Abstract
Background:
Since the approval of immune checkpoint inhibitors (ICIs) in extensive-stage small-cell lung cancer (ES-SCLC) patients, immunotherapy has been commonly prescribed in first-line and sometimes in subsequent-line treatment in clinical practice. However, real-world data on ICIs in ES-SCLC remain limited.
Objectives:
To delineate the current therapeutic landscape of ES-SCLC and assess the efficacy and outcomes of immunotherapy in different clinical settings.
Design and methods:
Patients with ES-SCLC who received at least two lines of therapy from February 2020 to February 2024 were retrospectively recruited. All enrolled subjects were divided into two groups, namely “ICIs cohort” and “chemotherapy-only cohort” according to the treatment regimens they received in the entire disease course. Patients in the ICIs cohort who received immunotherapy as first-line treatment were analyzed separately from those who received it in later lines. Survival analysis was conducted to evaluate the clinical significance of ICIs in different treatment settings using the Kaplan–Meier method. The utility of thoracic radiotherapy was also evaluated in ES-SCLC patients. Multivariate Cox regression was applied to identify independent predictors of survival in ES-SCLC.
Results:
A total of 214 patients were enrolled during the timeframe, of whom 81 received ICIs in first-line treatment and 78 received ICIs in subsequent-line treatment. In survival analysis, the ICIs cohort demonstrated significantly longer overall survival (OS) than the chemotherapy-only cohort, both in the first-line (17.0 vs 14.27 months; p = 0.045) and subsequent-line settings (16.87 vs 14.27 months; p = 0.017). In addition, the median OS was significantly prolonged in patients who underwent local thoracic radiotherapy compared to those who did not (20.53 vs 14.63 months; p = 0.005). Multivariate survival analysis validated that liver metastasis independently predicts inferior survival (p < 0.001). Meanwhile, immunotherapy administration (p = 0.002) and thoracic radiotherapy (p = 0.036) emerged as significant independent prognostic factors for prolonged survival in ES-SCLC patients.
Conclusion:
The incorporation of immunotherapy, either in first-line or subsequent-line treatment, significantly improved survival outcomes in ES-SCLC. Notably, local thoracic radiotherapy retained its significant survival benefit in ES-SCLC in the immunotherapy era.
Introduction
Lung cancer is the leading cause of cancer morbidity and mortality, with a 5-year survival rate of less than 20%. 1 Small-cell lung cancer (SCLC), which accounts for approximately 11% and 9% of lung cancers in men and women, respectively, is the most lethal neuroendocrine cancer subtype. 2 Due to the recalcitrant biological nature, this highly aggressive tumor exhibits rapid doubling and early widespread dissemination, 3 and about 80%–85% of patients present with extensive-stage small-cell lung cancer (ES-SCLC) at the time of initial diagnosis. 4
In ES-SCLC, the current standard of care is platinum-etoposide chemotherapy, routinely combined with the approved programmed death-ligand 1 (PD-L1) or programmed cell death-1 (PD-1) inhibitors, or given without immune checkpoint inhibitors (ICIs). 5 To date, the National Medical Products Administration has approved several ICIs for the first-line treatment of ES-SCLC, including the PD-L1 inhibitors atezolizumab, durvalumab, and adebrelimab, as well as the PD-1 inhibitors serplulimab, toripalimab, and tislelizumab. 6 Mechanistically, ICIs can restore the ability of T cells to recognize tumor cells and initiate immunotherapy by blocking the combination of PD-1 and PD-L1, thereby reversing tumor-mediated immunosuppression. 7
Despite the high sensitivity to initial treatment, therapeutic responses are not durable, and marked rapid recurrence is inevitably observed in the majority of ES-SCLC patients, and the clinical outcome for patients remains extremely poor. 8 The high overall response rates to initial treatment are as impressive as they are transient, since the median survival time is approximately 12 months or less, 9 median progression-free survival in first-line setting even after the administration of ICIs barely exceeds 5 months,10,11 and the 2-year survival rate is also less than 8%, 12 which leads to the perceptions of futility and nihilism.
In real-world clinical practice, the approval of ICIs in China has led to their increasing integration into first-line chemotherapy regimens for ES-SCLC. In addition, immunotherapy has also been employed for select relapsed ES-SCLC patients. 13 However, even with the introduction of immunotherapy, the management of ES-SCLC is still largely restricted by the limited treatment regimens and by the extreme clinical complexity of the population. In this context, real-world evidence (RWE) may help clarify the unresolved clinical questions that registration clinical trials have not yet addressed.
