Abstract
Background
Myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) is an uncommon neurological disease affecting the central nervous system (CNS). Numerous neurological disorders, including multiple sclerosis (MS), neuromyelitis optica spectrum disorder (NMOSD), acute transverse myelitis (ATM), and MOGAD, have been reported following the COVID-19 infection during the current COVID-19 pandemic. On the other hand, it has been suggested that patients with MOGAD may be at greater risk for infection (particularly in the current pandemic).
Objective
In this systematic review, we gathered separately 1) MOGAD cases following COVID-19 infection as well as 2) clinical course of patients with MOGAD infected with COVID-19 based on case reports/series.
Methods
329 articles were collected from 4 databases. These articles were conducted from inception to March 1st, 2022.
Results
Following the screening, exclusion criteria were followed and eventually, 22 studies were included. In 18 studies, a mean ± SD time interval of 18.6 ± 14.9 days was observed between infection with COVID-19 and the onset of MOGAD symptoms. Symptoms were partially or completely recovered in a mean of 67 days of follow-up.
Among 4 studies on MOGAD patients, the hospitalization rate was 25%, and 15% of patients were hospitalized in the intensive care unit (ICU).
Conclusion
Our systematic review demonstrated that following COVID-19 infection, there is a rare possibility of contracting MOGAD. Moreover, there is no clear consensus on the susceptibility of MOGAD patients to severe COVID-19. However, obtaining deterministic results requires studies with a larger sample size.
Introduction
Throughout history, the world has witnessed various endemics, epidemics, and pandemics. 1 In 2002, severe acute respiratory syndrome (SARS-CoV), a single-stranded RNA (ssRNA) virus belonging to the Coronaviridae family, emerged as an epidemic in Guangdong, China. The Middle East respiratory syndrome coronavirus (MERS-CoV) was first reported in Saudi Arabia and became endemic in the Middle East in the following decade. In December 2019, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) emerged from the Hunan seafood market in Wuhan, China, and in a short time, spread throughout the world, so that by late March 8th, 2021, 450 million people were reported to have been infected with COVID-19.2,3
The primary symptoms of COVID-19 infection have been reported to have respiratory and cardiac manifestations. Furthermore, headache, dizziness, acute demyelinating encephalomyelitis (ADEM), delirium, Guillain-Barre syndrome, acute transverse myelitis (ATM), hypogeusia, hyposmia, ischemic stroke, multiple sclerosis (MS), neuromyelitis optica spectrum disorder (NMOSD), and myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) have been identified as neurological manifestations of the virus.4-7
However, 1 question that needs to be asked is: “How do the neurotropic features of the SARS-CoV-2 virus trigger autoimmunity?” Several studies revealed that SARS-CoV-2 is able to cross the blood-brain barrier (BBB) and cause inflammation in the central nervous system (CNS), demyelination, and damage to axons.8-10 One of the autoimmune conditions triggered by COVID-19 infection was MOGAD. 11
The pathophysiology of autoimmune diseases could be ongoing for years, and infections, including COVID-19, may trigger these diseases.12,13 Literally defined, MOGAD is one of the autoimmune diseases of the CNS in which IgG antibodies target the myelin sheath. Although the phenotype of these patients is similar to those with MS and NMOSD, the main target of the MOG-IgG is the myelin oligodendrocyte glycoprotein. 14 It is possible to detect MOG-IgG synthesis within the CNS of patients with MOGAD from the onset of the disease, and this is linked to the severity of the disorder. 15 In MOGAD, complement deposition occurs in white matter, and reactive gliosis in the white and grey matter is associated with partial axonal preservation. Additionally, MOGAD differs from MS and NMOSD in terms of disability levels, CD4 infiltration, and MOG-IgG expression. 16
Appraising available data demonstrated the possibility of high hospitalization rates in patients with neurologic disorders contracting COVID-19.17-19 Furthermore, the possibility of hospitalization of MOGAD patients with COVID-19 has increased. That is why it is essential to review all cases in this regard.
