Abstract
Objectives:
Several randomized controlled trials (RCTs) indicated that first-line programmed cell death protein-1/death-ligand 1 inhibitors plus chemotherapy (PD-1/PD-L1 + chemo) led to survival benefits in extensive-stage small-cell lung cancer (ES-SCLC) compared with platinum-based chemotherapy. This study aims to identify the optimal PD-1/PD-L1 + chemo combination strategy.
Methods:
We included RCTs comparing PD-1/ PD-L1 + chemo versus chemo alone in ES-SCLC. Overall survival (OS), progression-free survival (PFS), objective response rate (ORR), and grade ⩾3 treatment-related adverse events were considered. Odds ratios (ORs), hazard ratios (HRs), and their 95% confidence intervals (CIs) were extracted.
Results:
Six RCTs with 2600 patients were analyzed in this Bayesian network meta-analysis. Results showed that adding PD-1/PD-L1 inhibitors to chemotherapy led to significant benefits in OS (HR = 0.72, 95% CI: 0.66–0.79), PFS (HR = 0.69, 95% CI: 0.63–0.75), and ORR (OR = 1.32, 95% CI: 1.12–1.56), and no differences in toxicity were found (OR = 1.09, 95% CI: 0.92–1.30). Serplulimab plus chemotherapy was found to provide the best OS (HR = 0.63, 95% CI: 0.49–0.82), the best PFS (HR = 0.47, 95% CI: 0.38–0.59), and the best ORR (OR = 1.7, 95% CI: 1.15–2.53). Moreover, although there were no difference between PD-L1 + chemo and PD-1 + chemo regarding OS (HR = 0.99, 95% CI: 0.91–1.08) and ORR (OR = 1.27, 95% CI: 0.91–1.78), PD-1 + chemo showed a significant benefit in PFS (HR = 0.82, 95% CI: 0.68–0.98) compared with PD-L1 + chemo.
Conclusions:
Serplulimab plus chemotherapy seems to be superior first-line immunotherapy combination for patients with ES-SCLC. PD-1 + chemo seems to outperform PD-L1 + chemo in PFS.
Keywords
Introduction
Small-cell lung cancer (SCLC), an aggressive carcinoma with high growth fraction, rapid progress, and early widespread metastases, accounts for about 15% of all lung cancer,1,2 and approximately two-third SCLC patients are diagnosed with extensive disease. 3 Platinum-etoposide chemotherapy can significantly palliate symptoms as well as prolong survival and has been the standard first-line treatment for patients with extensive-stage small-cell lung cancer (ES-SCLC). 4 Nevertheless, most patients rapidly and unavoidably develop resistance to chemotherapy, with a median progression-free survival (PFS) shorter than 6 months and a median overall survival (OS) time less than 1 year. 5 Due to the worse survival, more efficacious treatments are urgently needed in ES-SCLC patients.
Immunotherapy is one of the most important breakthroughs in cancer treatment, especially immune checkpoint inhibitors (ICIs) that block coinhibitory molecules such as programmed cell death protein-1 (PD-1) and related programmed death-ligand 1 (PD-L1), providing us with a new treatment option. In 2018, the IMpower133 study, a phase III study, has firstly revealed that atezolizumab, a PD-L1 targeted ICI, provides additional benefits in both median OS [12.3 versus 10.3 months, hazard ratio (HR) = 0.70, 95% confidence interval (CI): 0.54–0.91] and median PFS (5.2 versus 4.3 months, HR = 0.77, 95% CI: 0.62–0.96) when compared with platinum-based chemotherapy as the first-line treatment for patients with ES-SCLC. 6 Subsequently, another phase III study CASPIAN also demonstrates a survival benefit with another PD-L1 inhibitor durvalumab plus chemotherapy in prolonging median OS (13 versus 10.3 months, HR = 0.73, 95% CI: 0.59–0.91) than chemotherapy alone. 7 Based on the positive results of the two studies, PD-L1 inhibitor (atezolizumab and durvalumab) plus chemotherapy has been approved by the US Food and Drug Administration (FDA) as the first-line treatment for patients with ES-SCLC.8,9 Meanwhile, in addition to the above two ongoing studies, a series of randomized controlled trials (RCTs) concerning the application of ICIs in ES-SCLC has been published, including pembrolizumab, 10 nivolumab, 11 adebrelimab, 12 and serplulimab. 13 Nevertheless, this field is still evolving. In spite of survival benefit associated with ICIs, there are no head-to-head researches focusing on comparison among them. Due to the varied efficacy and safety profile among the ICIs trials, choosing the optimal combination strategy in clinical practice might be troubled. Chen et al. 14 has performed a network meta-analysis (NMA), and the results revealed that there was no statistical difference on PFS or OS among four agents of PD-1/PD-L1 inhibitors as the first-line treatment in patients with ES-SCLC. However, only four RCTs were included, and the results should be cautiously interpreted.
