Abstract
Background
Myelin oligodendrocyte glycoprotein antibody-associated disorder (MOGAD) is a rare central nervous system demyelinating disease with a variable course, including both monophasic and relapsing phenotypes. Effective treatment options, particularly for relapsing disease, remain limited. Ofatumumab (OFA), a subcutaneous anti-CD20 monoclonal antibody approved for multiple sclerosis, has not been studied in pediatric MOGAD. This study aims to assess its safety and efficacy in this population.
Objective
To describe the safety and efficacy of off-label OFA use in pediatric MOGAD patients.
Patients and Methods
We conducted a retrospective case series of consecutive pediatric patients with relapsing MOGAD who received OFA. Clinical data, including relapse history, annualized relapse rate (ARR), laboratory findings, and adverse events, were collected and analyzed.
Results
Three pediatric (1 female, 2 males) were included. Age at onset was 2.3, 6.1, and 2.5 years; disease duration before OFA was 6.3, 2.2, and 9.1 years; and age at OFA initiation was 8.6, 8.3, and 11.6 years. Two patients started OFA due to breakthrough relapses on mycophenolate mofetil (MMF); one switched from maintenance intravenous immunoglobulin (IVIG) for administrative convenience. Treatment durations were 12, 9, and 9 months. All patients achieved rapid B-cell depletion (CD19+ < 10 cells/μL), which was sustained in two. Serum MOG-IgG titers increased in two patients and became negative in one. Two patients remained relapse-free during OFA treatment; one experienced a single relapse at 3 months. ARR decreased in all three patients post-OFA initiation. OFA was well tolerated. Only one patient developed transient fever after the first injection, which resolved with symptomatic treatment and did not recur.
Conclusion
In this small case series, OFA was generally well-tolerated and associated with a reduction in ARR, suggesting a potential role in combination therapy for relapsing pediatric MOGAD. Given the use of combination treatments in some cases, including OFA+MMF and IVIG, the role of OFA alone remains unclear. Its subcutaneous route provides practical advantages in terms of convenience. Owing to the small sample size and relapse observed in one patient, these findings remain preliminary and warrant validation in larger, prospective studies.
Plain Language Summary
Myelin oligodendrocyte glycoprotein antibody-associated disorder (MOGAD) is a rare disease that affects the central nervous system and can cause problems with the brain and spinal cord. It can either occur as a single event or in relapses. The condition is difficult to treat, and effective treatment options for children are limited. Ofatumumab (OFA) is a medication approved for treating multiple sclerosis in adults and works by targeting certain cells in the immune system. This study looks at whether OFA could help children with MOGAD, especially those who have repeated episodes (relapses). We reviewed the medical records of three children who received OFA as part of their treatment for relapsing MOGAD. These children had been dealing with MOGAD for several years before starting OFA. After starting the medication, the children showed improvements. Specifically, two children remained free from further relapses, and one had only one relapse after a few months. In addition, the number of relapses in all three children decreased significantly. The treatment was well-tolerated, with only one child experiencing a mild side effect (a temporary fever) after the first dose. Although this is a small study with only a few children, the results suggest that OFA might be a safe and helpful option for treating relapsing MOGAD in children. However, more research with larger groups of patients is needed to confirm these early findings and determine how OFA can best be used for this condition.
1. Introduction
Myelin oligodendrocyte glycoprotein antibody-associated disorder (MOGAD) is a distinct inflammatory demyelinating disease of the central nervous system (CNS). Although rare, MOGAD affects all age groups, with a higher incidence observed in pediatric populations. The clinical presentation is heterogeneous, including acute disseminated encephalomyelitis (ADEM), optic neuritis (ON), transverse myelitis (TM), cortical encephalitis, cerebral lesions, and brainstem or cerebellar syndromes. 1 MOGAD may follow either a monophasic or relapsing disease course. According to multiple cohort studies, the relapse rate ranges from 17% to 36% over follow-up periods of 1 to 8 years. 2 Accumulated CNS damage from recurrent attacks is believed to be the primary contributor to long-term disability in MOGAD. 3 Among patients with relapsing disease, only 31% to 50% achieve full recovery after a relapse. 4 These findings underscore the importance of maintenance immunotherapy to reduce relapse risk and prevent disability. However, there are currently no approved treatments for MOGAD, and therapeutic decisions are often based on limited evidence derived primarily from retrospective studies, small cohort data, or expert consensus, due to the lack of large-scale, particularly prospective, investigations.
