Abstract
Background:
Emerging evidence indicates the importance of tertiary lymphoid structures (TLSs) in predicting the outcomes of nonsmall cell lung cancer (NSCLC) patients; however, their prognostic value and correlations with peripheral inflammatory prognostic indices in stage I patients have been less well studied.
Methods:
Stage I NSCLC patients were recruited retrospectively; the presence and location of TLSs (peritumoral [pTLSs] and intratumoral [iTLSs]) were determined via hematoxylin and eosin (H&E)-stained slides. Peripheral inflammatory indices, including the neutrophil-to-lymphocyte ratio (NLR), lymphocyte-to-monocyte ratio (LMR), prognostic nutritional index (PNI), and advanced lung cancer inflammation index (ALI), were obtained and compared among these subgroups. Disease-free survival (DFS) and overall survival (OS) were tested via Kaplan-Meier analysis, and risk factors for survival were determined via a Cox proportional hazards model.
Results:
A total of 24.73% and 92.73% of patients were positive for pTLSs and iTLSs, respectively. The absolute number of iTLSs was significantly greater than that of pTLSs (P < .001). Low preoperative LMR and ALI were detected only in patients with pTLSs but not in those without. Only pTLS was found to be a risk factor for both DFS and OS, and it was independently associated with OS (HR = 3.93, 95% confidence interval [CI] = 1.16-13.37; P = .028). Accordingly, patients with pTLSs had relatively poor DFS (log rank = 5.46, P = .019) and OS (log rank = 10.48, P = .001) rates.
Conclusions:
Among the heterogeneous results concerning the prognostic value of pTLSs and iTLSs in stage I NSCLC, our results for the first time indicated that the presence of pTLSs may predict poor outcomes in these patients and no correlation of iTLSs with the outcomes was validated; however, additional studies with large sample size are needed in future.
Keywords
Introduction
Lung cancer is still the leading cause of cancer-related death in China, with a mortality rate as high as 54.57/100 000 in men and women, 1 and nonsmall cell lung cancer (NSCLC) is still the main pattern of this disease. 2 To date, early diagnosis and radical resection are still the most important approaches for improving long-term survival in victims. Luckily, an obvious shift toward stage I along with improved survival has been observed in recent years due to the increasing popularity of low-dose computed tomography screening. 3 However, notably, the 5-year overall survival (OS) rate is 68% to 92% for stage I cases 4 and the benefit of adjuvant treatments for these patients is still controversial.5-10 Potential risk factors associated with long-term outcomes are still highly needed.
Previously, many studies have indicated that additional prognostic information for NSCLC patients can be obtained from single hematoxylin and eosin (H&E)-stained slides. Examples include tumor-infiltrating lymphocytes, 11 tumor growth patterns, 12 and the tumor-to-stroma ratio.13,14 Tertiary lymphoid structures (TLSs), which involve the ectopic expansion of lymphoid cells in tumors or adjacent normal areas, have been increasingly validated as prognostic factors in various malignancies,15-19 including NSCLC.20,21 However, the prognostic value of these structures in stage I patients is still under debate. For example, Dieu-Nosjean et al enrolled 74 early-stage patients (including 62 stage I patients) and found that although a high density of intratumoral TLSs (iTLSs) (marked by mature dendritic cells [DCs]) was correlated with favorable disease-specific survival (DFS) and OS, it was less likely to have prognostic value in the stage I subgroup. 22 Similarly, Fukuhara et al included 112 stage I patients and reported that TLSs (with no specific description of the TLS locations) were not associated with DFS. 23 In contrast, in a study that included 213 stage I patients, Rakaee et al reported that high peritumoral TLSs (pTLSs) were significantly correlated with superior OS. 20 On the basis of these data, the prognostic value of TLSs in stage I NSCLC patients, particularly the role of their locations, still needs further investigation.