In this study, we aim to evaluate the efficacy and safety of all the currently available first-line PD-1/PD-L1 combinations for patients with ES-SCLC. A meta-analysis was performed through indirect comparisons on the basis of the Bayesian framework approach, intending to identify the optimal ICI-combined chemotherapy (ICI-chemo) strategy for ES-SCLC patients.
Methods
Data source and search strategy
The electronic databases, namely, PubMed, Cochrane Library, and ClinicalTrials.gov databases were systematically searched for relevant literatures conducted until July 6, 2022. To include the updated outcomes, we also explored online proceedings from annual conferences including American Society of Clinical Oncology (ASCO), Chinese Society of Clinical Oncology (CSCO), European Society of Medical Oncology, and The World Conference on Lung Cancer. The following keywords were used for literature search: randomized clinical trial, small-cell lung cancer, ICI, PD-1, and PD-L1 (Supplemental Table S1). This study was registered in the Inplasy Register of Systematic Reviews (INPLASY202290121) to ensure transparency.
Selection criteria
The inclusion criteria were as follows: (1) RCTs that enrolled patients with ES-SCLC confirmed by either histologically or cytologically; (2) RCTs that used PD-1/PD-L1 inhibitor combinations as first-line treatment settings; (3) RCTs comparing first-line combinations of PD-1/PD-L1 inhibitors with platinum-etoposide chemotherapy versus platinum-etoposide chemotherapy alone in ES-SCLC; and (4) phase II or III trials reporting at least one of the following clinical outcomes: OS, defined as the time from randomization until death from any cause; PFS, defined as the time from randomization to disease progression or death from any cause; ORR, defined as the proportion of patients who achieved an objective response; treatment-related adverse events (TRAEs) of any-grade or grade ⩾3 TRAEs, which were defined and graded according to the National Cancer Institute Common Terminology Criteria for adverse events.
The exclusion criteria were as follows: (1) RCTs that were based on overlapping patients; and (2) RCTs with ambiguous clinical outcomes.
Data extraction and quality assessment
Data were independently extracted by two investigators (H.J. Li and H.D. Han), and any discrepancies were resolved by discussions with the other authors (C.L. Li, R.P. Wu, and Z.F. Wang). The trial name, publication sources, year of publication, sample size, trial phase, National Clinical Trials identification number, drugs and doses of experimental arm and control arm, and median follow-up were extracted from each study. The clinical outcomes extracted included median OS, median PFS, HRs with corresponding 95% CIs for OS and PFS, the incidence of ORR, any-grade TRAEs, and TRAEs of grade ⩾3. Cochrane Risk of Bias Tool (2.0) was used to evaluate the quality of the included RCTs, and the following items were deemed as necessary criteria for assessment: selection of the reported result, measurement of the outcome, missing outcome data, deviations from intended interventions, and randomization process. 15 The included studies were sorted into one of the following three categories: low risk, some concerns, and high risk.
Statistical analysis
After data were abstracted, all statistical analyses were executed using R software (version 4.2.1) and R Studio software. As already described, the outcomes selected to perform a standard meta-analysis and a Bayesian framework indirect comparison were OS, PFS, ORR, and grade ⩾3 TRAEs. We considered HRs to evaluate the association for PFS and OS with the relative 95% CIs. Furthermore, we considered odds ratios (ORs) with 95% CIs as an association measure for ORR and grade ⩾3 TRAEs. The χ2 test and I2 statistics were applied to evaluate the statistical heterogeneity of the included studies. If the p value for χ2 > 0.1 and I2 was <50%, a fixed-effects model would be used to count the pooled estimate. 16 Otherwise, a random-effects model would be selected to combine the studies. Chemotherapy was used as the control therapeutic arm in the indirect comparison, and this NMA indirectly evaluated the relative efficacy of different ICIs, via comparing ICI-chemo with chemotherapy. 17 The Bayesian NMA estimated the relative treatment effects through HRs, ORs, and corresponding 95% CIs. For each outcome measure, three independent Markov chains were run in parallel for 10,000 burn-ins and 100,000 sample iterations. The NMA also provided overall ranking probabilities of each ICI combination being the best among all treatments, via ranking the effects of all treatments in each iteration and then calculating the percentage of each treatment being ranked first across all iterations.
