Abstract
Background:
The 2025 American Diabetes Association guidelines emphasize the benefits of continuous glucose monitoring (CGM) for patients with diabetes receiving insulin therapy. CGM measures interstitial fluid glucose levels, offering an alternative to capillary (finger-stick) devices. This study aimed to evaluate the real-world impact of CGM devices on patients with uncontrolled type 2 diabetes (T2D) compared to traditional blood glucose monitoring (BGM) in a resource-limited population.
Methods:
This is a retrospective study of patients 18 years or older, with T2D, with a glycated hemoglobin (HbA1c) > 9% at enrollment during the study period of December 1, 2021, to February 12, 2023, at an inner-city hospital. Patients with T2D using CGM devices were compared to a historical cohort of patients from the same population using BGM devices in the same period. The primary outcome was the HbA1c at 3 months. In total, 64 patients were included in the analysis after screening: 27 in the CGM group and 37 in the non-CGM group.
Results:
For the primary end point of HbA1c at 3-months, the study found no significant difference between groups. After adjustment for baseline differences (age, HbA1c, creatinine, point-of-care glucose, and number of patients on injectables), average treatment effect (ATE) was −0.48% (SE = 0.27) in favor of the non-CGM group (P = .07). Potential outcome means were 8.8% (SE = 0.17) and 8.3% (SE = 0.2) for CGM and non-CGM groups respectively. Both groups achieved clinically meaningful reduction in HbA1c.
Conclusion:
Our study did not find that CGM titrated regimens resulted in a statistically significant difference in HbA1c change at 3 months compared to non-CGM based treatment. This may indicate that while diabetes technology can help achieve glucose goals in more controlled settings, optimal results in the real world is influenced by many factors, such as insurance coverage, patient adoption, and provider training. More research can be done on identifying factors that yield optimal results in CGM utilization in outpatient settings.
Plain language summary
Key Points Question: Can a Continuous Glucose Monitoring (CGM) technology titrated treatment plan lower glycated hemoglobin (HbA1c) more than a standard finger-stick blood glucose monitoring (BGM) based plan in a resource-limited population with type 2 Diabetes (T2D)? Findings: In this retrospective study involving 64 patients, those who used a CGM based care plan did not have a greater improvement in HbA1c compared to their Non-CGM cohort. Meaning: This study suggests that while CGM is a valuable tool in diabetes management, further research needs to be done on real-world factors that predict optimal benefit from its usage, especially in resource-limited settings.
Keywords
Introduction
Background
Type 2 diabetes (T2D) is one of the most prevalent chronic diseases in the United States. According to the Centers for Disease Control and Prevention (CDC), over 30 million Americans are living with diabetes. 1 In 2022, the prevalence of diabetes in East New York, where this study was done, was 17.1%, and Black, Hispanic, and Asian New Yorkers were at least twice as likely to have diabetes than their White counterparts. 2
Many people living with T2D who experience large glycemic variability are unable to achieve glycemic control with traditional blood glucose monitoring (BGM). The appropriate doses of injectable therapies are dependent and titrated based on the patient’s point-of-care testing of capillary glucose. Obtaining multiple glucose readings a day is necessary for optimal titrating but is painful for the patient and may also be difficult to obtain and trend for healthcare personnel. 3 As a result, many physicians base their titrations on glycated hemoglobin (HbA1c), an average measurement of glucose liable to miss outlying hypo- and hyperglycemia. Continuous glucose monitoring (CGM) is a significant advancement in diabetes management as it provides real-time data on glycemic control, diurnal glucose fluctuations, impending hypo-and hyperglycemia and quick feedback on treatment. 4 The resulting body of data can be utilized, theoretically, to create a superior treatment regimen.
The 2025 American Diabetes Association (ADA) guidelines recommend continuous glucose monitoring (CGM) for patients with diabetes, especially those on insulin or multiple injections, who are capable of using the device safely. 5 However, there is a relative paucity of real-world data evaluating CGM use in low-resource, racially diverse urban populations, where insurance coverage, technological literacy and other socioeconomic factors may impact both access and use. This study addresses this gap by comparing glycemic outcomes in patients with uncontrolled T2D utilizing CGM versus BGM in a medically underserved, socioeconomically disadvantaged community.