Our study gives an account of a systematic review of the status of MOGAD cases following COVID-19 infection. Our second aim is to provide an overview of the status of COVID-19 in patients with MOGAD.
Methods
Literature Search
We searched PubMed (Medline), Scopus, Web of Science, and Embase databases from inception to March 1st, 2022. Moreover, gray literature, including the references of the included articles and review studies, and conference abstracts, was searched. Our search strategy in databases included input such as: (“Myelin-Oligodendrocyte Glycoprotein” OR (Glycoprotein AND Myelin-Oligodendrocyte) OR “Myelin Oligodendrocyte Glycoprotein” OR “MOG Glycoprotein” OR “glycoprotein MOG” OR “MOG protein” OR “myelin oligodendroglial glycoprotein” OR “myelin-oligodendrocyte glycoprotein” OR “protein MOG”) AND (“COVID 19” OR “COVID-19 Virus Disease” OR “COVID 19 Virus Disease*” OR “COVID-19 Virus Disease*” OR (Disease AND “COVID-19 Virus”) OR (“Virus Disease” AND COVID-19) OR “COVID-19 Virus Infection*” OR “COVID 19 Virus Infection” OR (Infection AND “COVID-19 Virus”) OR (“Virus Infection” AND COVID-19) OR “2019-nCoV Infection” OR “2019 nCoV Infection*” OR (Infection AND 2019-nCoV) OR “Coronavirus Disease-19” OR “Coronavirus Disease 19” OR “2019 Novel Coronavirus Disease” OR “2019 Novel Coronavirus Infection” OR “2019-nCoV Disease” OR “2019 nCoV Disease” OR “2019-nCoV Diseases” OR (Disease AND 2019-nCoV) OR COVID19 OR “Coronavirus Disease 2019” OR (“Disease 2019” AND Coronavirus) OR “SARS Coronavirus 2 Infection” OR “SARS-CoV-2 Infection” OR (Infection AND SARS-CoV-2) OR “SARS CoV 2 Infection*” OR “COVID-19 Pandemic*” OR “COVID 19 Pandemic” OR (Pandemic AND COVID-19)). This syntax was customized and was searched based on each database.
Inclusion and Exclusion Criteria
All case reports and case series studies that had addressed MOGAD patients following COVID-19 infection were included. Besides, case reports and the case series on the presence of COVID-19 infection in MOGAD patients were encompassed in our review. Of note, all included articles were in English. Exclusion criteria consisted of review articles, irrelevant studies, animal studies, duplicated studies, other coronaviridae viruses, and study populations of MS and NMOSD-related studies.
Study Selection and Data Extraction
To select the studies, two researchers (HG and MSD) screened all the articles separately and in case of any disagreement, a third researcher (OM) resolved it. In the next step, two researchers (EMG and MSD) extracted the data from the articles. They reviewed their information in two separate tables (the first for MOGAD following COVID-19 and the second for MOGAD patients with COVID-19) reporting the following information: author, year and country, patients’ demographic data, disease duration, comorbidity, disease-modifying therapies (DMT), clinical and laboratory, findings associated with COVID-19 and MOGAD, the patients, hospitalization, treatments, follow-up, and outcomes.
Quality Assessment
To assess the quality of the articles, the Joanna Briggs Institute’s (JBI) critical appraisal tool for case reports/case series 20 was used separately by (OM) and (MSD), and any disagreement was resolved by a senior researcher (VS). Articles were evaluated according to the questions’ answers (Yes or No), and the ranking was done by evaluating “Yes” answers from 0 to 8 (0 to 10 for case series). Articles lower than the 4 ranking (5 ranking for case series) were considered low quality, and those higher than 4 ranking (5 ranking for case series) were presumed high-quality articles.
