Abstract
Background:
Recently, several new first-line immune checkpoint inhibitors (ICIs) plus chemotherapy have been approved for patients with extensive-stage small-cell lung cancer (ES-SCLC). However, direct comparisons between first-line treatments are lacking. Therefore, we indirectly compared the efficacy and safety of specific treatment strategies to inform physicians’ and patients’ clinical decisions.
Methods:
The Pubmed, Cochrane, Embase, and Web of Science databases were searched from 1 January 2000 to 27 November 2022, for randomized clinical trials (RCTs) assessing first-line immuno-chemotherapies for ES-SCLC. A fixed-effect multivariable meta-regression model was established for frequentist network meta-analysis and hazard ratios (HRs) with 95% confidence intervals (95% CI) were computed to compare the effects of immuno-chemotherapies on patient overall survival (OS) and progression-free survival (PFS), while risk ratios with 95% CI were used for treatment- and immune-related adverse events (AEs). The p score values were then used to rank treatments based on their odds of being the best treatment option. The research protocol was registered with the PROSPERO (CRD42022383254).
Results:
Seven studies involving 3822 patients were eligible for analysis. Serplulimab plus chemotherapy had better OS outcomes compared to chemotherapy (HR = 0.63; 95% CI: 0.49–0.82) and ipilimumab plus chemotherapy (HR = 0.67; 95% CI: 0.50–0.90). It additionally exhibited better PFS outcomes compared to chemotherapy (HR = 0.48; 95% CI: 0.39–0.60), adebrelimab (HR = 0.72; 95% CI: 0.53–0.97), atezolizumab (HR = 0.62; 0.46–0.85), durvalumab (HR = 0.60; 95% CI: 0.45–0.80), durvalumab and tremelimumab (HR = 0.57; 95% CI: 0.43–0.76), ipilimumab (HR = 0.57; 95% CI: 0.44–0.73), and pembrolizumab (HR = 0.64; 95% CI: 0.48–0.86) plus chemotherapy. Serplulimab plus chemotherapy was linked to the greatest odds of effectively reducing the odds of death (p score = 0.87) and progression (p score = 0.99) while exhibiting a good safety profile.
Conclusion:
Serplulimab plus chemotherapy exhibited the best survival outcomes with manageable AEs. Thus, serplulimab plus chemotherapy may represent the optimal best first-line treatment option for ES-SCLC patients.
Keywords
Introduction
Lung cancer is the deadliest malignancy in the world, with more than 179,000 deaths, and the second most diagnosed cancer, with approximately 220,000 diagnosed cases each year. 1 The aggressive neuroendocrine small cell lung cancer (SCLC) subtype comprises 10–15% of lung cancer cases, and about 70% of SCLC patients are first diagnosed exhibiting extensive disease.2–4 Extensive-stage SCLC (ES-SCLC) is associated with an extremely poor 7% 5-year survival rate. 5 Platinum-based chemotherapy has long been a standard first-line approach to treating ES-SCLC, but the median overall survival (OS) provided by this approach is poor at just 9–10 months.6–8
Immune checkpoint inhibitors (ICIs) have recently been established as first-line treatments for many cancers such as non-SCLC, esophageal cancer, and pleural mesothelioma.9–13 In 2018, the IMpower133 trial demonstrated for the first time that atezolizumab plus chemotherapy can provide significant efficacy in patients with ES-SCLC [median OS, 12.3; hazard ratio (HR) = 0.70; 95% confidence interval (CI): 0.54–0.91; p = 0.007; median progression-free survival (PFS), 5.2 months; HR = 0.77; 95% CI: 0.62–0.96; p = 0.020]. 14 The subsequent CASPIAN trial showed consistent results in ES-SCLC treated with durvalumab plus chemotherapy (median OS, 13.0 months; HR = 0.73; 95% CI: 0.59–0.91; p = 0.0047; median PFS, 5.1 months; HR = 0.78; 95% CI: 0.65–0.94). 15 Based on these trial results, the combination of these programed cell death ligand 1 (PD-L1) inhibitors has received worldwide approval as a first-line treatment option for ES-SCLC. 8 However, distinct efficacy outcomes were reported in trials focused on the first-line treatment of ES-SCLC with the programed cell death 1 (PD-1) inhibitors pembrolizumab (KEYNOTE-604) and nivolumab (ECOG-ACRIN EA5161) plus chemotherapy, or the use of the cytotoxic T-cell lymphocyte antigen-4 (CTLA-4) inhibitor ipilimumab (CA184-156).16–18
In 2022, adebrelimab and serplulimab were introduced as new ICIs that substantially enrich the available cancer treatment options, exhibiting robust antitumor activity and manageable adverse events (AEs) profile for ES-SCLC in phase III clinical trials (CAPSTONE-1 and ASTRUM-005 trials).19,20 Adebrelimab (SHR-1316) and serplulimab (HLX10) are novel humanized IgG4 monoclonal antibodies directed against PD-L1 and PD-1.19,21 Individually, these trial results are triggering profound changes in treatment decision-making for ES-SCLC, driving clinicians to update corresponding guidelines. However, parallel reports from several randomized controlled trials (RCTs) have left clinicians with questions regarding the comparative efficacy of the different available modern immunotherapy combinations, and there are significant clinical and cost factors that make it difficult to conduct future head-to-head comparisons of these regimens.
Selecting an optimal treatment strategy depends on the clinical presentation and tumor staging in a particular patient, and uncertainties remain with respect to the relative efficacy of specific treatments. Network meta-analyses provide a mechanism through which available evidence can be summarized while integrating direct or indirect comparisons of the efficacy of a range of treatment strategies. 22 The expanding landscape of ES-SCLC treatment options represents a major breakthrough in the appropriate management of this deadly disease, but only with further analyses will optimal treatment selection be possible. Here, we performed a network meta-analysis with the goal of assessing the relative efficacy results [OS, PFS, and objective response rate (ORR)] and safety [treatment- and immune-related AEs (TRAEs and irAEs)] from RCTs studying first-line immunochemotherapy regimens for ES-SCLC between 2000 and 2022.
