Abstract
Keywords
Introduction
Gliomas are the most malignant primary intracranial tumors, accounting for 40% of all primary brain tumors. The global incidence rate is approximately 6/100,000, the median survival period after surgery is approximately 15 months, and the most common first-line treatment regimen is surgery combined with radiotherapy and chemotherapy, and in recent years, the rapid development of nanotechnology has brought new hope for the treatment of gliomas, such as nanotechnology that can efficiently load chemotherapy drugs. 1 The inflammatory microenvironment is a general feature of gliomas that accelerates epigenetic changes in glioblastoma (GBM) and protects tumor cells from immune surveillance. Notably, systemic inflammation plays a critical role in the occurrence and development of glioma. 2 Research on glioma and inflammation has mainly focused on an inflammatory signal pathway and inflammatory cytokines, such as the interleukin family members, transforming growth factor-β1, and tumor necrosis factor-α. Evidence on the relationship between glioma and peripheral blood inflammatory indicators such as the neutrophil-to-lymphocyte ratio (NLR), indirect neutrophil-to-lymphocyte ratio (dNLR), platelet-to-lymphocyte ratio (PLR), and lymphocyte-to-monocyte ratio (LMR) remains limited.
NLR and PLR are generally considered inexpensive and repeatable inflammatory markers and are used as prognostic indicators for various cancers. The relationship between NLR and PLR in glioma has been widely studied; it may be an independent risk factor for identifying patients with glioma with poor prognosis. 3 Moreover, changes between the basic NLR and NLR are related to the prediction of glioma recurrence 4 and postoperative survival. 5 Some studies have found that inflammatory markers such as NLR, dNLR, PLR, and prognostic nutritional index (PNI) are correlated with Ki-67 expression and the pathological grade of glioma, which may help to predict the prognosis of glioma. 6 Furthermore, the dNLR has been discovered to hold the most significant value in differentiating between glioma and other tumors, while the NLR exhibits the highest accuracy in diagnosing glioma, potentially serving as an independent parameter for predicting glioma grade. 7 To assess the diagnostic utility of the NLR, dNLR, PLR, LMR, and PNI in glioma, a retrospective analysis was conducted on 750 patients with glioma, 44 patients with acoustic neuroma, 271 patients with meningioma, 102 patients with non-pathological epilepsy, and 682 healthy controls. The findings suggested that the combination of NLR and LMR had high sensitivity and specificity in diagnosing glioma, differentiating it from acoustic neuroma, meningioma, GBM, and in distinguishing between GBM and low-grade glioma. 8
However, controversy persists concerning the independent diagnostic value of NLR and PLR, with no definitive conclusion on the diagnostic efficacy of inflammatory markers like NLR, dNLR, PLR, and LMR in glioma cases. Therefore, this study primarily aimed to ascertain the prognostic and diagnostic significance of peripheral blood inflammation markers in patients with gliomas.
Materials and Methods
Patients and Examination
Retrospective analysis of patients with glioma and meningioma (in the control arm, patients with meningiomas were selected instead of “healthy people” to exclude potential glioma cases among these so-called healthy individuals, who had not undergone brain Magnetic Resonance Imaging [MRI]) was conducted at the Affiliated Hospital of Jining Medical University from December 2019 to December 2021. All patients included in this study met the following criteria: (1) glioma was histologically confirmed in resected or biopsy specimens according to WHO standards, and meningioma was confirmed histopathologically in resections; (2) patients received no tumor-specific pretreatments such as radiotherapy or chemotherapy prior to surgery; (3) patients had no blood diseases, infectious diseases, metabolic syndrome, severe heart, renal, or liver dysfunctions, other cancers, autoimmune, or inflammatory diseases, or were on drugs related to inflammatory status; (4) complete laboratory blood sample data were available; (5) after applying the inclusion and exclusion criteria, informed consent was obtained from 334 patients with various grades of gliomas and 345 patients with WHO grade I meningiomas (Figure 1). The study was authorized by the Ethics Committee of the Affiliated Hospital of Jining Medical University (protocol number 2022C210), and informed consent was waived because of the retrospective study design. The reporting of this study conformed to REMARK guidelines. 9 We have de-identified all patient details.

Flowchart of the current study.
