Abstract
Keywords
Introduction
Small cell lung carcinoma (SCLC) is an extremely aggressive malignancy, with approximately 70% of patients presenting with extensive-stage disease at the time of diagnosis.1,2 The median overall survival (OS) for patients with extensive-stage small cell lung cancer (ES-SCLC) receiving standard etoposide and platinum-based chemotherapy remains limited, at only 10.3 months. 3 Therefore, there is an urgent clinical need to develop more effective treatment strategies for this challenging disease.
Immunotherapy has significantly reshaped the treatment landscape of ES-SCLC over the past 3 decades, helping to overcome longstanding therapeutic challenges. Immune checkpoint inhibitors (ICIs) exert their effects by binding to specific targets, thereby suppressing inhibitory immune pathways, activating effector T cells, and enhancing anti-tumor immunity. 4 In addition, targeted therapies are showing promising progress. Current studies are investigating the synergistic benefits of combining chemotherapy with immunotherapy, antiangiogenic agents, or novel chemotherapeutic drugs.5 -8 In this context, we conducted a comprehensive review of existing randomized clinical trials and included 12 studies to assess the efficacy and safety of various chemotherapy-based combination regimens for ES-SCLC.
Methods
We conducted a comprehensive systematic review and network meta-analysis (NMA) following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Supplemental Table S1). 9
Search strategy
A systematic search was performed across PubMed, Embase, Cochrane Library, and Web of Science databases for randomized controlled trials (RCTs) published in English up to January 2025. The detailed search strategy is exhibited in Supplemental Table S2.
Selection criteria
Inclusion criteria are the following: (1) patients diagnosed with ES-SCLC; (2) Systemic therapy regimens based on chemotherapy including but not limited to immunotherapy, or targeted therapy; (3) regimens administered as first-line therapy for ES-SCLC; (4) phase II-III RCTs; and (5) use of prophylactic cranial irradiation (PCI) allowed at the discretion of the investigator. Exclusion criteria are the following: (1) retrospective studies, observational studies, or single-arm trials; (2) studies lacking sufficient data on treatment regimens or clinical outcomes.
Data extraction
Relevant data from each study were systematically extracted into a standardized spreadsheet. Extracted information included the study name, authors, treatment regimens, number of patients enrolled, number of patients with brain or liver metastases, hazard ratios (HRs) with corresponding 95% confidence intervals (CIs) for OS and progression-free survival (PFS) of in the overall population, and risk of bias. In addition, odds ratios (ORs) for objective response rate (ORR) and grade ⩾ 3 adverse events (AEs), as well as HRs for OS and PFS in subgroup analyses, were also collected.
Quality assessment
The risk of bias in the included RCTs was assessed using the Cochrane Collaboration’s tool, evaluating multiple methodological domains. 10 Each domain was rated as having a low, unclear, or high risk of bias. Any discrepancies between the 2 primary reviewers were resolved through consultation with a third reviewer.
Statistical analysis
Network plots were constructed using Stata version 17.0. A Bayesian NMA was performed using the Markov Chain Monte Carlo (MCMC) simulation method implemented through the gemtc package (version 1.0-1) and JAGS (version 4.3.1) within R software (version 4.2.1). A total of 100 000 iterations were run, with the initial 20 000 iterations designated as the burn-in phase. Given the limited availability of data for assessing heterogeneity across studies and the presence of only 1 RCT for most pairwise comparisons, a fixed-effect model was employed. Heterogeneity analysis was performed using the I2 test. 11 For I2 values <25%, 25% to 50%, and >50%, respectively, heterogeneity was considered as low, moderate, or high. Convergence of the MCMC chains was evaluated using trace plots, density plots, and diagnostic convergence statistics (Supplemental Figures S5 and S6). To compare treatment regimens, a frequentist approach was also applied. Treatment rankings were calculated using the surface under the cumulative ranking curve (SUCRA).