Results
Search Results
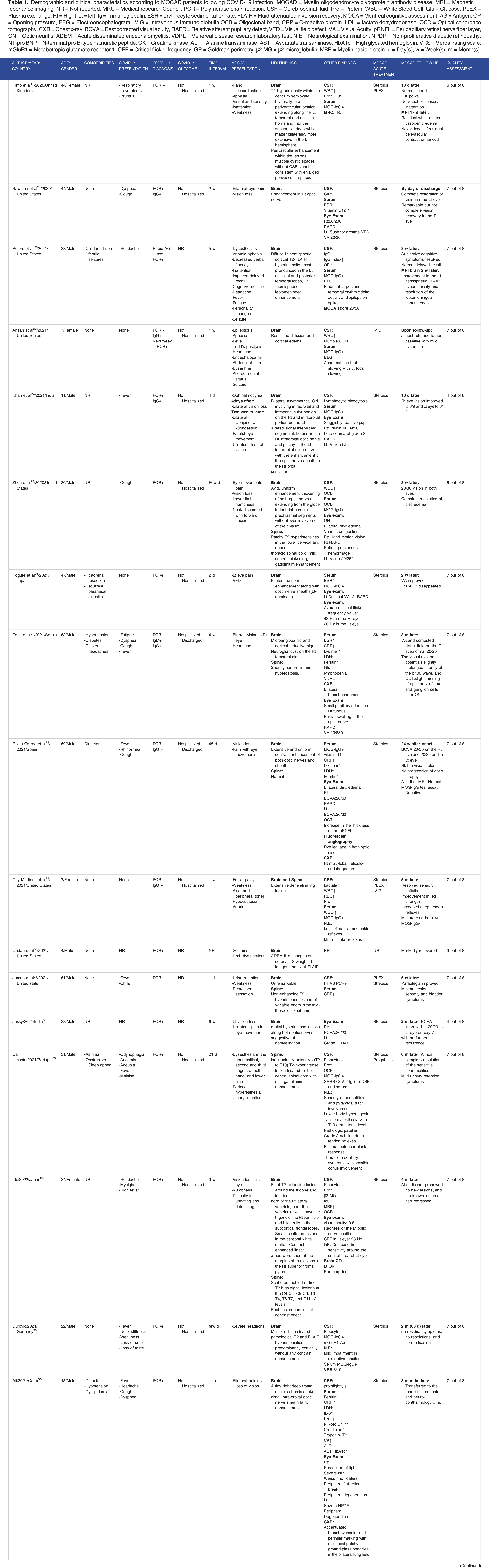
According to the PRISMA flow chart, all selected and eliminated articles are listed (Figure 1). Initially, 329 articles were obtained from various databases. Following elimination of duplicates, 234 articles remained. Afterward, by screening all of the articles, the full texts of 87 articles were collected, and finally, 22 articles were included. The data was extracted from the articles, and the results were laid out in Table 1 and Table 2. Table 1 provides information about patients who developed MOGAD following COVID-19, and Table 2 provides information on COVID-19 outcomes in MOGAD patients. In addition, quality assessment was carried out and is listed in the tables. PRISMA Flow Diagram: includes details our search and selection process applied during the systematic review. Demographic and clinical characteristics according to MOGAD patients following COVID-19 infection. MOGAD = Myelin oligodendrocyte glycoprotein antibody disease, MRI = Magnetic resonance imaging, NR = Not reported, MRC = Medical research Council, PCR = Polymerase chain reaction, CSF = Cerebrospinal fluid, Pro = Protein, WBC = White Blood Cell, Glu = Glucose, PLEX = Plasma exchange, Rt = Right, Lt = left, Ig = immunoglobulin, ESR = erythrocyte sedimentation rate, FLAIR = Fluid-attenuated inversion recovery, MOCA = Montreal cognitive assessment, AG = Antigen, OP = Opening pressure, EEG = Electroencephalogram, IVIG = Intravenous Immune globulin,OCB = Oligoclonal band, CRP = C-reactive protein, LDH = lactate dehydrogenase, OCD = Optical coherence tomography, CXR = Chest x-ray, BCVA = Best corrected visual acuity, RAPD = Relative afferent pupillary defect, VFD = Visual field defect, VA = Visual Acuity, pRNFL = Peripapillary retinal nerve fiber layer, ON = Optic neuritis, ADEM = Acute disseminated encephalomyelitis, VDRL = Venereal disease research laboratory test, N.E = Neurological examination, NPDR = Non-proliferative diabetic retinopathy, NT-pro BNP = N-terminal pro B-type natriuretic peptide, CK = Creatine kinase, ALT = Alanine transaminase, AST = Aspartate transaminase, HbA1c = High glycated hemoglobin, VRS = Verbal rating scale, mGluR1 = Metabotropic glutamate receptor 1, CFF = Critical flicker frequency, GP = Goldman perimetry, β2-MG = β2-microglobulin, MBP = Myelin basic protein, d = Day(s), w = Week(s), m = Month(s). Demographic and clinical characteristics according to MOGAD patients with COVID-19 infection, DMT = Disease Modifying Therapies, PCR = Polymerase chain reaction, NR = Not reported, IVIG = Intravenous Immune globulin.