The immunopathology of MOGAD involves both humoral and cellular mechanisms. MOG-specific IgG1 antibodies, produced by plasmablasts and plasma cells, are a hallmark feature. 1 CD4+ T cells are thought to initiate CNS inflammation, while B cells contribute critically by presenting antigens, promoting T cell activation, and producing MOG-specific antibodies. These immune interactions form the basis for the use of B cell–depleting therapies in selected patients with MOGAD. Among these, B cell–targeted treatments have demonstrated clinical efficacy. 5 Preliminary findings from observational studies suggest that rituximab (RTX), an anti-CD20 monoclonal antibody, may reduce relapse rates in patients with MOGAD. 4
RTX, the first murine/human monoclonal anti-CD20 antibody licensed against human diseases, is a unique therapeutic option. It targets CD20, a surface antigen predominantly expressed on B-cells from an early developmental stage, including pre-B-cells, but not on precursor hematopoietic stem cells or antibody-secreting plasma cells. However, it’s important to note that CD20 is not exclusively specific to B-cells, as a small subset of T cells, approximately 3-5% of the total CD3 T cells, also express CD20.6,7 RTX is administered intravenously and requires pretreatment with antihistamines and steroids. The most common adverse events associated with RTX treatment are infusion-related, accounting for about 10%. 5 Ofatumumab (OFA) are humanized anti-CD20 monoclonal antibody. 7 OFA binds to a small-loop epitope on CD20 and efficiently depletes B cells through complement-dependent cytotoxicity and antibody-dependent cell-mediated cytotoxicity, even when CD20 expression is low. 7 The subcutaneous administration of OFA, delivered into the hypodermis, may lead to more efficient and selective targeting of B cells residing in the lymphatic circulatory system than intravenous administration directly entering the systemic circulation. 8 These unique mechanisms of action and administration are intriguing and warrant further exploration.
OFA has been approved for the treatment of relapsing multiple sclerosis (MS) in adults; however, evidence regarding its safety and efficacy in other demyelinating disorders remains limited. Hiya et al reported a successful case of OFA treatment in a 17-year-old adolescent patient with refractory MOGAD, where the treatment prevented further relapses. 9 Additionally, Chen et al described two adult MOGAD patients who were also treated with OFA during the acute phase, showing quick improvement after three injections of the drug, despite the presence of Epstein-Barr virus infection. 10 The patients in the current study are younger than those reported in these two studies. This study aims to describe the safety profile and therapeutic potential of OFA in pediatric patients with MOGAD.
2. Subjects and Methods
2.1. Study Design and Patients
This retrospective case series was approved by the Institutional Review Board of Guangzhou Women and Children’s Medical Center. We consecutively identified all pediatric patients (aged <18 years) with a diagnosis of MOGAD who were treated with OFA at our center between January 2023 and July 2024 and had a minimum follow-up of 6 months after treatment initiation. Patients were excluded if they had an alternative diagnosis explaining the demyelinating events, concomitant AQP4-IgG positivity, or incomplete clinical, imaging, or follow-up data.
2.2. Diagnostic Criteria
Diagnostic criteria for MOGAD, proposed by Jarius et al, are as follows: (1) monophasic or relapsing acute optic neuritis, myelitis, brainstem encephalitis, or encephalitis or any combination of these syndromes; (2) Magnetic resonance imaging (MRI) or electrophysiological (visual evoked potentials in patients with isolated optic neuritis) findings compatible with central nervous system demyelination; and (3) seropositivity for MOG-IgG as detected using a cell-based assay employing full-length human MOG as target antigen. 11 All included patients also fulfilled the international 2023 Banwell diagnostic criteria for MOGAD.
2.3. Treatment and Monitoring
OFA was administered subcutaneously following the MS dosing schedule: 20 mg on days 0, 7, 14, and then every 4 weeks. 12 Peripheral CD19+ B-cell counts were monitored regularly by flow cytometry. B-cell depletion was defined as CD19+ count < 10 cells/μL. 13 Before initiation of OFA, all patients underwent standard safety screening, including hepatitis B virus serology, tuberculosis screening, and vaccination status assessment, in accordance with institutional protocols. Patients with active infection or contraindications to immunosuppressive therapy were excluded. Immunoglobulin levels were monitored longitudinally in all patients to assess for hypogammaglobulinemia. At the time of manuscript preparation, all patients remained on OFA treatment with ongoing 4-weekly dosing; therefore, post-discontinuation follow-up data were not available. Serum MOG-IgG titers, IgG/IgM levels, and clinical assessments for relapses and adverse events were tracked during follow-up. Relapses were defined as new neurological symptoms lasting >24 hours in the absence of fever. The MOG IgG and Aquaporin-4 (AQP4) IgG in serum were detected by the fixed cell-based assay commercial kit (Shaanxi Maiyuan Biotechnology Co., Ltd, Shanxi, China). These methods had been reported in detail in our previous study. 14
2.4. Measurement of CD19+ B Cell Counts
Peripheral blood CD19+ B cell counts were measured by flow cytometry using the BD Multitest IMK kit (catalog number 340503; BD Biosciences, CA, USA). The analysis was performed in the clinical laboratory of our hospital. This method has been described in detail in their previous study. 14
2.5. Outcomes
The primary outcomes were the annualized relapse rate (ARR) before and during OFA treatment and the occurrence of adverse events. Secondary outcomes included changes in CD19+ B-cell counts and MOG-IgG titers.