Notably, numerous studies have indicated that a series of inflammatory prognostic indices collected from peripheral blood tests, including the neutrophil-to-lymphocyte ratio (NLR), 24 lymphocyte-to-monocyte ratio (LMR), 25 prognostic nutritional index (PNI), 26 and advanced lung cancer inflammation index (ALI), 27 also play important roles in predicting outcomes in patients with NSCLC. Among these factors, the correlations of the NLR, LMR and ALI with patient outcomes have been further validated in stage I patients.28-30 Interestingly, heterogeneous immune cells, including DCs, 22 B cells, 31 CD4+ T cells, 32 CD8+ T cells, 20 and others, have been identified in TLSs in NSCLC33,34; however, some previous reports have indicated that systemic inflammatory cells are correlated with CD4+ immune cells in the tumor microenvironment (TME) 35 and that the NLR is inversely associated with tumoral and stromal CD8+ lymphocytes. 36 These data support a potential correlation of the aforementioned inflammatory prognostic indices with the presence of TLSs in NSCLC. Coincidentally, a low NLR was found to be associated with high TLSs in a previous study by Fukuhara et al, but they included 23.81% (35/147) patients who exceeded stage I. 23 The correlations of these indices with TLSs in stage I cases are still largely unknown.
On the basis of these data, we aimed to explore the prognostic value of TLSs in different locations by H&E in stage I NSCLC patients and determine their correlations with peripheral blood inflammatory prognostic indices.
Methods
Data collection
Stage I NSCLC patients who underwent thoracoscopic radical resection in Hainan Hospital of Chinese PLA General Hospital from December 2012 to June 2020 were enrolled. Patients were excluded if they met any of the following criteria: (1) were not naïve to neoadjuvant therapies; (2) had detectable remote lesions before surgery; (3) had in situ lesions; (4) did not have adenocarcinoma or squamous carcinoma; or (5) had follow-up problems. Clinical variables, including age, sex, type of resection, pathology, smoking/drinking history, comorbidities (cases with hypertension or type 2 diabetes), and body mass index, were collected from the archived digital records as described previously. 37 In addition, preoperative absolute counts of neutrophils, lymphocytes, monocytes, and albumin were also obtained from the digital system within 1 week before surgery via routine blood tests as described previously. 38 Peripheral inflammatory indices, including the neutrophil-to-lymphocyte ratio (NLR), lymphocyte-to-monocyte ratio (LMR), prognostic nutritional index (PNI), and advanced lung cancer inflammation index (ALI), were calculated according to previous reports.39,40 This study was approved by the ethics committee of Hainan Hospital of Chinese PLA General Hospital (ID: S2023-12), and mandatory written informed consent was not necessary because of its retrospective nature according to national regulations.
Identify TLSs in H&E-stained slides
Slides (4 µm) were cut from the archived formalin-fixed, paraffin-embedded samples and processed via standard histological methods with H&E staining. We partly followed the methods of Rakaee et al 20 to identify TLSs; in brief, first, a representative slide with a maximum tumor-stromal interface was selected; second, areas with diffuse infiltration of lymphocytes and the absence of distinguishable lymphoid follicles or aggregate structures were ruled out; and third, irregularly shaped lymphoid expansion architectures were ignored. The TLSs were located by using a light microscope at a magnification of 10× (Leica DM2500, Leica Corporation) in the whole field, and then a magnification of 200× was used to determine the absolute number of TLSs by a senior pathologist who was blinded to the clinical features. pTLSs were identified from an invasive border without any malignant components, and iTLSs were identified from an invasive border to the tumor center with all malignant components (Figure 1). Inconclusive areas were consulted with another senior pathologist to reach a conclusive result.

A representative image showing the presence of pTLSs and iTLSs. The left panel displays the whole tumor (10×). The right panel shows the enlarged fields of pTLSs (A) and iTLSs (B) (200×). The black dots indicate the invasive border of the tumor.
Study endpoints
Disease-specific survival and OS were set as the endpoints in this study, and the follow-up procedure was conducted as described previously; 37 the latest follow-up was terminated in August 2023.