Clinical Variables and Laboratory Testing
Demographic and clinicopathological variables, including age, sex, tumor grade, and histological type, were retrieved from patients’ medical records. As a routine preoperative examination, the laboratory department will complete the examination within 2 h after collecting blood samples and carry out routine blood tests, biochemistry, and other laboratory examinations to obtain white blood cell, neutrophil, lymphocyte, monocyte, and platelet counts. In addition, we calculated the NLR, dNLR (the calculation formula is: absolute neutrophil count /[white blood cell count - absolute neutrophil count] - PLR
The expression of IDH-1 and Ki-67 in glioma tissues was detected using immunohistochemistry (EnVision two-step method). All operations were performed according to the kit instructions. Glioma tissues were fixed in 10% neutral buffer formalin, dehydrated through an alcohol gradient, embedded in paraffin, and sliced to a thickness of 4-μm. The sections were then placed on a baking sheet at 68 °C for 20 min and underwent conventional xylene dewaxing and gradient alcohol dehydration. Subsequently, the sections were washed with water for 5 min and then rinsed three times with peripheral blood smear (PBS) (pH 7.4) for 3 min each. The repair solution used was ethylenediamine tetraacetic acid (PH 9.0), prepared at 200 ml with a 1:50 dilution according to the instructions. The dewaxed and hydrated tissue sections were positioned on a high-temperature resistant plastic section rack and placed in the repair solution, followed by a 2 × 3-min rinse with PBS. Each section was treated with one drop of 3% H2O2 and incubated at room temperature for 10 min to block endogenous peroxidase activity, then rinsed for 3 × 3 min with PBS. After PBS removal, each section was treated with 1 drop of primary antibody IDH-1/Ki-67 (1:200) (Fuzhou Maixin Biotechnology Development Co., LTD) and incubated at room temperature for 2 h, followed by a 3 × 5-min PBS rinse. Each section was then treated with one drop of polymer enhancer (reagent A) (Gene Tech (Shanghai) Company Limited) and incubated at room temperature for 20 min, followed by a 3 × 3-min PBS rinse. After another PBS rinse, each section received one drop of enzyme-labeled anti-mouse/rabbit polymer (reagent B) (Gene Tech (Shanghai) Company Limited) and was incubated at room temperature for 30 min, followed by a 3 × 5-min PBS rinse. After removing the PBS, each section was treated with one drop of freshly prepared 3,3′-Diaminobenzidine solution and observed under a microscope for 5 min. The sections were then stained with hematoxylin, differentiated with 0.1% hydrochloric acid(HCl), rinsed with running water, blued, dehydrated, and dried with a gradient alcohol. They were made transparent with xylene, sealed with neutral gum, dried, and observed. Brownish-yellow, brown, and tan colors indicated positive results.
Statistical Analysis
We adopted a case-control study design and used descriptive statistics to analyze the demographic characteristics of patients with gliomas and meningioma controls. Frequency and percentage were used to describe the classification variables, and the chi-square test was used to analyze whether the difference between the two groups was statistically significant. The continuous variables conforming to the normal distribution were expressed by mean ± standard deviation, and an independent sample t-test compared the statistical differences between the glioma and meningioma groups. The continuous variables that conformed to the skewed distribution were represented by the median (M), 25th, and 75th percentiles (P25 and P75), and the statistical differences between the three groups were compared using Kruskal–Wallis tests. Statistical differences between the two groups were compared using the Mann–Whitney U test. We divided the indicators horizontally according to their quantiles in meningioma controls, analyzed the correlation between inflammatory indicators and glioma risk using multiple logistic regression, and used odds ratio (OR) and 95% confidence interval (CI) as effect estimates. We drew a receiver operating characteristic (ROC) curve for statistically significant indicators in the logistic regression analysis and calculated the area under the curve (AUC). Considering the detection value of the maximum Youden index [(sensitivity + specificity) - 1] as the critical value (cutoff value), the diagnostic efficacy of single and combined detection of each index was calculated. Furthermore, we analyzed the correlation between inflammatory indicators and glioma grade, IDH-1, and Ki-67 Proliferation Index. Statistical analysis was conducted using SPSS26.0. The significance level was set at α=0.05, with P < .05 deemed statistically significant.
Results
Baseline Characteristics of the Selected Participants
Table 1 describes the demographic information of glioma cases and meningioma controls. A chi-square test was conducted on the two groups of patients to analyze whether the difference was statistically significant; 334 patients with gliomas(of the 431 glioma patients, 97 were excluded, including 26 with preoperative radiotherapy or chemotherapy, 31 with indefinite pathological diagnosis, 15 with incomplete clinical data, 20 with other malignancies, and 5 with inflammatory diseases, and the remaining 334 patients were included in statistical analysis)(Figure 1), comprising 175 males (52.4%) and 159 females (47.6%), were selected for the experiment. The average age of the participants was 52 ± 13 years. Further, 90 patients had a history of smoking (26.9%), 85 patients had a history of drinking (25.4%), and their BMI was 24.01 ± 3.13 kg/m². A total of 335 patients with meningioma were selected, comprising 74 men (21.4%) and 271 women (78.6%). The average age of the participants was 55 ± 10 years, 43 (12.5%) had a history of smoking, and 39 (11.3%) had a history of drinking, with a BMI of 24.84 ± 3.30 kg/m². In summary, the male sex, smoking, and drinking ratios of patients with glioma were significantly higher than those of meningioma controls (P < .05), while the age and BMI of patients with glioma were significantly lower than those of patients with meningioma (P < .05).
Baseline Characteristics of the Participants.

Continuous variables were presented as mean ± standard deviation or median (interquartile range), according to its distribution; categorical variables were presented asnumbers (percentage);p-values were calculated by Student's t-test or chi-square test for different variables. P-value in bold indicates significance at P < .05.