3. Results
OFA was administered to three pediatric patients with MOGAD, designated as Case 1 to Case 3 for presentation clarity.
Case 1: A female patient presented at 2.3 years of age with an acute episode of gait disturbance and irritability. Her prior development was age-appropriate, with no relevant personal or family history of neurological or autoimmune disorders. Cerebrospinal fluid (CSF) analysis showed normal white blood cell (WBC) and protein levels. CSF polymerase chain reaction (PCR) testing was negative for common pathogens including enterovirus, influenza A/B, herpes simplex virus (HSV), Epstein–Barr virus (EBV), and cytomegalovirus (CMV). Oligoclonal bands (OCBs) were absent in both CSF and serum. Brain MRI demonstrated multifocal T2-weighted hyperintensities involving the bilateral basal ganglia, right cerebral white matter, and right cerebellum, consistent with acute demyelination. Spinal MRI was normal. Serum AQP4 and MOG antibodies were tested before initiation of any immunotherapy and were negative. A diagnosis of an ADEM-like episode was made. She was treated with intravenous methylprednisolone (IVMP) followed by oral prednisone, in combination with intravenous immunoglobulin (IVIG). Her symptoms improved, and follow-up brain MRI demonstrated partial resolution of the lesions.
The first relapse occurred 20 months after disease onset, presenting with left-sided ON and encephalitic symptoms. This relapse was managed at an external institution; detailed investigations were unavailable, though the patient reportedly received IVMP. A second relapse occurred at 27 months, presenting with left-sided ON and ataxia. Brain MRI demonstrated multifocal T2-weighted hyperintensities involving the right cerebellar hemisphere, left parieto-occipital white matter, and right temporoparietal region. Orbital MRI with gadolinium contrast revealed focal enhancement of the left optic nerve. Repeat testing for serum AQP4 and MOG antibodies before initiation of any immunotherapy, serum MOG IgG was 1:32, while AQP4-IgG remained negative. A diagnosis of MOGAD was made at that time. She received IVMP and IVIG during the acute phase, and MMF was subsequently initiated as maintenance therapy for relapse prevention at 250 mg once daily and titrated to 250 mg twice daily (approximately 20 mg/kg/day).
Despite 3.4 years of MMF treatment, disease activity persisted, with an ARR of 0.6. At the time of the fourth relapse, she presented with hand tremor. Brain MRI demonstrated multifocal T2-weighted hyperintensities involving bilateral subcortical white matter of the temporal and occipital lobes, the medulla oblongata, right pontine brachium, and right cerebellar hemisphere, with increased clarity of the right pontine lesion compared to previous scans. Orbital MRI revealed bilateral optic nerve enhancement, while spinal MRI showed no abnormalities (Figure 1A and C). Serum MOG-IgG tested negative at this time. Following the fourth relapse, OFA was added to MMF at age 8.6 years. Over 12 months of OFA treatment, B-cell depletion was achieved and sustained (CD19+ B cells ≤5.02 cells/μL). Follow-up MRI at 6 and 12 months showed no new or enlarging lesions (Figure 1B and D). Although serum MOG-IgG converted from negative at baseline to a titer of 1:320 after 9 months of OFA therapy, the patient remained relapse-free, with the ARR decreasing from 0.6 prior to OFA to 0.0 during treatment (Figure 2 & Table 1). MRI changes before and after ofatumumab treatment. MRI of case 1: (A) Axial T2-weighted FLAIR brain MRI image before ofatumumab treatment, showing hyperintense lesions in the bilateral subcortical white matter of the temporal and occipital lobes; (B) Axial T2-weighted FLAIR brain MRI image after 12 months of ofatumumab treatment, showing a reduction in the size of the hyperintense lesions in the bilateral subcortical white matter of the temporal and occipital lobes, with no new lesions; (C) Axial T1-weighted post-contrast optic nerve MRI image before ofatumumab treatment, showing bilateral optic nerve enhancement. (D) Axial T1-weighted post-contrast optic nerve MRI image after 12 months of ofatumumab treatment, showing slight reduction of bilateral optic nerve enhancement. MRI of case 2: (E) Axial T2-weighted FLAIR brain MRI image before ofatumumab treatment, showing hyperintense lesions in the right frontal subcortical white matter, left temporal subcortical white matter, and right basal ganglia; (F) Axial T2-weighted FLAIR brain MRI image at 3 months after ofatumumab treatment, showing marked reduction of the previously noted hyperintense lesions; (G) Axial T2-weighted FLAIR brain MRI image at 6 months after ofatumumab treatment, showing near-complete resolution of the prior lesions with no new lesions identified; (H) Axial T1-weighted post-contrast optic nerve MRI image before ofatumumab treatment, showing normal appearance of the bilateral optic nerves without swelling or enhancement. (I) Axial T1-weighted postcontrast optic nerve MRI image at 3 months after ofatumumab treatment, showing diffuse swelling of the bilateral optic nerves without significant contrast enhancement. (J) Axial T1-weighted post-contrast optic nerve MRI image at 6 months after ofatumumab treatment, showing reduced optic nerve swelling without contrast enhancement. MRI of case 3: (K) Axial T2-weighted FLAIR brain MRI image before ofatumumab treatment, showing hyperintense lesions in the bilateral basal ganglia and left thalamus; (L) Axial T2-weighted FLAIR brain MRI image after 6 months of ofatumumab treatment, showing a reduction in the size of the high signal lesions in the bilateral basal ganglia and left thalamus, with no new lesions Disease course and treatment regimen before and after the initiation of ofatumumab (time 0) in patients with MOGAD. IVIG: intravenous immunoglobulin; MMF: mycophenolate mofetil; OFA: ofatumumab; RTX: rituximab Characteristics of the Patients Abbreviations: MOG, myelin oligodendrocyte glycoprotein; MMF, mycophenolate mofetil; mo, moths; OFA, ofatumumab; RTX, rituximab; yrs, years.