Statistical tests
All of the data were analyzed via SPSS 20.0 (SPSS Inc., Chicago, IL, USA) and GraphPad Prism 5 (GraphPad Software Inc., San Diego, CA, USA). Patients were divided into pTLS, iTLS-low or -high subgroups on the basis of the median absolute counts in the whole cohort. The differences in the distributions of the clinical variables among these subgroups were tested via Chi-square tests, and the differences in the NLR, LMR, PNI, and ALI among them were determined via independent-samples t tests or Mann-Whitney U tests when a Gaussian distribution was not met. Correlations between pTLSs and iTLSs were determined via the Pearson test. Differences in DFS and OS among these subgroups were analyzed by the Kaplan-Meier method. Risk factors for the outcome were calculated via the Cox proportional hazards model. A 2-sided P value < .05 was considered statistically significant.
Results
Description of patient features
A total of 275 patients were included (Figure 2), with 148 women and 127 men. The median age of the patients was 57 years (years) (range: 23-84 years), and the median follow-up was 51 months (m) (range: 9-121 m). At the end of the follow-up, 4 and 8 patients died in stages IA and IB, respectively. The median number of pTLSs was 0 (range: 0-50), and the median number of iTLSs was 6 (range: 0-46), with 20 patients having 0; thus, 24.73% (68/275) and 92.73% (255/275) of the patients were positive for pTLSs and iTLSs, respectively. The absolute number of iTLSs was significantly greater than that of pTLSs (P < .001). As shown in Table 1, male patients with squamous carcinoma, a smoking history or comorbidities, and IB were more common in the high-pTLS subgroup.
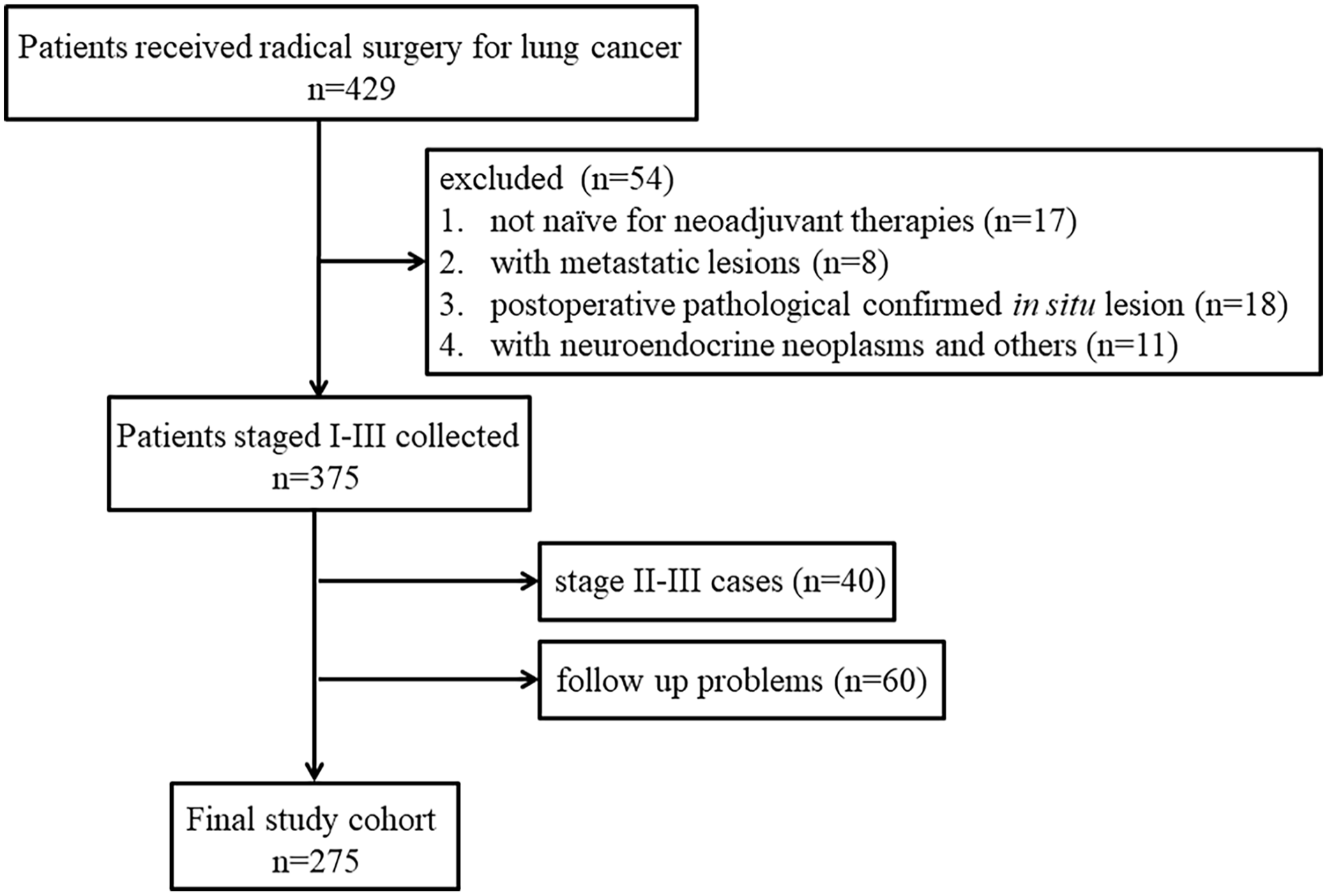
Patient enrollment.