Methods
Study Setting
This study was done in the medication management clinic of One Brooklyn Heath-Brookdale, New York, a hospital that provides inpatient and outpatient care in an inner city, urban neighborhood. Median household income in the area was $36,790 in 2022, about 53% less than citywide median household income of $77,550. The poverty rate in the area was 39.1%, compared to 18.3% citywide. 6 The medication management clinic is a pharmacist-led clinic where qualified pharmacists and a prescribing primary care physician practice under a collaborative practice agreement to manage chronic diseases.
Ethics Approval and Written Informed Consent
The study was conducted in accordance with the Declaration of Helsinki and was approved by the One Brooklyn Health Institutional Review Board (no. OBH I-22-19) on April 25th, 2022, with the need for written informed consent waived.
Study Design
This is a retrospective cohort study. The study includes patients 18 years of age and older with a diagnosis of T2D, glycated hemoglobin (HbA1c) above goal at enrollment, and CGM-titrated care. These patients were retrospectively compared to patients with non-CGM based care on age, baseline injectables, baseline HbA1c, serum creatinine, and point-of-care (POC) glucose. Included patients needed to have at least two visits between December 1st, 2021, to February 12, 2023. Participants were excluded if they had less than 1 visit for the CGM titrated cohort or less than 1 scan daily or no available data, less than 2 visits within the first month for the non-CGM cohort, or less than 1 HbA1c recording in a 6-month period for both cohorts. A comprehensive chart review of medical records was conducted with de-identified, summative data, with no randomization or blinding of the subjects.
Patient visits were conducted by pharmacy residents and Clinical Pharmacists. All participants were informed that they could freely choose CGM or opt for traditional blood glucose monitoring.
Any medication titration required was performed by Clinical Pharmacists as per the collaborative practice agreement. Education via didactic teaching or provision of visual aids was conducted by clinical pharmacists and/or pharmacy residents or students. For patients in the CGM arm, patients’ phones were configured with the CGM application or CGM receivers were set up as readers if they did not have a compatible smart phone. Patients were trained on how to appropriately scan the device using their receiver or cell phone. The importance of re-scanning was emphasized at appointments. Trained interpreters were used as needed.
Visit cadence for follow-up was individualized based on acuity and needs of the patient. Most often, follow-up visits were scheduled every 4 weeks with some patients requiring more frequent follow-up every 2 weeks. Oftentimes those who required more frequent visits were ones who experienced hypoglycemia due to self-titration of their insulin or skipping of meals. At each visit, medication regimens were evaluated and titrated based on the patient’s ambulatory glucose profile (AGP) report. A standard template and protocol were used to guide shared clinical decision making.
Patients using BGM titrated plans used similar diet, exercise, and medication management counseling as the CGM group, with only the CGM-specific treatment omitted. They had at least 2 visits in the first month of enrollment to the clinic and titrations were based on their reported self-monitored blood glucose (SMBG).
Patients’ individualized goals were determined by shared decision making in line with American Diabetes Association guidelines, <7% for most nonpregnant adults; and less stringent targets (i.e. < 8%) for older adults with complex/intermediate health. 7
The treatment protocol and patient education was structured and standardized as the medication management clinic is built into a teaching pharmacy program, and a reasonable amount of fidelity in the process from patient to patient can be surmised.
HbA1c was tested with whole blood at a central lab by turbidimetric immunoinhibition methodology. Interpretative ranges were based on institution policy and used consistently for all testing. Given the facility’s inability to measure HbA1c > 15.5% at the time, all patients who had HbA1c exceeding that, were documented as 15.5%.
Primary, Secondary, and Safety Endpoints
The primary endpoint was the change in HbA1c at 3 months. Secondary endpoints included HbA1c at 6, and 9 months, number of patients achieving their individualized HbA1c goal, percentage change in HbA1c from baseline to 3 months, diabetes-related hospitalizations, and emergency room visits. Safety endpoints were patient-reported adverse events.
Statistical Analysis
This study evaluated the effectiveness of CGM via HbA1c levels measured as a percentage. Since this is a pilot study, statistical power was not calculated. All potential candidates from the initiation of CGM-based care were considered, maximizing sample size.