Case 2: A male patient presented at 6.1 years of age with acute fever and headache. His prior development was age-appropriate, with no relevant personal or family history of neurological or autoimmune disorders. CSF analysis showed WBC was 145x106/L with a lymphocytic predominance of 73%, and an increased protein level of 0.61 g/L.CSF PCR was negative for enterovirus, influenza A/B, HSV, EBV and CMV. OCBs were absent in both CSF and serum. Brain MRI demonstrated T2-weighted hyperintensities in the bilateral frontal white matter. Spinal MRI revealed a longitudinally extensive transverse myelitis lesion extending from C5 to T5, without gadolinium enhancement. Orbital MRI was unremarkable. Serum AQP4 and MOG antibodies were tested before initiation of any immunotherapy and were negative. The initial diagnosis was ADEM. He was treated with IVMP followed by oral prednisone combined with IVIG. His symptoms improved, and follow-up brain MRI demonstrated partial resolution of the lesions.
A relapse occurred 4 months after disease onset, presenting with ADEM-like phenotype. Brain MRI showed new T2-hyperintense lesions in in the right caudate head and lentiform nucleus. Repeat serology was positive for MOG-IgG (titer 1:32) before initiation of immunotherapy, establishing the diagnosis of MOGAD. The acute episode was managed with IVMP and IVIG; however, maintenance immunotherapy was initially declined by the parents. Following a third relapse, MMF was initiated at 250 mg once daily and gradually titrated to 375 mg twice daily (approximately 22 mg/kg/day).
Despite 11 months of MMF treatment, relapse continued. At the time of the fourth relapse, he presented with acute onset of bilateral lower limb paralysis, hand tremors, and altered consciousness. Brain MRI showed new multifocal T2-weighted hyperintensities involving the left dentate nucleus, left temporoparietal region, right parietal lobe, bilateral frontal white matter, and bilateral basal ganglia (Figure 1E). Spinal and orbital MRI remained unremarkable (Figure 1H). Reversal pattern visual evoked potentials revealed reduced P100 amplitudes bilaterally, with preserved latencies, indicating bilateral optic nerve dysfunction. Serum MOG-IgG tested negative at this time.
OFA was added to MMF at 8.3 years of age. B-cell depletion (CD19+ < 2 cells/μL) was achieved after initiation and sustained throughout the 9-month treatment period, with corresponding B-cell counts of 1.00 and 2.00 cells/μL at months 6 and 9, respectively. Notably, one month after starting OFA, serum MOG-IgG increased from negative to a titer of 1:320. At month 3 of OFA therapy, the patient developed a breakthrough relapse characterized by bilateral optic neuritis (Figure 1F and I). Serum MOG-IgG titer at relapse was 1:320. Peripheral B-cell counts were not available at the time of relapse because blood sampling was declined by the parents. OFA was continued according to the standard dosing schedule; however, given the acute visual involvement and the established efficacy of IVIG in MOGAD, adjunctive maintenance IVIG was added rather than relying on anti-CD20 therapy alone (Figure 2 & Table 1). Subsequent MRI at 6 months post-OFA showed no new or enlarging lesions compared to baseline (Figure 1G and J). No further relapses occurred thereafter, and the ARR decreased from 1.8 pre-treatment to 1.3 during the OFA treatment period.