The current study assessed the outcomes of ES-SCLC receiving immunotherapy in the first-line or subsequent-line treatment administration. A retrospective sample of 214 patients with ES-SCLC was recruited, of whom 81 patients received initial ICIs, and 78 patients received sequential ICIs as part of their treatment regimen.
Our study aims to comprehensively characterize the current treatment landscape of ES-SCLC and specifically evaluate the clinical significance of ICIs in different clinical settings. Given the paucity of RWE, we believe that this retrospective data may offer valuable insights into the management of ES-SCLC patients in routine practice.
Materials and methods
Study design and participants
This was a retrospective study to depict the current treatment landscape of ES-SCLC and compare the real-world clinical outcome of immuno-combined treatment versus treatment without immunotherapy in patients who were available to both first-line and subsequent-line treatment. Consecutive patients with histologically confirmed ES-SCLC between February 2020 and February 2024 were identified in a database approved by the Ethics and Scientific Committees of Beijing Chest Hospital, Capital Medical University. All patients were histologically or cytologically confirmed, and the clinical stage was determined by brain magnetic resonance imaging, computed tomography of the chest and abdomen, radionuclide bone scanning, or positron emission tomography scanning. The inclusion criteria were as follows: (i) ⩾18 years of age; (ii) Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 0–2, or those with PS >2 due to acute tumor-related complications who were deemed capable of tolerating therapy; (iii) newly diagnosed with treatment-naïve ES-SCLC according to the Veterans Administration Lung Study Group staging system; and (iv) received both first-line and subsequent-line treatment in our hospital. The exclusion criteria were as follows: (i) with a history of other malignant tumors; (ii) received previous surgical resection; (iii) lacked critical information; and (iv) PS ⩾3 points not due to tumor compression (Figure 1). Patients’ demographic and clinical characteristics, including age, sex, PS, smoking history, pathology, and sites of disease metastasis at diagnosis, and previous treatment, were collected from the patients’ electronic medical records. Detailed data on specific treatment schedules for immunotherapy and thoracic radiotherapy for some patients were lacking in the medical records and, therefore, could not be systematically analyzed. This study was conducted following the Declaration of Helsinki (as revised in 2013) and was approved by the Ethics and Scientific Committees of Beijing Chest Hospital of Capital Medical University (code: LW-2024-015). The ethics committee waived individual consent due to the retrospective nature of the study. The findings of this study were reported by the STrengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 14

Flow chart of the study design. Patients with ES-SCLC were screened according to the inclusion and exclusion criteria.
Treatment and follow-up
All enrolled patients with recorded first-line and subsequent-line treatment information were included in the treatment patterns analysis. Based on the treatment regimens used, patients were divided into groups with or without ICIs (including the PD-L1 inhibitors atezolizumab, durvalumab, and adebrelimab, as well as the PD-1 inhibitors serplulimab, toripalimab, tislelizumab, and camrelizumab). The selection of treatment regimens was determined based on the following factors: patients’ PS, past medical history, and patients’ economic capacity in accordance with current guidelines and clinical experience. The landscape of first-line and subsequent-line treatment options during the entire disease course is depicted in Figure 2(a) and (c).

(a) Stacked bar graphs of treatment choices with different numbers of treatment lines. “Other” options include chemotherapy combined with multiple therapies, anti-angiogenic therapy alone, immunotherapy alone, etc. (b) Radargrams of progressive organs in the chemotherapy and immunotherapy groups. (c) Sankey diagram of patient treatment options. “Other” denotes treatment regimens not individually specified in the figure: First-line: lobaplatin and etoposide, nedaplatin and etoposide, etc., and combined immune checkpoint inhibitor regimens; subsequent-line: uncommon treatment regimens, including platinum-free chemotherapy, combinations of other therapies, and monotherapy.
The final follow-up date was December 31, 2024. Follow-up data were obtained by retrospective review of medical records, clinic visits, and telephone interviews. Overall survival (OS) was defined as the time from the start of the first-line treatment to death from any cause, with surviving patients censored at the time of the last follow-up. We evaluated OS differences between patients receiving ICIs versus those managed without ICIs.