Method and materials
Study selection
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement was used to prepare the present meta-analysis (Supplemental Table S1). 23 The PubMed, Embase, Cochrane, and Web of Science databases were systematically searched for all articles published between 1 January 2000 and 27 November 2022 using the following search terms: ‘extensive small-cell lung cancer, immunotherapy, PD-1, PD-L1, chemotherapy, and randomized clinical trial’ (Supplemental Table S2). To be eligible for inclusion, studies had to meet the following criteria: (1) patients diagnosed with ES-SCLC; (2) studies compared ICIs plus chemotherapy to chemotherapy alone as first-line therapy; (3) studies were RCTs; (4) the primary endpoint was OS and PFS; (5) studies were published in English. When multiple articles were derived from the same study, we selected the latest article or the study with the most comprehensive data. Studies not matching the inclusion criteria were excluded. American Society of Clinical Oncology and European Society of Medical Oncology (ESMO) conference abstracts were also searched for additional relevant studies. Two researchers (YWZ and KL) independently evaluated eligibility for study inclusion based on established inclusion and exclusion criteria. Discrepancies were addressed via discussion with a third investigator (HZ).
Data and statistical analysis
Based on PICO (patients, interventions, comparisons, and outcomes), data extracted from the final RCTs were included and a table compiling study-specific information was generated. Risk of bias for individual RCTs was assessed with the Cochrane Collaboration tool. 24 A network meta-analysis used frequency and fixed-effect multivariable meta-regression models to compare relative efficacy [HRs for OS and PFS, odds ratios (ORs) for ORR with corresponding 95% CIs] and safety [risk ratios (RRs) for all-grade and grade 3 or higher TRAEs, all-grade and grade 3 or higher irAEs, AEs resulting in treatment discontinuation, and treatment-related death with corresponding 95% CIs] outcomes associated with different treatment regimens in patients with ES-SCLC.25,26 The primary efficacy analyses were focused on comparing serplulimab plus chemotherapy with all other treatments and comparing all treatments with chemotherapy alone. Safety analyses primarily focused on comparing all treatments versus chemotherapy and further analyzed the incidence of the most common grade 3 or higher irAEs caused by different combination treatments. The probability of a treatment strategy becoming the best treatment and exhibiting the best safety profile was assessed by using p score values to rank treatments. In addition, we first performed multivariable meta-regression analyses with patient characteristics, and then analyses of OS outcomes for particular subgroups of patients (brain metastases status, smoking status, and PD-L1 expression). Results were synthesized with forest plots. Heterogeneity refers to the differences between studies that are performed with I2 values (If I² < 50%, no heterogeneity is considered to exist; if the value of I² > 50%, there is a certain heterogeneity). 27 Funnel plots, Egger et al.’s test, and the Begg test were used to analyze the potential for publication bias. 28 Symmetrical graphs or p values > 0.05 were indicative of the absence of publication bias. 28 Analyses were performed using R (version 4.2.2, http://www.r-project.org) with the ‘meta and netmeta’ package, STATA (version 17.0, https://www.stata.com/) with the ‘metan’ package, RevMan (version 5.4.1, https://training.cochrane.org/online-learning/core-software/revman), and Graphpad Prism (version 9.4.1, https://www.graphpad.com/).
Results
Screening results and study characteristics
A preliminary literature search identified 712 articles, with 507 remaining following duplicate removal. An additional 105 articles were omitted upon title and abstract review, and 7 articles were included in the final network meta-analysis, with these studies being focused on the CA184-156 (phase III), 18 ECOG-ACRIN EA5161 (phase II), 17 KEYNOTE-604 (Phase III), 16 IMpower133 (phase III),14,29 CASPIAN (phase III),30,31 CAPSTONE-1 (phase III), 19 and ASTRUM-005 20 trials, respectively (Figure 1). A total of 3822 patients were involved, with these ES-SCLC patients having undergone first-line treatment with ipilimumab plus chemotherapy (n = 478), nivolumab plus chemotherapy (n = 80), pembrolizumab plus chemotherapy (n = 228), atezolizumab plus chemotherapy (n = 201), durvalumab plus chemotherapy (n = 268), durvalumab and tremelimumab plus chemotherapy (n = 268), adebrelimab plus chemotherapy (n = 230), Serplulimab plus chemotherapy (n = 389), and chemotherapy (n = 1680; Supplemental Table S3). The network plot is shown in Supplemental Figure S1. When we pooled the baseline characteristics of patients from each study, we found these populations to be largely similar except for the CAPSTONE-1 and ASTRUM-005 trials, which were predominantly focused on Asian populations, unlike other studies. There were no significant differences in other patient characteristics, indicating that these seven studies are comparable (Table S4 in Supplemental Materials). The CASPIAN study was open-label and therefore subject to a high risk of bias, while most of the other studies exhibited a low bias risk (Supplemental Figure S2). The research protocol was registered in PROSPERO, an international prospective register of systematic reviews (registration code CRD42022383254; https://www.crd.york.ac.uk/prospero/#searchadvanced).
