Abstract
Background
Extensive-stage small cell lung cancer (ES-SCLC) is a highly aggressive malignancy with poor prognosis. This study aimed to assess the efficacy of combining immunotherapy (IT) with Anlotinib in ES-SCLC patients.
Methods
This study was a multicenter retrospective cohort analysis. Survival outcomes were evaluated using Kaplan-Meier curves and Cox proportional hazards regression models.
Results
A total of 147 patients were included in the analysis. The median overall survival (mOS) for the cohort was 15.5 months (95% CI: 13.9-17.1). Patients in the chemotherapy(CT) plus IT group had an mOS of 17.8 months, compared to 12.6 months in the CT-alone group (p = 0.055). When stratified into CT + IT + Anlotinib, CT + IT, and CT-alone groups, the mOS were 18.5, 16.3, and 12.6 months, respectively, with the CT + IT + Anlotinib group demonstrating significantly improved OS compared to CT-alone (p = 0.044). The ORR and DCR for the entire cohort were 71.4% and 85.7%, respectively. Subgroup analysis revealed ORRs of 74.1% (CT + IT + Anlotinib), 73.9% (CT + IT), and 70.1% (CT-alone), with corresponding DCRs of 92.6%, 91.3%, and 82.5%. Multivariate analysis revealed that radiotherapy (RT, p = 0.003) and IT (p = 0.021) were independent prognostic factors for OS, while liver metastasis (p = 0.023) and RT (p = 0.018) were associated with PFS. Patients receiving RT in combination with CT showed markedly improved OS (17.5 vs 12.5 months; p = 0.002) and PFS (7.3 vs 6.3 months; p = 0.004). The incidence of adverse events was comparable across all groups (p = 0.721).
Conclusion
The combined application of Anlotinib with IT and the combination of CT with RT both significantly improved survival outcomes in patients with ES-SCLC while maintaining a favorable safety profile. These findings warrant further investigation in future studies.
Introduction
Lung cancer remains one of the most prevalent and lethal malignancies globally. 1 According to the 2024 global cancer burden statistics, approximately 2.48 million new cases of lung cancer were diagnosed worldwide in 2022, resulting in 1.82 million deaths, representing 12.4% of all cancer-related fatalities. 2 In China, lung cancer accounted for 1.06 million new cases and 733 000 deaths in the same year, ranking first in both incidence and mortality among malignancies. 2 Small cell lung cancer (SCLC), a highly aggressive subtype, constitutes approximately 15% of all lung cancer cases. 1 The Veterans Administration Lung Study Group (VALSG) staging system classifies SCLC into limited-stage SCLC (LS-SCLC) and extensive-stage SCLC (ES-SCLC). 3 SCLC is distinguished by its rapid growth and early metastasis, with most patients presenting at the extensive stage at diagnosis. 4 Traditional first-line treatment involves 4-6 cycles of platinum-based chemotherapy (eg, etoposide with cisplatin/carboplatin). Although initial response rates reach 50%-70%, drug resistance develops rapidly, leading to a median OS of less than one year and a 5-year survival rate below 7%.5,6
Vascular endothelial growth factor (VEGF) is a critical mediator of angiogenesis, a process essential for tumor growth and metastasis. 7 Targeting VEGF has demonstrated efficacy in treating multiple malignancies, including colorectal, gastric, breast, renal, hepatic, and non-small cell lung cancers. Evidence indicates that SCLC progression is heavily dependent on microvessel formation. 8 Moreover, VEGF overexpression in SCLC has been associated with poorer outcomes, highlighting angiogenesis inhibition as a potential therapeutic strategy.Immune checkpoint inhibitors (ICIs), which target programmed cell death protein 1 (PD-1) and programmed cell death-ligand 1 (PD-L1), have emerged as a promising therapeutic approach for multiple cancers. In ES-SCLC, recent prospective studies have shown that combining ICIs with CT can improve median overall survival (mOS) to 12-15 months, signaling the start of the immunotherapy era for this disease.9-12 As a result, immunotherapy (IT) has become a key component of first-line treatment for ES-SCLC, establishing a new standard of care. Combining anti-angiogenic therapy with IT has shown synergistic effects across various cancers, 13 as vascular normalization can enhance immune cell infiltration and alter the tumor microenvironment.14,15 Anlotinib, a multi-target anti-angiogenic agent, has the potential to boost the efficacy of ICIs through such mechanisms.16,17 This study aims to investigate the clinical efficacy and safety of combining IT with Anlotinib in ES-SCLC patients within a real-world clinical setting.
