Abstract
Background
The Swedish National Diabetes Register (NDR) initiated registration of the FreeStyle Libre® system and other continuous glucose monitoring (CGM) systems in June 2016. We investigated change in HbA1c for people with type 2 diabetes (T2DM) using FreeStyle Libre in Sweden.
Methods
We included adults with T2DM, registered in the NDR after January 1, 2014, and an index date for first use of FreeStyle Libre of June 2016 or later. Methodology was a before/after comparison of HbA1c within 6 months before the index date versus HbA1c around 6 and 12 months after the index date.
Results
711 adults with T2DM using FreeStyle Libre had HbA1c measurements within the study period. Mean HbA1c was significantly reduced at 6 months (−0.50%-unit) and at 12 months (−0.52%-unit) in this group. Degree of change was negatively correlated to baseline HbA1c. Reductions in HbA1c were observed in incident users of FreeStyle Libre with T2DM who were truly naïve to CGM or had unknown prior experience of CGM, and aged 25–74 years.
Conclusions
This real‐world study on the Swedish NDR shows that people with T2DM using FreeStyle Libre system for 6 and 12 months significantly reduced their HbA1c.
Keywords
Introduction
In Sweden, approximately 5.5% of the population have diabetes, 1 the majority of whom have type 2 diabetes (T2DM). The Swedish National Diabetes Register (NDR) was founded in 1996 and is an integral part of diabetes care in Sweden, covering both primary and secondary care. The NDR aims to monitor and help improve diabetes care, reducing diabetes-related morbidity and enabling comparisons between a number of important clinical outcome measures. Nationwide registration of people with diabetes in Sweden is encouraged at least once a year. By January 2019, the register covered 435,093 adults recorded as having diabetes during the preceding 12 months, The NDR includes approximately 90% of all patients aged ≥18 years diagnosed with T2DM in Sweden. Healthcare providers report continuously directly to the NDR or via electronic patient records from routine clinical practice. 1
In June 2016, the NDR initiated documentation of the usage of sensor-based continuous glucose monitoring (CGM) including flash glucose monitoring with the FreeStyle Libre® system (Abbott Diabetes Care, Witney, Oxon, UK) amongst adults with diabetes and thus created the opportunity to systematically investigate the impact of the FreeStyle Libre system in Sweden. Since 2018, the Swedish National Board of Health and Welfare has recommended sensor-based glucose monitoring for people with type 1 diabetes (T1DM), and also for people with T2DM who are treated with multiple daily insulin injections and have problems with recurrent hypoglycemia or hyperglycemia.2,3
Landmark studies have proven that good glucose control, as measured by reduced glycated hemoglobin A1c (HbA1c), is strongly associated with lowered risk of diabetes complications for adults with T2DM. The UK Prospective Diabetes Study4,5 demonstrated that a lowering of HbA1c is associated with clinically significant reductions in microvascular complications and long-term macrovascular disease. Randomized clinical trials (RCTs) indicate that, in comparison to self-monitored blood glucose (SMBG) testing, real-time CGM systems can reduce HbA1c in people with T2DM on insulin therapy. 6 In studies using the FreeStyle Libre system to date, the REPLACE 7 RCT, showed no change in HbA1c over / 26 weeks comparing the FreeStyle Libre system with SMBG in the total study population, whereas one other RCT 8 and several real-world observational studies9–11 have shown significant reductions in HbA1c amongst adults with T2DM on insulin or non-insulin therapy using the FreeStyle Libre system.
The aims of this study were: (a) to assess the data collected within the NDR, regarding both the incident and prevalent users of the FreeStyle Libre system amongst individuals with T2DM in Sweden since mid-2016, stratified by type of diabetes treatment and (b) to analyze changes in recorded HbA1c levels in people with T2DM, before and after initiating the FreeStyle Libre system, including subgroup analyses according to prior metabolic control and age.
Methods
This retrospective cohort study used data extracted from the NDR covering the period from 1st January 2014 to the 25th June 2019. The study included adults (≥18 years old) with T2DM with a diabetes clinic visit recorded in the NDR after 1st January 2014 and recorded use of the FreeStyle Libre system with an index date of June 2016 or later. The Index date is the date of the first registration where the FreeStyle Libre system use is recorded in the NDR for a person with T2DM. There were no specific exclusion criteria. The T2DM diagnosis was based on clinician’s diagnosis in primary or hospital-based diabetes out-patient clinics.