Results
Characteristics of the included RCTs
We identified a total of 1133 records through the databases and conference proceedings during the preliminary literature search. After eliminating the duplicates and no pertinent articles through title and abstract screening, 107 studies were considered eligible for full-text review, and finally, six RCTs for a total of eight full-text studies and one abstract met our eligibility criteria (Figure 1), including IMpower133,6,18 CASPIAN,7,19,20 KEYNOTE-604, 10 EA5161, 11 CAPSTONE-1, 12 and ASTRUM-005. 13 A total of 2600 patients were enrolled to receive the following seven treatments: chemotherapy (chemo), atezolizumab plus chemotherapy (Atezo-chemo), durvalumab plus chemotherapy (Durva-chemo), pembrolizumab plus chemotherapy (Pem-chemo), nivolumab plus chemotherapy (Nivo-chemo), adebrelimab plus chemotherapy (Ade-chemo), and Serplulimab plus chemotherapy (Serp-chemo). Among them, three explored the efficacy of PD-L1 inhibitor (atezolizumab or durvalumab or adebrelimab) plus chemotherapy versus chemotherapy alone, and the other three explored the efficacy of PD-1 inhibitor (pembrolizumab or nivolumab or serplulimab) plus chemotherapy versus chemotherapy alone. The network plot is depicted in Figure 2. Detailed information on the included RCTs has been summarized in Table 1.

Literature search and selection.

Network of the comparisons
Baseline characteristics of RCTs included in the network meta-analysis.

Data are presented as experimental/control unless indicated otherwise.
ASCO, American Society of Clinical Oncology; AUC, area under curve; ESMO, European Society for Medical Oncology; HR (95% CI), hazard ratio (95% confidence interval); NR, not reported; ORR, objective response rate; OS, overall survival; PD-1, programmed cell death protein-1; PD-L1, programmed death-ligand 1; PFS, progression-free survival; TRAEs, treatment-related adverse events.
Quality assessment of the included RCTs
With the performance of quality assessment according to the criteria of Cochrane Risk of Bias Tool (2.0), we detected that all of the included RCTs in this study satisfied the criteria items including allocation concealment, random sequence generation, binding of outcome assessments, and binding of participants and personnel, with results presented in Supplemental Figure S1.
Overall survival
The addition of a PD-1/PD-L1 inhibitor to platinum-etoposide chemotherapy led to a statistically significant benefit in OS in patients with ES-SCLC (HR = 0.72, 95% CI: 0.66–0.79) (Figure 3(a)), and patients receiving either PD-L1 + chemo (HR = 0.73, 95% CI: 0.64–0.82) or PD-1 + chemo (HR = 0.72, 95% CI: 0.62–0.83) exhibited significantly longer OS than those receiving chemo alone. Moreover, Serp-chemo yielded the best OS benefit compared with chemotherapy (HR = 0.63, 95% CI: 0.49–0.82) (Figure 4(a)), and the analysis showed no statistically significant difference in OS between any two of Atezo-chemo, Pem-chemo, Nivo-chemo, Ade-chemo, Durva-chemo, and Serp-chemo (Figure 4(a)). There were also no statistical difference between PD-L1 + chemo and PD-1 + chemo in OS (HR = 0.99, 95% CI: 0.91–1.08) (Figure 4(c)).

Meta-analysis results for (a) OS, (b) PFS, (c) ORR, and (d) grade ⩾3 TRAEs in patients with ES-SCLC.

Efficacy and safety profiles of the Bayesian network meta-analysis in patients with ES-SCLC. (a) HRs and 95% CIs for overall survival (upper triangle in blue) and progression-free survival (lower triangle in yellow), (b) ORs and 95% CIs for objective response rate (upper triangle in blue) and grade ⩾3 TRAEs (lower triangle in yellow), and (c) OS, PFS, ORR, and grade ⩾3 TRAEs according to used immunotherapy molecule. The results are presented as column-defined treatment versus row-defined treatment.