New MOGAD Cases Following COVID-19 Infection
18 new cases of MOGAD following COVID-19 infection were included. The female-to-male ratio was 1:2.6 and the mean ± SD age of the participants was 32 ± 20.4 years. The most common comorbidities of the patients were hypertension and diabetes, and a positive PCR result was obtained in 77.7% of cases. Additionally, the most common symptoms associated with COVID-19 were fever and cough, and only 2 patients were hospitalized.
The mean ± SD time interval between the time of contracting COVID-19 and the onset of MOGAD symptoms was 18.6 ± 14.9 days. As for MOGAD, the majority of lesions were located in the optic nerve and the cerebral cortex. Lesions were also found in spinal MRIs, ranging in size from small to extensive. Most MOGAD cases presented visual problems at the onset. 61.1% of patients were positive for serum MOG-Ab, and their acute treatment of patients was as follows: 88% (15 out of 17) of them received steroids, 18% (3 out of 17) received intravenous immune globulin (IVIG), and 18% (3 out of 17) carried out plasma exchange (PLEX). A partial or complete recovery of symptoms was achieved after a mean of 67 days of follow-up.
COVID-19 Infection in MOGAD Patients
Overall, 27 patients were enrolled. The mean ± SD age of patients was 42 ± 17.1 years, with a female-to-male ratio of 2:1. The mean ± SD of COVID-19’s duration was 6.4 ± 5.3 days in patients with a history of MOGAD. Moreover, the most common comorbidity among them was hypertension, and about 44.4% were undergoing rituximab therapy. Based on available data, 5 out of 6 patients showed positive PCR for COVID-19, 25% were hospitalized, and 15% were admitted to ICU. The most common symptoms of COVID-19 in the MOGAD population were anosmia, fever, and cough.
Discussion
Post-viral Infection Neurological Complications
Since the beginning of the COVID-19 pandemic in late 2019, many studies worldwide encompassed the effects of this infection on all aspects of human health.42,43 Regarding the reports of MOGAD following COVID-19 infection, it was essential to know how the virus can trigger autoimmunity and how it acts on patients with autoimmune disorders. Several studies indicated that other viruses can also trigger various neurological complications. According to Mailles et al. study, bornavirus, Lassa fever virus(LFV), Ebola, zika, chikungunya, and herpes simplex virus (HSV) cause encephalitis, 44 a dengue virus causes Guillain-Barré syndrome, 45 NMOSD and ATM46,47 The SARS-CoV-2 virus is no exception to the norm and is expected to cause neurological complications. 48
The Effects of COVID-19 on the CNS
With regards to the infection’s transmission to the CNS, 2 hypotheses are possible. In the hematogenous route, the invasive agent crossed the BBB through the bloodstream. The COVID-19 infection invades the CNS following binding to angiotensin-converting enzyme (ACE2) receptors present in the outer membrane of lung cells and several other tissues, or is transported with other immune cells to enter the CNS. Afterward, it invades endothelial and epithelial cells, or leukocytes. In the retrograde route, the virus invades neurons with axonal transport machinery. Consequently, these 2 routes causes inflammation in the CNS.10,49 Therefore, not only do glial cells secrete IL-6, IL-12, IL-15, and TNFα, but also the occurrence of a cytokine storm is possible. 50
By the same token, COVID-19 associated with acute respiratory distress syndrome (ARDS) exhibits pathological characteristics such as pulmonary edema and hyaline membrane formation. These factors may cause gas exchange disorders and hypoxia in the central nervous system. Anaerobic metabolites in the mitochondria and acid metabolites in the brain are excessively accumulated in hypoxia, resulting in the swelling of the cells of the brain and obstruction of blood flow to the brain. 51
