Abstract
Background
The neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), monocyte-to-lymphocyte ratio (MLR), systemic immune-inflammatory index (SII), systemic inflammation response index (SIRI), and Onodera’s prognostic nutritional index (OPNI) have been reported as prognostic markers for various cancers. We evaluated the prognostic value of the NLR, PLR, MLR, SII, SIRI, and OPNI for poorly-to moderately-differentiated cervical squamous cell carcinoma (CSCC).
Patients and Methods
We retrospectively analyzed the cases of 109 patients with early-stage poorly-to moderately-differentiated CSCC who underwent radical surgery at our institution in 2014-2017. The optimal cutoff points for the NLR, PLR, MLR, SII, SIRI, and OPNI were determined by receiver operating characteristic curves. Overall survival was analyzed by the Kaplan-Meier method. We performed a multivariate analysis using the Cox proportional hazard regression model to determine the independent prognostic indicators for early-stage poorly-to moderately-differentiated CSCC.
Results
The appropriate cutoff points were: NLR, 1.72; PLR, 111.96; MLR, .24; SII, 566.23; SIRI, 1.38; and OPNI, 52.68. The OS of the patients with a high OPNI (P = .04), low SII (P = .03), or low SIRI (P = .01) was significantly better. The uni- and multivariate analyses identified only the OPNI as an independent prognostic marker for early-stage poorly-to moderately-differentiated CSCC (P = .04 and P = .02).
Conclusion
The OPNI is an independent prognostic marker for early-stage poorly-to moderately-differentiated CSCC; the NLR, PLR, MLR, SII, and SIRI are not.
Keywords
Introduction
Cervical cancer is the most common gynecologic malignancy, accounting for a significant number of cancer-related fatalities, particularly in developing countries.1,2 Radical hysterectomy and pelvic lymph node dissection are the conventional surgical procedures for patients with early-stage cervical squamous cell carcinoma (CSCC). However, there remains a significant disparity in prognosis among individuals with early-stage cervical cancer who have undergone surgery. This may be due to the fact that patients have different tumor differentiation and microenvironments. New prospective medications and biomarkers for the diagnosis, prognosis, and treatment of CSCC are thus urgently needed.
Recent investigations have demonstrated that inflammatory indices related to the tumor microenvironment have high prognostic value for some solid cancers. Systemic inflammatory responses affect cancer patients’ nutrition, function, and prognosis.3,4 Poor nutritional status is significantly associated with poor prognosis in a variety of cancers. 5 Onodera et al established the concept of Onadera’s prognostic nutritional index (OPNI), which has become 1 of the most widely used markers of nutritional status. 6 The OPNI has also been demonstrated to have predictive and prognostic value in a variety of cancers.7-10 There have been few studies of the OPNI in cervical cancer, however. 11
In the U.S. National Comprehensive Cancer Network (NCCN) guidelines, poor tumor differentiation is not regarded as a high-risk feature for the prognosis of cervical cancer. 12 However, clinical experience has suggested that individuals with early-stage poorly-to moderately-differentiated CSCC are more likely to develop recurrences and metastases than patients with well-differentiated CSCC. A biomarker is thus needed for early-stage poorly-to moderately-differentiated CSCC. To the best of our knowledge, there has been no investigation of the relationship between clinical features and the OPNI in patients with early-stage poorly-to moderately-differentiated CSCC. We conducted the present study to explore the predictive significance of the OPNI and other parameters in this patient population.
Patients and Methods
Patient Population and Study Design
We retrospectively analyzed the cases of the 109 patients with early-stage poorly-to moderately-differentiated CSCC who underwent radical cervical cancer treatment during the period from May 2014 to December 2017 at Changzhi People’s Hospital and Yuncheng Central Hospital in Shanxi Province, China. The ages of the patients who underwent the treatment ranged from 27 to 71 (53.95 ± 9.6) years.