Data completeness
In this study, we have focused on understanding the association between new incident users of the FreeStyle Libre system and two distinct variables within the NDR. These are HbA1c values and prior use of CGM. Data were collected in line with international consensus standards on HbA1c reporting in mmol/mol and converted into % units according to the IFCC reference system for national standardization.12,13 As with all registries, missing values in each of these categories will occur if the information is unknown, or if the assessment was not conducted or recorded by the responsible healthcare professional. Within the NDR cohort of interest, the relevant data completeness is provided in Supplementary Figure S1.
Incident use of the FreeStyle Libre system in individuals with type 2 diabetes
All individuals with T2DM and an NDR index date for first use of the FreeStyle Libre system from June 2016 to June 2019 were identified within each calendar year. These new incident users were then categorized based on their known or possible use of CGM (other than FreeStyle Libre) prior to their FreeStyle Libre index date. These categories were: (a) truly naïve, with confirmed absence of use of CGM prior to the index date; (b) new incident users with unknown prior status; and (c) new incident users with documented use of CGM prior to the index date. Because of the diagnosis of T2DM, the expectation is that new incident users with unknown prior status (group b) are most likely new incident users. The identification and selection process for new incident users is described in the Supplementary data in Supplementary Figure S2.
Numbers of new incident and prevalent users of FreeStyle Libre with T2DM categorized by prior use of continuous glucose monitoring.
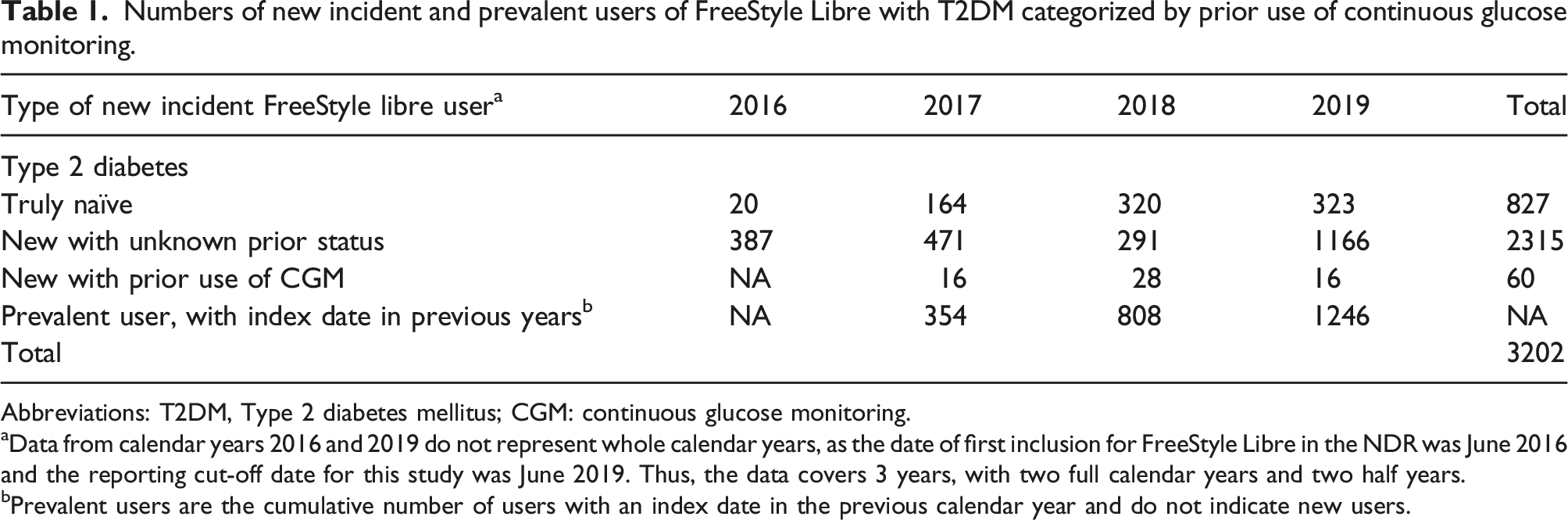
Abbreviations: T2DM, Type 2 diabetes mellitus; CGM: continuous glucose monitoring.
aData from calendar years 2016 and 2019 do not represent whole calendar years, as the date of first inclusion for FreeStyle Libre in the NDR was June 2016 and the reporting cut-off date for this study was June 2019. Thus, the data covers 3 years, with two full calendar years and two half years.
bPrevalent users are the cumulative number of users with an index date in the previous calendar year and do not indicate new users.