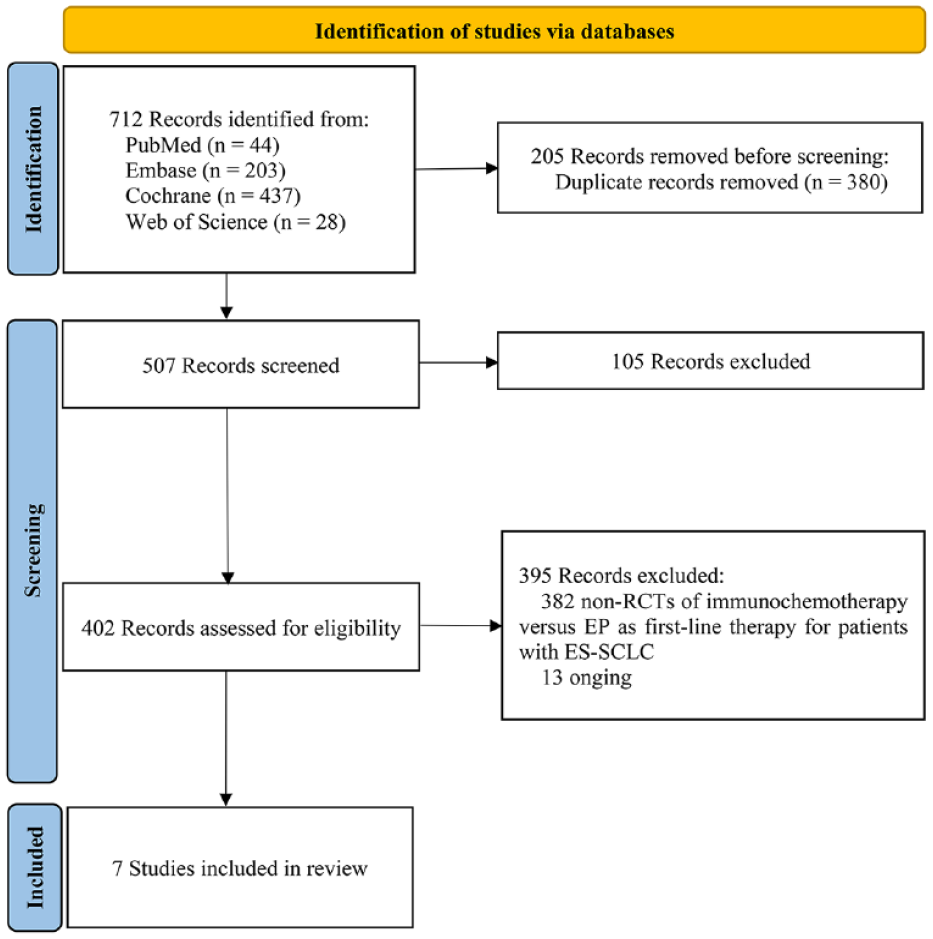
Study selection process.
Efficacy
Overall survival
OS was reported in seven studies analyzing eight treatment strategies (I2 = 29.9%; 95% CI: 0–68.3%). With respect to the HRs for OS, serplulimab plus chemotherapy lowered the risk of death by 37% and 33%, relative to chemotherapy (HR = 0.63; 95% CI: 0.49–0.82) and ipilimumab plus chemotherapy (HR = 0.67; 95% CI: 0.50–0.90), respectively. Serplulimab plus chemotherapy did not exhibit superiority to durvalumab and tremelimumab plus chemotherapy (HR = 0.78; 95% CI: 0.57–1.07), pembrolizumab plus chemotherapy (HR = 0.81; 0.58–1.13), atezolizumab plus chemotherapy (HR = 0.83; 95% CI: 0.59–1.17), nivolumab plus chemotherapy (HR = 0.86; 95% CI: 0.53–1.40), adebrelimab plus chemotherapy (HR = 0.88; 95% CI: 0.62–1.23), or durvalumab plus chemotherapy (HR = 0.89; 95% CI: 0.65–1.12; Figure 2 and Table 1).

Forest plot for hazard ratio of survival effect.
Network meta-analyses for hazard ratio of survival.