Materials and Methods
Data Collection
This research was designed as a retrospective cohort study and all patient data were de-identified to ensure confidentiality. Medical records of patients diagnosed with ES-SCLC who received first-line CT between January 2020 and January 2024 were reviewed across three tertiary public hospitals. Baseline characteristics were systematically collected. All patients underwent enhanced computed tomography scans of the neck, chest, and abdomen, as well as magnetic resonance imaging (MRI) of the brain to evaluate intracranial conditions. When necessary, additional assessments, including positron emission tomography-computed tomography (PET-CT) and bone scans, were performed to establish baseline staging. The reporting of this study conforms to the STROBE guidelines. 18
Patient Selection
Inclusion Criteria: 1. Patients received first-line treatment. 2.Patients aged ≥18 years. 3. Pathologically confirmed diagnosis of SCLC. 4. Eastern Cooperative Oncology Group Performance Status (ECOG PS) score between 0 and 2. 5. Receipt of at least two cycles of CT. 6. Patients treated with ICIs or Anlotinib received a minimum of two treatment cycles and underwent at least one efficacy evaluation. 7. Availability of complete clinical data prior to treatment and comprehensive follow-up records. Exclusion Criteria: 1. Lack of a definitive pathological diagnosis. 2. Presence of concurrent other types of malignant tumors. 3. ECOG PS score greater than 2. 4. Receipt of fewer than two cycles of CT or fewer than two cycles of ICI or Anlotinib treatment. 5. Absence of efficacy evaluation or loss to follow-up. 6. Presence of blood disorders, autoimmune diseases, or other significant comorbid conditions. Patients were consecutively selected based on inclusion/exclusion criteria from three participating centers.
Observational index
The primary outcome measure was overall survival (OS), while the secondary outcomes included progression-free survival (PFS), objective response rate (ORR), disease control rate (DCR) and the safety profile of combination therapy. The assessment relied on imaging tests, with efficacy assessments conducted at a minimum of one time after every two treatment cycles. An overall response encompassed a complete response (CR), partial response (PR), stable disease (SD), or progressive disease (PD). The formula for the ORR was: ORR = (CR + PR) / (CR + PR + SD + PD) × 100%; while the DCR was calculated as: DCR = (CR + PR + SD) / (CR + PR + SD + PD) × 100%. OS was defined as the duration from the initiation of CT to all-cause mortality or the date of the last follow-up. PFS was defined as the interval from CT initiation to tumor progression or death from any cause. Patients without any recorded events by the final follow-up or those lost to follow-up were considered censored. Tumor response was assessed based on the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. Adverse events were graded according to the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0 established by the United States National Cancer Institute. The latest follow-up for this study was conducted on January 31, 2024.
Statistical Analysis
The Shapiro-Wilk (S-W) test was used to assess the normality of the data distribution. Quantitative variables following a normal distribution were presented as mean ± standard deviation(
Results
Data Collection
A Total of 365 Patients Were Initially Screened for Eligibility in This Study. Exclusions Included 150 Patients with LS-SCLC, 21 Patients who Received Fewer Than two Cycles of CT, IT, or Anlotinib, 16 Patients who did not Receive First-Line IT or Anlotinib, and 31 Patients with Incomplete or Uncertain OS or PFS Data. Ultimately, 147 Patients Satisfied the Inclusion Criteria and Were Included in the Final Analysis: 27 Received CT + IT + Anlotinib, 23 Received CT + IT, and 97 Received CT Alone. (Figure 1)

Diagram of patient's selection process.