The differences of clinical variables amongst pTILs and iTILs low or high subgroups.

pTILs: peritumoral tertiary lymphoid structures; iTILs: intratumoral tertiary lymphoid structures.
Significant statistical difference.
Correlation between pTLSs and iTLSs
As shown in Figure 3, there was a positive correlation between the densities of the pTLSs and iTLSs (R = 0.12, P = .051), but the correlation was not statistically significant.
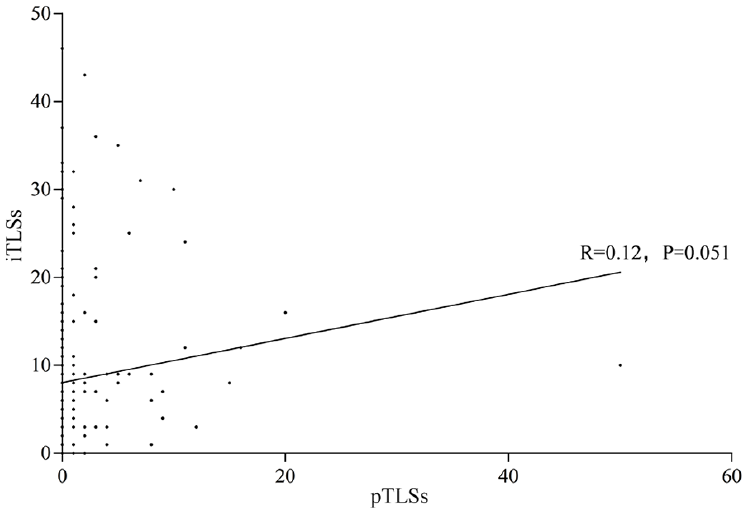
The correlation between pTLSs and iTLSs was checked via the Pearson test.
Distribution of peripheral inflammatory prognostic indices among the absent or present pTLS subgroups
As shown in Figure 4, significant differences in the preoperative LMR and ALI were detected among patients with pTLSs but not among those without. Patients with pTLSs were more likely to have a relatively low LMR and ALI.

Differences in inflammatory prognostic indices, including the NLR (A, E), LMR (B, F), PNI (C, G), and ALI (D, H), between the low- and high-TLS subgroups.
Survival differences among the absent or present pTLS subgroups
As shown in Figure 5, significant survival differences were detected only among the absent or present pTLS subgroups, as patients with pTLSs had significantly worse DFS (log rank = 5.46, P = .019) and OS (log rank = 10.48, P = .001). No such differences were found in the iTLS low- or high-level subgroups.