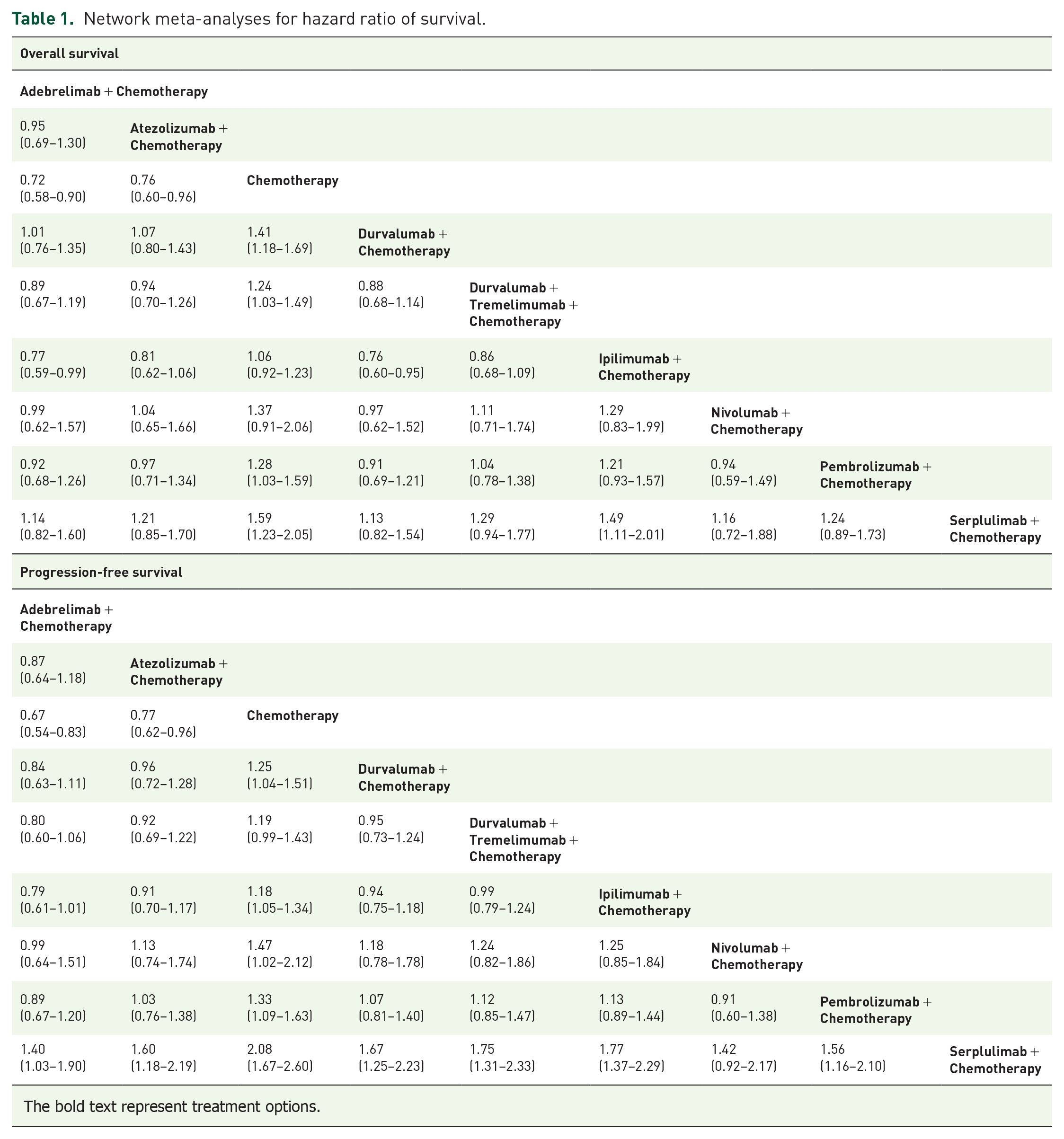
The bold text represent treatment options.
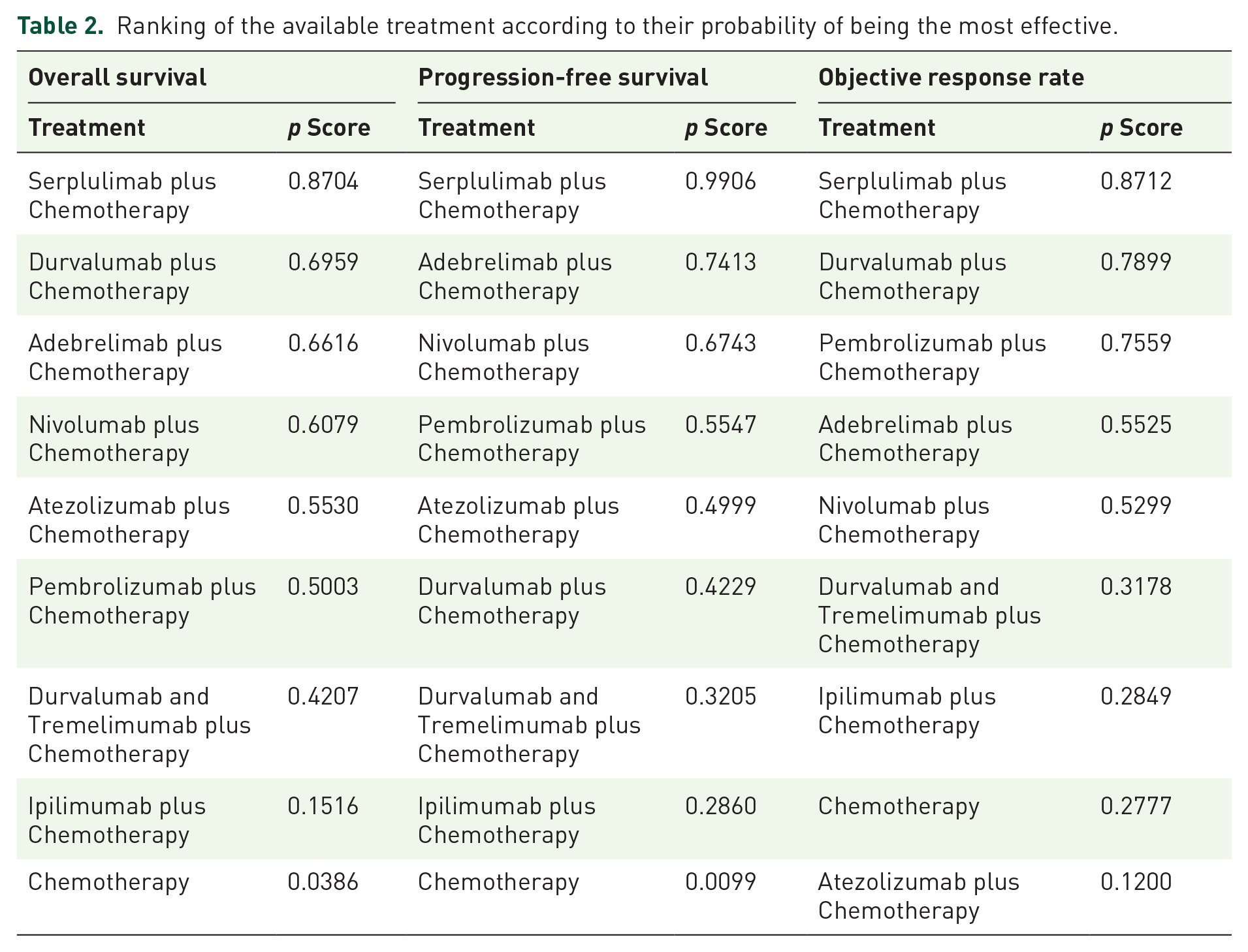
Most treatments exhibited significant survival advantages over chemotherapy alone, with this benefit being greatest for serplulimab plus chemotherapy (HR = 0.63; 95% CI: 0.49–0.82; p score = 0.87), followed by durvalumab plus chemotherapy (HR = 0.71; 95% CI: 0.59–0.85; p score = 0.70), adebrelimab plus chemotherapy (HR = 0.72; 95% CI: 0.58–0.90; p score = 0.66), nivolumab plus chemotherapy (HR = 0.73; 95% CI: 0.49–1.10; p score = 0.61), atezolizumab plus chemotherapy (HR = 0.76; 95% CI: 0.60–0.96; p score = 0.55), pembrolizumab plus chemotherapy (HR = 0.78; 95% CI: 0.63–0.97; p score = 0.50), durvalumab and tremelimumab plus chemotherapy (HR = 0.81; 95% CI: 0.67–0.98; p score = 0.42), ipilimumab plus chemotherapy (HR = 0.94; 95% CI: 0.81–1.09; p score = 0.15) (Supplemental Figure S3A and Table 1). The p score values indicated that the best OS results were achieved with treatments (Table 2).