The study’s inclusion criteria were: (1) postoperative histopathology that revealed a diagnosis of early-stage poorly-to moderately-differentiated CSCC; (2) no coeval tumors; (3) stage IA to IIA disease according to the 2009 International Federation of Gynecology and Obstetrics (FIGO) staging system; (4) no treatment for CSCC was conducted within 1 week before the surgery; and (5) complete medical records and follow-up records were available. Patient demographics and clinical characteristics were retrieved from the patients' medical records: age, body mass index (BMI), FIGO stage, tumor lymph node metastasis status, vascular infiltration, and peripheral blood test results (including routine blood tests, liver and kidney function, and the tumor marker SCCA [squamous cell carcinoma antigen]) within 1 week before surgery. We used these parameters to calculate the following inflammatory and nutritional indices: the neutrophil-to-lymphocyte ratio (NLR), the platelet-to-lymphocyte ratio (PLR), the monocyte-to-lymphocyte ratio (MLR), the OPNI, the systemic inflammatory response index (SIRI), and the systemic immunoinflammatory index (SII).
The patients’ overall survival (OS) was calculated from the date of the patient’s first surgery to the date of death (or the date of last follow up). We used OS as the end point of this study, with death as a positive event, because OS better reflects patients’ long-term survival prognosis.
The exclusion criteria were: (1) the presence of hematologic illness, autoimmune disease, organ dysfunction, acute or chronic infection, and other diseases that may impact hematologic indexes; and (2) no history of other malignant tumors.
Data Collection
All of the information analyzed in this study was gathered from the patients' medical records and pathology reports. The NLR was calculated as the neutrophil count/lymphocyte count (109/L). The PLR was calculated as the platelet count/lymphocyte count (109/L). The MLR was calculated as the monocyte count/lymphocyte count (109/L). The SII was calculated as the neutrophil count × platelet count/lymphocyte count (109/L). The SIRI was calculated as the neutrophil count × monocyte count/lymphocyte count (109/L). The OPNI was calculated as serum albumin (g/L) + 5 × lymphocyte count (109/L).
Demographic and Clinical Characteristics of the Included Patients With Cervical Carcinoma.

Ethical Statement
This study was reviewed and approved by the ethics committees of Changzhi People’s Hospital (no. 2022K004) and Yuncheng Central Hospital (no. YXLL2022027) in Shanxi Province, China and was conducted in accord with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. The reporting of this study conforms to the STROBE guidelines, 13 and all patient information was de-identified. Written informed consent for their data to be used was obtained from all of the patients.
Statistical Analyses
Receiver operating characteristic curves were used to determine the optimal cutoff points for the above-described variables, which were then used to divide patients into high and low groups. The demographic and clinical parameters in relation to OS were analyzed by univariate log-rank analyses. A multivariate analysis was performed to analyze the correlations between OS and the variables to further determine an independent prognostic index by using the Cox proportional hazard model. For the survival analysis, we used GraphPad Prism ver. 8.0.2 (GraphPad Software, San Diego, CA, USA) to create survival curves. The characteristics of the patients in the cohorts were analyzed using the Wilcoxon rank-sum test for continuous variables and the Pearson χ2-test or Fisher’s exact test for qualitative variables. Associations were assessed by a determination of hazard ratios (HRs) and 95% confidence intervals (95% CIs). Probability (P)-values <.05 were considered significant. SPSS 22.0 statistical software (IBM, Armonk, NY, USA) and GraphPad Prism 8.0.2 were used for the statistical analyses.
Results
Patients and General Characteristics
Table 1 summarizes the data of the patients’ age, red blood cell (RBC) count, hemoglobin (HGB), platelets (PLT), lymph node metastases, vascular infiltration, SCCA, FIGO stage, BMI, NLR, PLR, MLR, OPNI, SII, and SIRI.
ROC Curves
By calculating the maximum Jorden index, we observed that the optimal cutoff points were as follows: NLR = 1.72, PLR = 111.96, MLR = .24, OPNI = 52.68, SII = 566.23, and SIRI = 1.38 (Figures 1A–F). Receiver operating characteristic (ROC) curves of the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), monocyte-to-lymphocyte ratio (MLR), systemic immune-inflammatory index (SII), systemic inflammation response index (SIRI), and Onodera’s prognostic nutritional index (OPNI). The area under the curve (AUC) values were as follows: NLR = .5801, PLR = .5681, MLR = .5454, SII = .5470, SIRI = .5688, and OPNI = .5909.