Results
Systematic review and study characteristics
A total of 6446 records were initially identified through literature searches. After screening and eligibility assessment, 12 RCTs comprising 5840 patients with SCLC met the inclusion criteria and were included in the analysis, evaluating 14 different treatment regimens (Figure 1). Among these, 4 studies assessed the efficacy of programmed death-ligand 1 (PD-L1) inhibitors, adebrelimab, atezolizumab, durvalumab, or socazolimab in combination with chemotherapy compared with chemotherapy alone. Five studies investigated the combination of programmed death-1 (PD-1) inhibitors nivolumab, pembrolizumab, serplulimab, tislelizumab, or toripalimab with chemotherapy. Additional studies evaluated other combinations, including cytotoxic T lymphocyte–associated antigen-4 (CTLA-4) monoclonal antibody (ipilimumab), PARP inhibitor (veliparib), dual immune checkpoint blockade with PD-L1 and CTLA-4 inhibitors (durvalumab and tremelimumab), and a PD-L1 inhibitor combined with an antiangiogenic agent (benmelstobart and anlotinib), all compared with chemotherapy. The detailed characteristics of included studies have been depicted in Table 1. The network plots of treatment comparisons are shown in Figure 2 and the risk of bias assessment is illustrated in Supplemental Figure S1.

Literature search and selection.
Characteristics of studies in the Network Meta-Analysis.

Network of the comparisons.
OS and PFS
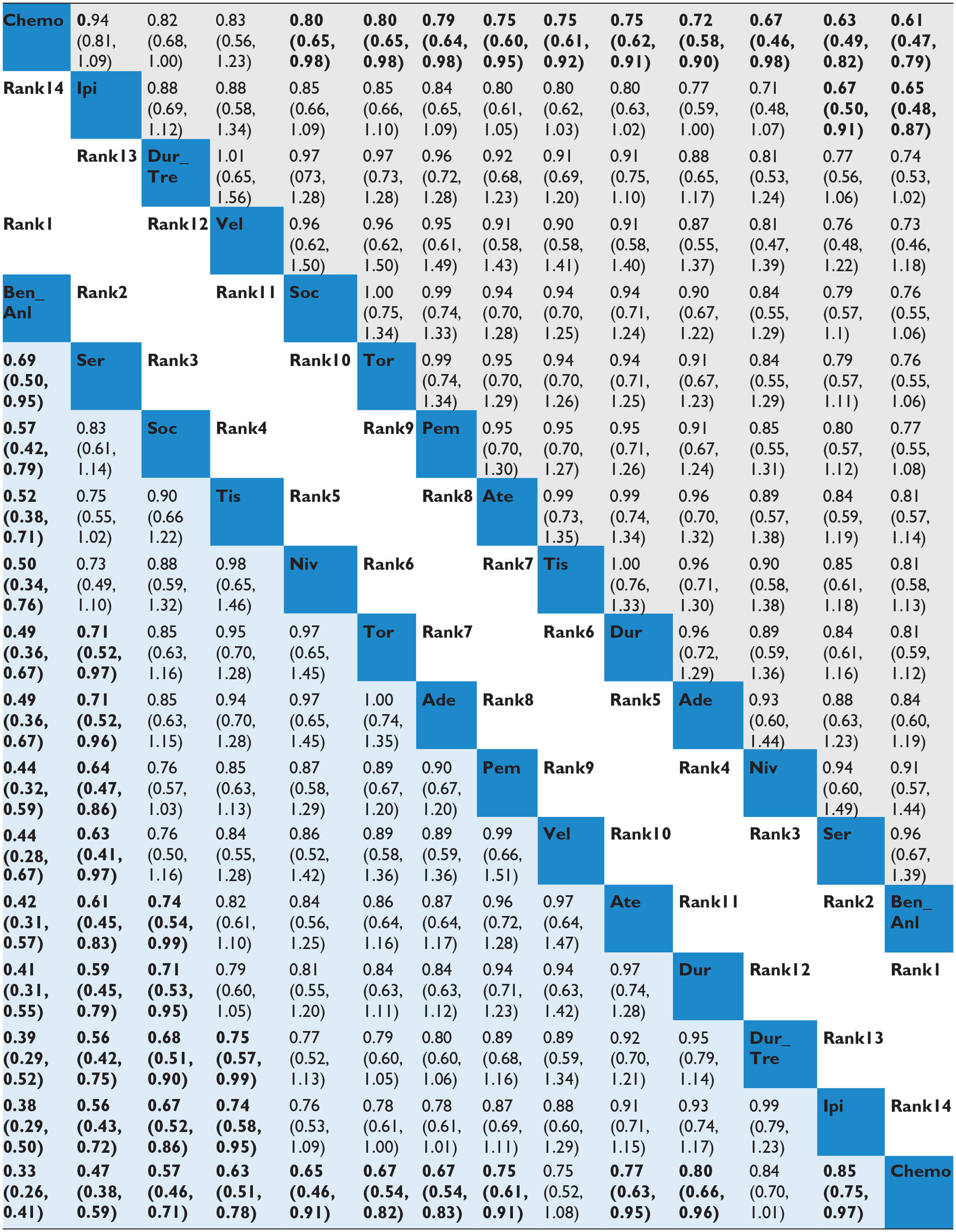
