site measured.
Mean of right and left.
CCA, common carotid artery; CHD, coronary heart disease; CVD, cardiovascular disease; ICA, internal carotid artery; IMT, intima–media thickness; MI, myocardial infarction.
Prediction of cardiovascular events by cIMT in epidemiological studies
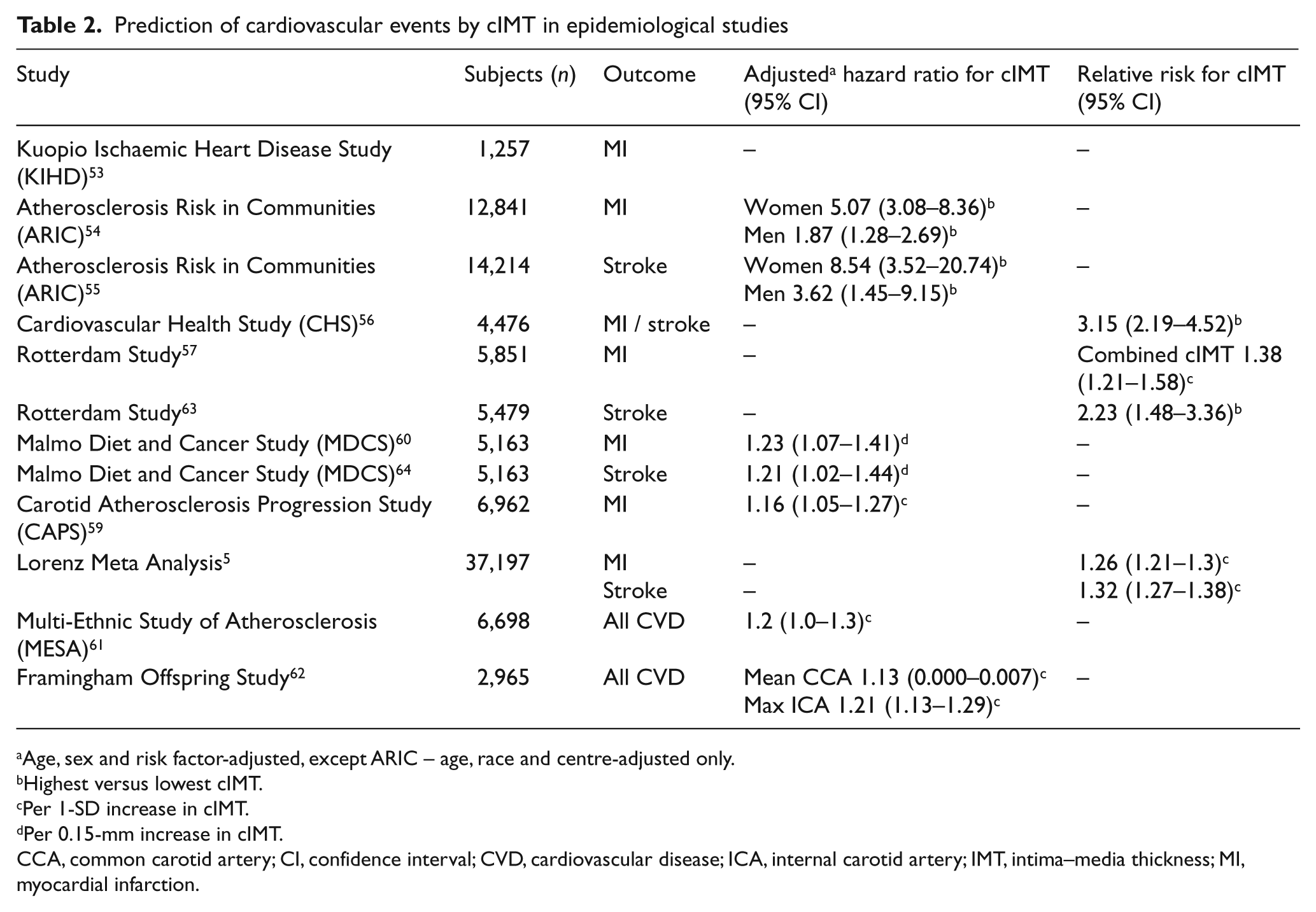
Age, sex and risk factor-adjusted, except ARIC – age, race and centre-adjusted only.