Survival Analysis
The Kaplan-Meier analysis showed that a high OPNI, a low SII, and a low SIRI were each significantly associated with better OS (P = .04, P = .03, P = .01, respectively) (Figure 2). There were no significant differences in OS between the high and low patient groups based on the NLR, the PLR, or the MLR. Age > 62 years, A low RBC count, and positive lymph node metastasis status were each significantly associated with poor OS in the patients (P = .018, P = .005, and P = .001, respectively) (Figure 3). A-E: Overall survival (OS) in relation to the NLR, PLR, MLR, OPNI, SII, and SIRI in patients with early-stage, poorly-to moderately-differentiated CSCC. D-F: Kaplan-Meier analyses showed that a high OPNI, a low SII, and a low SIRI were each significantly associated with better OS. A–C: The CSCC patients’ OS in relation to age, RBC count, and lymph node metastasis. Age ≥ 62 years, a low RBC count, and positive lymph node metastasis (LNM) were each significantly associated with poor OS.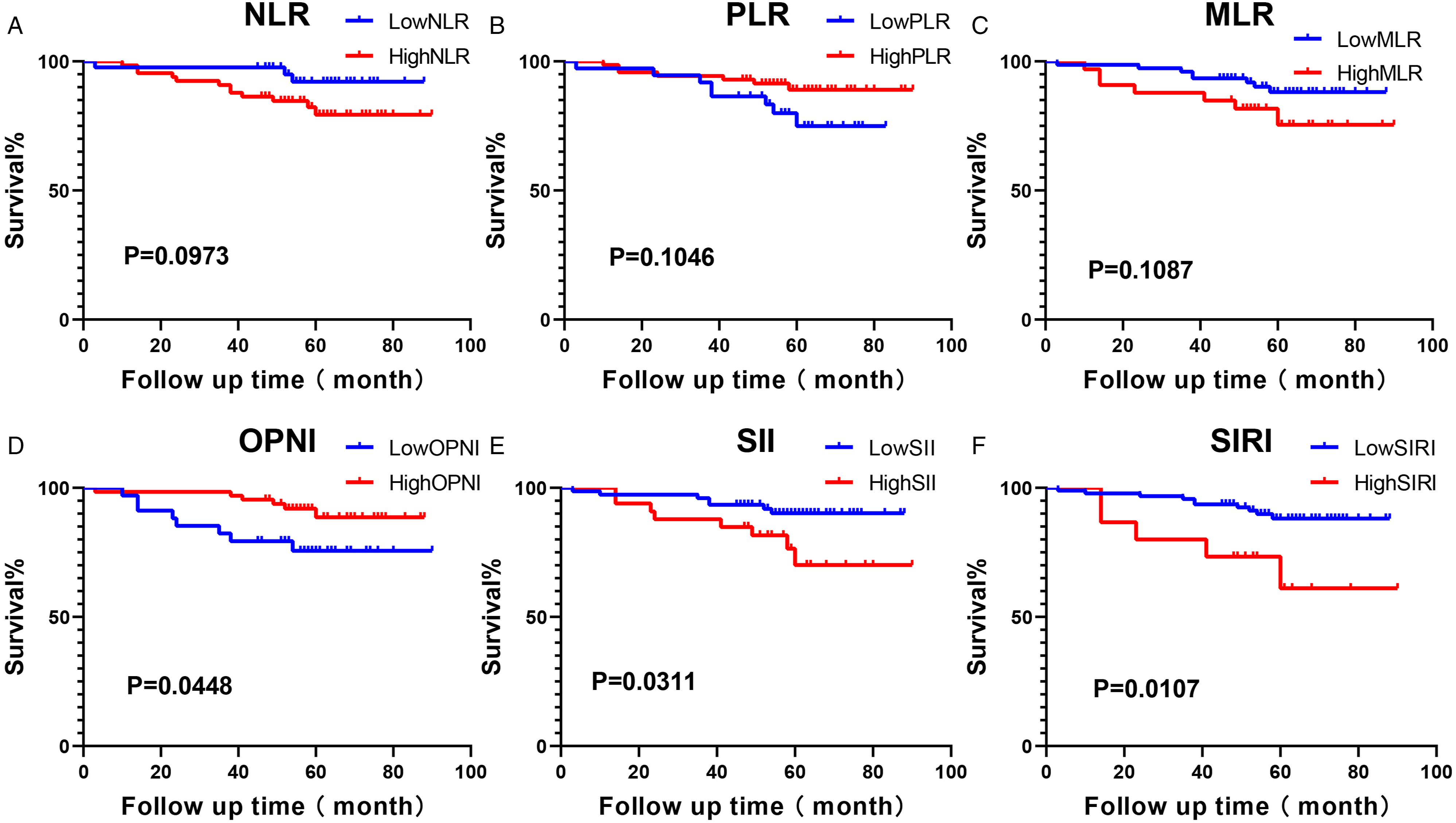

Uni- and Multi-Variate Analyses
Univariate and Multivariate Analysis of Cervical Carcinoma Patients.