Ranking of the available treatment according to their probability of being the most effective.
Progression-free survival
PFS was reported in seven studies analyzing eight treatment strategies (I2 = 76.5%; 95% CI: 44.5–86.7%). Serplulimab plus chemotherapy reduced the risk of progression by 52%, 43%, 43%, 40%, 38%, 36%, and 28%, compared with chemotherapy (HR = 0.48; 95% CI: 0.39–0.60), ipilimumab plus chemotherapy (HR = 0.57; 95% CI: 0.43–0.76), durvalumab and tremelimumab plus chemotherapy (HR = 0.57; 95% CI: 0.44–0.73), durvalumab plus chemotherapy (HR = 0.60; 95% CI: 0.45–0.80), atezolizumab plus chemotherapy (HR = 0.62; 95% CI: 0.46–0.85), pembrolizumab plus chemotherapy (HR = 0.64; 95% CI: 0.48–0.86), and adebrelimab plus chemotherapy (HR = 0.72; 95% CI: 0.53–0.97), respectively. Serplulimab plus chemotherapy was not significantly superior to nivolumab plus chemotherapy (HR = 0.71; 95% CI: 0.46–1.08; Figure 2 and Table 1).
Most treatments exhibited significant survival advantages over chemotherapy alone, with serplulimab plus chemotherapy reducing this risk by the greatest amount (HR = 0.48; 95% CI: 0.39–0.60; p score = 0.99), followed by adebrelimab plus chemotherapy (HR = 0.67; 95% CI: 0.54–0.83; p score = 0.74), nivolumab plus chemotherapy (HR = 0.68; 95% CI: 0.47–0.98; p score = 0.67), pembrolizumab plus chemotherapy (HR = 0.75; 95% CI: 0.61–0.92; p score = 0.56), atezolizumab plus chemotherapy (HR = 0.77; 95% CI: 0.62–0.96; p score = 0.50), durvalumab plus chemotherapy (HR = 0.80; 95% CI: 0.66–0.97; p score = 0.42), durvalumab and tremelimumab plus chemotherapy (HR = 0.84; 95% CI: 0.70–1.01; p score = 0.32), and ipilimumab plus chemotherapy (HR = 0.68, 95% CI: 0.47–0.98; p score = 0.29) (Supplemental Figure S3B and Table 1). The p score values for PFS are shown in Table 2.
Objective response rate
ORR was reported in seven studies analyzing eight treatment strategies (I2 = 31.5%; 95% CI: 0–68.9%). Most treatments exhibited significant survival advantages over chemotherapy alone, with serplulimab plus chemotherapy reducing this risk by the greatest amount (OR = 0.48; 95% CI: 0.39–0.60; p score = 0.99), followed by adebrelimab plus chemotherapy (OR = 0.67; 95% CI: 0.54–0.83; p score = 0.74), nivolumab plus chemotherapy (OR = 0.68; 95% CI: 0.47–0.98; p score = 0.67), pembrolizumab plus chemotherapy (OR = 0.75; 95% CI: 0.61–0.92; p score = 0.56), atezolizumab plus chemotherapy (OR = 0.77; 95% CI: 0.62–0.96; p score = 0.50), durvalumab plus chemotherapy (OR = 0.80; 95% CI: 0.66–0.97; p score = 0.42), durvalumab and tremelimumab plus chemotherapy (OR = 0.84; 95% CI: 0.70–1.01; p score = 0.32), and ipilimumab plus chemotherapy (OR = 0.68, 95% CI: 0.47–0.98; p score = 0.29) (Supplemental Table S5, Figure S4, and Table 1). The p score values for PFS are shown in Table 2.