Our NMAs evaluated 14 treatment regimens for OS and PFS. Regarding OS (Figure 3A and Table 2A), several combination therapies demonstrated significantly improved survival outcomes compared with chemotherapy alone. These included adebrelimab + chemo (0.72; 0.58-0.90), atezolizumab + chemo (0.75; 0.6-0.95), benmelstobart + anlotinib + chemo (0.61; 0.47-0.79), durvalumab + chemo (0.75; 0.62-0.91), nivolumab + chemo (0.67; 0.46-0.98), pembrolizumab + chemo (0.79; 0.64-0.98), serplulimab + chemo (0.63; 0.49-0.82), socazolimab + chemo (0.8; 0.65-0.98), tislelizumab + chemo (0.75; 0.61-0.92) and toripalimab + chemo (0.80; 0.65-0.98). All these combinations showed statistically significant improvements in OS compared with standard chemotherapy.

Forest plot of meta-analysis. (A) HRs for OS. (B) HRs for PFS. (C) ORs for ORR. (D) ORs for grade ⩾ 3 AEs.
Network meta-analyses for HRs of OS. Significant results are shown in bold.

In terms of PFS (Figure 3B and Table 2B), several combination therapies demonstrated statistically significant improvements compared with chemotherapy alone. These included benmelstobart + anlotinib + chemo (0.33; 0.26-0.41), serplulimab + chemo (0.47; 0.38-0.59), socazolimab + chemo (0.57; 0.46-0.71), tislelizumab + chemo (0.63; 0.51-0.78), nivolumab + chemo (0.65; 0.46-0.91), toripalimab + chemo (0.67; 0.54-0.82), adebrelimab + chemo (0.67; 0.54-0.83), pembrolizumab + chemo (0.75; 0.61-0.91), atezolizumab + chemo (0.77; 0.63-0.95), durvalumab + chemo (0.80; 0.66-0.96) and ipilimumab + chemo (0.85; 0.75-0.97) . Among these, the combination of benmelstobart, anlotinib, and chemotherapy provided the most pronounced benefit in PFS and outperformed all other regimens. In addition, serplulimab + chemo and socazolimab + chemo also demonstrated superior efficacy compared with most other treatment combinations.