Highest versus lowest cIMT.
Per 1-SD increase in cIMT.
Per 0.15-mm increase in cIMT.
CCA, common carotid artery; CI, confidence interval; CVD, cardiovascular disease; ICA, internal carotid artery; IMT, intima–media thickness; MI, myocardial infarction.
In the Kuopio Ischaemic Heart Disease Risk Factor Study, 1257 men free of vascular disease and aged 43–60 years at baseline were followed-up for 3 years. 53 Intimal thickening of the carotid artery wall was found to be predictive of incident myocardial infarction (MI), with a 0.1-mm increase in IMT representing an 11% increase in risk of MI.
The Atherosclerosis Risk in Communities (ARIC) study recruited 12,841 men and women aged 45–64 years with no previous history of CVD, and measured IMT in the CCA, internal carotid artery and carotid bifurcation (mean of the individual IMT measurements for each section of the far wall of the carotid artery).54,55 After adjusting for age, race and centre, extreme mean IMT > 1 mm, when compared to IMT < 1 mm, was associated with an increased incidence of CVD for both women and men (hazard ratio (HR) 5.07 (95% CI 3.08–8.36) and 1.87 (95% CI 1.28–2.69), respectively). 54 However, after additional adjustment for conventional cardiovascular risk factors, IMT was associated with increased cardiovascular risk only in the group of subjects with the highest IMT (0.8 mm and above when compared with the lowest tertile of IMT).
The Cardiovascular Health Study examined cIMT in relation to future risk of MI and stroke in 4476 subjects with no history of CHD and a mean age of 72.5 years. 56 After a median follow-up of 6.2 years and after adjustment for major conventional cardiovascular risk factors, subjects in the higher quintiles of maximal IMT had an increased relative risk (RR) for MI or stroke compared with those in the lower quintiles. A linear increase in RR of MI or stroke was evident across quintiles, with a higher RR in the highest quintile compared with the lowest quintile (risk factor adjusted RR 3.15 (95% CI 2.19–4.52).
In the Rotterdam Study, a prospective study of men and women aged 55 years and above, data were collected on cardiovascular risk indicators and incident MIs, with carotid ultrasound performed on 5851 subjects. Increased cIMT was a strong predictor of MI after adjustment for age and sex. IMT was measured in three different sections of the carotid artery (common carotid, bifurcation and internal carotid) and a combined cIMT value was derived which included all three measurements. When adjustment for traditional risk factors was included in addition to age and sex, IMT measured in the CCA, at the bifurcation and the combined measure were all significantly associated with incident MI after 4.6 years (RR per 1-SD increase in IMT 1.37, 1.28 and 1.38, respectively). 57 However, an earlier study from the same group revealed that when added to models with conventional risk factors, cIMT did not significantly improve predictive ability of cIMT for CHD. 58
In the Carotid Atherosclerosis Progression Study, 6962 people drawn from a primary health care scheme underwent baseline IMT measurement (at all carotid segments) and 5056 participants were followed-up for MI, stroke and death, with an average follow-up of 4.2 years. An increased unadjusted HR for IMT was found at all segments for all outcomes, suggesting that IMT was predictive of MI, stroke and death. Following adjustment for age, sex and vascular risk factors, IMT at the CCA and bifurcation remained predictive for MI (HR 1.16 (95% CI 1.05–1.27) and 1.16 (95% CI 1.05–1.28), respectively) and the combined endpoint of MI, stroke or death. 59
In just over 5000 participants from the Malmo Diet and Cancer Study, a 10-year cohort study of people living in Malmo, Sweden, cIMT, carotid plaque and carotid stenosis were associated with future coronary events (fatal and non-fatal MI or death from CHD). These relationships persisted following adjustment for conventional risk factors (HR for 0.15-mm increment in CCA-IMT 1.23 (95% CI 1.07–1.41)). The relationship was reduced slightly following additional adjustment for carotid plaque but remained significant for those with higher cIMT. 60
In 2008, the MESA study published data looking at coronary artery calcium and cIMT in relation to incident vascular disease and found that cIMT was predictive of events, although not to the same extent as coronary artery calcium. The age, race and sex-adjusted HR for cIMT was 1.3 (95% CI 1.1–1.4), which reduced to 1.2 (95% CI 1.0–1.3) upon multivariate adjustment. 61 More recent data from the Framingham Offspring Study have also examined the predictive ability of cIMT in their cohort. 62 They measured both the maximal IMT in the internal carotid artery and the mean IMT in the CCA in 2965 men and women enrolled in the cohort. They established that both mean common carotid IMT and maximal internal carotid IMT could predict future vascular events. The HR for CVD with a 1-SD increase in mean cIMT of CCA was 1.13 (95% CI 0.000–0.007). Similarly, for the maximum cIMT of internal CCA, the HR was 1.21 (95% CI 1.13–1.29). However, only internal carotid IMT improved clinical risk classification significantly, with a net reclassification index (NRI) of 7.6% (p < 0.001) compared with that of the CCA (0.0%, p = 0.99).