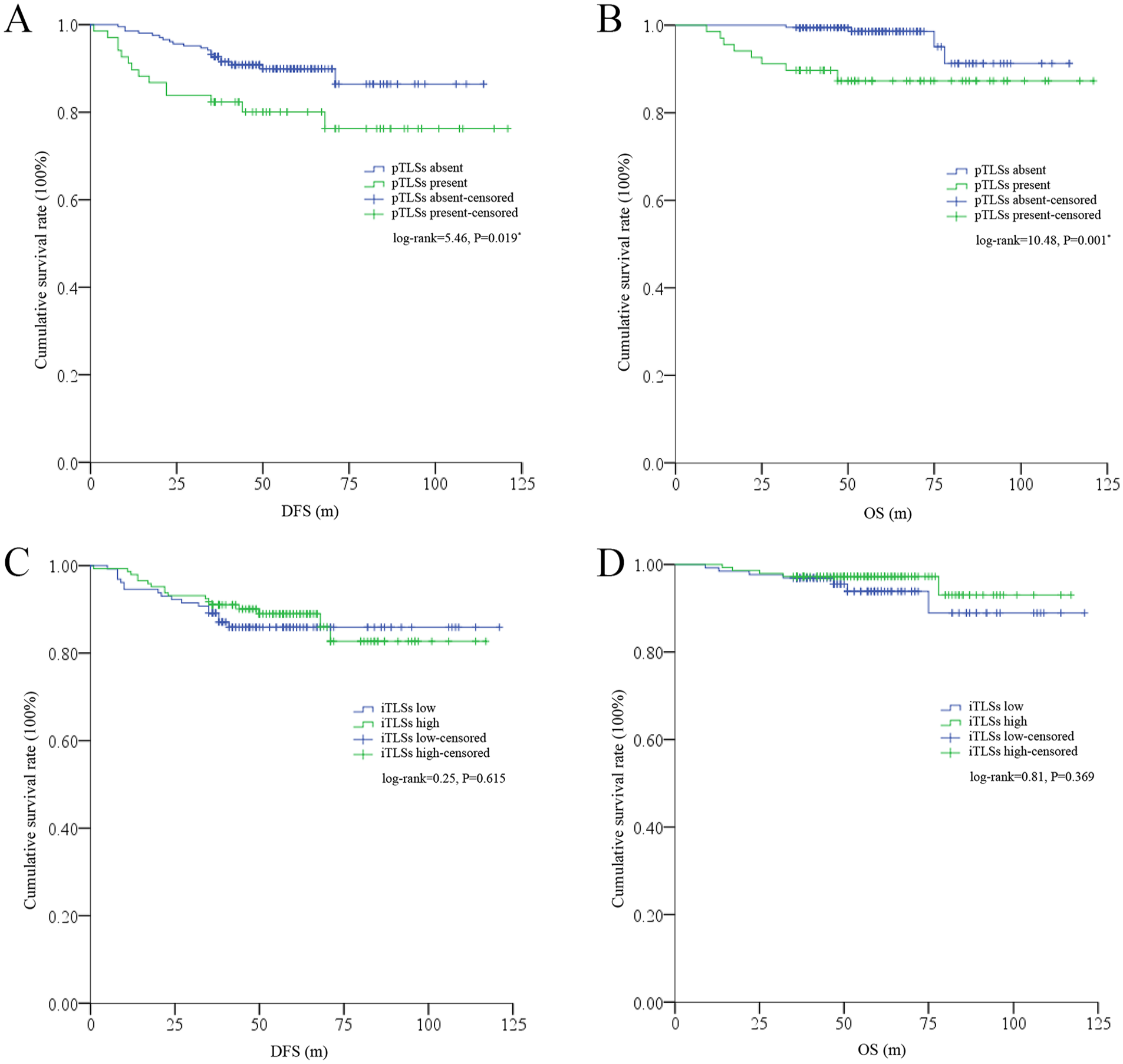
Survival differences between the low- and high-TLS subgroups. (A) DFS differences between the low- and high-pTLS subgroups. (B) OS differences between the low- and high-pTLS subgroups. (C) DFS differences between the low- and high-iTLS subgroups. (D) OS differences between the low- and high-iTLS subgroups.
Determination of risk factors for DFS and OS by univariate and multivariate tests
Univariate tests revealed that TNM stage and pTLSs were common risk factors for both DFS and OS, whereas smoking history was a statistically significant risk factor for DFS and was a definite risk factor for OS (Table 2). Together, these factors were included in multivariate tests, and the results indicated that pTLSs were an independent risk factor only for OS (HR = 3.93, 95% confidence interval [CI] = 1.16-13.37; P = .028) (Table 3).