ORR and AEs
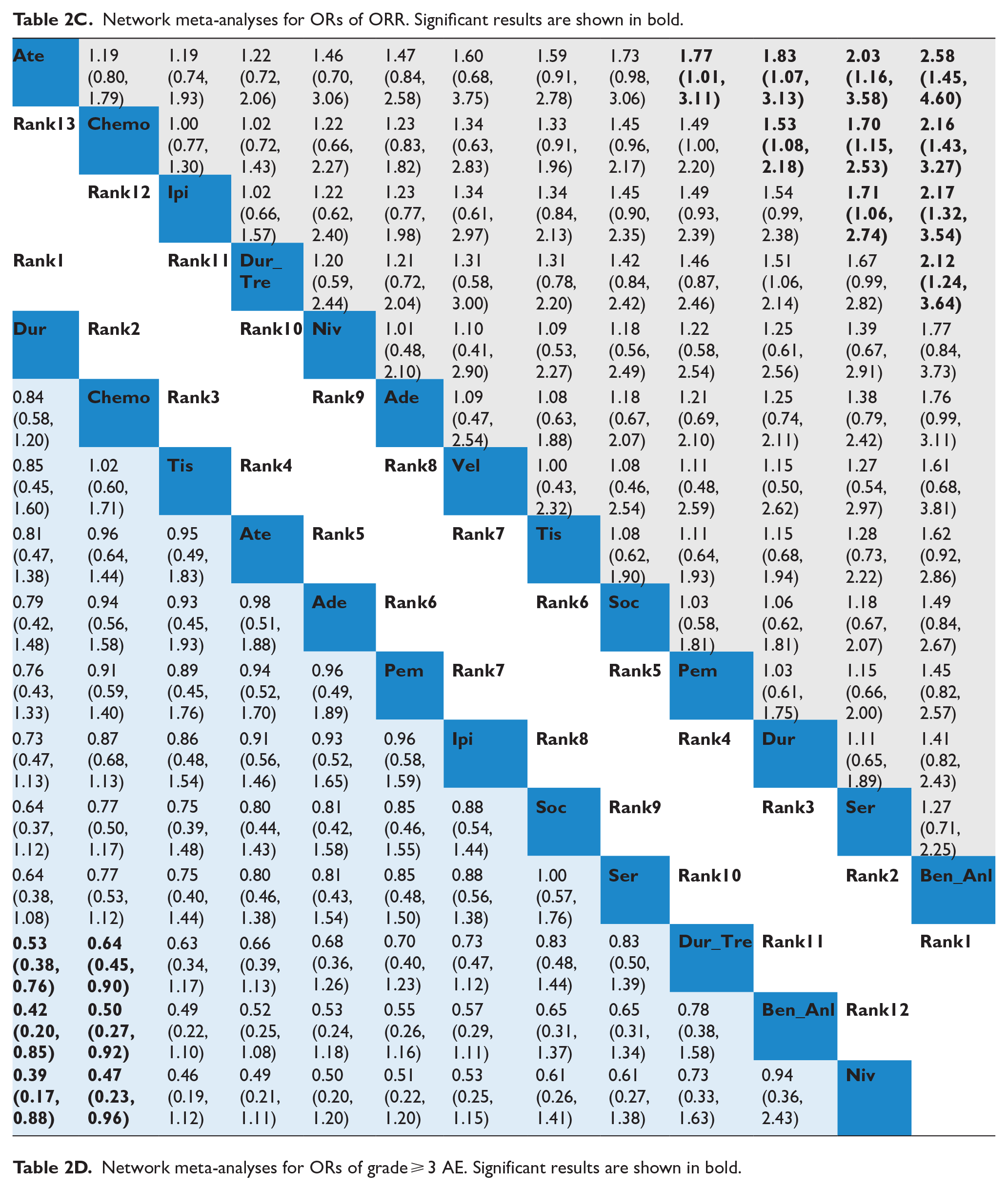
Regarding ORR (Figure 3C and Table 2C), the combination of benmelstobart + anlotinib + chemo demonstrated the most significant improvement (OR = 2.16; 1.43-3.27), followed by serplulimab + chemo (OR = 1.70; 1.15-2.53) and durvalumab + chemo (OR = 1.53; 1.08-2.18), all showing superior responses compared with chemotherapy alone.
Network meta-analyses for ORs of ORR. Significant results are shown in bold.
In terms of safety (Figure 3D and Table 2D), combination therapies were associated with a higher incidence of AEs. Specifically, nivolumab + chemo (OR = 2.14; 1.04-4.42), benmelstobart + anlotinib + chemo (OR = 2.01; 1.09-3.73) and durvalumab + tremelimumab + chemo (OR = 1.57; 1.11-2.22) showed a significantly increased risk of grade ⩾ 3AEs compared with chemotherapy alone.