Progression-free survival
Patients who received PD-1/PD-L1 inhibitor combinations revealed consistently better PFS than standard chemotherapy (HR = 0.69, 95% CI: 0.63–0.75) (Figure 3(b)), and relative to chemo, patients receiving either PD-L1 + chemo (HR = 0.75, 95% CI: 0.67–0.84) or PD-1 + chemo (HR = 0.62, 95% CI: 0.54–0.71) exhibited significantly longer PFS. Serp-chemo yielded the best PFS benefit compared with chemotherapy (HR = 0.47, 95% CI: 0.38–0.59) (Figure 4(a)); furthermore, Serp-chemo was discerned to offer marked PFS benefits on comparison with Ade-chemo (HR = 0.71, 95% CI: 0.52–0.96), Pem-chemo (HR = 0.64, 95% CI: 0.47–0.86), Atezo-chemo (HR = 0.61, 95% CI: 0.45–0.84), and Durva-chemo (HR = 0.59, 95% CI: 0.44–0.79) (Figure 4(a)). In addition, treatment regimens containing anti-PD-1 were found to yield superior PFS benefit when compared with regimens containing anti-PD-L1 (HR = 0.82, 95% CI: 0.68–0.98) (Figure 4(c)).
Objective response rate
Relative to standard chemotherapy, PD-1/PD-L1 inhibitor combinations revealed better ORR (OR = 1.32, 95% CI: 1.12–1.56) (Figure 3(c)), and although no statistical difference in ORR was found in PD-L1 + chemo (OR = 1.2, 95% CI: 0.96–1.49), PD-1 + chemo exhibited a potential advantage (OR = 1.52, 95% CI: 1.18–1.96). Serp-chemo was observed to be the best treatment with regard to ORR (OR = 1.7, 95% CI: 1.15–2.53), followed by Durva-chemo (OR = 1.54, 95% CI: 1.08–2.19) and Pem-chemo (OR = 1.48, 95% CI: 1.01–2.2) (Figure 4(b)). And any combination treatment of Serp-chemo (OR = 2.03, 95% CI: 1.16–3.6), Durva-chemo (OR = 1.84, 95% CI: 1.08–3.14), and Pem-chemo (OR = 1.78, 95% CI: 1.01–3.13) had shown statistical superiority on ORR when compared with Atezo-chemo (Figure 4(b)). Moreover, no difference was perceived regarding ORR between PD-L1 + chemo and PD-1 + chemo (OR = 1.27, 95% CI: 0.91–1.78) (Figure 4(c)).
Toxicity
Safety and toxicity were determined according to any-grade TRAEs and grade ⩾3 TRAEs. Toxicity of Durva-chemo was found to be the lowest for all PD-1/PD-L1 inhibitor combinations with the fewest grade ⩾3 TRAEs, especially when compared with Nivo-chemo (OR = 0.4, 95% CI:0.18–0.85) (Figure 4(b)). Nevertheless, the addition of PD-1/PD-L1 inhibitor to standard chemotherapy tended to elevate the toxicity when compared with standard chemotherapy (OR = 1.09, 95% CI: 0.92–1.30), especially when PD-1 inhibitor was added (OR = 1.30, 95% CI: 1.01–1.68) (Figure 3(d)). And there was no significant difference between PD-L1 + chemo and PD-1 + chemo in grade ⩾3 TRAEs (OR = 1.39, 95% CI: 0.98–1.97) (Figure 4(c)).
Rankings
Ranking analysis was performed based on the Bayesian ranking profiles (Supplemental Table S2). The ranking results were accordant with the pooled results obtained using HRs and ORs, revealing the stability and reliability of the framework (Figure 5). For patients with ES-SCLC, superior efficacy was achieved for Serp-chemo, and the treatment ranked first for OS (cumulative probability of 44.5%), PFS (92.4%), and ORR (45.3%), albeit ranked sixth in safety analysis with relative more grade ⩾3 TRAEs (43.9%). For patients with ES-SCLC according to immunotherapy molecule type, PD-1 + chemo was most likely to rank first for OS (57.9%), PFS (98.5%), and ORR (92.3%), whereas PD-L1 + chemo was most likely to rank first for TRAEs of grade ⩾3 (70.3%).