Assessing change in HbA1c amongst incident FreeStyle Libre users with T2DM
HbA1c is a recorded variable for people with diabetes in the NDR and in T2DM is typically measured once a year but sometimes more often, We compared the latest laboratory measured HbA1c value within 6 months prior to index date per person with T2DM versus the HbA1c value recorded between day 91–day 272 after the index date that was closest to the 6-month timepoint (day 181.5) and also between day 272–day 455 after the index date that was closest to the 12 month timepoint (day 363.5). HbA1c measurements were available within the defined before and after periods for a subset of the total study population who were incident FreeStyle Libre users. Based on these criteria, change in HbA1c was evaluated for all new incident users with T2DM based on: baseline HbA1c prior to the index date, and age of the subjects. Data for change in HbA1c are presented as absolute mean change in % HbA1c units from baseline at 6 months, not % change as a proportion of baseline.
Ethical approval
The study protocol has been approved by the Swedish Ethical Review Authority, Dnr 2019-03089
Results
Number of new incident users of the FreeStyle Libre system with type 2 diabetes identified in the Swedish National Diabetes Register between June 2016 and June 2019.

Data are presented as absolute number of subjects (% of total).
Abbreviations: T2DM: Type 2 diabetes mellitus; NDR: Swedish National Diabetes Register.
aNew incident users are defined as having an NDR index date for first registration of the FreeStyle Libre system between June 2016 and June 2019.
Number of new incident users of the FreeStyle Libre system with HbA1c readings before and after index date a .

aNew incident users are defined as having an NDR index date for first registration of the FreeStyle Libre system between June 2016 and June 2019.
bInsulin use or non-insulin use was confirmed where possible based on NDR registration data.
Incident use of the FreeStyle Libre system in people with T2DM
In T2DM, there were 827 (26%) truly naïve adults FreeStyle Libre users (Table 1) as defined by the selection process in Supplementary Figure S2. The most common profile for individuals with T2DM and an index date in the NDR was as new to FreeStyle Libre but with unknown prior status (n = 2243, 72%). As indicated previously, most of the users in this group are likely to be naïve to CGM prior to FreeStyle Libre. The number of adult users with T2DM who were new to FreeStyle Libre but with prior experience of CGM was 60 (2%).
Incident users with T2DM and HbA1c measurements within the defined evaluation period
Based on the definitions above, we identified 711 (22%) incident users with T2DM who had HbA1c measurements registered in NDR that aligned with the 6-months post-index timepoint and 538 (17%) with HbA1c readings at the 12-month timepoint (Table 3). These incident users were the subject of subsequent evaluation of change in HbA1c following first registration of the FreeStyle Libre system in the NDR. Change in HbA1c based on prior metabolic control and age group categorization focused on incident users with T2DM with either confirmed use of insulin therapy (n = 655) or confirmed non-insulin therapy (n = 35) (Table 3).
Change in HbA1c in T2DM after first registration of the FreeStyle Libre system in the Swedish National Diabetes Register
Figure 1 shows incident users with T2DM with a recorded HbA1c before and after the index date and a significant reduction of −0.50% HbA1c units at 6 months (n = 711) and −0.52%-unit at 12 months (n = 538) following their first registration of the FreeStyle System in the NDR (p < 0.0001 in each case; Figure 1). The decrease was similar for truly naïve users, with reductions at 6 months of −0.67%-unit and −0.66%-unit at 12 months (p < 0.0001 in each case). Users with unknown prior use of CGM also experienced a decrease in HbA1c at 6 months (−0.43%-unit; p < 0.001) and at 12 months (−0.49%-unit: p < 0.0001). In the small group of T2DM users with documented prior use of CGM, there were no significant changes in HbA1c from baseline either at 6 or 12 months. Change in HbA1c in type 2 diabetes at 6 and 12 months after first registration of the FreeStyle Libre system.
Change in HbA1c at 6 months stratified by baseline HbA1c
Change in HbA1c at 6 months in T2DM treated with insulin stratified by baseline HbA1c.

Data for change in HbA1c are presented as absolute mean change in % HbA1c units from baseline at 6 months, not % change as a proportion of baseline.
Abbreviations: 95% CI, 95% confidence interval. T2DM: Type 2 diabetes mellitus; CGM: continuous glucose monitoring.
aInsulin use was confirmed where possible based on NDR registration data.
bPaired sample t Test for comparison of mean baseline HbA1c with mean HbA1c at 6 months following first registration in NDR of the FreeStyle Libre system.
Change in HbA1c at 6 months stratified by age
Change in HbA1c at 6 months in T2DM stratified by age range.