Subgroup analysis
Multivariable meta-regression analysis was performed on the characteristics of the patients in the included studies, and it was found that the aggregated p value was <0.05 (Supplemental Table S7), which suggested that it was necessary to conduct further subgroup analysis. Subgroup analyses were conducted to examine differences in OS according to brain metastasis status [six studies including seven different interventions (brain metastases: I2 = 27.0%; 95% CI: 0–70.7% and non-brain metastases: I2 = 60.6%; 95% CI: 0–85.9%)], smoking history [five studies including six different interventions (smoker: I2 = 53.2%; 95% CI: 0–79.3% and non-smoker: I2 = 29.9%; 95% CI: 0–71.6%)], and PD-L1 expression level [four studies including four different interventions (PD-L1 expression <1%: I2 = 0%; 95% CI: 0–67.9% and PD-L1 expression >1%: I2 = 0%; 95% CI: 0–67.9%)]. Serplulimab plus chemotherapy did not significantly lower mortality risk relative to other study arms in non-smokers or patients with PD-L1 expression levels <1% (Supplemental Figure S5). The p score values indicated that serplulimab plus chemotherapy was associated with the best OS outcomes in patients with brain metastases (p score = 0.88) and smokers (p score = 0.86). Additionally, durvalumab and tremelimumab plus chemotherapy, adebrelimab plus chemotherapy, and atezolizumab plus chemotherapy reduced the risk of death in non-smokers (p score = 0.83), individuals exhibiting PD-L1 expression ⩾1% (p score = 0.69), and individuals exhibiting PD-L1 expression < 1% (p score = 0.84), respectively (Supplemental Table S6).
Safety
Safety was assessed based on the TRAE and irAE incidence rates, with these AEs being classified based on the Medical Dictionary of Regulatory Activity. The least safe treatment strategies were ranked based on p score.
Treatment-related AEs
TRAEs were reported in seven studies including eight different treatment strategies and 3797 patients (All-grade TRAEs: I2 = 47.0%; 95% CI: 0–75.9% and grade 3 or higher TRAEs: I2 = 13.5%; 95% CI: 0–61.9%). Serplulimab plus chemotherapy (RR = 1.25; 95% CI: 1.08–1.43; p score = 0.99) and nivolumab plus chemotherapy (RR = 1.24; 95% CI: 1.01–1.55; p score = 0.89) were the only alternatives exhibiting an elevated likelihood of all-grade and grade 3 or higher TRAEs relative to chemotherapy, whereas these differences were not significant for other treatments [Figure 3A, Tables 3(a) and 4, Supplemental Figure S6A, and Supplemental Table S8].

Forest plot for risk ratio of safety.
Network meta-analyses for risk ratio of grade 3 or higher adverse events.

Ranking of the available treatments according to their probability of being the worst safe.

irAEs, immune-related adverse events; TRAEs, treatment-related adverse events.
Immune-related AEs
Rates of irAEs were reported in six studies including seven different treatment strategies and 3637 patients (All-grade irAEs: I2 = 46.1%; 95% CI: 0–75.6% and grade 3 or higher irAEs: I2 = 20.0%; 95% CI: 0–66.3%). Durvalumab and tremelimumab plus chemotherapy were associated with the highest risk of all-grade irAEs (RR = 13.71; 95% CI: 6.49–28.98; p score = 0.98) and grade 3 or higher irAEs (RR = 36.00; 95% CI: 4.97–260.67; p score = 0.93), while adebrelimab plus chemotherapy and atezolizumab plus chemotherapy were associated with a lower risk for all-grade irAEs (RR = 1.61; 95% CI: 1.14–2.29; p score = 0.27) and grade 3 or higher irAEs (RR = 4.21; 95% CI: 1.44–12.28; p score = 0.49), respectively [Figure 3B, Tables 3(a) and 4, Supplemental Figure S6B, and Supplemental Table S9].
We further assessed particular types of irAEs from seven trials. To focus on AEs with the greatest clinical relevance, which primarily focused on those accounting for >1% of all-grade irAEs and >0.5% of grade 3 or higher irAEs. The most prevalent all-grade irAEs was hypothyroidism [(11.11%; 95% CI: 8.62–13.60%); (10.10%; 95% CI: 7.86–12.34%); (9.02%; 95% CI: 5.58–12.47%)] in PD-1, PD-L1, and PD-L1 with CTLA-4 plus chemotherapy groups, respectively; and diarrhea/colitis (31.80%; 95% CI: 27.62–35.97%) in CTLA-4 plus chemotherapy groups. The most common grade 3 or higher irAEs were hyperglycemia (0.65%; 95% CI: 0.02–1.29%), pneumonitis (1.01%; 95% CI: 0.27–1.75%), diarrhea/colitis (8.99%; 95% CI: 6.722–11.25%), diarrhea/colitis (3.38%; 95% CI: 1.21–5.56%) in PD-1, PD-L1, CTLA-4, and PD-L1 with CTLA-4 plus chemotherapy groups, respectively (Supplemental Figure S7).
Treatment-related AEs leading to discontinuation and death
The incidence of TRAEs leading to discontinuation (I2 = 36.2%; 95% CI: 0–70.7%) and treatment-related death (I2 = 0%; 95% CI: 0–56.3%) was reported in seven studies including eight different treatment strategies and 3797 patients. Ipilimumab plus chemotherapy (RR = 9.52; 95% CI: 4.85–18.69; p score = 0.99) and durvalumab with tremelimumab plus chemotherapy (RR = 6.00, 95% CI: 1.36–26.55; p score = 0.86) were associated with the highest odds of TRAEs resulting in the discontinuation of treatment and death relative to chemotherapy, respectively, followed by durvalumab with tremelimumab plus chemotherapy (RR = 3.54, 95% CI: 1.96–6.39; p score = 0.79) and atezolizumab plus chemotherapy (RR = 2.97, 95% CI: 0.12–72.46; p score = 0.61). Other treatments were not associated with an elevated risk of such TRAEs resulting in treatment discontinuation or death (Figure 3C and D and Table 4). Joint analyses of the relative efficacy and safety of these treatments revealed that serplulimab plus chemotherapy and adebrelimab plus chemotherapy were the most suitable treatment regimens analyzed in this study (Figure 4 and Supplemental Figure S8).

Joint evaluation of efficacy and safety of the different therapeutic strategies for ES-SCLC.