Patients’ demographics were summarized using descriptive statistics such as mean with standard deviation, median with interquartile range, frequency with percentage and compared using two-sample t-test, the Pearson’s chi-square test, Fisher’s exact test, and the Wilcoxon rank sum test. A propensity score method was utilized to minimize confounding effects and improve statistical efficiency. Inverse Probability Weighing was used rather than propensity score matching to avoid reducing the sample size.
Our primary outcome, measured as HbA1c at 3 months post CGM initiation, was analyzed using multiple regressions using the average treatment effect (ATE) approach. All analyses were done in SAS 9.4®.
Results
Participants
A total of 90 patients’ charts were screened, and 64 eligible patients were included in the analysis with 27 patients in the CGM group and 37 patients in non-CGM group. Twenty-six patients were excluded due to self-discontinuation or being lost to follow-up or. There were no differences between the groups with respect to sex and race with P-value of .92 and .59 respectively. However, there was a difference in age at baseline with younger patients included in the CGM group as compared to the non-CGM group with the mean (SD) age of 55.6 (11.5) and 63 (12.4) years, respectively (P = .02; Table 1).
Baseline Demographic Characteristics of the Study Sample.

Parameter that was controlled in the statistical model.
The majority of baseline clinical characteristics were similar between the groups except for means (SD) of HbA1c (CGM, 11% (2.1); non-CGM, 9% (2.1); P < .001), serum creatinine (SCr) (CGM, 0.9 mg/dL (0.3); non-CGM, 1.2 mg/dL (0.6); P = .05), and point of care (POC) glucose (CGM, 220 mg/dL (83.4); non-CGM, 172 mg/dL (52.2); P = .02). Also, the groups were different in number (%) of patients on injectable (CGM, n = 26 (96); non-CGM, n = 24 (65); P = .002), number of injectables per patient (P < .001), and number (%) of patients on basal insulin (CGM, 24 (89); non-CGM, 13 (35); P < .001; Table 2).