This study included 147 patients diagnosed with ES-SCLC who underwent first-line CT. Detailed baseline clinical characteristics are summarized in Table 1. The median age of the cohort was 65 years (range: 44-79 years), and 81% (119/147) of the patients were male. Most patients (94.6%, 139/147) had an ECOG PS score of 0-1. At the time of initial diagnosis, the proportions of patients with metastases to the brain, liver, and bone were 21.8%, 22.4%, and 27.2%, respectively. Additionally, 45.6% of the patients received RT, and 34.0% underwent IT during their treatment course.
Baseline Characteristics of Patients According to First-Line Therapy.

Baseline Characteristics of Patients According to First-Line Therapy.

All patients included in the study received first-line CT. Among them, 139 patients were treated with platinum-based regimens: 52 received carboplatin, and 87 received cisplatin in combination with etoposide. Additionally, 5 patients were treated with nedaplatin combined with etoposide, and 3 received irinotecan combined with cisplatin. A total of 27 patients received Anlotinib as part of their first-line treatment. Anlotinib was administered in a 21-day cycle, taken orally once daily from day 1 to day 14, with an initial dose of 10-12 mg. In cases of severe adverse reactions, the dose was gradually reduced, with adjustments from 12 mg to 8 mg as needed.
Regarding IT, 50 patients underwent first-line IT. Of these, 21 were treated with PD-1 inhibitors, including tislelizumab (n = 10), serplulimab (n = 3), camrelizumab (n = 3), sintilimab (n = 2), pembrolizumab (n = 2), and toripalimab (n = 1). The remaining 29 patients received PD-L1 inhibitors, such as durvalumab (n = 23), atezolizumab (n = 4), and avelumab (n = 2).
The radiation dose for each patient was determined based on the recommendations of the Chinese Society of Clinical Oncology (CSCO) or the National Comprehensive Cancer Network (NCCN) guidelines. The total prescribed dose ranged from 30 Gy to 60 Gy, with a median of 45 Gy. Radiation was delivered in daily fractions of 2-3 Gy, administered five days per week. To ensure accuracy in treatment delivery, all plans were evaluated to verify that the planning target volume consistently received at least 95% of the intended dose.
Survival Analysis
The follow-up period ended on January 31, 2024, with a median duration of 25.0 months (range: 5-38 months). Among the cohort, 50 patients (34.0%) received combined CT and IT, while 97 patients (66.0%) underwent CT alone. By the end of follow-up, 122 patients (83.0%) had reached the primary endpoint: 40 patients (80.0%) in the CT + IT group and 82 patients (84.5%) in the CT-only group. Disease progression occurred in all patients during the follow-up period. Survival analysis showed a mOS of 15.5 months (95% CI: 13.9-17.1) for the entire cohort. The 1-year and 5-year OS rates were 71.8% and 3.0%, respectively. Although the differences in OS (17.8 months vs 12.6 months; p = 0.055) and PFS (7.1 months vs 6.5 months; p = 0.157) between the CT + IT and CT-only groups were not statistically significant, the data suggested a trend toward improved OS in the CT + IT group (Figure 2, Table 2).

The kaplan-meier survival curves.
the Survival Rate of OS and PFS.