Case 3: A male patient presented at 2.5 years of age with acute onset of fever and seizure. His prior development was age-appropriate, with no relevant personal or family history of neurological or autoimmune disorders. CSF analysis revealed a WBC count of 18 × 106/L; differential classification was not performed due to the limited number of cells. CSF protein was 0.21 g/L.CSF PCR testing was negative for enterovirus, influenza A/B, HSV, EBV and CMV. Brain MRI demonstrated multiple T2-weighted hyperintense lesions involving the bilateral cerebral cortex and subcortical regions, as well as the left lentiform nucleus and thalamus. Spinal and orbital MRI findings were unremarkable. Spinal and Orbital MRI was unremarkable. Serum AQP4 and MOG antibodies were tested before initiation of any immunotherapy and were negative. The initial diagnosis was ADEM. He was treated with IVMP followed by oral prednisone combined with IVIG. His symptoms improved, and follow-up brain MRI demonstrated partial resolution of the lesions.
A relapse occurred one year after disease onset, presenting with ADEM-like phenotype. Brain MRI showed new T2-hyperintense lesions in right caudate head and lentiform nucleus. Spinal MRI showed a longitudinally extensive T2-hyperintense lesion was identified from C4 to T11, with gadolinium enhancement. Orbital MRI was unremarkable. Both CSF and serum OCBs were negative. Repeat testing for serum AQP4 and MOG antibodies before initiation of immunotherapy, serum MOG IgG was 1:64. A diagnosis of MOGAD was made at that time. He received IVMP and IVIG during the acute phase, and RTX was subsequently initiated as maintenance therapy for relapse prevention.
During 3.7 years of RTX treatment, he experienced two relapses. At the fifth relapse, an allergy shock occurred during RTX treatment, leading to its discontinuation. Maintenance IVIG was initiated and continued for 21 months during which no further relapses occurred. At 11.6 years of age, the patient was switched to subcutaneous OFA for convenience, and IVIG was discontinued.
After 9 months of OFA treatment, B-cell depletion was achieved initially (CD19+ = 10.00 cells/μL) and sustained at month 6 (0.00 cells/μL), though B-cell repopulation was observed by month 9 (30.00 cells/μL). The patient remained relapse-free throughout the OFA treatment period, with the ARR decreasing from 0.5 pre-treatment to 0.0 (Figure 2 & Table 1). Serum MOG-IgG decreased from a baseline titer of 1:32 to negative by month 6. Follow-up MRI at 6 months showed no new or enlarging lesions (Figure 1K and L).