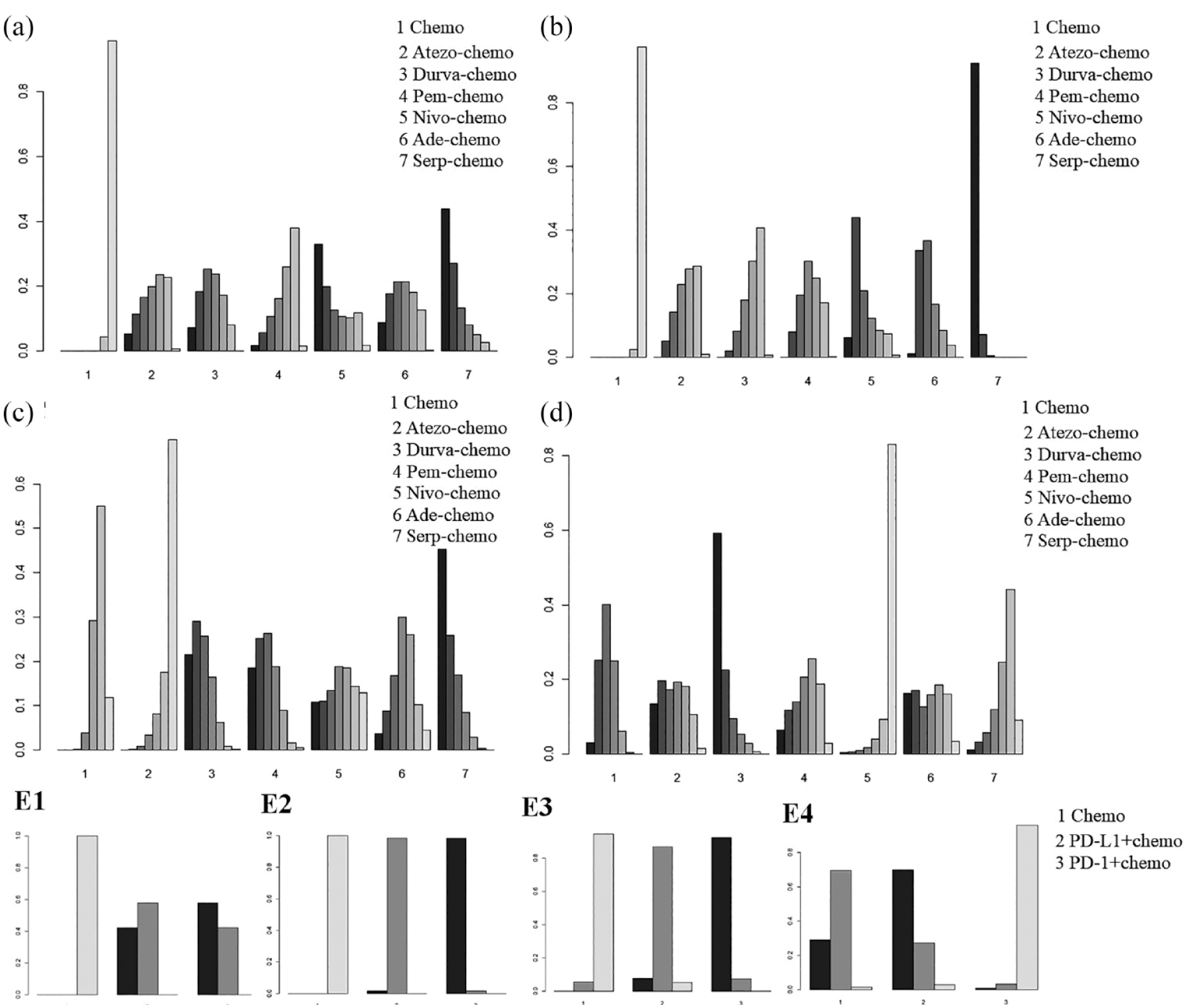
Bayesian ranking profiles of (a) OS, (b) PFS, (c) ORR, and (d) grade ⩾3 TRAEs for the entire study population and according to used immunotherapy molecule [(E1) OS, (E2) PFS, (E3) ORR, and (E4) grade ⩾3 TRAEs].