Statistical analysis
Statistical analyses were performed using the Statistical Package for Social Science (SPSS) software (version 23, International Business Machines Corporation, USA) and GraphPad Prism software (Version 8, GraphPad Software, USA). The Kaplan–Meier method was used to analyze the survival probability, and the log-rank test was used to calculate the significance of differences. The Cox proportional hazard model was applied for the univariate and multivariate analyses to calculate the hazard ratio (HR) and 95% confidence interval (95% CI). The proportional hazards assumption was verified using Schoenfeld residuals. No major violations were detected.
Results
Baseline patient characteristics
Between February 2020 and February 2024, a total of 214 patients who received both first-line and subsequent-line treatment at our center were retrospectively recruited. The baseline clinical characteristics of the patients are shown in Table 1. The median age of the patients was 62 years (range 40–85 years), with 173 males and 41 females. The population was characterized by frequent smoking history (77.1%) and generally good PS (ECOG 0–1: 88.3%). Metastatic involvement of organs at diagnosis included liver (25.2%), bone (30.8%), brain (29.9%), and adrenal glands (29.4%). In first-line treatment, the most commonly used regimen in the chemotherapy group was CE (n = 81, 37.9%), followed by EP (n = 29, 13.6%). The most commonly used regimen for chemotherapy combined with immunotherapy was CE plus PD-L1 inhibitors (n = 60, 28.0%), followed by CE plus PD-1 inhibitors (n = 9, 4.2%). During the entire treatment course, 119 patients (55.6%) received immunotherapy, and the remaining 95 patients (44.4%) constituted the non-ICIs cohort. In first-line immunotherapy, PD-L1 inhibitors (n = 66, 81.5%) were more frequently administered than PD-1 inhibitors (n = 15, 18.5%). Consistently, PD-L1 inhibitors (n = 49, 62.8%) demonstrated higher utilization rates than PD-1 inhibitors in subsequent-line immunotherapy. Notably, 41.5% (n = 89) of patients underwent local thoracic radiotherapy during their treatment course.
Baseline clinical characteristics of patients with ES-SCLC.

Other, including lobaplatin and etoposide, nedaplatin and etoposide, etc., and combined immunotherapy regimens.
CE, carboplatin and etoposide; CEA, carcinoembryonic antigen; ECOG, Eastern Cooperative Oncology Group; EP, cisplatin and etoposide; ES-SCLC, extensive-stage small-cell lung cancer; ICIs, immune checkpoint inhibitors; NSE, neuron-specific enolase; PD-1, programmed cell death protein 1; PD-L1, programmed cell death ligand 1; pro-GRP, progastrin-releasing peptide.
Landscape of first-line and subsequent-line treatment options
In the first-line treatment, 133 patients (62.1%) were administered chemotherapy alone, while 81 patients (37.9%) received a combination of chemotherapy and immunotherapy (Figure 2(a) and (c)). Following initial therapy, all patients experienced disease progression or metastasis to various organs and underwent subsequent-line treatment. A comparative analysis of progression patterns revealed distinct metastatic distributions among treatment cohorts. In the chemotherapy-alone group (n = 133), lung progression predominated (n = 106, 79.7%), followed by brain (n = 41, 30.8%), liver (n = 20, 15.0%), bone (n = 13, 9.8%), and adrenal gland metastases (n = 10, 7.5%). Notably, the ICIs cohort (n = 81) demonstrated different progression patterns, with higher frequencies of brain (n = 46, 46.9%), liver (n = 19, 23.5%), and adrenal metastases (n = 15, 18.5%), though lung involvement remained the most frequent site (n = 46, 56.8%; Figure 2(b)). In the current study, subsequent-line therapy refers to second-line and beyond systemic therapy. The regimens for these subsequent-line treatments displayed significant heterogeneity, with combination therapies being the most prevalent and chemotherapy serving as the cornerstone. In second-line treatment, chemotherapy remained predominant (n = 65, 30.4%). The proportions of combination therapies, including chemotherapy plus radiotherapy (n = 33, 15.4%), chemotherapy plus immunotherapy (n = 34, 15.9%), and chemotherapy plus anti-angiogenic therapy (n = 33, 15.4%), were relatively balanced. Notably, treatment patterns evolved in third-line and later treatments, with increased adoption of chemotherapy plus anti-angiogenic therapy and a growing trend toward triple-combination regimens (chemotherapy plus immunotherapy plus anti-angiogenic therapy) compared to second-line treatment (Figure 2(a) and (c)).