Subgroup-level NMA
We performed subgroup analyses of OS across various clinical and demographic characteristics. By sex, in male patients (Supplemental Figure S2A), serplulimab + chemo (0.63; 0.48-0.84), benmelstobart + anlotinib + chemo (0.64; 0.48-0.85) and adebrelimab + chemo (0.72; 0.57-0.92) showed significantly improved OS compared with chemotherapy alone. In female patients (Supplemental Figure S2B), benmelstobart + anlotinib + chemo (0.30; 0.12-0.75), serplulimab + chemo (0.56; 0.3-1.06) and durvalumab + chemo (0.65; 0.45-0.93) demonstrated survival benefits. By age, among patients aged ⩾65 years (Supplemental Figure S2C), benmelstobart + anlotinib + chemo (0.53; 0.35-0.81), atezolizumab + chemo (0.59; 0.42-0.82) and serplulimab + chemo (0.60; 0.40-0.89) provided improved outcomes. For those under 65 (Supplemental Figure S2D), serplulimab + chemo (0.62; 0.45-0.86), benmelstobart + anlotinib + chemo (0.65; 0.46-0.92) and socazolimab + chemo (0.69; 0.52-0.91) were associated with better efficacy. By smoking status, in smokers,(Supplemental Figure S2E), durvalumab + chemo (0.75; 0.62-0.91), adebrelimab + chemo (0.75; 0.59-0.95), tislelizumab + chemo (0.76; 0.60-0.97) outperformed chemotherapy. In nonsmokers (Supplemental Figure S2F), benmelstobart + anlotinib + chemo (0.38; 0.20-0.74) and adebrelimab + chemo (0.59; 0.37-0.95) showed significant benefits. In the patients with baseline Eastern Cooperative Oncology Group Performance Status (ECOG PS) of 0 (Supplemental Figure S2G), serplulimab + chemo (0.44; 0.23-0.84) and socazolimab + chemo (0.50; 0.30-820) demonstrated better efficacy. In addition, in the patients that baseline ECOG PS of 1 (Supplemental Figure S2H), benmelstobart + anlotinib + chemo (0.63; 0.47-0.84), serplulimab + chemo (0.65; 0.49-0.86) and adebrelimab + chemo (0.69; 0.55-0.87) presented increased OS. By baseline lactate dehydrogenase (LDH) level, for patients with LDH within the upper limit of normal (ULN) (Supplemental Figure S2I), benmelstobart + anlotinib + chemo (0.55; 0.37-0.82), adebrelimab + chemo (0.59; 0.42-0.82) and tislelizumab + chemo (0.72; 0.53-0.98) were associated with better survival. For those with LDH levels above ULN (Supplemental Figure S2J), benmelstobart + anlotinib + chemo (0.61; 0.43-0.87) remained superior to chemotherapy.
Among patients without brain metastases (Supplemental Figure S3A), all treatment regimens, except for ipilimumab + chemo, showed improved OS compared with chemotherapy alone. The most pronounced benefit was observed with serplulimab + chemo (0.60; 0.48-0.75), followed closely by benmelstobart + anlotinib + chemo (0.60; 0.45-0.79), adebrelimab + chemo (0.68; 0.55-0.85), atezolizumab + chemo (0.74; 0.58-0.94), durvalumab + chemo (0.76; 0.62-0.92), pembrolizumab + chemo (0.75; 0.6-0.94), tislelizumab + chemo (0.75; 0.61-0.93), socazolimab + chemo (0.79; 0.64-0.97) and durvalumab + tremelimumab + chemo (0.80; 0.66-0.98). In patients without liver metastases (Supplemental Figure S3C), several regimens were associated with significantly improved OS compared with chemotherapy, including benmelstobart + anlotinib + chemo (0.51; 0.36-0.72), adebrelimab + chemo (0.61; 0.46-0.81), serplulimab + chemo (0.62; 0.48-0.80), durvalumab + chemo (0.68; 0.53-0.88), durvalumab + tremelimumab + chemo (0.75; 0.58-0.96), tislelizumab + chemo (0.76; 0.59-0.97) and socazolimab + chemo (0.76; 0.60-0.97). For patients with liver metastases (Supplemental Figure S3D), only 2 regimens demonstrated a statistically significant survival advantage over chemotherapy: serplulimab + chemo (0.58; 0.40-0.84) and tislelizumab + chemo (0.65; 0.44-0.95).