A number of data originating from virus and human antigen sets demonstrating molecular mimicry have been used to support the mimicry concept. In the presence of virus proteins, activated B cells proliferate, differentiate into plasma cells, which are then capable of producing antibodies. They are produced to combat the infection, but mistakenly attack similar proteins that share extensive amino acid homology with human antigens. 52
Moreover, infection with viruses is associated with an inflammatory response, activating myelin-specific T cells, and this can result in early or delayed degeneration of myelin. The literature has historically reported that the genetically similar SARS-CoV-1 53 and the MERS-CoV, 53 are associated with central demyelination. In several experimental studies, the murine coronavirus infection of susceptible mice was found to result in demyelinating lesions similar to MS, as well as the presence of the virus' RNA sequences and its antigen. 54 Furthermore, the CNS may be compromised through ischemia–hypoxia or latent immune-mediated mechanisms in response to severe respiratory infections. In addition to its neurotropic and neuroinvasive properties, SARS-CoV-2 can also cause neurological damage directly, by binding to ACE2 receptors that are ubiquitous, including the central nervous system, or by passing through the Virchow Robin spaces through blood circulation. It is also believed that delayed brain damage is caused by an undesired immune response following acute infection, which leads to demyelination of the brain. 54
According to similar studies, infection causes a CNS-inflammatory disease mediated by CD4+ T cells that targets MOG protein, which then induces the recruitment and activation of MOG-specific B cells in peripheral lymph nodes draining into the CNS, which in turn produces MOG antibodies. It is also possible for a direct CNS infection to cause a peripheral circulation of CNS antigens, which in turn results in an immune response against MOG in demyelinating diseases. By using molecular mimicry, encephalitogenic T cells could be activated in the periphery after infection, resulting in inflammation in the central nervous system and an immune response against MOG. 55
Moreover, COVID-19 may induce autoimmunity in various ways, including through molecular mimicry, viral persistence, epitope spreading, and neutrophil extracellular trap (NET) formation. 56 A NET is a structure that resembles a net and consists of DNA-histone complexes and pro-inflammatory proteins released by neutrophils when they become activated. 57 As a result of these events, demyelination and degeneration of neurons occur, and neurodegenerative diseases, including MS, NMOSD, ATM, etc. are caused. 47
MOGAD Mechanism and Patients’ Characteristics
MOGAD is a rare neuro-inflammatory disorder,
58
and there are several hypotheses that explain why MOGAD is triggered by COVID-19 infection (Figure 2). This virus is able to stimulate the demyelination of neurons and lead to MOG-Ab production by plasma cells.59,60 Also, endothelial cells in multiple organs, including the brain, express ACE2, which are the main targets of SARS-COV-2. Symptoms of MOG-related demyelination may be caused by lymphocytic endothelitis associated with COVID-19 infection, which may facilitate the entry of MOG-IgG into the brain.
37
Possible mechanism of COVID-19 in the CNS and causing MOGAD. Created with: Biorender.com.