Clinical outcomes in ES-SCLC receiving immunotherapy
As of December 31, 2024, the median OS for the entire study cohort was 14.27 months (95% CI, 14.09–17.51). Throughout the treatment course, ICIs cohort achieved superior OS of 16.67 months (95% CI, 14.529–18.811) compared to 14.27 months (95% CI, 12.117–16.423) in non-ICIs cohort, with a risk ratio (HR) = 0.704 (95% CI, 0.521–0.952, p = 0.021; Figure 3(a); Table 2). Subgroup survival analyses were performed to evaluate the clinical significance of immunotherapy when used as either first-line or subsequent-line treatment. Results indicated that patients who received immune-combined therapy as initial treatment regimen had a median OS of 17.0 months (95% CI, 13.716–20.284), compared with those who never received immunotherapy (14.27 months, 95% CI, 12.117–16.423), with an HR of 0.709 (95% CI, 0.506–0.994; p = 0.045; Figure 3(b); Table 2). Further analysis revealed a significant survival advantage for subsequent immunotherapy-treated patients, with a median OS of 16.87 months (95% CI, 12.898–20.842) compared to 14.27 months (95% CI, 12.117–16.423) in non-ICIs cohort (HR = 0.668, 95% CI, 0.478–0.934, p = 0.017; Figure 3(c), Table 2). In addition, when comparing the timing of immunotherapy initiation, the median OS did not differ significantly between patients receiving first-line immunotherapy and those treated in the subsequent-line setting (17.0 vs 16.10 months, HR = 0.939, 95% CI, 0.605–1.457, p = 0.778; Figure 3(d), Table 2).

OS Kaplan–Meier curves for the entire cohort. (a) Immunotherapy was received during treatment or not. (b) Based on whether immunotherapy is received in the first-line treatment. (c) Based on whether immunotherapy is received in the subsequent-line treatment. (d) Comparison of first-line immunotherapy and subsequent-line immunotherapy.
Univariate and multivariate Cox regression analyses of OS in patients with ES-SCLC.
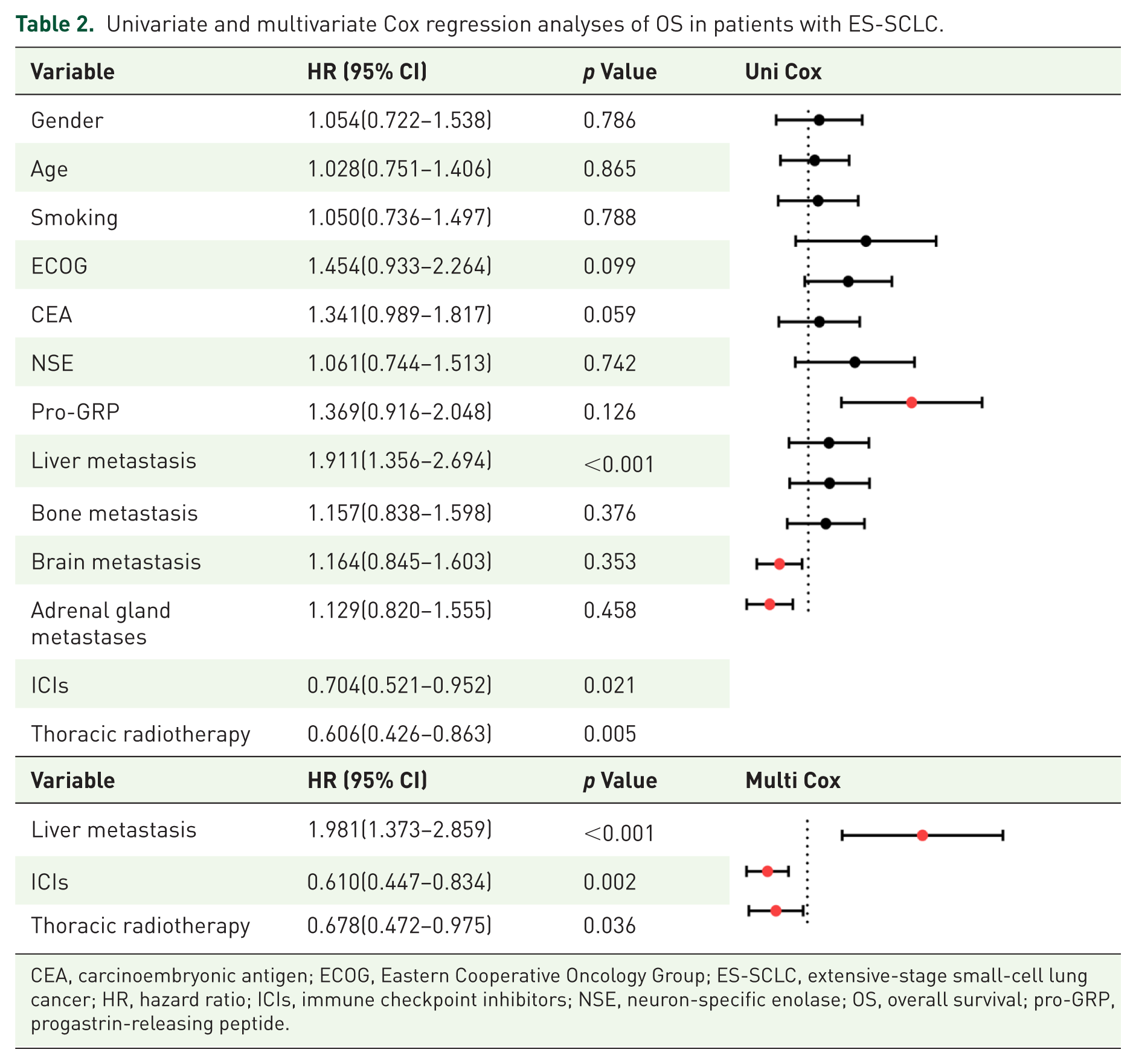
CEA, carcinoembryonic antigen; ECOG, Eastern Cooperative Oncology Group; ES-SCLC, extensive-stage small-cell lung cancer; HR, hazard ratio; ICIs, immune checkpoint inhibitors; NSE, neuron-specific enolase; OS, overall survival; pro-GRP, progastrin-releasing peptide.
Local radiotherapy for ES-SCLC