The NMA for PFS in subgroup populations included 8 treatment regimens. Among patients without brain metastases (Supplemental Figure S4A), the following combinations demonstrated significantly improved PFS compared with chemotherapy alone: benmelstobart + anlotinib + chemo (0.33; 0.26-0.42), socazolimab + chemo (0.55; 0.44-0.69), tislelizumab + chemo (0.63; 0.51-0.77), adebrelimab + chemo (0.66; 0.53-0.81), pembrolizumab + chemo (0.68; 0.55-0.85), atezolizumab + chemo (0.75; 0.60-0.93) and durvalumab + chemotherapy (0.80; 0.66-0.97). In patients without liver metastases (Supplemental Figure S4C), several regimens demonstrated superior PFS relative to chemotherapy: benmelstobart + anlotinib + chemo (0.31; 0.23-0.41), socazolimab + chemo (0.57; 0.45-0.74), tislelizumab + chemo (0.60; 0.47-0.77), pembrolizumab + chemo (0.64; 0.49-0.83), adebrelimab + chemo (0.64; 0.5-0.83) and atezolizumab + chemo (0.72; 0.55-0.94) showed better efficacy than chemotherapy. In patients with liver metastases (Supplemental Figure S4D), only 2 regimens showed significant improvements in PFS compared with chemotherapy: benmelstobart + anlotinib + chemo (0.36; 0.24-0.53) and socazolimab + chemo (0.49; 0.31-0.77) .
Rank probability
The treatment rankings of each outcome were determined based on SUCRA values. For OS (Figure 4A), benmelstobart + anlotinib + chemo achieved the highest SUCRA value (0.8806), indicating it was the most favorable regimen, followed by serplulimab + chemo (0.8356), nivolumab + chemo (0.7180), and adebrelimab + chemo (0.6368). In terms of PFS, benmelstobart + anlotinib + chemo also ranked highest with an SUCRA value of 0.9991, suggesting it was most likely to improve PFS (Figure 4B). This was followed by serplulimab + chemo (0.9032), socazolimab + chemo (0.7787) and tislelizumab + chemo (0.6625). For ORR (Figure 4C), the highest SUCRA values were again observed for benmelstobart + anlotinib + chemo (0.9343), followed by serplulimab + chemo (0.7840) and durvalumab + chemo (0.6979). Regarding grade ⩾ 3AEs (Figure 4D), durvalumab + chemo was associated with the lowest toxicity. In contrast, nivolumab + chemo (0.1098), benmelstobart + anlotinib + chemo (0.1190) and durvalumab + tremelimumab + chemo (0.2239) were linked to a higher risk of severe AEs compared with chemotherapy.

SUCRA probabilities of different treatments for efficacy and safety outcomes. The first rank shows the highest probabilities respectively in ranking first for improving OS (A) or prolonging PFS (B) or increasing ORR (C) or being the most likely to cause the grades ⩾ 3 AEs (D).