Determination of the risk factors for DFS and OS by univariate analyses.

Significant statistical difference.
Determination of the risk factors for DFS and OS by multivariate analyses.

with significant statistical difference.
Discussion
In this study, pTLSs, rather than iTLSs, were found to be significantly associated with survival in stage I NSCLC patients and were identified as independent risk factors for OS. Notably, patients with pTLSs had significantly worse DFS and OS than their counterparts did.
Previously, a group of studies indicated the presence or density of TLSs with different locations in NSCLC and explored their value in prognostication; however, the results were highly conflicting. For example, Rakaee et al included 553 stage I-III patients and reported that a high frequency of TLSs (mainly iTLSs according to the description) was associated with improved DFS and OS 20 ; similarly, Fukuhara et al enrolled 147 stage I-IV patients and reported that a high TLS density (also a high iTLS density according to the description) was correlated with better DFS. 23 In line with these findings, Giatromanolaki et al included 103 stage I-IV patients and individually explored the prognostic values of pTLSs and iTLSs; their results indicated that high densities of both pTLSs and iTLSs were linked to better OS. 21 In contrast, Brunet et al included 114 metastatic patients who were enrolled in the BIP study (NCT02534649) and reported that the presence of TLSs (iTLSs + pTLSs) had no value in predicting progression-free survival (PFS). 41 Conventionally, these heterogeneous results may be explained by the fact that these studies included patients with mixed stages; surprisingly, these controversies are still present in the stage I scenario. For example, some reports have suggested that the presence of iTLSs is positively associated with outcomes.20,42 However, the aforementioned studies did not support these results;22,23 furthermore, He et al suggested that these structures were not associated with OS in a large sample study (n = 326). 43 In our study, we found no correlations between iTLSs and DFS or OS, which was partly in line with the findings of previous studies.22,23 In addition, we found that the presence of pTLSs was significantly correlated with worse survival; interestingly, the role of pTLSs in survival has been supported by several other studies. For example, Sofopoulos et al included 112 breast cancer patients and reported that the presence of pTLSs was correlated with poor DFS and OS, 44 whereas Zhang et al enrolled 170 hepatocellular carcinoma patients and suggested that the presence of pTLSs predicts inferior DFS and OS. 45 These reports suggest that the function of pTLSs in cancer may differ from that of iTLSs.
In fact, some important questions remain unanswered regarding the cellular constitutions and functions of TLSs. Previous studies have indicated that the TLSs consists of a group of immune cells, including DCs, 22 B cells, 31 CD4+ T cells, CD8+ T cells,20,32 T regulatory cells (Tregs), 33 mast cells, 46 and macrophages, 47 notably, these cells and their secreted cytokines and chemokines may act as “double-edged swords” in the TME during the development of cancer. Correspondingly, the functions of TLSs may be controlled by the comprehensive coordination of these factors at different locations. In our study, the presence of pTLS was negatively correlated with survival, which can also be partly explained by these cells and cytokines. For example, previous studies have shown that mast cells can induce epithelial-to-mesenchymal transition and migration by regulating the IL-8/Wnt/β-catenin pathway or promoting cell proliferation in NSCLC.48,49 Noticeably, these cells are multifaceted with distinct phenotypes in the tumor compared with adjacent normal areas, 50 and further, although it was not validated in NSCLC, these cells in adjacent normal mucosa but not in the invasive front can promote disease progression. 51 In addition, fibroblasts, which are the main source of several cytokines, such as IL-33, 52 are critically involved in the formation of TLSs. 53 Interestingly, the level of IL-33 is significantly greater in adjacent normal tissues than in tumor areas in lung cancer 54 ; moreover, IL-33 can fuel the growth and metastasis of cancer cells.55,56 In addition, IL-33 has a functionally important role in recruiting neutrophils, which are the main leukocytes in NSCLC. 57 Notably, numerous reports have shown that neutrophils are key players in promoting cancer progression and metastasis.58,59 Moreover, IL-33 can promote IL-10 secretion by macrophages, 60 which have broad functions in regulating immunosuppression, 61 promoting cell growth, 62 and enhancing treatment resistance in lung cancer. 63 Actually, the formation of pTLSs may greatly be manipulated by the TME in the invasive border. As some of the aforementioned immune cells; for example, the mast cells, were activated by the cancer cell-derived extracellular vesicles and the neutrophils were stimulated by the hypoxic TME.64,65 On the basis of these data, it is likely that that the presence of pTLSs may act as “a lighthouse in the sea” in the TME, which can facilitate the migration and expansion of cancer cells. Although the density of iTLSs was significantly greater than that of pTLSs in our study, considering the complexity and diversity of the immune and inflammatory context of the TME, their role in cancer could be to promote antitumor immunity, contribute to immunosuppression, or be just bystander sentinels in such a scenario. 66 For example, CD8+ T-cell secretion of IL-10 was found to be correlated with a good prognosis in patients with lung cancer and was further validated in patients with stage I disease67,68; however, some other studies have suggested that T-cell-derived IL-10 can promote lung cancer cell growth and migration.62,69 These results indicate that more studies concerning the role of iTLSs in NSCLC are needed in the future.