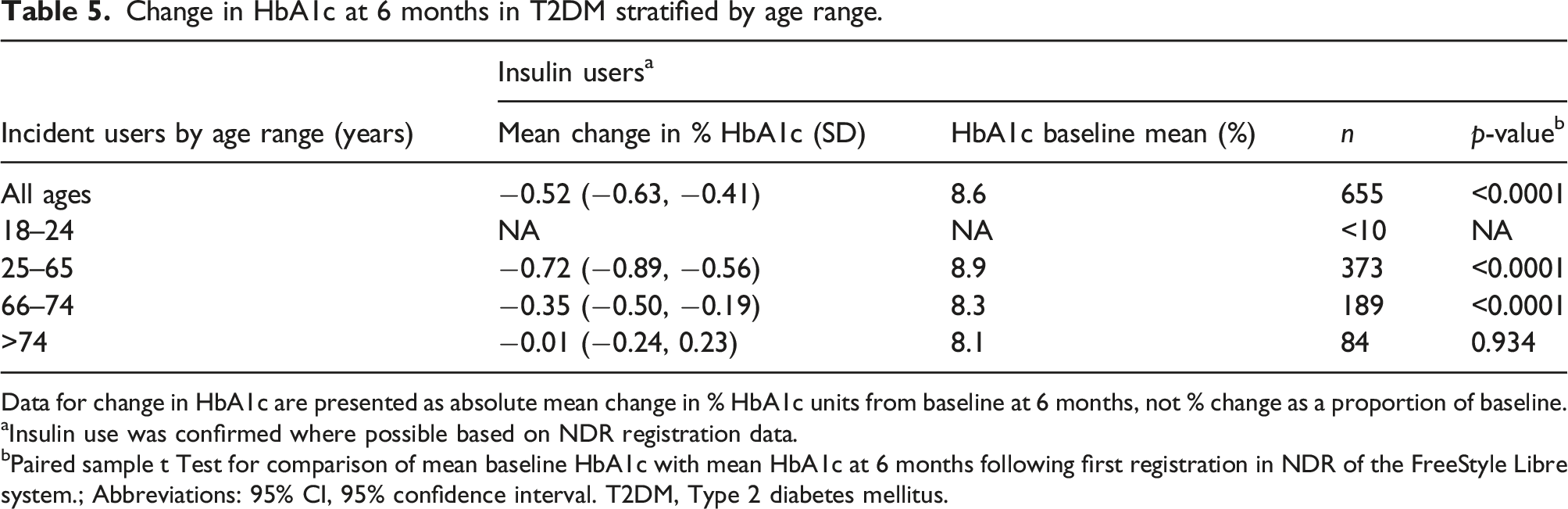
Data for change in HbA1c are presented as absolute mean change in % HbA1c units from baseline at 6 months, not % change as a proportion of baseline.
aInsulin use was confirmed where possible based on NDR registration data.
bPaired sample t Test for comparison of mean baseline HbA1c with mean HbA1c at 6 months following first registration in NDR of the FreeStyle Libre system.; Abbreviations: 95% CI, 95% confidence interval. T2DM, Type 2 diabetes mellitus.
Discussion
The main findings of this retrospective cohort study using the Swedish NDR are revealing as they relate to change in HbA1c amongst incident users of the FreeStyle Libre system with T2DM. Across the study period 3202 adults with T2DM had at least one registered use of the FreeStyle Libre system in the NDR between 1st June 2016 and 25th June 2019 (Table 1). Amongst this population, HbA1c measurements were available within NDR such that it was possible to analyze this aspect of glycemic performance for 711 adults with T2DM before and after the index date for FreeStyle Libre use. Equally, HbA1c data in NDR enabled an assessment of metabolic control prior to the index date, such that the association between change in HbA1c and use of flash glucose monitoring in adults with T2DM could be evaluated in this context.