Relationship Between Peripheral Blood Inflammatory Markers and Glioma Grade
According to the fourth edition of the WHO classification of central nervous system tumors published in 2016, 10 334 gliomas were categorized as follows: 130 were WHO Grade II (38.9%), 74 were WHO Grade III (22.2%), and 130 were WHO Grade IV (38.9%). We further studied the tumor classifications and explored the differences in the expression of inflammatory factors such as lymphocytes, NLR, dNLR, and PLR. Notably, a negative correlation was found between glioma grade and the median lymphocyte count. The median of lymphocytes in WHO II, III and IV patients(× 109 /L) was 1.97 (1.57-2.38), 1.68 (1.4-2.07), and 1.62 (1.28-1.98) respectively, and the difference between the three groups was statistically significant (P < .001); Kruskal–Wallis tests showed that the interaction between glioma grade (WHO II–IV) and NLR (P < .001), dNLR (P < .001), PLR (P < .002), and LMR (P < .025) was statistically significant (Table 2). Histograms were drawn for lymphocytes, NLR, dNLR, and PLR between the WHO II and WHO III, WHO II and WHO IV, and WHO III and WHO IV groups. We found that the median lymphocyte count, NLR, and dNLR were significantly different between the WHO II and WHO III and WHO II and WHO IV groups (P < .05) but not between the WHO III and WHO IV groups (Figure 2A–C). PLR was statistically significant between the WHO II and WHO IV, WHO III, and WHO IV groups (P < .05) but not between the WHO II and WHO III groups (Figure 2D). Taken together, our data suggest that peripheral blood inflammatory markers may be related to the glioma grade.

(A-D) Histograms of the comparisons in wHO grade ii versus grade III, grade ii versus grade iv and grade III versus grade iv for lymphocyte, NLR, dNLR and PLR; * P < .05, ** P < .01, *** P < .001, ns stands for not significant. PLR, platelet-to-lymphocyte ratio.
Correlations Between Peripheral Blood Inflammatory Markers and Glioma Grade.

P-values for comparisons of the differences between glioma WHO grades group using the Kruskal-Wallis tests, P-value in bold indicates significance at P < .05.
LMR, lymphocyte-to-monocyte ratio; PLR, platelet-to-lymphocyte ratio.
Relationship Between Peripheral Blood Inflammatory Markers and Glioma Types
As shown in Table 3, we used the Mann–Whitney U test to analyze a statistical difference existed between the glioblastoma and non-glioblastoma groups in lymphocytes, NLR, dNLR, PLR, and LMR. The results showed that the median levels of lymphocytes and LMR were significantly lower than those in patients without glioblastoma, whereas the levels of neutrophils, NLR, dNLR, and PLR were significantly higher than those of patients without glioblastoma (P < .05).
Correlations Between Peripheral Blood Inflammatory Markers and Glioma Types.

P-values for comparisons of the differences between the glioblastoma group and the non-glioblastoma group using the Mann-Whitney U tests, P-value in bold indicates significance at P < .05.
LMR, lymphocyte-to-monocyte ratio; PLR, platelet-to-lymphocyte ratio.
Relationship Between Peripheral Blood Inflammatory Markers and the Location of Glioma
Studies had shown that the location of glioma may be related to specific biological signatures, and the anatomical distribution of glioma biomarkers had a high degree of variability, and this high heterogeneity may have a clinical role. There was study shown that patients with frontal lobe high-grade gliomas(HGGs) may have better survival expectations. 11 However, it is unclear whether peripheral blood inflammatory indicators are related to tumor location. Next, we used the Kruskal–Wallis test to analyze whether changes were observed in peripheral blood inflammatory indicators in patients with glioma in the frontal lobe, temporal lobe, and other parts (Table 4). The results showed that the neutrophil count (P = .782), NLR (P = .7704), dNLR (P = .771), LMR (P = .864), and levels of inflammatory markers in different locations were not statistically significant (P > .05).
Correlations Between Peripheral Blood Inflammatory Markers and the Location of Glioma.

P-values for comparisons of the differences between glioma location using the Kruskal-Wallis tests, p-value in bold indicates significance at p < .05.
LMR, lymphocyte-to-monocyte ratio; PLR, platelet-to-lymphocyte ratio.
Relationship Between Peripheral Blood Inflammatory Markers and IDH-1 Mutation in Glioma
IDH-1 is considered the earliest genetic event in the process of glioma formation and is an essential molecular marker of low-grade glioma. 12 Some studies have predicted the survival rate of patients with glioblastoma by studying preoperative inflammatory markers and IDH mutation status. Our analysis showed that a low NLR was only related to the prognosis of the IDH wild-type GBM group. Some study showed that high levels of NLR, monocyte to lymphocyte ratio (MLR) and PLR were risk factors for the prognosis of GBM patients, and preoperative hematological markers may predict the survival status of individual GBM patients 3 years after treatment, helping clinicians to make good clinical decisions. 13 We analyzed the differences in inflammatory markers between IDH-1 (-) (N = 157) and IDH-1 (+) (N = 54) using Mann–Whitney U tests and found no difference in the inflammatory indicators in patients with glioma IDH-1 (-) and IDH-1 (+) (P > .05) (Table 5 and Figure 3).

Photomicrographs of glioma tissue stained for IDH-1: (A) mutant type, (B) wild type, original magnification: ×400.
Correlations Between Peripheral Blood Inflammatory Markers and Glioma IDH-1.

P-values for comparisons of the differences between glioma IDH-1(-) and IDH-1(+) group using the Mann-Whitney U tests, P-value in bold indicates significance at P < .05.
LMR, lymphocyte-to-monocyte ratio; PLR, platelet-to-lymphocyte ratio.