In this study, we evaluated the clinical significance of local thoracic radiotherapy and whole-brain radiotherapy (WBRT) separately. Among all enrolled patients, 55 patients (25.7%) underwent local thoracic radiotherapy either during or after completion of their first-line therapy, achieving a median OS of 20.53 months (95% CI, 16.684–24.376). By contrast, patients who did not receive local thoracic radiotherapy had a median OS of 14.63 months (95% CI, 13.132–16.128, p = 0.005; Figure 4(a); Table 2). The addition of WBRT was also evaluated. Results demonstrated that patients who received WBRT (n = 83, 38.7%) exhibited a longer median OS of 17.97 months (95% CI, 15.453–20.487) versus 14.27 months (95% CI, 12.590–15.943) for those who did not receive WBRT, but it did not reach statistical significance (p = 0.264, Figure 4(b)). Furthermore, we conducted comparative survival analyses between patients receiving local thoracic radiotherapy with or without ICIs. Contrary to our hypothesis, patients receiving localized thoracic radiotherapy combined with immunotherapy (n = 20, 36.3%) showed numerically lower survival rates compared to radiotherapy alone, though this difference did not reach statistical significance (18.53 vs 22.0 months, p = 0.188; Figure 4(c)). A larger sample size may be required to establish the therapeutic benefit of the combination of the two. Kaplan–Meier analysis revealed no significant survival benefit when combining WBRT with immunotherapy compared to WBRT alone (20.0 vs 17.97 months, p = 0.278; Figure 4(d)).

(a) KM curves of OS in different groups of local thoracic radiotherapy. (b) KM curves of the OS of WBRT. (c) Survival outcomes comparing local thoracic radiotherapy alone versus combined with ICIs. (d) Survival outcomes comparing WBRT alone versus combined with ICIs.
Univariate and multivariate prognostic analyses for ES-SCLC patients
Our univariate Cox regression analysis identified three clinically significant prognostic factors for OS. Specifically, patients with liver metastases had a nearly 1.9-fold higher risk of death compared to those without liver metastases (HR = 1.911, 95% CI, 1.356–2.694, p < 0.001). Conversely, two therapeutic interventions demonstrated protective effects. The risk of death was significantly reduced by 29.6% in patients treated with ICIs throughout the treatment course (HR = 0.704, 95% CI, 0.521–0.952; p = 0.021). In addition, patients who received local thoracic radiotherapy in the first line had a 39.4% lower risk of death (HR = 0.606, 95% CI, 0.426–0.863; p = 0.005). Other variables, including tumor markers and additional metastatic sites, failed to show significant prognostic value in our cohort (all p > 0.05). Multivariable analysis confirmed that liver metastases, the use of immunotherapy throughout the treatment course, and receiving local thoracic radiotherapy in the first-line remained as independent prognostic indicators after adjusting for potential confounders (Table 2).