In this case series, three pediatric MOGAD patients (1 female, 2 males) aged 8.3–11.6 years treated with add-on OFA for 9–12 months. Indications included breakthrough relapses despite immunotherapy (n=2) or preference for subcutaneous administration over intravenous immunoglobulin (n=1). All patients achieved initial B-cell depletion (CD19+ < 10 cells/μL), which was sustained throughout the treatment period in two (Figure 3). Regarding serum MOG IgG, it increased in two patients and turn negative in one. During OFA treatment, two patients remained relapse free, while one experienced a single relapse. All patients had a decrease in ARR after OFA treatment compared with that before OFA treatment. Peripheral blood CD19 ⁺ B cell counts in four pediatric MOGAD patients following ofatumumab treatment. CD19 ⁺ B cell levels were measured at baseline and during follow-up. Blue arrows indicate the timing of ofatumumab administration, including loading and maintenance doses. The dashed horizontal line denotes the threshold for B cell depletion (CD19⁺ <10 cells/μL)
During OFA treatment, one patient reported adverse events. Case 2 experienced transient fever on the day after the first OFA injection. All adverse events resolved after symptomatic treatment. Serum IgG and IgM levels were measured at baseline and during follow-up (Figure 4). In Case 2, IgG levels showed a mild decline at 6 months but returned to the normal range by 9 months. IgM levels decreased in all patients over time, with one patient showing a value below the lower limit of normal (0.48 g/L). No infections or life-threatening injection-related reactions were reported during the study period. Serum IgG and IgM levels in four pediatric MS patients receiving ofatumumab treatment. (A) Serum IgG levels were measured at baseline and during follow-up. (B) Serum IgM levels were measured at baseline and during follow-up. Blue arrows indicate the timing of ofatumumab administration, including both loading and maintenance doses. Dashed horizontal lines represent the lower limits of normal (IgG: 6.7 g/L; IgM: 0.48 g/L)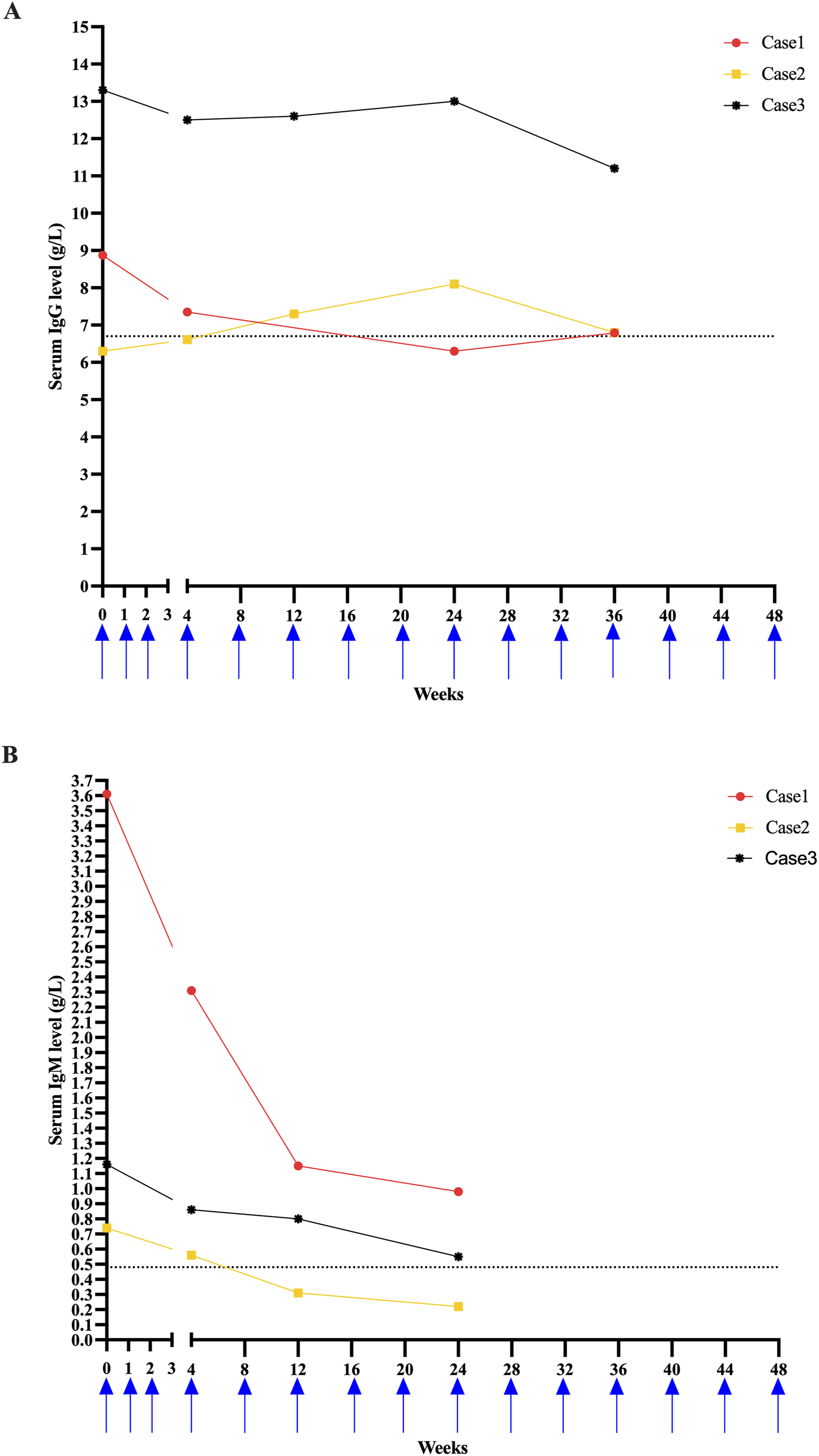
4. Discussion
In this retrospective case series, we assessed the clinical use of OFA in three pediatric MOGAD patients. OFA was initiated either due to breakthrough relapses or for treatment convenience, with treatment durations of 9 to 12 months. B-cell depletion was achieved and maintained in all patients, with two remaining relapse-free and one experiencing a single relapse. Serum MOG-IgG titers increased in two patients and converted to negative in one. All patients showed a reduction in ARR following OFA treatment. OFA was generally well tolerated, with only one patient experiencing mild, self-limited adverse events.