The study population was categorized into three groups based on their receipt of IT and Anlotinib: the CT + IT + Anlotinib group (27 patients, 18.4%), the CT + IT group (23 patients, 15.6%), and the CT-only group (97 patients, 66.0%). Short-term efficacy was assessed after 2 cycles of treatment. Among patients in the CT + IT + Anlotinib, CT + IT, and CT-alone groups, CR was observed in 1, 1, and 4 patients, respectively; PR was achieved in 19, 16, and 64 patients; SD was documented in 5, 4, and 12 patients; and PD occurred in 2, 2, and 17 patients. The ORR and DCR for the entire cohort were 71.4% and 85.7%, respectively. Subgroup analysis revealed ORRs of 74.1% (CT + IT + Anlotinib), 73.9% (CT + IT), and 70.1% (CT-alone), with corresponding DCRs of 92.6%, 91.3%, and 82.5% (Table 3). While no statistically significant difference in OS was observed between the CT + IT group and the CT-only group (16.3 months vs 12.6 months, p = 0.328), the CT + IT + Anlotinib group demonstrated a significantly longer OS compared to the CT-only group (18.5 months vs 12.6 months, p = 0.044). The 1-year OS rates in the CT + IT + Anlotinib, CT + IT and CT alone group were 96.3%, 82.6% and 61.8%, respectively; the 2-year OS rates were 11.7%, 6.8% and 12.0%, respectively. In contrast, PFS did not significantly differ among the three groups (7.3 months, 6.9 months, and 6.5 months, respectively).The 6-month PFS rates in the CT + IT + Anlotinib, CT + IT and CT alone group were 88.9%, 69.6% and 60.8%, respectively; the 12-month PFS rates were 7.4%, 21.7% and 7.2%, respectively. (Figure 2, Tables 2–3).
Survival Outcomes of Three Groups.

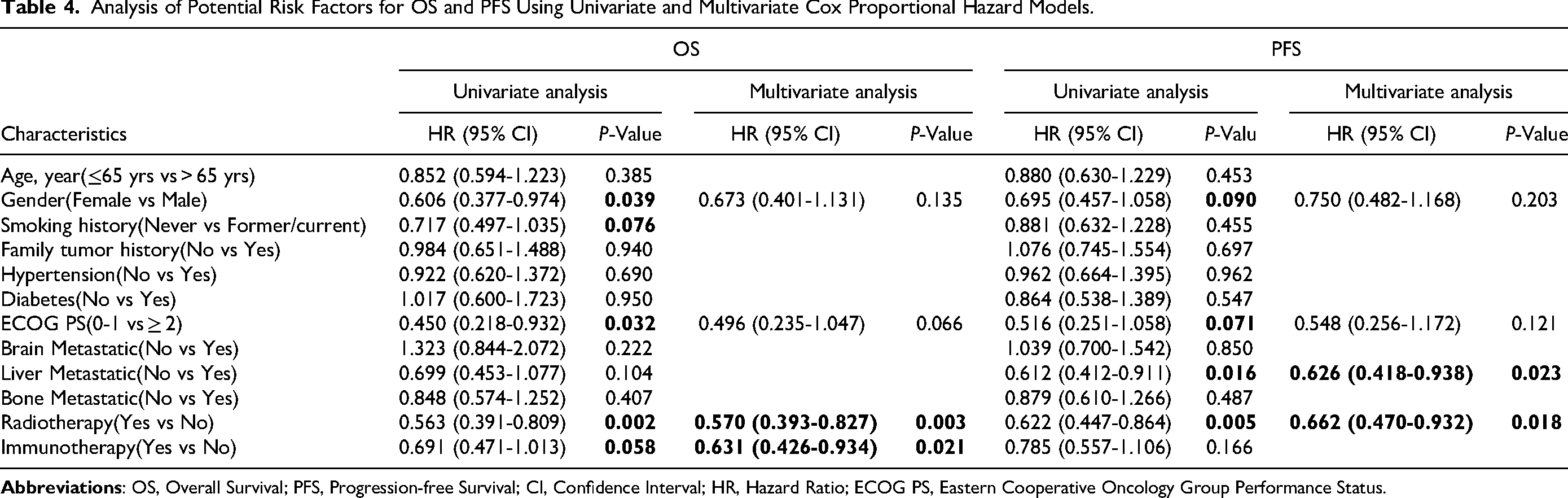
Univariate survival analysis identified gender (p = 0.039), ECOG PS (p = 0.032), radiotherapy (RT) (p = 0.002), and IT (p = 0.048) as significant prognostic factors for OS. Similarly, gender (p = 0.049), liver metastasis (p = 0.016), and RT (p = 0.005) were found to significantly influence PFS. Multivariate analysis further confirmed RT (HR: 0.570; 95% CI: [0.393-0.827]; p = 0.003) and IT (HR: 0.631; 95% CI: [0.426-0.934]; p = 0.021) as independent prognostic factors for OS. Additionally, liver metastasis (HR: 0.626; 95% CI: [0.418-0.938]; p = 0.023) and RT (HR: 0.662; 95% CI: [0.470-0.932]; p = 0.018) were independently associated with PFS (Table 4).