Discussion
Current research in SCLC has progressed beyond the traditional combination of immunotherapy and chemotherapy, now encompassing a broader range of combination strategies. In our NMAs, which included 12 RCTs involving 5840 patients with ES-SCLC, the combination of benmelstobart, anlotinib, and chemotherapy demonstrated the highest probability of delivering superior survival outcomes in terms of both PFS and OS in the overall population, Serplulimab combined with chemotherapy also ranked highly, showing considerable potential to improve both PFS and OS. Notably, the benmelstobart plus anlotinib plus chemotherapy regimen showed significant OS benefits in several subgroups, including women, nonsmokers, patients aged ⩾65 years, those with a baseline ECOG PS of 1, and patients without baseline liver metastases. Meanwhile, serplulimab plus chemotherapy was the most effective regimen for prolonging OS in subgroups such as patients without brain metastases, those with liver metastases, men, individuals under 65 years of age, and those with baseline ECOG PS of 0. It is noteworthy that our NMA included socazolimab plus chemotherapy, which was not included in previous NMAs. This combination demonstrated a more pronounced improvement in OS within specific subgroups, such as male patients under 65 years of age with ECOG PS of 1.21,22 In addition, the NMA included a comparative evaluation of the PARP inhibitor veliparib, which has been previously associated with drug resistance and the induction of cellular stress responses in SCLC due to PARP overexpression. However, veliparib failed to demonstrate any significant survival benefit. 20
The combination of benmelstobart, anlotinib, and chemotherapy represents a notable advancement in improving survival outcomes for patients with ES-SCLC. This finding highlights the potential of integrating a multi-targeted antiangiogenic agent with immunotherapy and chemotherapy to significantly enhance treatment efficacy in ES-SCLC. The observed anti-tumor effects may stem from several underlying mechanisms. Small cell lung cancer is typically classified as an “immune desert” tumor, characterized by minimal immune cell infiltration and a poor response to immunotherapy. In addition, SCLC is highly vascularized, and its abnormal tumor vasculature results in increased permeability and impaired perfusion. This exacerbates tumor hypoxia and necrosis, which in turn hinders the infiltration of tumor-specific cytotoxic T lymphocytes into the tumor microenvironment, limiting their ability to mediate effective immune responses. 23 Moreover, hypoxia contributes to the secretion of cytokines and chemokine, promoting the recruitment of immunosuppressive cells. It also induces the upregulation of immune checkpoint molecules such as CTLA-4 and PD-L1, further dampening anti-tumor immunity. Collectively, these mechanisms underscore the rationale for combining antiangiogenic therapy with immunotherapy to overcome resistance and improve outcomes in SCLC. This mechanism limits the activity of immunosuppressive cells and contributes to immune escape. 24 Antiangiogenic agents help normalize tumor vasculature, modulate the tumor microenvironment, and reduce the release of immunosuppressive factors by downregulating vascular endothelial growth factors, 25 these effects can act synergistically when combined with immunotherapy, enhancing anti-tumor efficacy. 26
However, our NMA revealed that the combination of benmelstobart, anlotinib, and chemotherapy was associated with a higher incidence of AEs, ranking second in terms of grade ⩾3 AEs. Notably, previous studies have reported a positive correlation between the occurrence of AEs and improved treatment efficacy in patients receiving immunotherapy and targeted therapies.27,28 This may explain why the benmelstobart-anlotinib-chemotherapy regimen demonstrated both superior clinical efficacy and a higher incidence of severe AEs. The promising results of this combination have spurred further investigation. For example, the BEAT-SC study compared a 4-drug regimen of bevacizumab, atezolizumab, and chemotherapy against atezolizumab plus chemotherapy in treatment-naïve ES-SCLC patients. While the combination extended PFS, it did not translate into a significant OS benefit. 29 In addition, a single-arm study reported a median OS of 17.3 months in patients treated with induction chemotherapy followed by camrelizumab, apatinib, and chemotherapy, further supporting the potential synergy between ICIs and anti-VEGFR (vascular endothelial growth factor receptor) agents in SCLC treatment. 30