Publication bias
The funnel plot (symmetric), Egger’s (p = 0.208), and Begg’s (p = 0.174) tests did not exhibit any evidence of significant publication bias pertaining to these studies of first-line treatments (Supplemental Figure S9).
Discussion
Treating SCLC, with its high metastatic potential and poor prognosis, has long remained a difficult but persistent clinical challenge. The emergence of ICIs has gradually expanded the treatment options for various malignancies, and these novel immunotherapeutic regimens are increasingly playing a greater role in the treatment of ES-SCLC. Patients that once faced a poor prognosis of fewer than 8 months can now achieve a median OS number of more than 15 months.19,20,32 First-line treatment decisions carry significant weight in determining each patient’s oncology care plan. The high rates of second-line treatment utilization by patients underscore the need for efficient first-line options to maximize long-term survival. On the other hand, AEs data underscore that each treatment is absolutely important for improving patient outcomes and quality of life (QoL). This is the first systematic review and network meta-analysis to our knowledge to include the most comprehensive data pertaining to ES-SCLC patients undergoing first-line immuno-chemotherapy treatment.
In this network meta-analysis, which incorporated seven high-quality RCTs focused on the first-line immuno-chemotherapeutic treatment of ES-SCLC, we found that serplulimab plus chemotherapy is the most efficacious treatment strategy for lowering the odds of death, followed by durvalumab plus chemotherapy and adebrelimab plus chemotherapy. Serplulimab plus chemotherapy was also the most effective treatment strategy for lowering the odds of progression, followed by adebrelimab plus chemotherapy and nivolumab plus chemotherapy. ORR comparisons revealed that serplulimab plus chemotherapy, durvalumab plus chemotherapy, and pembrolizumab plus chemotherapy offer the highest probability of achieving a radiological response. Therefore, serplulimab plus chemotherapy and adebrelimab plus chemotherapy exhibit better survival efficiency. However, the addition of PD-1 inhibitors with serplulimab or pembrolizumab resulted in a statistically non-significant positive effect on OS compared to PD-L1 inhibitors or the dual inhibition of PD-L1 and CTLA-4, which may be explained by the wide variation in patient outcomes included in the study
Atezolizumab or durvalumab plus chemotherapy have shown improvements in OS, PFS, ORR, and QoL compared to chemotherapy and have rapidly replaced the use of general-purpose first-line chemotherapy.8,14,15,29,31 However, serplulimab and adebrelimab produced by Chinese institutions have further benefited the median OS and addressed the gap in available clinical PD-1 inhibitors available for first-line ES-SCLC patient treatment. 20 Evidence pertaining to superiority that is not significant when comparing checkpoint blockade strategies is of value in settings when head-to-head RCT comparisons do not exist. Although immunotherapeutic regimens have resulted in improved survival in patients with ES-SCLC, only a portion of patients actually benefit from immunotherapy. The main reason for this outcome is that most SCLC tumors are of the immune desert type, with low levels of infiltrating CD8+ T cells in the tumor microenvironment and high levels of FOXP3+ Tregs and myeloid suppressor cells, together with low levels of MHC-I expression that result in limited ICI treatment efficacy. 33 In addition, studies have also found that SCLC is a highly heterogeneous disease and can be separated into different molecular subtypes based on differences in transcription factor expression, with particular subtypes exhibiting different immune microenvironmental characteristics. 34 Of these, the Y subtype or inflammatory SCLC does not express the three transcription factors A/N/P and is more sensitive to immunotherapy. 34 The subgroups of patients who are most likely to benefit from immunotherapy warrant further exploration.
There is ongoing debate as to whether tobacco exposure, newly diagnosed brain metastases, and PD-L1 expression levels affect responses to ICIs treatment in ES-SCLC.35–37 When patients were stratified according to smoking status, brain metastases status, and PD-L1 expression levels, serplulimab plus chemotherapy exhibited statistically significant efficacy improvements in smokers, individuals without brain metastases, and individuals with PD-L1 expression levels ⩾1% relative to chemotherapy. When ranking interventions based on p scores, serplulimab plus chemotherapy ranked first with respect to the OS of patients with brain metastases, without brain metastases, and smokers. However, durvalumab with tremelimumab plus chemotherapy, adebrelimab plus chemotherapy, and atezolizumab plus chemotherapy were respectively ranked first with respect to the OS of smokers, patients with PD-L1 expression ⩾1%, and patients with PD-L1 expression <1%, respectively. Unlike previous meta-analyses, we have highlighted for the first time the relative survival benefits of specific treatment strategies in specific subgroups of ES-SCLC patients defined based on disease etiology, metastasis, and predictive biomarkers.38,39 When determining whether an innovative drug or treatment strategy can be widely used in the clinic, it is critical to ensure that these treatment options are safe and effective in patients with specific characteristics.