These findings prompt a deeper examination of the immunopathological mechanisms underlying MOGAD and the current treatment strategies, particularly regarding the role and limitations of B cell–targeted therapies. Though MOGAD is also a relapse inflammation demyelinating syndrome as MS and AQP4 antibody positive NMOSD, its pathophysiology is distinct. 15 In MOGAD, MOG-reactive T cells activate B cells to produce MOG antibodies, leading to antibody-mediated demyelination. This is accompanied by Th17-driven immune response and blood-brain barrier disruption. The phagocytosis of damaged myelin by macrophages further amplifies local inflammation. Collectively, cellular and humoral immunity, along with innate immunity, are enhanced by proinflammatory cytokines and chemokines, driving the disease progression, and causing the fusion of lesions, ultimately resulting in extensive confluent demyelination. 15
Despite the clear association between relapse and the accumulation of disability in MOGAD patients, there is currently a lack of randomized controlled trials (RCTs) or evidence-based guidelines specifically addressing the treatment of MOGAD relapses. The 2020 Chinese Expert Consensus on the Diagnosis and Treatment of MOGAD recommended low dose oral corticosteroids, azathioprine (AZA), methotrexate, MMF and RTX for relapse prevention following a second attack. 16 Similarly, the 2023 Clinical Practice Guidelines for MOGAD in Japan recommended oral corticosteroids, AZA, MMF, RTX, IVIG, or tocilizumab for relapsing MOGAD, with RTX, IVIG or tocilizumab being preferred in highly active cases. 17 Data from the six largest retrospective studies on the treatment of relapsing MOGAD, as reported by Marignier et al revealed that, at a median follow-up of 9 to 16 months after the start of treatment, the relapse-free rate was highest in IVIG (69%), followed by the RTX (50%), MMF (47%) and AZA (39%). 18 Another study involving 121 MOGAD patients (24.8% pediatric) showed a 37% overall reduction in relapse rate with RTX; 63% received it as first-line therapy, while 26% switched to RTX after other steroid-sparing agents. 19 B cell depletion treatment using RTX in MOGAD showed efficacy, but its efficacy is variable. Approximately 30% of patients experience a relapse within weeks of the first RTX, infusion and 78.9% of breakthrough relapses occur despite peripheral B-cell depletion.19,20
In our study, two patients with MOGAD were treated with OFA after experiencing uncontrolled relapses on MMF. One patient achieved relapse-free status after 9 months of OFA therapy, despite a rise in serum MOG titer. The other experienced a relapse at 3 months, with a rise in MOG-IgG despite sustained B-cell depletion. A third patient received OFA primarily for the convenience of subcutaneous administration as an alternative to IVIG, and remained relapse-free with seroreversion to MOG-IgG negativity. OFA is advantageous in clinical practice due to its subcutaneous administration, which allows patients to manage treatment at home, reducing hospital visits. Our findings suggest that OFA, like RTX, can be effective in preventing relapses in MOGAD and enhances treatment convenience. However, our study also highlights the possibility of relapse occurring even under B cell depletion. In our cohort, two patients exhibited paradoxical increases in serum MOG-IgG titers from negative to 1:320 despite sustained peripheral B-cell depletion following OFA therapy. A similar phenomenon has been reported in patients treated with rituximab, underscoring a known limitation of anti-CD20 therapies. Both OFA and rituximab selectively deplete CD20-positive B cells but do not target antibody-secreting plasma cells, particularly long-lived plasma cells residing in bone marrow and secondary lymphoid tissues. These cells lack CD20 expression and can therefore continue to produce pathogenic antibodies despite effective B-cell depletion, providing a plausible explanation for the observed titer increase.21-23 Importantly, transient increases in pathogenic autoantibody titers following anti-CD20 therapy have been reported in NMOSD. 24 Nakashima et al demonstrated a significant early rise in anti-AQP4 antibody titers two weeks after rituximab initiation, in parallel with marked elevations in serum BAFF levels. 24 The authors proposed that BAFF rebound following B-cell depletion may promote antibody production from short-lived plasma cells, leading to a transient increase in circulating autoantibodies despite effective depletion of CD20-positive B cells. This BAFF-driven mechanism offers a biologically plausible explanation for the paradoxical increase in MOG-IgG titers observed in our patients under OFA therapy. From a clinical perspective, these findings highlight a potential dissociation between peripheral B-cell depletion and humoral immune activity in MOGAD. The persistence of seropositivity or paradoxical antibody increases under anti-CD20 therapy suggests that serological monitoring alone may not reliably reflect immunological disease control, and that relapse risk cannot be excluded solely based on peripheral B-cell depletion. This is consistent with prior rituximab-treated MOGAD cohorts, in which relapses have been reported despite apparent robust B-cell depletion. In addition, the subcutaneous administration of OFA, delivered into the hypodermis, has been proposed to result in more efficient and selective targeting of B cells within the lymphatic circulation compared with intravenous administration, which enters directly into the systemic circulation. This raises the possibility that OFA may differentially affect B-cell compartments within lymphoid tissues. 8 However, whether this translates into more effective depletion of germinal center B cells or plasma cell precursors remains unclear. At present, direct comparative data on tissue-level B-cell and plasma cell depletion between subcutaneous OFA and intravenous rituximab are lacking and warrant further investigation.
Additionally, Hiya et al reported a case of a 17-year-old adolescent patient with refractory MOGAD who was treated with subcutaneous OFA, resulting in complete prevention of relapses. 9 The patient’s serum MOG-IgG was negative before OFA treatment and was not followed up after OFA initiation. Similarly, Chen et al described two adult MOGAD patients who were treated with OFA during the acute phase, achieving significant improvement in clinical symptoms after three doses of OFA, with a decrease in serum MOG-IgG titers from 1:32 to 1:10 in one patient, while another patient’s MOG-IgG decreased from negative to 1:100 and then to 1:10. 10 These findings are consistent with our observations, where some patients show a decrease in serum MOG-IgG titers after OFA therapy, although paradoxical increases in titers may occur in some patients. This suggests that the relationship between serum MOG-IgG titers and clinical response to OFA treatment is not always straightforward. Notably, the patients in our cohort were substantially younger than those reported by Hiya et al and Chen et al, and age-related differences in immune maturation may partly contribute to variability in susceptibility to ofatumumab and in anti-MOG antibody dynamics, although this requires further investigation.