Discussion
This retrospective study revealed the landscape of first-line and subsequent-line treatment options and systematically evaluated the clinical significance of ICIs for ES-SCLC in a real-world setting. Patients receiving chemoimmunotherapy as first-line treatment demonstrated notable improvements in OS. Importantly, even when administered as subsequent-line therapy, the incorporation of immunotherapy can also significantly prolong patients’ survival. In addition, our study confirmed the continued importance of thoracic radiotherapy in the era of immunotherapy, validating its crucial role in the management of ES-SCLC.
It has been confirmed that the incidence of SCLC is significantly associated with tobacco consumption. Long-term tobacco exposure can induce a high tumor mutation burden (TMB), which may create an opportunity for ICI therapy. 15 SCLC exhibits a robust response to initial chemotherapy, which can facilitate the release of immune antigens and enhance the T-cell response. 16 Collectively, these findings provide a theoretical foundation for integrating immunotherapy and chemotherapy in SCLC. Given the evidence supported by a series of randomized clinical trials, checkpoint inhibitors have been approved in ES-SCLC with moderate improvements in median OS in recent years. 17 Currently, either chemotherapy or chemoimmunotherapy is recommended in the first-line treatment for ES-SCLC according to National Comprehensive Cancer Network guidelines. 12 In the current real-world retrospective study, only subjects with available information on both first-line and subsequent-line treatments were recruited. The proportion of brain metastases was much higher in our study compared to the corresponding clinical trials, Impower-133 18 and CASPIAN. 19 Among all enrolled patients, 133 (62.1%) were administered chemotherapy as first-line treatment, while 81 (37.9%) received chemoimmunotherapy as initial treatment. The landscape of treatment distribution in our center indicated that chemotherapy remains a predominant first-line option. The majority of included patients did not receive immunotherapy, primarily because the high economic cost of ICIs, such as atezolizumab and durvalumab, significantly influences the choice of treatment regimen. The financial burden often leads to the selection of more cost-effective, albeit potentially less efficacious, treatment options. In addition, some patients abandon immunotherapy due to their disease status and physical condition. For those who received chemoimmunotherapy in our study, atezolizumab and durvalumab are the most commonly used treatment regimens, while serplulimab, tislelizumab, and sintilimab are also sometimes prescribed. Survival analysis revealed that the addition of ICIs in first-line treatment significantly prolonged patients’ OS compared to that of the chemotherapy-only group. This result is consistent with the findings of previous prospective studies and other reported real-world data, supporting chemoimmunotherapy as the preferred first-line standard regimen.
According to the report that the most dominant pattern of tumor progression was at the initial thoracic lesion in SCLC. 20 Comparative analysis of progression patterns was conducted in our study, and the results revealed distinct metastatic distributions in the two treatment cohorts. Among patients who received chemotherapy as initial treatment, primary lung lesion enlargement was the most common pattern of tumor progression (79.7%), followed by brain metastases (30.8%), liver metastases (15.0%), bone metastases (9.8%), and adrenal gland metastases (7.5%). Notably, patients administered with first-line chemoimmunotherapy demonstrated different progression patterns, with higher frequencies of brain metastases (46.9%), liver metastases (23.5%), and adrenal metastases (18.5%), though thoracic lesions remained the most frequent site of disease progression (56.8%). This discrepancy may be explained by the longer survival of patients who had received treatment with the addition of ICIs.