cIMT and stroke
As well as an association with MI, several studies have found an association between cIMT and both prevalent and incident stroke. In the Rotterdam Study, a cIMT > 0.84 mm was predictive of incident stroke and remained so after adjustment for conventional risk factors (RR 2.23 unadjusted; 2.42 adjusted) when compared to the lowest tertile. This relationship was found to be stronger than the relationship between plaque and stroke. 63 In the Malmo Diet and Cancer Study, common carotid IMT was associated with the incidence of stroke, independent of the presence of carotid plaque (adjusted HR 1.21 (95% CI 1.02–1.44)). 64 In ARIC, increasing IMT was associated with increased risk of stroke, although the relationship was not linear and was attenuated somewhat after the addition of common stroke risk factors into the model. 55 In the Cardiovascular Health Study, the RR of stroke was increased in the highest quintile of IMT, although this was not entirely linear across the quintiles. The associations between IMT and a combined endpoint of MI or stroke were at least as strong as for traditional risk factors, including systolic BP and smoking. 56 In contrast, however, the MESA study did not demonstrate an association between cIMT and incident stroke, with an adjusted overall HR per 1-SD increment of 1.1 (95% CI 0.8–1.3). 65
cIMT in higher risk groups
Most large population-based studies of IMT have investigated prediction of risk in a largely healthy population. Considerably fewer have considered cIMT as a predictor of cardiovascular events in specific high-risk groups. An example of such a group is people with type 2 diabetes. There is little evidence on the relationship between IMT and incident CVD risk in this population, despite current use as a surrogate marker in trials involving people with type 2 diabetes.41,42,66–68 A recent systematic review identified 24 studies on cIMT in patients with type 2 diabetes and impaired glucose tolerance, 69 the results of which indicated that people with type 2 diabetes had a higher cIMT than comparable non-diabetic people, with an average difference of 0.13 mm. However, all the studies were cross- sectional and no prospective data are available on the relationship between IMT and subsequent cardiovascular events in this group. In 2009, a study of 150 subjects with diabetes mellitus demonstrated an association between cIMT and prevalent CVD determined by CT coronary angiography, with those with normal coronary arteries having the lowest cIMT (0.58 mm) and those with obstructive stenosis having the highest cIMT (0.75 mm). 70 As has been previously discussed, it has been recommended that cIMT should not be measured in high-risk groups for the purposes of risk prediction. However, the USPSTF statement suggests that there is a gap in the available evidence 6 and thus more research is required before any judgement can be made on this issue.
IMT and clinical risk prediction
In 2007, Lorenz et al. undertook a systematic review and meta-analysis, including data from many of the large epidemiological studies already discussed. Although they identified a number of potential sources of heterogeneity, including ultrasound protocols, the meta-analysis indicated that carotid IMT was a strong predictor of future vascular events. 5 The age and sex-adjusted estimated RR for cIMT in prediction of MI was 1.26 (95% CI 1.21–1.30) per 1-SD CCA-IMT difference and for stroke was 1.32 (95% CI 1.27–1.38) per 1-SD CCA-IMT difference. Estimates were slightly lower per 0.01-mm difference in cIMT. 5 Therefore, its use in clinical risk prediction should be considered further.
As an emerging cardiovascular risk predictor, the role of IMT in addition to traditional risk prediction methods has been considered by several studies, an approach recommended by the USPSTF statement. 6 Whilst the univariate relationship between cIMT and future vascular events would suggest potential usefulness for the cIMT in risk prediction, evidence for an additional benefit over and above existing risk prediction models (e.g. Framingham risk scoring), is less certain.