Analysis of Potential Risk Factors for OS and PFS Using Univariate and Multivariate Cox Proportional Hazard Models.
Recognizing RT as an independent prognostic factor for both OS and PFS, we performed survival analyses stratified by RT status. Of the total cohort, 67 patients (45.6%) received RT, while 80 patients (54.4%) did not. The survival analysis revealed that patients who underwent combined RT and CT experienced significantly longer OS (17.5 months vs 12.5 months; p = 0.002) and PFS (7.3 months vs 6.3 months; p = 0.004) compared to those who received CT alone (Figure 2).
Across the entire cohort, 99 (81.2%) patients experienced treatment-related adverse events (trAEs). Among these, 76 (51.0%) cases were classified as grade 1-2, while 45 (30.2%) cases were grade 3-4. The incidence of grade 3-4 adverse events was 37.0%, 43.5%, and 35.1% in the CT + IT + AL group, CT + IT group, and CT group, respectively, with no statistically significant differences observed among the three groups (p = 0.721). Notably, no grade 5 adverse events were reported (Table 5).
Treatment-Related Adverse Event.

Treatment-Related Adverse Event.

CT, chemotherapy; IT, immunotherapy; AL, Anlotinib.
ES-SCLC remains a lethal malignancy with limited therapeutic advances despite global efforts. In recent years, significant advancements have been made in the treatment of ES-SCLC, particularly with the integration of immunotherapy (IT) into the standard platinum-based chemotherapy regimen. Several pivotal clinical trials have validated the role of PD-1/PD-L1 inhibitors in ES-SCLC. In the CASPIAN trial, durvalumab combined with platinum-etoposide significantly improved mOS to 13.0 months versus 10.3 months with CT alone (HR 0.75; 95% CI 0.62-0.91; p = 0.0032), with grade ≥3 adverse events (AEs) occurring in 62% versus 62% of patients. 11 Similarly, IMpower133 demonstrated that adding atezolizumab to carboplatin-etoposide extended mOS to 12.3 months compared to 10.3 months (HR 0.76; 95% CI 0.60-0.95; p = 0.0154), with grade 3–4 AEs reported in 56.6% versus 56.1% of patients. 10 The ASTRUM-005 trial further showed that serplulimab plus CT achieved an mOS of 15.4 months versus 10.9 months (HR 0.63; 95% CI 0.49-0.82; p < 0.001), with a 33.2% incidence of grade ≥3 AEs in the experimental arm. 9 CheckMate 032 and KEYNOTE-028 reported objective response rates (ORR) of 10%–25% with nivolumab or pembrolizumab monotherapy in relapsed SCLC, albeit with higher immune-related AEs (eg, pneumonitis in .5-8%).19,20 These studies highlight that combination therapy substantially improves patient survival compared to historical outcomes achieved with CT alone. As a result, PD-1/PD-L1 inhibitors combined with CT have been established as the recommended first-line treatment for ES-SCLC in clinical guidelines. Despite these advances, the clinical benefits of IT in real-world ES-SCLC patients remain under investigation. Hence, we conducted a real-world study to evaluated the effectiveness and safety of first-line CT combined with IT in ES-SCLC. Our analysis found no statistically significant differences in OS (17.8 months vs 12.6 months; p = 0.055) and PFS (7.1 months vs 6.5 months; p = 0.157) between patients receiving CT + IT (n = 50) and those receiving CT alone (n = 97). However, a trend toward improved OS was observed in the CT + IT group (p = 0.055). The discrepancies between our findings and those of previous studies may be attributed to the limited sample size and the retrospective nature of the study, which is inherently prone to selection bias.