Relationship Between Peripheral Blood Inflammatory Markers and the Ki-67 PI in Glioma
The Ki-67 Proliferation Index is significantly correlated with histopathological grading and is a crucial auxiliary marker of glioma grading. 14 We used Mann–Whitney U tests to analyze the expression difference of inflammatory markers between the Low Ki-67 PI and High Ki-67 PI groups and found that the median lymphocyte level in patients with glioma Low Ki-67 PI was 1.94, which was significantly higher than that in patients with High Ki-67 PI (the median lymphocyte level was 1.65) (Table 6 and Figure 4). In contrast, neutrophil count, NLR, dNLR, PLR, and red cell distribution width-standard deviation were significantly lower than those in patients with a High Ki-67 PI. The difference between the levels of inflammatory markers and Ki-67 PI was statistically significant (P < .05).
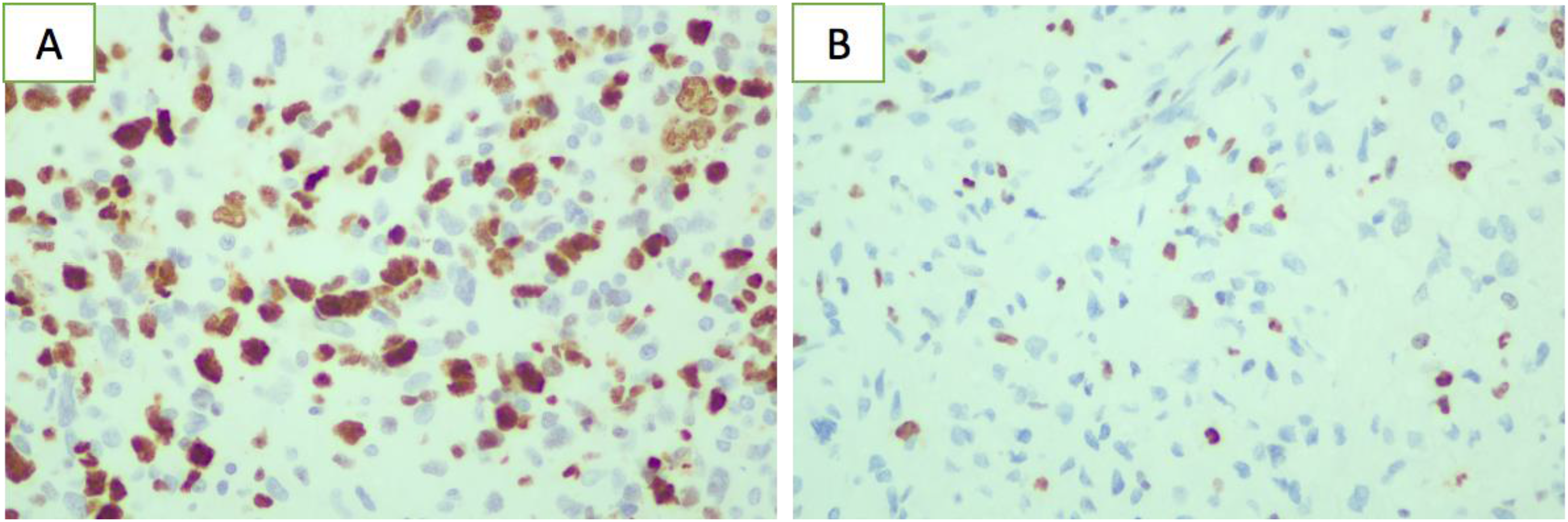
Photomicrographs of glioma tissue stained for expression of ki-67 pi: (A) high ki-67 pi, (B) low ki-67 pi, original magnification: ×400.
Correlations Between Peripheral Blood Inflammatory Markers and Ki-67 PI.
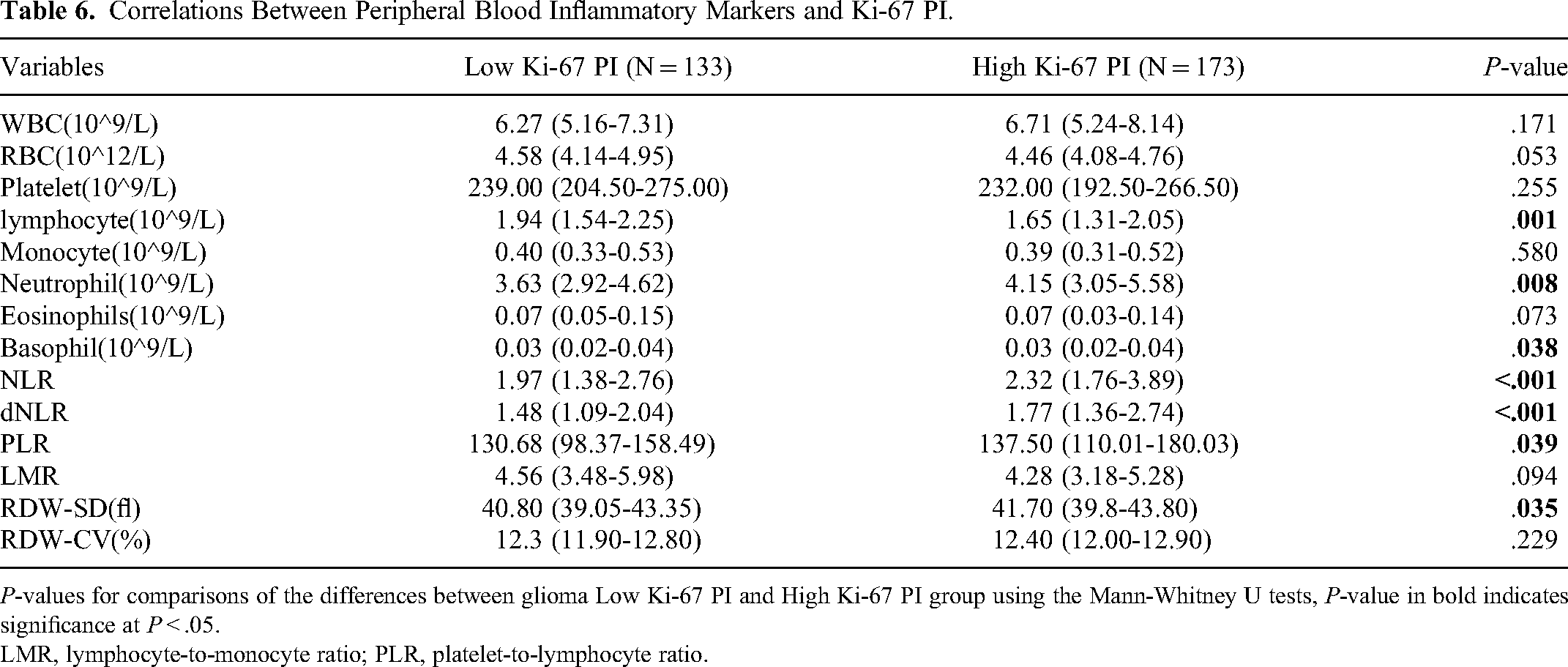
P-values for comparisons of the differences between glioma Low Ki-67 PI and High Ki-67 PI group using the Mann-Whitney U tests, P-value in bold indicates significance at P < .05.