Another hypothesis is that the demyelination of neurons produces antigens that can cross the BBB and enter the bloodstream. This is why MOG antigens are detected mainly by immune cells and lead to the production of MOG-Abs. 61 The stimulation of B and T cells by viral infection is the final hypothesis proposed by Reindl et al. wherein the subsequent encounter of the virus leads to the development of IgG associated with MOG-Ab, and the virus then binds to cells in the CNS. 55 Following this binding, antibody-dependent cell-mediated cytotoxicity (ADCC) occurs, 62 Finally, macrophages and T-cells enter the CNS, 63 activate the complement cascade, 64 and cytokine storm is developed. 65 Furthermore, the MOG-Ab causes white and gray matter to be affected, 66 while also triggering gliosis, 67 demyelinating lesions around small to medium-sized venules, 68 as well as acute plaques in the cortical, intracortical, and subpial areas.68-71 Narayan et al. reported that this disorder causes lesions in the optic nerve, 72 brain, 73 and spinal cord. 74 Characteristics of MOGAD patients include optic neuritis, 75 ADEM, 76 myelopathy, 77 and brainstem syndrome 77 associated with MOG-Ab 78 and preserving aquaporin 4 (AQP4)-IgG. 71
MOGAD Diagnosis Following COVID-19 Infection
Studies by Netravathi et al. described the symptoms of MOGAD patients as seizures, encephalitis, leukodystrophy, meningitis, cranial neuropathy, myeloradiculopathy, 77 and fever. 79 In our review, most symptoms of MOGAD patients were ON, vision loss, weakness, headache, and seizure following a period of COVID-19 infection. Moreover, the symptoms of MOGAD appeared after a mean ± SD of 16.6 ± 14.9 days after the onset of COVID-19, while in other demyelinating diseases, onset symptoms occurred following about 10 days.80-84 According to a systematic review of CNS demyelination published by Ismail et al. in 2021, 54 3 cases of MOGAD have been reported. Although our research supports the findings of the previous systematic review, we report more cases than the previous studies.
Routine treatments for acute MOG antibody disease are intravenous methylprednisolone, 85 PLEX, 86 and IVIG. 87 Administration of Steroids for the chronic phase and IVIG for the acute phase were common treatments for patients with MOGAD. Adequate follow-ups were performed after discharge, and all of them regained relative improvement.
Interestingly, according to a Canadian study by Kaseka et al., the number of admissions due to MOGAD decreased in COVID-19 lockdowns. 88 The study concluded that by controlling viral infections, the incidence of MOGAD can be reduced to some extent.
MOGAD Patients With COVID-19 Infection
A number of studies have found that the risk and susceptibility of COVID-19 infection among populations with neurological disorders were different.89-91 There was a 25% hospitalization rate and 15% ICU admission rate for MOGAD patients with COVID-19, respectively, whereas 14% of MOGAD patients were hospitalized after COVID-19. In this study, no COVID-19 mortality for MOGAD patients was reported, whereas the mortality rate of COVID-19 in the normal population was 1.5-3.5%. 92 Reindl et al. also explained that MOG antibodies exacerbated CNS inflammation caused by a viral infection that allowed the infiltration more immune cells into the brain and spinal cord by MOG-IgG. 55
13 MOGAD patients with COVID-19 were reported in an original study by Sen et al. study, in which 2 patients were reported with symptoms of severe COVID-19 infection and one patient was hospitalized. The number of relapses is nearly equal before and after COVID-19. 93 These results are consistent with those reported in other case reports and case series which indicate that COVID-19 is mild in these patients and few hospitalizations are associated with it.
Limitations
In this systematic review, at first, we comprehensively assessed all case reports on new cases of MOGAD following COVID-19 infection. Secondly, COVID-19 infection outcomes in MOGAD patients were collected. Although our knowledge of MOGAD patients' pre-and post-COVID-19 conditions is primarily limited, this is the first systematic review that has been carried out in this regard. Furthermore, the limited number of case reports and case series, and the heterogeneity of the collected data contributed to the incapability of conducting a meta-analysis.
Conclusion
In conclusion, there is no clear consensus that there is an association between COVID-19 and MOGAD. Moreover, a larger sample size is required to clarify this issue. The prospect of obtaining more studies with larger sample sizes about the status of MOGAD patients following COVID-19 and also the status of MOGAD patients during the development of COVID-19 infection serves as a continuous incentive for future research.
Footnotes
Author Contributions
All the authors listed in the manuscript have participated actively in preparing the final version of this case report.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