Baseline Clinical Characteristics and Medication Use of the Study Sample.
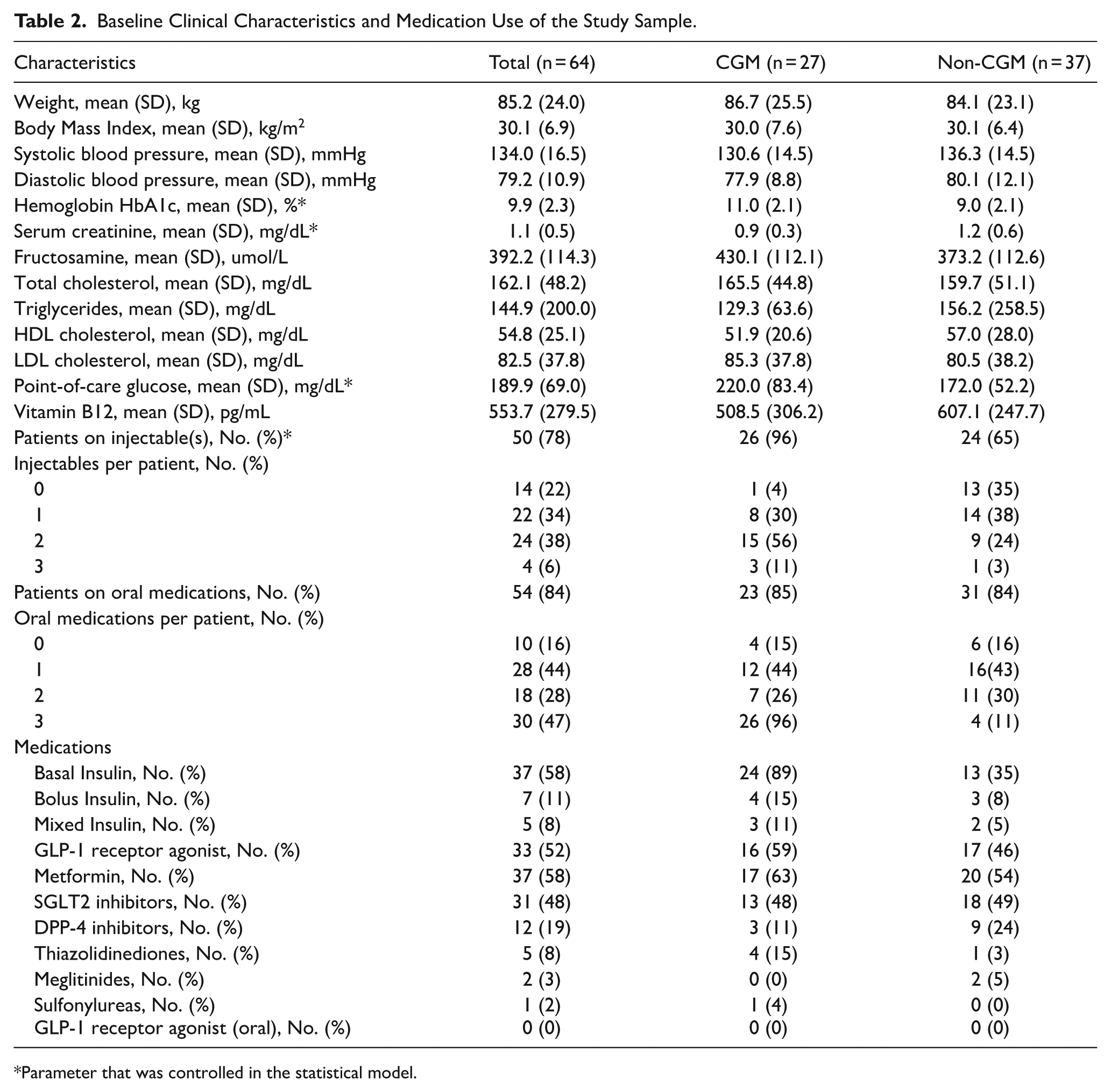
Parameter that was controlled in the statistical model.
Outcomes
Primary Endpoint
For the primary endpoint HbA1c at 3-months, the study found no significant difference between groups. Independent variables age, HbA1c, SCr, POC glucose, and number of patients on injectables were controlled in the statistical model. After adjustment for baseline differences, (ATE) (standard error (SE) was −0.48% (0.27) in favour of the non-CGM group (P = .07). The observed HbA1c means (SE) at 3 months were 8.8% (0.17) and 8.3% (0.2) for CGM and non-CGM groups, respectively.
Secondary Endpoints
HbA1c at 6-months period was not different between groups with ATE (SE) of −0.9% (0.66) and P = .18. At the 9-months interval, the analysis showed a significant difference with ATE (SE) of −1.4% (0.71) and P = .049 in favour of the non-CGM group.
With respect to percentage change in HbA1c from baseline to 3-,6-, and 9-months periods, the study found no difference between groups with adjusted average differences of .29 (P = .25), .17 (P = .8), and .26 (P = .73) respectively. The numbers of patients achieving their individual HbA1c goal during the study period were similar between the groups with P = .72.
Safety Endpoint
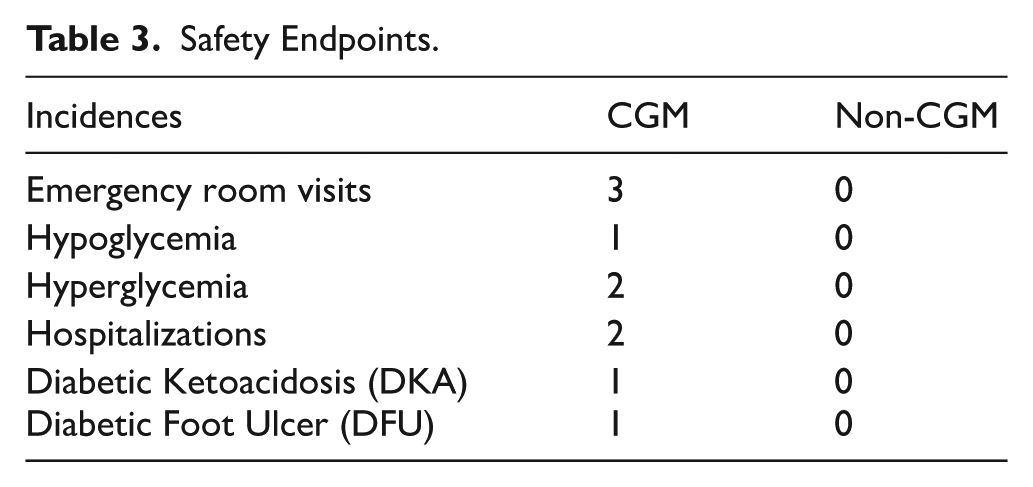
Patients in the CGM group tolerated CGM devices well. There were a few incidences of diabetes related to re-hospitalizations or emergency room visits in the CGM group compared to patients in the non-CGM group (Table 3). No other serious adverse drug reactions were reported in medical records.