LMR, lymphocyte-to-monocyte ratio; PLR, platelet-to-lymphocyte ratio.
Distribution of Peripheral Blood Inflammatory Markers in Glioma and Meningioma
As shown in Table 7 and Figure 5 (A–F), differences existed in the distribution of peripheral blood inflammatory markers between meningiomas and gliomas. According to the Mann–Whitney U test, the median levels of eosinophils and LMR in patients with glioma were lower than those in patients with meningioma. WBC, monocyte, and neutrophil counts, NLR, and dNLR were significantly higher than those in patients with meningioma (P < .05). Patients with gliomas exhibited increases in the number of WBC, monocytes, neutrophils, NLR, and dNLR, along with a decreased LMR, compared with those with meningioma. A statistically significant difference was noted in the distribution of inflammatory factors between these two types of brain tumors (P < .05).

(A-F) Histograms of the comparisons in glioma versus meningioma for WBC, monocyte, neutrophil, NLR, dNLR and LMR; * P < .05, ** P < .01, *** P < .001, ns stands for not significant. LMR, lymphocyte-to-monocyte ratio.
Distribution of Peripheral Blood Inflammatory Markers in Glioma and Meningioma.
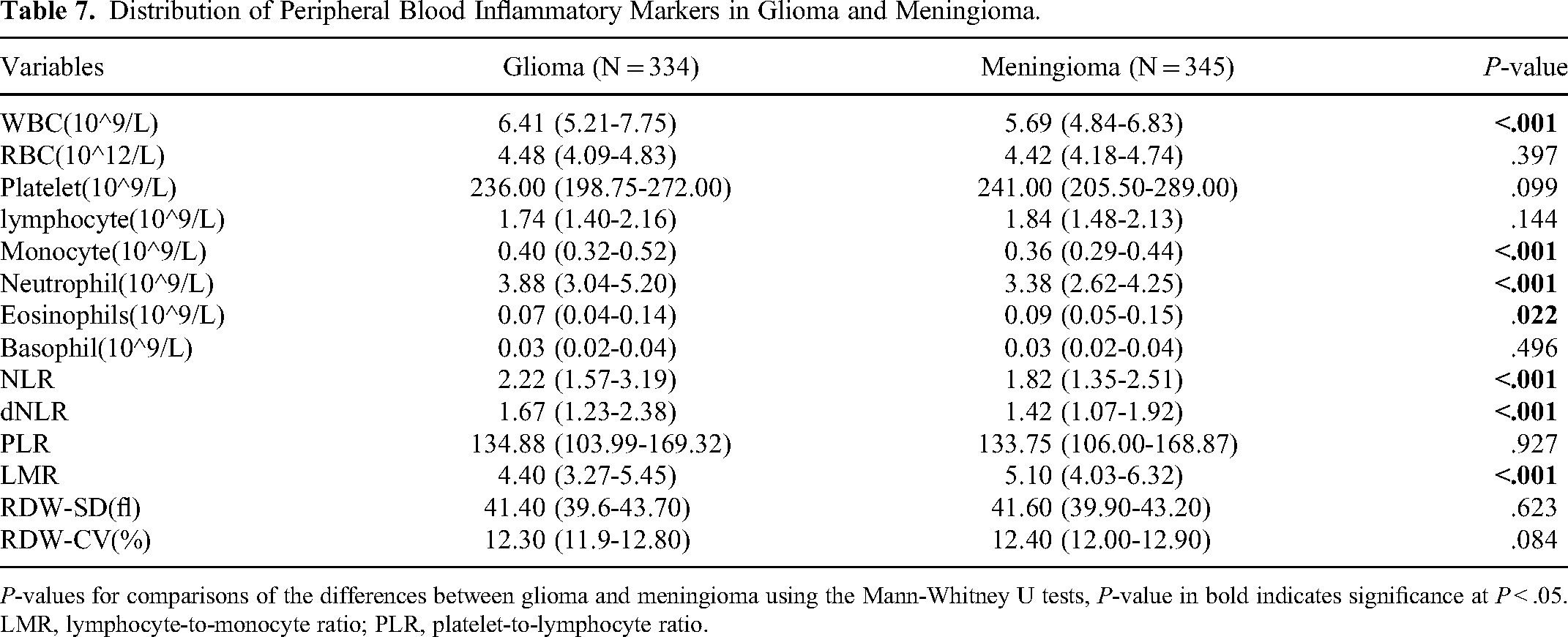
P-values for comparisons of the differences between glioma and meningioma using the Mann-Whitney U tests, P-value in bold indicates significance at P < .05.