At the time of diagnosis, patients with ES-SCLC frequently present with metastatic disease, with the liver and brain being among the most common sites of metastasis. These metastatic sites are important prognostic indicators. 31 Evidence suggests that the immunosuppressive tumor microenvironment associated with liver metastases can diminish the effectiveness of immunotherapy. 32 Indeed, several studies have reported that first-line ICIs did not significantly improve OS or PFS in patients with liver metastases.33,34 However, our NMAs revealed that serplulimab combined with chemotherapy improved OS, while benmelstobart combined with anlotinib and chemotherapy enhanced PFS in ES-SCLC patients with liver metastases, compared with chemotherapy alone. Nonetheless, it is important to note that the survival benefits in this subgroup were less pronounced than those observed in patients without liver metastases. Traditionally, the brain has been considered an “immune-privileged” site due to the protective roles of the blood-brain barrier (BBB) and blood-tumor barrier (BTB). However, emerging research suggests that the brain possesses constrained—rather than entirely silent—immune activity. 35 ICIs do not directly target intracranial tumor cells; instead, they activate peripheral T cells that can subsequently cross the BBB to exert anti-tumor effects. 36 A limited number of studies have shown that adding atezolizumab to chemotherapy can improve median PFS in patients with brain metastases. However, these improvements have not translated into significant OS benefits. 37 Our NMA confirmed that the addition of first-line immunotherapy particularly when combined with targeted agents to chemotherapy significantly prolonged both PFS and OS in patients without brain metastases. In contrast, patients with brain metastases did not experience similar clinical benefits, indicating that more intensive or alternative treatment strategies may be necessary for this subgroup. Emerging evidence suggests that combining brain radiotherapy (RT) with ICIs may have a synergistic therapeutic effect.38,39 In our NMA, a subset of patients received PCI following chemoimmunotherapy. However, due to the limited number of cases and lack of stratified outcome reporting, no definitive conclusions could be drawn regarding its impact. The RISE study specifically evaluated the role of RT after chemoimmunotherapy in ES-SCLC and proposed that consolidation RT protocols should be adapted to align with modern chemoimmunotherapy regimens. 40 Importantly, it also highlighted the potential negative impact of RT-induced lymphopenia, which may compromise the efficacy of immunotherapy. Therefore, limiting the RT target volume, especially to avoid unnecessary exposure of lymphocyte-rich tissues may enhance treatment tolerability and help preserve immune function, thereby sustaining therapeutic benefits. Thus, considering the complex interplay between immune modulation and central nervous system involvement, the integration of brain RT with ICIs and chemotherapy in patients with ES-SCLC and brain metastases remains an area of active investigation. 41 Further large-scale, well-designed clinical trials are warranted to define the optimal sequencing, timing, and patient selection for this multimodal approach.
Limitations
First, there was variability in trial designs, eligibility criteria, baseline characteristics (eg, ECOG status, metastasis burden), and prior treatments across the included studies. This heterogeneity may introduce bias and limit the comparability of results in various trials. Second, Most of the trials did not stratify or report outcomes based on predictive biomarkers such as PD-L1 expression, tumor mutational burden (TMB), or immune microenvironment classification. This limits the ability to personalize therapeutic strategies based on molecular or immunological markers. Third, the EA5161 trial was a phase II study and lacked comprehensive tabular data reporting. Consequently, interpretations regarding the efficacy and safety of nivolumab should be made with caution.
Conclusion
The treatment landscape of SCLC has evolved into a new era marked by multimodal strategies, particularly the integration of immunotherapy with chemotherapy. Emerging therapies such as antibody-drug conjugates and novel targeted agents are expanding the range of therapeutic options. Overall moving forward, it is crucial to optimize combination regimens, validate predictive biomarkers, and investigate the sequencing and integration of various treatment modalities to enhance clinical outcomes.
Supplemental Material
sj-docx-1-onc-10.1177_11795549251364320 – Supplemental material for Efficacy and Safety of First-Line Chemotherapy-Based Combination Therapy for Patients With Extensive-Stage Small Cell Lung Cancer: A Systematic Review and Network Meta-Analysis
Supplemental material, sj-docx-1-onc-10.1177_11795549251364320 for Efficacy and Safety of First-Line Chemotherapy-Based Combination Therapy for Patients With Extensive-Stage Small Cell Lung Cancer: A Systematic Review and Network Meta-Analysis by Qingfang Zhao, Xiaomin Liu, Minghua Yu and Jinglong Chen in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
Not applicable.
Ethical considerations
Not applicable.
Author contributions
QZ drafted the manuscript. QZ and XL extracted, analyzed, and interpreted the data of the article. MY revised the article critically for important intellectual content. JC has approved the version to be published and agreed to be accountable for all aspects of the work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.