Discussion
So far, the most impressive characteristic about ES-SCLC is the bleak prognosis. With PD-L1 inhibitors atezolizumab and durvalumab approved by the FDA in combination with platinum-based chemotherapy, the advent of immunotherapies of the first-line treatment for ES-SCLC finally arrived.21,22 Lately, the clinical world of oncologists were excited again by results of RCTs including KEYNOTE-604, 10 EA5161, 11 CAPSTONE-1, 12 and ASTRUM-005, 13 which showed that PD-L1 inhibitor (adebrelimab) and PD-1 inhibitors (pembrolizumab, nivolumab, and serplulimab) led to improved survival benefits when concurrently combined with chemotherapy, highlighting the therapeutic value of combinations of PD-1/PD-L1 inhibitor with platinum-etoposide chemotherapy. However, although a consistent and reproducible pattern of efficacy improvement has been noted, it is warranted to carry out additional studies to provide clarity on the benefit of PD-1/PD-L1 inhibitor combinations in this setting. And many of the oncologists have long considered different ICIs (no matter targeting PD-1 or PD-L1) as equally effective and clinically interchangeable options. Hence, establishing the optimal combination strategy still addresses an unmet clinical need in the first-line setting.
Since it will be unlikely to see head-to-head comparison studies, our study represents an attempt to indirectly compare these combination approaches to identify any potential differences in both activity and toxicity profiles. Based on a thorough review of current random clinical trials, we included six phase II/III studies comparing combinations of PD-1/PD-L1 inhibitors with platinum-etoposide chemotherapy versus platinum-etoposide chemotherapy alone in the first-line setting for patients with ES-SCLC. Envisaging the translation of the observed results in the clinical practice, the following two major findings in this NMA seemed worthy of attention.
First, our study found that Serp-chemo yielded the best OS benefit (HR = 0.63, 95% CI: 0.49–0.82), the best PFS benefit (HR = 0.47, 95% CI: 0.38–0.59), and the best ORR benefit (OR = 1.7, 95% CI: 1.15–2.53) compared with chemotherapy. Comprehensively, efficacy was significantly superior in Serp-chemo, which ranked the first for OS, PFS, and ORR across all PD-1/PD-L1 inhibitor combinations. The ASTRUM-005 study 13 revealed that Serp-chemo experimental arm achieved a statistically significant benefit in median OS with an extension of 4.5 months (15.4 versus 10.9 months, HR = 0.63, 95% CI: 0.49–0.82, p < 0.001), broke the benefit range of previous PD-L1 inhibitors,21,22 and set a new record of OS for first-line treatment of ES-SCLC, significantly reducing the risk of death by 37% (HR = 0.63, 95% CI: 0.49–0.82, p < 0.001). The 6-month and 12-month PFS rates in Serp-chemo experimental arm were 2.5 and 4 times higher than chemotherapy control arm, respectively (48.1% versus 19.7%, 23.8% versus 6.0%), demonstrating robust and durable ability of tumor control, with 52% lower risk of disease progression or death (5.7 versus 4.3 months, HR = 0.48, 95% CI: 0.38–0.59, p < 0.001). The pronounced anti-tumor efficacy of serplulimab may be contributed to the molecular structure and function. Serplulimab, a fully humanized IgG4 monoclonal antibody against PD-1 receptor, occupies a solvent-accessible overlapping surface area of 445 Å2 (55% of PD-L1 surface) on PD-1. 23 Serplulimab showed potent PD-L1 and PD-L2 blocking activity and was efficient in enhancing T-cell responses and cytokine production in vitro. And no antibody-dependent cellular cytotoxicity or complement-dependent cytotoxicity was observed with serplulimab when using syngeneic PD-1-expressing activated T cells as target T cells, suggesting that serplulimab was unlikely to deplete PD-1-positive cells in patients. 23 Based on the promising research results, The National Medical Products Administration has accepted the marketing application of serplulimab for first-line treatment of ES-SCLC, and the CSCO clinical guidelines for the Diagnosis and Treatment of SCLC, updated and released in 2022, had regarded serplulimab combined with chemotherapy as the grade III recommendation (class 1A evidence), which would potentially be a first-line treatment option for ES-SCLC. In addition, the safety of serplulimab is worthy of attention. Though safety analysis in ASTRUM-005 study 13 showed that serplulimab demonstrated good security, the results of our NMA suggested that serplulimab was associated with relatively more TRAEs, ranking sixth for grade ⩾3 TRAEs across all PD-1/PD-L1 inhibitor combinations. Some meta-analyses24,25 have revealed a positive association between occurrence of adverse events and improved treatment efficacy in patients treated with ICIs in several solid malignancies including lung cancer. It could explain the phenomenon that serplulimab ranked the first in clinical efficacy but with relatively more higher-graded TRAEs. In short, serplulimab displayed substantially superior survival benefits and may be a promising optimal combination strategy for patients with ES-SCLC.