LMR, lymphocyte-to-monocyte ratio; PLR, platelet-to-lymphocyte ratio.
Correlation Between Peripheral Blood Inflammatory Markers and Glioma Risk
After adjusting for sex, age, BMI, smoking, drinking, and other factors, the level was divided according to the third quantile of meningioma control. Multivariate logistic regression was employed to analyze the correlation between inflammatory indicators and the risk of glioma. The OR and 95% CI served as effect estimates. An OR value of 1 suggests that the factor does not influence the disease's occurrence; an OR greater than 1 indicates a risk factor; an OR less than 1 suggests a protective factor.
The WBC count, neutrophil count, NLR, and dNLR were significantly and positively correlated with the risk of glioma (Table 8). Compared with that in the first quantile, the risk of glioma in the third quantile increased by 1.84 (1.24, 2.74), 2.01 (1.34, 3.02), 1.94 (1.29, 2.93), and 1.93 (1.29, 2.90) times, respectively. Furthermore, LMR levels were negatively correlated with the risk of glioma. Notably, compared with that in the first third quartile, the risk of glioma in the third quantile was 0.52 (0.35, 0.79). Moreover, the expression of inflammatory markers was associated with the risk of glioma (P < .05).
Correlation Between Peripheral Blood Inflammatory Markers and Glioma Riska.

aModel was adjusted for sex, age, BMI, smoking and drinking status.
LMR, lymphocyte-to-monocyte ratio; PLR, platelet-to-lymphocyte ratio.
Prognostic Value of Peripheral Blood Inflammatory Markers for Glioma
Peripheral blood inflammatory indicators were clearly correlated with the clinicopathological features of patients with glioma. Furthermore, we aimed to determine whether they could be involved in the diagnosis of patients with glioma. That is, when some patients did not undergo in-depth brain MRI, surgery, or other tests, we could identify patients with glioma through changes in some indicators in the peripheral blood. After adjusting for sex, age, and other factors, ROC curve analysis was performed on the experimental data, and the AUC was obtained from the ROC curve. The area under the ROC curve ranges from 0–1. The closer the value approaches 1, the stronger the diagnostic efficiency. Generally, the higher the AUC, the more robust the differential ability of the diagnostic test. The AUC for a completely worthless test is 0.5, while for the ideal diagnostic test it is 1. The diagnostic value ranges from low at 0.5 to 0.7, medium from 0.7 to 0.9, and >0.9, indicating the highest diagnostic value. The results indicated that the AUC for WBC was 0.72 (95% CI [0.68,0.76]), and the cutoff value for WBC was 0.48, with a sensitivity of 0.65 and a specificity of 0.70. The AUC for neutrophils was 0.72 (95% CI [0.68,0.76]), and the cutoff value for neutrophils was 0.46, with a sensitivity of 0.67 and a specificity of 0.68. The AUC for NLR was 0.71 (95% CI [0.68, 0.75]), and the cutoff value for NLR was 0.50, with a sensitivity of 0.61 and a specificity of 0.76. The AUC for dNLR was 0.71 (95% CI [0.67,0.75]), and the cutoff value for dNLR was 0.52, with a sensitivity of 0.61 and a specificity of 0.76. The AUC for LMR was 0.71 (95% CI [0.67,0.75]), and the cutoff value for LMR was 0.45, with a sensitivity of 0.67 and a specificity of 0.70. In conclusion, the AUCs for WBC, neutrophil, NLR, dNLR, and LMR under the ROC curve ranged from 0.71 to 0.72, which could be utilized for diagnosing glioma. However, owing to chronic inflammatory reactions and various malignant tumors, these markers could increase to varying degrees. These markers have high sensitivity but relatively low specificity; therefore, they are not independent gold standards for the diagnosis of glioma. Thus, it is crucial to identify other glioma-related serum markers and their combined detection

ROC curves showed the diagnostic value of WBC, neutrophil, NLR, dNLR, LMR and their combinations in patients with glioma (A:WBC; B: neutrophil; C:NLR; D: dNLR; E: LMR; F:their combinations). LMR, lymphocyte-to-monocyte ratio; ROC, receiver operating characteristic.
Diagnostic Value of Peripheral Blood Inflammatory Markers in Patients with Gliomaa.