The ARIC investigators recently published data examining IMT measurements and their incremental contribution to risk prediction. They found that adding cIMT and plaque information to traditional risk factors improved the prediction of vascular events compared with traditional risk factors alone. A total of 23% of cases were reclassified, with a net reclassification index (NRI) of 9.9%. 71 This represented a clinical NRI of 21.7% (clinical NRI is the NRI only for those at intermediate risk of disease). In addition, they examined whether CCA-IMT was as good as all-segment IMT (A-IMT) in the prediction of vascular events, when added to traditional risk factors and plaque information. 72 They determined that there was no difference in the NRI and clinical NRI of CCA-IMT or A-IMT added to plaque when compared with traditional risk factors only, suggesting that CCA-IMT would be sufficient for use in clinical risk prediction in addition to plaque.
Unfortunately, data concerning the use of IMT in addition to traditional risk scores is not clear cut. Recently published data from the Carotid Atherosclerosis Progression Study (CAPS) found that when added to the Framingham risk score and SCORE models, IMT did not significantly improve risk prediction in their cohort. 73 Overall, 8.8% of people were reclassified following IMT measurement and, of these, 30% were classified correctly. As such, the net reclassification was −1.41%. This suggests that IMT may not be useful in risk prediction, despite its association with incident vascular disease. 73
In 2010, Simon et al. published a review examining the impact of IMT and plaque on risk prediction. 74 They found that whilst IMT independently predicted CHD, it made only a modest improvement to risk prediction and suggested that carotid plaque may be a more useful tool in predicting CHD.
A recent systematic review which examined several novel markers of risk in addition to traditional risk assessment concluded that there was strong evidence that cIMT improved risk prediction above traditional assessment. 75 Twelve studies were identified, and nine showed an increase in C statistic when cIMT was added to the traditional risk assessment. Six of the studies identified reported the NRI and, of those, five showed a positive NRI, with only one showing a negative NRI (range −1.4 to 11.6%).
In the setting of CVD prevention, it is important to consider the result of identifying individuals with a raised cIMT. Patient motivation and physician action following cIMT measurement and how these may lead to preventive interventions at an individual level are key areas to be explored. A small study by Korcarz et al. examined the effect of measuring cIMT in a clinical setting on both physician and patient motivations. Physicians were more likely to prescribe aspirin and lipid-lowering medications whilst patients were more aware of their increased cardiovascular risk and were therefore more likely to want to take lipid-lowering medications. 76 In a study focusing on carotid plaque rather than cIMT, physicians were more likely to prescribe aspirin and lipid-lowering therapy if carotid plaque was identified, but patients, despite reporting that they perceived they had an increased cardiovascular risk if a plaque was recorded, did not report increased motivation to improve lifestyle factors. 77
Conclusions
At present, the evidence is unclear as to whether IMT could be a useful tool for predicting vascular risk in clinical practice. B-mode ultrasound is a non-invasive and inexpensive method for measuring IMT. Many large epidemiological studies have shown a strong relationship between IMT and incident CVD, but the evidence for the use of cIMT in clinical practice is incomplete. Current evidence suggests that for people at intermediate risk according to Framingham risk scoring, IMT may add useful information on vascular risk but findings are inconsistent between studies and, in some cases, the improvement in classification is modest. Most studies have looked only at general healthy populations who have no prior history of CVD and there is not the same body of evidence regarding its use in people who have already experienced a cardiovascular outcome, or in those who are already at a higher risk.
Although the overall evidence to date suggests that cIMT has the potential to be a clinically useful predictor of vascular risk, there is a continued need for cIMT to be tested over and above traditional risk scores in large cohorts and at-risk populations. A single unified protocol for the measurement of cIMT could allow wider adoption of cIMT in clinical practice. Furthermore, evidence that measuring cIMT in a clinical setting changes physician management and patient motivation would add considerable weight to the value of cIMT as a risk predictor. This needs to be included as an additional focus in future research on cIMT.
Footnotes
Acknowledgements
Acknowledgement is due to Professor John Deanfield and his team at University College London for kindly providing a figure for use in this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