Second, though no relevant statistically significant differences were observed between PD-L1 + chemo and PD-1 + chemo regarding OS and ORR in the NMA, PD-1 + chemo showed a significant benefit in PFS (HR = 0.82, 95% CI: 0.68–0.98) compared with PD-L1 + chemo. The underlying mechanism remains to be fully elucidated, but one possible reason could be attributed to the interaction of PD-1 and PD-L2 that could also inhibit the activation of T cells. And to our knowledge, PD-1 inhibitor can simultaneously block the binding of PD-1 to both PD-L1 and PD-L2, inhibiting the immune escape pathway more comprehensively, whereas PD-L1 inhibitor can only inhibit the binding of PD-1 to PD-L1, and cancer cells can thereby escape antitumor immune response through the PD-1/PD-L2 axis. 26 However, meta-analyses performed by other researchers showed no difference between PD-L1 + chemo and PD-1 + chemo in terms of survival benefit.27,28 This may resulted from limited sample size and number of included RCTs in these studies. Recently, survival data in previously published RCTs were updated and the latest RCTs including CAPSTONE-1 and ASTRUM-005 were published, leading to the possibility of more comprehensive comparison and identification of the optimal ICI-chemo strategy. Our observation that PD-1 + chemo yielded statistical superiority on PFS than PD-L1 + chemo is meaningful for patients with ES-SCLC to choose optima treatments. Nevertheless, further researches including head-to-head comparison are required to substantiate this finding and explore the underlying mechanism.
In conclusion, our NMA suggested that Serp-chemo seemed to be superior first-line PD-1/PD-L1 inhibitor combination for patients with ES-SCLC with relatively more grade ⩾3 TRAEs. Furthermore, PD-1 + chemo exhibited potentially better survival outcomes than PD-1 + chemo with comparable safety profiles.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231189430 – Supplemental material for Efficacy and safety of first-line PD-1/PD-L1 inhibitor combinations for extensive-stage small-cell lung cancer: a Bayesian network meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359231189430 for Efficacy and safety of first-line PD-1/PD-L1 inhibitor combinations for extensive-stage small-cell lung cancer: a Bayesian network meta-analysis by Huijuan Li, Hedong Han, Chuling Li, Ranpu Wu, Zhaofeng Wang, Yimin Wang, Ping Zhan, Tangfeng Lv, Fang Zhang, Yong Song and Hongbing Liu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359231189430 – Supplemental material for Efficacy and safety of first-line PD-1/PD-L1 inhibitor combinations for extensive-stage small-cell lung cancer: a Bayesian network meta-analysis
Supplemental material, sj-docx-2-tam-10.1177_17588359231189430 for Efficacy and safety of first-line PD-1/PD-L1 inhibitor combinations for extensive-stage small-cell lung cancer: a Bayesian network meta-analysis by Huijuan Li, Hedong Han, Chuling Li, Ranpu Wu, Zhaofeng Wang, Yimin Wang, Ping Zhan, Tangfeng Lv, Fang Zhang, Yong Song and Hongbing Liu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-3-tam-10.1177_17588359231189430 – Supplemental material for Efficacy and safety of first-line PD-1/PD-L1 inhibitor combinations for extensive-stage small-cell lung cancer: a Bayesian network meta-analysis
Supplemental material, sj-jpg-3-tam-10.1177_17588359231189430 for Efficacy and safety of first-line PD-1/PD-L1 inhibitor combinations for extensive-stage small-cell lung cancer: a Bayesian network meta-analysis by Huijuan Li, Hedong Han, Chuling Li, Ranpu Wu, Zhaofeng Wang, Yimin Wang, Ping Zhan, Tangfeng Lv, Fang Zhang, Yong Song and Hongbing Liu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