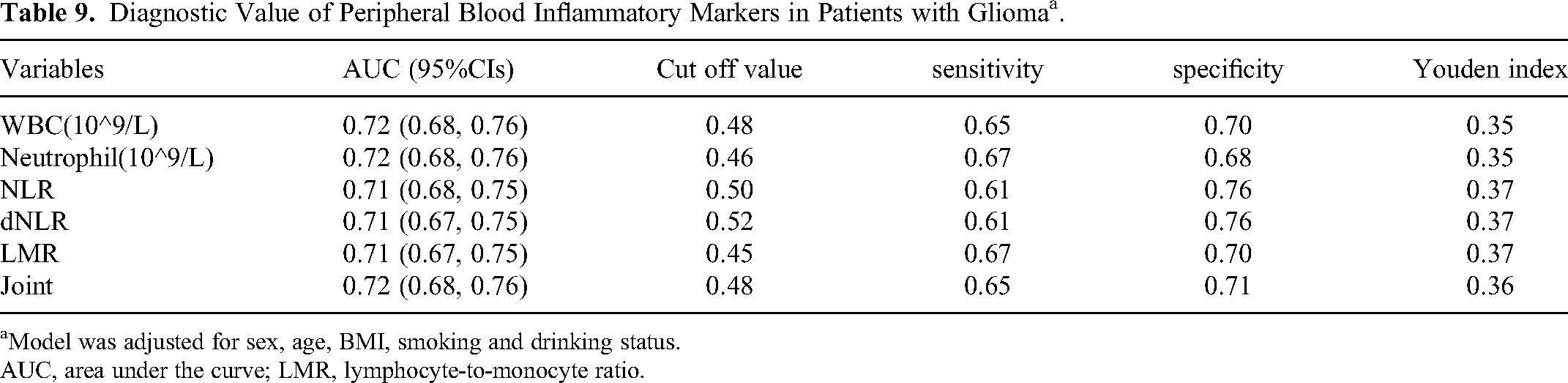
aModel was adjusted for sex, age, BMI, smoking and drinking status.
AUC, area under the curve; LMR, lymphocyte-to-monocyte ratio.
Discussion
Recent studies have shown that the glioma microenvironment exhibits cytokine and chemokine profiles similar to those observed during chronic inflammation. 15 Peripheral inflammatory parameters, such as NLR, dNLR, LMR, and PLR, may prove to be reliable, sensitive, and cost-effective markers for glioma diagnosis, glioma grade prediction, treatment response monitoring, and predicting recurrence. 7 Moreover, the correlation between differential expression of inflammatory parameters and gliomas has not been clarified. We further analyzed the evaluation value of NLR, dNLR, PLR, LMR, WBC count, and other parameters for glioma by retrospectively analyzing the differences in the expression of inflammatory parameters between glioma and meningioma.
Currently, researchers believe that NLR, LMR, and NLR + LMR are helpful for diagnosis and differential diagnosis of glioma, and the combination of peripheral blood inflammatory indicators may be a tool for the preoperative diagnosis of glioma. 8 Inflammatory reactions are closely associated with glioma development and prognosis. Experiments have been conducted to evaluate the influence of inflammatory indicators on the prognoses of different glioma molecular subtypes. High NLR, low LMR, and high dNLR were associated with poor prognosis, whereas PLR had no prognostic significance. 16 However, in addition to the value of peripheral blood inflammatory indicators in the diagnosis and prognosis of glioma, the correlation between the expression of inflammatory indicators in glioma and tumor grade, location of onset, and pathological markers remains unclear. Nonetheless, the sensitivity and specificity of the different inflammatory indicators in gliomas have not yet been clarified. Future research should further investigate the diagnostic efficacy of different indicators and indicator combinations and screen for effective and cheap specific markers.
Specifically, in our study, with the improvement in the pathological grade of glioma, the median number of neutrophils in patients with WHO grades II, III, and IV(× 109 /L) was 3.63, 3.9, and 4.2, respectively; the median lymphocyte(× 109 /L) was 1.97, 1.68, and 1.62, respectively (Table 2). Consequently, the elevation in NLR and dNLR observed in patients with glioma stems from the increase in neutrophil count coupled with a relative decline in lymphocyte count. Our data revealed that the neutrophil-dependent inflammatory response was intensified, while the lymphocyte-mediated antitumor immune response was diminished. Furthermore, previous studies had corroborated high NLR was independently associated with 30-day postoperative mortality and shorter overall survival (OS) among surgically treated patients with spinal metastasis. 17 Neutrophils are the most abundant circulating leukocytes in humans. Their complex roles in gliomas include their ability to play a tumor-promoting or antitumor role and their ability to show various polarized phenotypes. 18 Studies have shown that neutrophils in human peripheral blood can infiltrate tumors, play an essential role in tumor angiogenesis, promote the malignant phenotype of gliomas, and regulate glioma grading. The decrease in peripheral blood lymphocytes indicated that the antitumor immune function of the cells was reduced, thus providing a relatively stable environment for the growth and migration of tumor cells. Weng et al found that the NLR and radiological characteristics of peripheral blood inflammatory markers were positively correlated with the proliferation index in patients with glioma, whereas the proliferation index was highly correlated with tumor grade. 19 In summary, a positive correlation was noted between NLR, dNLR, and tumor grade, with the highest expression in glioblastoma.