Notably, the OS of the CT + IT group in our study (17.8 months) exceeded the reported in all major prospective trials, including IMpower133, CASPIAN, CAPSTONE-1, and ASTRUM-005, where OS ranged between 12.3 and 15.4 months.9,11,12,21 Similarly, a multicenter retrospective study by Xie et al, 22 which included 83 propensity score-matched (PSM) patient pairs, reported a mOS of 17.3 months in the EP + ICI cohort compared to 13.4 months in the EP cohort (p = 0.001). This aligns closely with our findings, supporting the robustness of our results. Moreover, the PFS of the CT + IT group in our study (7.1 months) also surpassed that reported in these trials, where the mPFS ranged from 4.5–5.8 months for CT + IT and 4.3–5.6 months for CT alone. To explore this phenomenon, we examined prognostic factors and found that multivariate analysis indicated that RT was an independent prognostic factor for both OS and PFS in ES-SCLC. Further analysis revealed that RT significantly improved OS (17.5 months vs 12.5 months; p = 0.002) and PFS (7.3 months vs 6.3 months; p = 0.004) . The LEAD study, presented at the European Lung Cancer Congress (ELCC), highlighted the clinical feasibility and potential of integrating systemic and local therapies. 23 Similarly, a study by Tian et al, 24 utilizing data from the National Cancer Database, evaluated 14,367 ES-SCLC patients, of whom 12,348 received CT combined with RT (CT + RT). Multivariate analysis revealed a higher risk of death in the CT-only group compared to the CT + RT group (HR 1.74; 95% CI [1.64-1.84]; p < 0.001). PSM analysis further showed superior outcomes in the CT + RT group, with an mOS of 12.1 months compared to 8.2 months in the CT-only group, 12-month OS rates of 50.5% versus 28.5%, and 5-year OS rates of 7.6% versus 2.0% (HR 1.80; 95% CI [1.67-1.95]; p < 0.001). Additionally, an Italian multicenter retrospective study demonstrated that CT + RT significantly improved the 12-month PFS rate (61% vs 31%; p < 0.001) and 12-month OS rate (80% vs 61%; p = 0.027) compared to CT alone. 25 Consolidative RT has been consistently shown to reduce the risk of intrathoracic progression, while CT + RT markedly improves both PFS and OS.4,26,27 Consistent with these findings, our study showed that patients in the CT + RT group achieved significantly better OS (17.5 months vs 12.5 months; p = 0.002) and PFS (7.3 months vs 6.3 months; p = 0.004) compared to those receiving CT alone. The superior survival outcomes in our study compared to those in prospective trials may be attributed to the high proportion of patients (45.6%) in our cohort who received RT, which likely contributed to improved clinical outcomes.