Relationships Between the OPNI and Other Clinicopathological Parameters
The Relationships Between the Onodera’s Prognostic Nutritional Index and the Clinicopathological Factors in Patients With Cervical Cancer.

Discussion
Our analyses revealed that patients with early-stage poorly-to moderately-differentiated CSCC who had a high OPNI value achieved better OS compared to those with a low OPNI value. Uni- and multivariate analyses identified the OPNI as an independent marker for this cancer. Malignancy with a higher tumor load may be accompanied by an immunosuppressive tumor microenvironment and malnutrition, both of which can result in low OPNI values. As a result, the OPNI may represent the tumor load and is clearly associated with the prognosis of volume reduction in cervical cancer patients.
The multivariate analysis in a 2016 study revealed that the OPNI was an independent predictive factor for progression-free survival (PFS) and OS in cervical cancer patients receiving concurrent chemoradiation, 14 and Gangopadhyay reported that the OPNI was significantly associated with a clinical complete response to radiotherapy in locally advanced cervical cancer. 15 Similarly, Ida et al. 16 demonstrated that the OPNI was an independent prognostic factor for 12- and 24-month survival and OS in recurrent cervical cancer. However, few studies have evaluated the prognostic significance of the OPNI for patients with early-stage poorly-to moderately-differentiated CSCC. To the best of our knowledge, the present study is the first to report the prognostic value of more markers, for example, the NLR, PLR, MLR, SII, SIRI, and OPNI in patients with early-stage poorly-to moderately-differentiated CSCC. Our findings validate the results of the above-described studies, suggesting that the OPNI could be useful for guiding treatment decisions for patients with early-stage poorly-to moderately-differentiated CSCC.
Accumulating evidence has shown that the tumor immune microenvironment plays a pivotal role in the progression of various malignancies, as tumors can actively subvert the immune system to bring about immune escape, determining the tumor’s ultimate fate.17,18 Numerous indices have been used to measure the immuno-nutritional status of cancer patients, and various immuno-nutritional biomarkers have been observed to be prognostic markers for various solid tumors such as gastric, hepatocellular, colorectal, pancreatic, and epithelial ovarian tumors.19-23 It has also been shown that malnutrition suppresses the tumor immune microenvironment. Malnutrition reduces the ratio of CD4+ to CD8+ T cells as well as the appearance of peripheral immature T cells. 24 Low levels of CD19 lymphocytes can also indicate some nutritional deficiencies. 25 The relationship between circulating lymphocyte counts and malnutrition has thus been shown to be important in identifying nutrient deficiencies. Critically ill patients exhibit a decreased rate of albumin synthesis resulting in hypoalbuminemia, which is often caused by the combined effects of inflammation and malnutrition. Inflammation alone increases the fractional catabolic rate and alters the distribution of albumin between the intra- and extra-vascular compartments. 26
A study by Matsuo et al. 27 demonstrated that among women with squamous cervical carcinoma, grade 2 (44.7%) to 3 (48.1%) lesions were more common than grade 1 (7.2%) lesions, and high-grade tumors frequently have a poor prognosis. That study also demonstrated that the tumor grade is a predictive factor for squamous cervical cancer, particularly in early stages. Higher tumor grades are associated with worse OS. In the multivariable analysis, the grade 2 tumors (HR 1.21, P < .001) and grade 3 tumors (HR 1.45, P < .001) were independently associated with decreased cause-specific survival compared to the grade 1 tumors. The purpose of our study was thus to identify immuno-nutritional indices that can be used to predict the survival of patients with early-stage poorly-to moderately-differentiated CSCC before surgery.
Our results revealed a strong link between lymph node metastases and patient prognosis. The positive lymph node metastases group was significantly associated with poor OS compared to the negative lymph node metastases (LNM) group (HR 4.424, P = .025). A similar study showed that the 5-year recurrence-free survival rate of patients with early-stage cervical cancer was 88% in the non-LNM group and reduced to 57% in the LNM group. 28 LNM is 1 of the most important prognostic factors of cervical cancer, and the results of our present study validate the findings of prior investigations. 29 The 2009 FIGO staging system did not include lymphatic metastasis as a risk factor, but in the 2018 FIGO staging system lymphatic metastasis was classified in stage III. Our results thus further confirm the significance of the 2018 FIGO system.