PLR and LMR have been shown to predict the prognosis of many malignant tumors, such as lung cancer 20 and colorectal cancer. 21 However, the use of the PLR and LMR to evaluate gliomas is rare. Here, as the pathological grading of glioma improved, the median LMR levels for WHO grade II, III, and IV patients were 4.74, 4.41, and 4.16, respectively; median PLR levels were observed at 126.38, 128.59, and 140.37. LMR expression showed a negative correlation with the pathological grade of glioma, while PLR exhibited a positive correlation (Table 2). Kemerdere et al found that the expression of LMR differed across glioma grades, especially in high-grade gliomas, which is consistent with our current results. 22 Compared with that of the meningioma control group, the median LMR level of meningioma was 5.10, while the levels of WBC, monocytes, neutrophils, NLR, and dNLR were significantly higher. In this study, the PLR, LMR, NLR, and dNLR were identified as biomarkers for glioma grading, and their application in the differential diagnosis and prognosis prediction of glioma was supported (Table 7).
In glioblastomas, tumor-associated macrophages account for 50% of the tumor volume and play an essential role in tumor maintenance and development. Macrophages exhibit critical regional differences between the periphery of invasive and hypoxic tumor cores. 23 However, the relationship between peripheral blood inflammatory factors and the location of gliomas remains unclear. Therefore, we carried out relevant statistical analyses to prove for the first time that the expression of peripheral blood inflammatory indicators was similar regardless of the location of the glioma (Table 4).
Pathological examination continues to be the gold standard for diagnosing gliomas during clinical treatment. Ki-67 PI and IDH-1 are significant indicators of meningioma progression. Some studies have demonstrated that Ki-67 PI is significantly related to the pathological grading of gliomas, and there exists a potential biological relationship between Ki-67 PI and inflammatory markers such as NLR and PLR. 6 These results were consistent with our findings. Some research showed that IDH-1 mutation was an independent factor in prolonging the OS and progression-free survival (PFS) of patients with GBM. 24 We further analyzed the relationship between IDH-1 and peripheral blood inflammatory indicators. However, our results showed no difference in inflammatory indicators between patients with IDH-1(-) and IDH-1(+) glioma (P > .05).
Currently, the diagnosis and screening of glioma mainly rely on imaging examinations; however, imaging examinations are expensive, and it is difficult to differentiate between some primary brain tumors and gliomas. 25 Therefore, it is important to establish a multi-element prognostic nomogram for glioblastoma based on peripheral blood detection, conventional MRI, and clinical factors, which will improve prognostic evaluation. 26 Therefore, the identification of new biomarkers with noninvasive, diagnostic, and prognostic characteristics is urgently needed. Circulating biomarkers have potential clinical applications in population screening, tumor subtype classification, tumor status monitoring, and the delivery of individualized treatments generated by tumor genotyping. However, there is currently no clinical application for circulating biomarkers that are sensitive to glioma. 27 The current study analyzed the correlation between inflammatory indicators and glioma risk, producing an ROC curve. The experimental results indicate that the WBC count, neutrophil count, NLR, and dNLR were significantly positively correlated with glioma risk, while LMR levels were negatively correlated. These five indicators—WBC count, neutrophil count, NLR, dNLR, and LMR—demonstrated significant diagnostic efficacy for gliomas, and these biomarkers exhibited considerable clinical value. First, due to their low cost, they can be broadly applied in glioma screening, enhancing the diagnostic rate. Second, for brain tumors that are challenging to diagnose via imaging, combining inflammatory indicators with MRI can aid in diagnosis. Third, these biomarkers are instrumental in guiding the radiotherapy and chemotherapy of glioma. A previous study indicated that neutrophilia at any time interval before and after radiotherapy, NLR prior to radiation, and thrombocytopenia post-radiation are predictive of poor glioma prognosis. 28 Pretreatment inflammatory markers can predict the efficacy of bevacizumab in recurrent glioma, 29 however, the main limitations of the study were its retrospective design and the small and diverse sample.
There were some limitations in our retrospective study. (1) Our study only collected data from a relatively small proportion of glioma patients in our hospital, one single clinical center. Therefore, the need for a multicenter study and a larger number of glioma patients to verify our preliminary results would be more convincing; (2) Patients with glioma were not followed up in this study, and the specific prognosis was unknown.
Conclusion
NLR, dNLR, PLR, and other inflammatory markers correlated with WHO grade and Ki-67 PI in patients with glioma. Additionally, WBC, neutrophil, NLR, and dNLR were significantly positively correlated with glioma risk, while LMR levels were inversely correlated with this risk. We also demonstrated that the peripheral blood inflammatory index, serving as a noninvasive biomarker, exhibits relatively high sensitivity and specificity for diagnosing glioma, differentiating glioma from meningioma, diagnosing GBM, and distinguishing GBM from low-grade glioma. This index can be utilized as a routine screening tool in patients with glioma. These findings may further elucidate the role of the peripheral blood inflammation index in glioma development. In the future, we will follow up more patients to conduct relevant studies, and study the correlation between preoperative inflammatory markers and patient prognosis, which has an important guiding role for clinical work.
Footnotes
Acknowledgements
The authors would like to thank the PhD Research Foundation of Affiliated Hospital of Jining Medical University for its support to our research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval Consent to Participate:
The study was approved by the Ethics Committee of Affiliated Hospital of Jining Medical University (protocol number 2022C210) and informed consent was waived due to the retrospective study design.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by PhD Research Foundation of Affiliated Hospital of Jining Medical University (Grant NO. 2021-BS-006) and 2022 key research and development program of Jining Science and Technology Bureau (policy guidance)(No.2022YXNS031).