Beyond first-line treatment, ICIs have also been evaluated in patients with relapsed SCLC. In subsequent treatment, KEYNOTE-028, 21 KEYNOTE-158, 22 and CheckMate032 23 studies concluded that ICIs are of clinical benefit in recurrent/metastatic SCLC, but KEYNOTE-604 24 and CheckMate331 25 subsequently overturned this indication. In real-world practice, immunotherapy has been commonly prescribed as first-line and sometimes as subsequent-line treatment in ES-SCLC patients. In the current study, chemotherapy accounts for 30.4% of all second-line treatment options and continues to be the most frequently used regimen. In addition, chemoimmunotherapy based on a PD-L1 inhibitor accounted for 15.9%, followed by chemotherapy plus anti-angiogenic therapy at 15.4%. The usage of immunotherapy can also be observed in the third line and beyond, occasionally. It is noteworthy that our findings suggest that patients who received immunotherapy in the subsequent-line treatment had longer survival time than those who had never used immunotherapy. Our result is in accordance with previously reported real-world retrospective data, which has also suggested that patients with ES-SCLC can benefit from the use of ICIs in the subsequent-line treatment.26,27
Subgroup analysis in this study suggests no marked difference in survival outcomes between the first-line ICIs group and the subsequent-line ICIs group. Our study confirmed that chemoimmunotherapy is the preferred standard initial treatment regimen for ES-SCLC. Moreover, we further demonstrated that the addition of immunotherapy significantly improves the prognosis of patients. This finding underscores the principle that, for ES-SCLC, immunotherapy is beneficial even if initiated later in the treatment course—better late than never. However, due to the exploratory nature of the current study, no formal adjustment for multiple comparisons was applied, and further prospective large cohort investigations are warranted to validate our findings.
In this study, local thoracic radiotherapy significantly prolonged the survival time of patients with ES-SCLC and maintained an independent prognostic value in a multivariate analysis. This result is consistent with the findings of certain prospective trials that have shown that thoracic radiotherapy not only improves the local control rate and symptomatic remission but also may result in OS benefit in ES-SCLC.28 –31 Our RWE further supports the clinical significance of thoracic radiotherapy in the management of ES-SCLC. Several retrospective studies have demonstrated that thoracic radiotherapy combined with ICIs significantly improves patient survival even in the era of immunotherapy, suggesting that there may be a synergistic effect between radiotherapy and immunotherapy.32 –35 Radiotherapy, as an adjunct to ICIs, has been shown to enhance the effect of immunotherapy to a certain extent, and relevant studies have confirmed that radiotherapy can change the tumor immune microenvironment.36 –38 However, there is still a lack of prospective data supporting the optimal dose of radiotherapy, target range, and timing of combination with ICIs. Relevant randomized controlled trials need to be conducted in the future to further optimize the comprehensive treatment model for ES-SCLC.
Multifactorial analysis in this study showed that liver metastasis was an independent risk factor for ES-SCLC. Patients with SCLC liver metastasis are affected by multiple factors in the tumor microenvironment, such as the unique immunosuppressive microenvironment of the liver, 39 and therefore have the worst prognosis among advanced lung cancers and respond poorly to chemotherapy and immunotherapy. This study demonstrates that both immunotherapy and thoracic radiotherapy have the potential to offer significant survival protection. In contrast to prospective trials, the real-world data from this study further confirm that the benefits of ICIs can be extended to populations not covered by prospective clinical trials. It is also noteworthy that the protective effect of chest radiotherapy exists independently of ICIs, supporting its continued value in the era of immunotherapy.
This study also has some limitations. First, the high heterogeneity of ICIs subtypes, treatment cycles, and combination regimens makes it difficult to distinguish drug-specific effects. Moreover, the absence of PD-L1 and TMB data made it impossible to characterize the tumor immune microenvironment. Future investigations incorporating these markers are warranted to validate our findings and to elucidate the potential role of immune-related biomarkers. Lastly, our data lack the critical information of treatment-related adverse events, primarily due to doctors’ limited awareness of immune-related adverse events in early immunotherapy cases.
Conclusion
In real-world practice, we conducted a comprehensive evaluation of the ICI’s application in ES-SCLC patients in different clinical settings. Our study supports the incorporation of immunotherapy, either in initial or in subsequent treatment, which significantly improved survival outcomes in ES-SCLC. Notably, local thoracic radiotherapy retained its significant survival benefit in the immunotherapy era.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251387570 – Supplemental material for Real-world evidence for immunotherapy in the first-line or subsequent-line setting in extensive-stage small-cell lung cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359251387570 for Real-world evidence for immunotherapy in the first-line or subsequent-line setting in extensive-stage small-cell lung cancer by Ruizi Ren, Ying Wang, Qunhui Wang, Baohua Lu, Yuan Gao, Hongxia Li, Hengrui Zhang, Yong Zhang and Tongmei Zhang in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors thank all the participants in this study. Also, thanks to Biorender for Figure 1 (![]() ).
).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