In routine clinical practice, the FIGO staging system has been used to evaluate the prognosis of CSCC patients. An advanced FIGO stage in CSCC generally predicts a poor prognosis. However, the present study’s univariate analysis showed that the FIGO stage had no prognostic significance for early-stage poorly-to moderately-differentiated CSCC, probably because the 2009 FIGO staging system did not include lymphatic metastasis.
The SII and the SIRI had prognostic value in our univariate analysis, but they showed no significant relationship with OS in the multivariate analysis. In a similar study, Chao et al. 30 observed that compared with the inflammatory indices NLR, PLR, and MLR, the SIRI could be a more promising marker for monitoring the response to therapy among patients with curable cervical cancer. Further, Huang et al. 31 showed that the SII was better at predicting the prognosis of cervical cancer patients undergoing radical resection compared to the NLR, PLR, and MLR; the SII was also an independent prognostic factor for OS in their cohort. There is a discrepancy between our results and those of previous studies; this discrepancy may be due to the differing pathological types among the patients examined.
Our present univariate analysis indicated that a lower RBC predicted worse OS. We thus speculate that increasing the RBC of patients with early-stage poorly-to moderately-differentiated CSCC may contribute to a better prognosis.
In our opinion, age is a vital clinical feature that can affect patients’ nutritional and immune inflammation status. In the present study, the cutoff point for age obtained by the ROC curve analysis best reflected the nutritional and immune inflammation status of the patients and had a greater weight in the subgroup analysis. Accordingly, the results are more reliable.
Of course, there are some study limitations to consider. This was a retrospective study with potential recall bias, and the sample size was limited to the patients treated at 2 hospital during the period from 2014 to 2018. It was difficult to achieve unified quality control of radical hysterectomies performed before 2014 in our region, and the surgical quality of patients treated before 2014 was not uniform. The incidence of positive events in the patients with early-stage poorly-to moderately-differentiated CSCC treated after 2018 has been low due to the better prognosis. In addition, certain confounding variables could not be controlled in this study, and some data are absent. Despite these limitations, this study was a two-center retrospective analysis that included a homogeneous group of histologically identical patients who were treated in the same way. Additional prospective studies with a substantial quantity of available data are needed to confirm our findings. In the future, we hope to implement a prospective randomized controlled trial of preoperative albumin intake to further explore the clinical value of the OPNI.
Conclusion
Our data showed that preoperative OPNI could be useful as an independent prognostic index for early-stage poorly-to moderately-differentiated CSCC, as it can reveal the tumor burden and predict the clinical prognosis to some extent. The OPNI is a simple and inexpensive indicator that can be used in clinical practice to assess a patient’s immunological and nutritional health. This index should be regarded a predictive and prognostic factor for patients with early-stage poorly-to moderately-differentiated CSCC.
Footnotes
Acknowledgments
We thank our gynecology colleagues at Changzhi People’s Hospital and Yuncheng Central Hospital, the patients, and their family members for their support of this study.
Authors’ Contributions
Yan Han was the one who came up with the idea for the study, wrote the report, and analyzed the data. The study’s conceptualization and design were aided by Liang Zong, Wenqing Hu, and Zehua Wang. Jianfei Guo was responsible for material preparation, data collecting, statistical analysis, and manuscript writing. The data was gathered by Weiqin Lv and Yun Shang. The paper was revised by Fan Yang, Xiaomin Zhang, Keyuan Xiao, Shangyunduo Zhang, and Xiaoqi Pan. The final manuscript was reviewed by all authors.
Declaration of Conflicting Interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Innovation Fund of Changzhi People’s Hospital.
Correction (May 2024):
Article has been revised to update first affiliation.
Ethical Statement
This study was reviewed and approved by the ethics committees of Changzhi People’s Hospital and Yuncheng Central Hospital in Shanxi Province, China (No. 2022K004, YXLL2022027). All procedures performed in studies involving human participants were in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. Informed consent was obtained from the legal representatives of eligible patients in accordance with local regulations.
Consent for Publication
Written informed consent was obtained from the patient for publication.