As evidence for treatment in pediatric MOGAD remains limited, particularly for newer agents such as OFA, ongoing clinical trials will play a vital role in shaping future therapeutic approaches. Several randomized controlled trials are currently underway to explore effective immunotherapeutic strategies for MOGAD. These include the MOGwAI trial (NCT05349006), which investigates the efficacy of azathioprine as a maintenance therapy. Another ongoing trial evaluates satralizumab, an interleukin-6 receptor inhibitor, for its efficacy, safety, pharmacokinetics, and pharmacodynamics in MOGAD patients (NCT05271409). Tocilizumab, another anti-IL-6 receptor monoclonal antibody, is being assessed for its safety and efficacy in a separate study (NCT06452537). Additionally, a phase III trial (NCT05063162) is investigating rozanolixizumab, a subcutaneously administered anti-FcRn monoclonal antibody, as a targeted treatment to prevent relapses in adult patients with MOGAD. These studies represent a critical step toward establishing evidence-based disease-modifying treatments for MOGAD, which currently lacks approved therapies.
The most common adverse effect of OFA treatment was injection-related systemic reactions. The APLIOS phase II trial reported an incidence of 32.0%, predominantly after the first injection. 13 Larger phase III ASCLEPIOS I/II trials recorded a lower rate of 20.1%. 25 In our study, one patient developed transient fever on the day of the first injection, consistent with an injection-related systemic reaction, which resolved with symptomatic treatment. In Case 2, IgG levels declined modestly at 6 months but normalized by 9 months. IgM levels decreased in all patients over time, falling below the lower limit of normal (0.48 g/L) in one individual. These findings are consistent with previous reports indicating that OFA may have a relatively milder impact on IgG-producing B cells in lymphoid tissues compared to intravenously delivered anti-CD20 agents such as RTX.26,27 Despite moderate IgM suppression, no serious infections or adverse events were observed, suggesting that OFA was generally well tolerated in pediatric patients. Nonetheless, the long-term clinical implications of declining immunoglobulin levels, particularly IgM, warrant ongoing surveillance in future studies involving larger pediatric cohorts.
5. Limitations
This study has several important limitations. First, this is a retrospective case series without a control group, which precludes causal inference and limits the ability to attribute reductions in ARR specifically to OFA. Second, OFA was used in combination with other immunotherapies in some patients, including MMF and IVIG, introducing potential confounding effects and further limiting attribution of clinical benefit to OFA alone. Third, the small sample size and single-center design restrict the generalizability of our findings.
6. Conclusion
Ofatumumab demonstrated a acceptable safety profile and potential clinical benefit in this small pediatric MOGAD cohort. Its subcutaneous administration provides practical advantages in terms of administration convenience. However, the small sample size and relapse observed in one patient necessitate caution in interpretation. These preliminary findings underscore the need for validation through larger, prospective, controlled studies.
Footnotes
Ethical Considerations
This study was conducted in accordance with the principles outlined in the Declaration of Helsinki. Approval was obtained from the Ethics Committee of Guangzhou Women and Children’s Medical Center ([20194071]).
Consent to Participate
Written informed consent for participation and publication was obtained from the legal guardians of all included patients.
Consent for Publication
Written and signed consents were obtained from the patient’s parents or legal guardians. The patient’s parents or legal guardians explicitly consent to publish their children’s personal details, clinical details, and associated figures of themselves that could identify them.
Author Contributions
Wenlin Wu, Haixia Zhu, Chi Hou, and Yanping Ran made substantial contributions to the conception and design of the study, data analysis and interpretation, and drafting of the manuscript.
Wenxiao Wu, Wen-Xiong Chen, Xiaolan Mo, Kelu Zheng, and Yulin Tang contributed to data analysis and interpretation, reviewed and revised the manuscript, and approved the final version.
Houliang Deng, Huiling Shen, and Liang Wei contributed to data acquisition, participated in manuscript revision, and approved the final version.
Yuanyuan Gao and Xiaojing Li supervised the study, contributed to the study design and interpretation, and critically revised the manuscript for important intellectual content.
All authors reviewed all manuscript versions, agreed on the journal of submission, approved the final version to be published, and agreed to be accountable for all aspects of the work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Guangzhou Municipal Science and Technology Basic and Applied Research Project (Grant No. 202201011838), the Guangzhou Health Science and Technology Project, Guangzhou Municipal Health Commission, China (Grant No. 20261A011024) and the the Self-funded Scientific Research Project of the Health Commission of Guangxi Zhuang Autonomous Region, China (Contract No. Z-B20251156). The authors had full control of the data and information submitted for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding author.