In recent years, peripheral inflammatory indices have been increasingly reported as easily accessible and reliable prognostic factors in NSCLC patients.24-27 With respect to stage I patients, several indices, such as the NLR, LMR and ALI, have been extensively validated in previous studies as significant predictors of survival; generally, a high preoperative NLR and low LMR and ALI are conventionally associated with poor outcomes in these patients. 30 ,70-75 Interestingly, TLSs are clustered with CD4+ 32 and CD8+ T cells, 20 and previous reports have indicated that peripheral systemic inflammatory cells or inflammatory prognostic indices such as the NLR can potentially be correlated with these cells in tumors.35,36 Coincidentally, a low NLR was found to be associated with high iTLSs in the study of Fukuhara et al, which may partly support the prognostic value of iTLSs in these patients. 23 Notably, iTLSs in this study were not a significant predictor of DFS in stage I patients. 23 In our study, we did not find any differences in the NLR, LMR, PNI, or ALI among the iTLS low or high subgroups but we did detect significantly low LMR and ALI in patients with high pTLSs. Together with the role of these markers in the prognosis of stage I patients,30,74 we believe that they may also contribute to the prognostic value of pTLSs in stage I patients.
Our study also presented several limitations. First, we collected data retrospectively from a single hospital with relatively small sample sizes, and some bias cannot be excluded; in particular, both the relatively low presence of pTLSs and the wide CIs of it in the multivariate Cox regression analysis indicated a low statistical power and the necessities of enlarged sample size. Second, a group of previous studies has indicated that some other features of TLSs, particularly the maturity or specific immune cells that they contain, are important in determining their prognostic value in NSCLC. However, some of these results (mainly those of iTLSs), for example, the role of the maturation of TLSs in predicting the efficacy of chemotherapy, are still controversial.20-23,41-43 This additional information may be helpful in our study, as we not only focused on iTLSs but also further addressed pTLSs, specifically in stage I patients. Third, some key genetic alterations, such as epidermal growth factor receptor (EGFR) and anaplastic lymphoma kinase (ALK), have a great impact on the presence and density of TLSs in NSCLC,76,77 which could be more reasonable if stratification analysis could be performed on the basis of these characteristics of the patients in our study. Nonetheless, we advocate more detailed studies to validate our results in the future.
Conclusion
Overall, among the heterogeneous results concerning the prognostic value of pTLSs and iTLSs in stage I NSCLC, our results for the first time indicated that the presence of pTLSs may predict poor outcomes in these patients. In addition, we found no such correlation of iTLSs with the outcomes; however, these results should be further validated with large sample size in future taken the aforementioned limitations into consideration.
Footnotes
Author Contributions
HY designed the work. XTH, YQW, and LPH obtained the data. XTH and ZXX analyzed the data. XTH drafted the article. All authors read and approved the final manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of Data and Materials
The data sets generated or analyzed during the current study are available from the first author (Xue TH) on reasonable request.
Ethics Approval and Consent to Participate
This study was approved by the ethics committee of our hospital (ID: S2023-12), and mandatory written informed consent was not necessary because of its retrospective nature according to national regulations.