Safety Endpoints.
Discussion
This study evaluates the effect of CGM on glycemic control in a racially diverse, low-resource population, mostly using public insurance. While studies have shown CGM to improve glycemic control in both type 1 and type 2 diabetes, there is a paucity of studies looking into aspects of the real-world use of CGM in patients with T2D, especially in non-White populations living in economically disadvantaged urban areas. In this study, people with uncontrolled T2D using CGM in a medication management clinic in a medically underserved, low-income area of New York City were retrospectively compared to patients in the same clinic who used a BGM-based approach.
Our study found that both CGM and BGM used to self-monitor glucose were related to a clinically significant reduction in HbA1c, generally considered to be a decrease of 0.3%-0.5% or more. Despite initial expectations that using CGM as a tool would yield superior results, the CGM titrated regimens did not result in a statistically significant difference in HbA1c change at 3-6 months over BGM after adjustment for the covariates (HbA1c, Age, S.Cr, point-of-care glucose, and number of patients on injectables at baseline). However, both groups saw improved glycemic control, with CGM and non-CGM cohorts seeing 3-month average reductions in HbA1c of 2.2% and 2.9%, respectively.
The findings also showed no statistically significant difference in HbA1c change in using CGM instead of BGM at 6 months, similar to some studies,8,9 but diverging from others.10,11
At the 9-month interval, we found a statistically significant difference in favour of the non-CGM group (P = .049). 8 While the reasons for this are unclear, we can hypothesize it to be due to ‘CGM fatigue’ where decision fatigue from dealing with increased amounts of data and alerts diminishes long-term engagement with the device.12,13 Further research can be done to explore the sustainability of CGM use in diverse populations.
While the majority of baseline characteristics were similar between the groups, the CGM arm had a higher mean baseline HbA1c (11% vs 9%) and more frequent use of injectable therapies (96 % vs 65%). Additionally, 89% of the CGM group were on basal insulin compared to 35% for their non-CGM counterparts. Whether these differences reflect more stringent insurance requirements for approval and/or clinical urgency in initiating CGM for patients with a higher HbA1c, can be a subject for further studies in the future.
The CGM group also had a lower mean age at baseline than the non-CGM group. It can be surmised that the use of technology was challenging to adopt for some older adults, despite the potential benefits of CGM in this population. 14 Among those who did use CGM in the resource-limited setting of this study, technology remained a barrier as explored in other studies, considering 51.9% of patients in the CGM arm did not own a smart phone compatible with their mobile application and had to use a separate reader which they sometimes forgot to carry. 15
One of the limitations of the study is a small sample size, partly stemming from difficulty in obtaining CGMs due to limited insurance coverage. Since this is a pilot study, statistical power was not calculated. However, this may limit generalizability of the results to other settings, especially those with more resources. In this study setting, the local enrollment rates in public health insurance (41.9%) exceeded national rates according to state comptroller data, and 32.4% of the population were enrolled in Medicaid for low-income adults. 16 Unsurprisingly, more than 80% of the patients in the CGM arm were insured by Medicaid/Medicare, which has stricter guidelines for CGM approval than commercial insurance. At the commencement of the study, obtaining prior approval took a trained staff member, an average of 15.6 days (range 0-76 days). It may be considered sufficiently time and labor-intensive to be a barrier to the use of CGM in usual primary care settings.
Another limitation was the short study period of 3 months as a primary outcome. This period was selected because the highest number of follow-up visits usually occurred during this time, ensuring that adherence was broadly high across both groups. Patients and providers were often most engaged at this time, thus maximizing intervention and implementation fidelity. Additionally, the difficulty of maintaining longitudinal follow-up in an inner-city, low-resource population, necessitated the need to focus on shorter follow-up periods. However, it can be hypothesized that, given the time required to obtain, educate, and fully adopt CGM use, its impact on diabetic control may not be accurately reflected in a short time period.
Third, the pharmacist-primary care physician collaborative setting provided more clinical support to the patients than may be available to the usual primary care environment, limiting generalizability of the study findings.