Previous studies have highlighted the synergistic potential of combining anti-angiogenic agents with immune checkpoint inhibitors (ICIs) in cancer treatment. The IMpower150 trial demonstrated that the combination of an anti-PD-L1 agent (Atezolizumab), bevacizumab, and with platinum-based chemotherapy significantly enhanced outcomes in patients with advanced non-small cell lung cancer (NSCLC). 28 Similarly, combining ICIs with anti-angiogenic inhibitors has shown promising ORRs in renal cell carcinoma and urothelial carcinoma patients.29,30 Fan et al reported that the PD-1 inhibitor camrelizumab, when combined with the anti-angiogenic agent apatinib, improved both ORR and progression-free survival (PFS) in patients with ES-SCLC. 31 The abnormal tumor microenvironment (TME), influenced by vascular endothelial growth factor (VEGF)-driven angiogenesis, contributes to immunosuppression and limits the efficacy of PD-1/PD-L1 blockade.32,33 Anlotinib, a multi-target tyrosine kinase inhibitor (TKI), has been shown to normalize tumor vasculature by enhancing pericyte and endothelial cell distribution. It also promotes immune cell infiltration by downregulating PD-L1 expression on endothelial cells, reducing the recruitment of immunosuppressive cells, and enhancing T-cell activity, thereby improving the effectiveness of IT.34-36 Yang et al demonstrated that first-line maintenance therapy combining anlotinib with anti-PD-1/PD-L1 antibodies provided significant clinical benefits in ES-SCLC patients, achieving a PFS of 13.6 months (95% CI: 11.2-15.6 months) and an OS of 19.5 months (95% CI: 14.5-24.5 months), both statistically significant. 37 In our study, IT combined with CT did not significantly improve OS compared to CT alone (17.8 months vs 12.6 months, p = 0.055). However, adding Anlotinib to the IT + CT regimen led to a statistically significant improvement in OS (18.5 months vs 12.6 months, p = 0.044), underscoring the potential synergy between anti-angiogenic agents, IT, and CT in treating ES-SCLC. Additionally, the 1-year OS rates were 96.3% (CT + IT + Anlotinib), 82.6% (CT + IT), and 62.2% (CT alone), while the 6-month PFS rates were 88.9%, 69.6%, and 60.8%, respectively. Short-term efficacy evaluation revealed ORRs of 74.1%, 73.9%, and 70.1%, with corresponding DCRs of 92.6%, 91.3%, and 82.5%. It can be observed that the survival benefit in the combination therapy group was numerically slightly improved. Moreover, we found that the 12-month PFS rate in the CT + IT group was significantly higher than in the other two groups (7.4%, 21.7%, and 7.2%), which might be attributable to bias from the small number of enrolled patients (n = 23). Although the beneficial interaction between anti-angiogenic therapy and IT has been recognized, the precise mechanisms and optimal conditions for this synergy remain to be fully elucidated.
Our retrospective multicenter analysis demonstrated comparable incidences of treatment-related adverse events (trAEs) across the CT + IT + Anlotinib, CT + IT, and CT-only groups. Most trAEs observed during treatment were mild to moderate (grade 1-2) and manageable. Commonly reported adverse events included fatigue, nausea, rash, and hemoptysis. Importantly, no patients discontinued treatment due to trAEs. These findings reaffirm the safety of integrating IT with Anlotinib in the management of ES-SCLC.
The primary strengths of this study include its demonstration of the efficacy and safety of IT combined with Anlotinib in patients with ES-SCLC, as well as further suggest potential synergistic effects between these two therapies. Moreover, the combination of CT and RT showed significant survival benefits. However, several limitations must be addressed. First, as a retrospective study, it is subject to inherent confounding factors and potential selection bias. Second, the relatively small sample size of patients receiving Anlotinib may have introduced bias into the conclusions. Third, the limited cohort of patients treated with IT, along with the use of diverse anti-PD-1/PD-L1 agents, may have influenced the analysis results. Fourth, while nearly half of the patients underwent RT, the inclusion of different modalities—such as thoracic RT, brain RT, and bone RT—might have affected the outcomes. Lastly, our retrospective design cannot definitively establish synergy, because it is not possible to statistically isolate and test an interaction effect between IT and Anlotinib using our current dataset.However, the observed survival trends, combined with mechanistic evidence, provide a strong rationale for future prospective trials comparing CT + IT versus CT + IT + Anlotinib.
Conclusions
Our study highlights the potential synergistic effects of combining Anlotinib with IT in the treatment of ES-SCLC, demonstrating improved patient survival with a favorable safety profile. Furthermore, the integration of CT and RT significantly enhances survival outcomes in ES-SCLC patients. Despite certain limitations, these combination therapies may merit further exploration in future research.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