In addition to efficacy considerations, AEs are a key selection factor for the first-line immuno-chemotherapeutic treatment of ES-SCLC, as they can have a pronounced impact on patient prognosis and QoL. In our comparative evaluation of the clinical utility of different treatment regimens, we evaluated the probability of TRAEs, irAEs, permanent discontinuation due to TRAEs, and treatment-related deaths for each therapy. The combination of PD-1 inhibitors with nivolumab plus chemotherapy was associated with a higher risk of TRAEs. Since the ECOG-ACRIN EA5161 study did not report a detailed table of AEs, we found that the most common TRAEs for this combination strategy were gastrointestinal reactions or hematologic events, which were most likely chemotherapy-driven, and cisplatin was more toxic than carboplatin about previously published studies.11,40–42 Therefore, our analysis provides a valuable reference regarding the relative benefits and limitations of these different treatment options, including chemotherapy. In addition, we need to know whether treatment with PD-1 inhibition may be generally well tolerated among older patients and those with liver dysfunction.43,44
The dual inhibition of PD-L1 and CTLA-4 with durvalumab and tremelimumab plus chemotherapy is associated with elevated irAE risk, potentially owing to Treg-related mechanisms. As Tregs are among the most common immunosuppressive cells within the tumor microenvironment, they serve as key regulators of immunological homeostasis and protect against autoimmune pathologies. These Tregs also express high immune checkpoint levels including PD-1 and CTLA-4, and ICIs targeting these checkpoints may thus cause irAEs. 45 Increased toxicity rates are not the result of a single event type, and are instead linked to AEs across many classes of organs. These irAEs exhibit particular clinical significance. In this meta-analysis, the most commonly observed grade 3 or higher irAEs were hyperglycemia, pneumonitis, diarrhea/colitis, and diarrhea/colitis in the PD-1, PD-L1, CTLA-4, and PD-L1 with CTLA-4 plus chemotherapy groups, respectively. Close monitoring and early detection of the signs and symptoms related to these AEs may help clinicians manage them appropriately. If not caught early, these autoimmune diseases often manifest in more serious and potentially fatal forms. Therefore, clinical vigilance is essential to ensure the early identification and treatment or prevention of irAEs. Dual ICI treatment regimens are associated with a higher risk of TRAEs resulting in discontinuation or death. The increased toxicity associated with the addition of tremelimumab may be the result of fewer patients achieving the target exposure for all utilized clinical agents and associated tolerability of a sufficient treatment dose. Our results help to highlight the additive and non-overlapping toxicities associated with particular treatment options and may therefore help to prevent the high rates of high-toxicity events seen in other tumor types while suggesting appropriate high-quality dose-matching strategies for oncologists and patients.
Overall, this multiple-comparative study found that serplulimab plus chemotherapy was associated with the best safety and controllable safety as well as the highest incidence of TRAEs for hematologic and irAEs for endocrine disorders. Unlike previous meta-analyses that studied the treatment of patients with ES-SCLC, our network meta-analysis compared a broader range of first-line immune-chemotherapy treatments in this population, including serplulimab, adebrelimab, durvalumab, atezolizumab, pembrolizumab, nivolumab, and ipilimumab. In addition, we summarized the incidence and profile of AEs most relevant to ICIs.38,46,47 Therefore, our study can help clinicians make better decisions regarding the optimal use of a variety of promising treatment options by fully considering the associated clinical benefits and toxicity characteristics of these regimens in patients with ES-SCLC.
Our network meta-analysis of landmark clinical trials for ES-SCLC is subject to certain limitations. First, because this study entailed separate indirect comparisons of RCTs, inconsistencies between the studies may have affected the results, with subtle differences in the characteristics of the subjects included in the study (i.e. ECOG-ACRIN EA5161 did not detail the baseline characteristics of the patients; the ASTRUM-005 and CAPSTONE-1 studies were dominated by Asians; some studies were not stratified according to liver metastasis status or PD-L1 expression levels, etc.). However, the remaining relevant factors (interventions, outcome measures, and methods) of the included studies were similar and no significant heterogeneity was found in these analyses. Therefore, the included studies are comparable. Second, OS benefits may be influenced by follow-up time, cancer-independent mortality, and exposure to subsequent treatment. However, PFS depends on the subjective assessment of responses, the timing of radiological assessment, and the different mechanisms of action of each investigational agent. As the included studies are subject to some subtle differences in these respects, the results need to be interpreted with caution. However, in the field of network meta-analysis, OS and PFS remain the most objective endpoints used for indirect comparisons. Third, the ECOG-ACRIN EA5161 trial was the only included phase II study, with a limited sample size and no detailed tabular report. A phase III RCT with a larger sample size is thus needed. Therefore, interpretations of the efficacy and safety of nivolumab should be made with caution.
Conclusion
This network meta-analysis offers robust and reproducible evidence supporting the efficacy and safety of immuno-chemotherapies as first-line treatment for ES-SCLC patients. This study demonstrated that serplulimab plus chemotherapy ranked first, exhibiting better OS, PFS, and ORR outcomes with manageable AEs such that it may be the best treatment option for ES-SCLC. In addition, there were no significant differences in AEs between adebrezumab plus chemotherapy and other drugs, and it produced a relatively high survival ranking. Thus, adebrezumab plus chemotherapy is an effective treatment for ES-SCLC. There were no significant differences in treatment outcomes among subgroups. However, subgroup rankings did indicate that serplulimab plus chemotherapy had the highest efficacy in patients with brain metastases and smokers, while adebrelimab plus chemotherapy and atezolizumab plus chemotherapy were more effective for individuals with PD-L1 expression ⩾1% and <1%, respectively. These important findings provide a reference for clinicians and may guide clinical practice.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231206147 – Supplemental material for Comparative efficacy and safety of novel immuno-chemotherapy for extensive-stage small-cell lung cancer: a network meta-analysis of randomized controlled trial
Supplemental material, sj-docx-1-tam-10.1177_17588359231206147 for Comparative efficacy and safety of novel immuno-chemotherapy for extensive-stage small-cell lung cancer: a network meta-analysis of randomized controlled trial by Youwen Zhu, Kun Liu, Hong Zhu, Hui Cao and Yangying Zhou in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359231206147 – Supplemental material for Comparative efficacy and safety of novel immuno-chemotherapy for extensive-stage small-cell lung cancer: a network meta-analysis of randomized controlled trial
Supplemental material, sj-docx-2-tam-10.1177_17588359231206147 for Comparative efficacy and safety of novel immuno-chemotherapy for extensive-stage small-cell lung cancer: a network meta-analysis of randomized controlled trial by Youwen Zhu, Kun Liu, Hong Zhu, Hui Cao and Yangying Zhou in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