Despite its limitations, our study shows that crafting effective care plans using either BGM or CGM as a tool can improve diabetes control. Therefore, the decision to use CGM in a resource-limited setting should be through shared decision-making, considering the patient’s circumstances, capabilities, means, and preferences. 17 Additionally, the investment of time required to establish a CGM regimen should be foreseen, which may include delays due to insurance coverage and time required for successful patient adoption, and a bridging BGM-based care plan should be in place. For sustainability through longer periods, patients’ motivation and interest in CGM may periodically be assessed to avoid technology burnout. 18
Our study did not look at additional benefits of CGM, such as reduction of hypoglycemia, increase in Time in Range (TIR), inability to check BGM, or increased patient satisfaction, all of which may be additional reasons for adopting CGM over BGM, rather than solely HbA1c reduction when resources are limited. More research should be done to identify factors that may yield optimal results in CGM utilization in low-resource urban settings. It’s possible that controlled trials with strict protocols may find their results to be less generalizable to such settings, leaving a need for studies like ours that primarily look at real-world scenarios where patients and providers alike are limited by time and resources.
Conclusion
Our study did not find using CGM as a tool was associated with a statistically superior glycemic outcome over BGM at short periods, when significant real-world barriers were present. More research should be done on identifying factors that optimize CGM use. In the long run, exploring factors that influence sustainable integration of CGM use in diverse, resource-strapped settings may provide insights into creating equitable opportunities for all to obtain and use advanced diabetes technologies when needed.
Footnotes
Acknowledgements
We gratefully acknowledge the assistance of the Dr. Aimee Affable, PhD, MPH, Associate Professor of Community Health Sciences, Downstate Health Science University for helping to draft the protocol; Dr. Chris Paras, DO, FACP, Interim Executive Director, One Brooklyn Health - Brookdale for providing overarching guidance; Dr. Ankhnu Uaskhem, Attending Provider at Medication Management Clinic, One Brooklyn Health - Brookdale for assistance with creating treatment plans; Kenneth Chiu, MPH, MD/MBA student at SUNY Upstate Medical University for helping to draft the analysis plan for the protocol, and the following providers for identifying eligible patients, collecting data and creating treatment plans based on CGM/BG data: Sami Barakat, PharmD, BCACP, AAHIVP, Clinical Pharmacy Specialist, Northwell Health; Justin Ramnarain, PharmD, BCACP, Clinical Pharmacy Specialist, Northwell Health; Da Som Kim, PharmD, BCACP, CDTM Pharmacist, Kings County HHC; Kathleen Horan, PharmD, BCACP, Assistant Clinical Professor of College of Pharmacy & Health Sciences, St. John’s University; Yuliana Toderika, PharmD, BCACP, Associate Professor of Pharmacy Practice, Long Island University and The Arnold & Marie Schwartz College of Pharmacy and Health Sciences. Contributors were not compensated for their work outside normal salary.
ORCID iDs
Ethical Considerations
Ethical approval was obtained from One Brooklyn Health Institutional Review Board (One Brooklyn Health, (1 Brookdale Plaza, Brooklyn, NY 11212). Ethics Approval number: OBH I-22-19. Date approved: April 25th, 2022.
Consent to Participate
The requirement of written informed consent from the subjects prior to study initiation was waived by the Institutional Review Board.
Consent for Publication
Not Applicable.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Qudsia Banu: The work was supported as a faculty fellow by the National Institute on Minority Health and Health Disparities (Award Number S21MD012474). The funder has no part in the design and conduct of this study. Rose Calixte: The work was supported as a faculty fellow by the National Institute on Minority Health and Health Disparities (Award Number S21MD012474). The funder has no part in the design and conduct of this study. No other funding were reported for other authors
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Banu received grant funding from the National Institute on Minority Health and Health Disparities to their institution for the conduct of the submitted study. Dr. Calixte also received grant funding from the National Institute on Minority Health and Health Disparities. No other authors declared any potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data available: Yes. Data types: Deidentified participant data. How to access data: Contact corresponding author. When available: With publication Supporting Documents. Document types: Statistical/analytic code. When available: With publication. Who can access the data: Researchers whose proposed use of the data has been approved. Types of analyses: For study purpose. Mechanisms of data availability: After approval of a proposal with a signed data access agreement. Any additional restrictions: None.