For incident users of FreeStyle Libre with T2DM, the NDR data reveal a significant association between FreeStyle Libre use after the index date and reductions in laboratory measured HbA1c. There was an observed decrease in HbA1c across the total incident population of −0.5%-unit at 6 months which was maintained at 12 months. Within the incident user group with T2DM, those truly naïve to prior use of CGM experienced reductions in HbA1c of −0.67%-unit at 6 months and −0.66%-unit at 12 months. People with unknown prior use of CGM also achieved reductions in HbA1c, both at 6 months (−0.43%-unit) and at 12 months (−0.49%-unit). This significant but lower reduction compared to truly naïve users is arguably because of the presence in this group a significant proportion of people with no previous experience of CGM. This is supported by the observation that the group of incident users with documented prior experience of CGM did not experience a significant change in HbA1c at either timepoint (Figure 1). However, it must be noted that the user group with prior experience of CGM is a small proportion (<7%) of the total incident user population at both timepoints. These significant reductions in HbA1c observed in T2DM in our study are comparable with those previously reported in smaller-scale RCTs and real-world studies for adults treated on insulin or non-insulin therapy, who have no documented prior experience of flash glucose monitoring.8,9,11
Truly naïve users with T2DM on insulin treatment will benefit from using the system as a result of the technical and psychological support that comes from short-term and longer-term awareness of trends in their daily glucose levels, allied to the immediate feedback that they get about impending hypoglycemia or prandial excursions.14,15 Some of the users with unknown prior use of CGM will see the same benefit, as they will in reality also be naïve. Others in this group will have prior undocumented experience with CGM and can have developed behaviors that aid in improved daily glucose control, independent of the device itself.
The value of good glucose-control behaviors independent of the application of FreeStyle Libre is also supported by the data on HbA1c change observed for the separate groups of users stratified by baseline HbA1c prior to the index date (Table 4) and insulin use status. Incident users of the FreeStyle Libre system with better initial control, as evidenced by baseline HbA1c <8.0%, did not achieved any significant change 6 months after the index date, irrespective of their insulin or non-insulin treatment status. It is possible that the glycemic control objective for this group is focused on avoiding hypoglycemia whilst maintaining their HbA1c, rather than reducing it further. In our study, reductions in HbA1c at 6 months after the index date were more notable for incident users with higher starting baseline values ≥8.0% and greatest for those with HbA1c ≥12.0%. These observations are in line with smaller real-world studies indicating that the degree of change in HbA1c for people with diabetes using the FreeStyle Libre system is directly correlated with baseline HbA1c in T2DM.8,9,16 It is clear from our data that significant reductions in HbA1c across the total population of incident users at 6 months after the FreeStyle Libre index date are driven by substantial reductions in those individuals with higher baseline HbA1c measurements.
Our analysis of the data from the NDR show that the benefits of reduced HbA1c after initiating the FreeStyle Libre system are extended across adults aged 25–74 years old with T2DM who are confirmed insulin users (Table 5). Non-insulin users did not show a change in HbA1c, however this group contained mainly people with T2DM and good baseline glycemic control with HbA1c <8.0% (Table 4) where change was also not significant in the insulin user group. Our study could not confirm a previous prospective study on 49 individuals with T2DM on non-insulin therapy and a baseline HbA1c of 7.8% which did show a significant reduction (−0.46%-unit; p < 0.001) at 24 weeks. 11 However, comparisons cannot be realistically drawn between a prospective RCT and our retrospective real-world analysis. Another important outcome from our data is that improvements in glycemic control amongst adults with T2DM on insulin aged 66–74 years are achievable using the FreeStyle Libre system. This extends previous studies reporting significant reductions in HbA1c using CGM in subjects with a mean age of 67 years with T2DM. 17 Use of sensor-based glucose monitoring systems in older and elderly people with diabetes has focused on reducing the adverse risks and consequences of hypoglycemia and hyperglycemia in this high-risk population18,19 rather than directly reducing HbA1c. Although our data cannot be interpreted in this context, they do indicate that improvements in long-term glycemic control are possible for older people with T2DM. Although the reductions in HbA1c appear to be attenuated in the 66–74 years age group compared to the 25–65 year old subjects (−0.35% versus −0.72%), it is not possible to conclude whether this is due to a genuine age-dependent response to using the FreeStyle Libre system, or whether it reflects the known correlation between the scale of reduction with starting HbA1c,9,16 since the baseline HbA1c of the 66–74 years age group is 8.3% compared to 8.9% amongst the 25–65 years age group. Further research on the response of younger versus older people with T2DM using the FreeStyle Libre system is likely to be valuable. In this context, it must be acknowledged that HbA1c is only one aspect of glycemic control, and that other measures of glycemic health in T2DM are improved by using the FreeStyle Libre system, such as frequency and time spent in hypoglycemia, 7 which could be of more value in older populations, compared to change in HbA1c.
Limitations and strengths
It is important to acknowledge that our study has limitations. First, of the 3202 people with T2DM identified as users of the FreeStyle Libre system, only 711 (22%) had HbA1c test readings recorded in the NDR with the 6-month post-index timepoint, falling to 538 (17%) at 12 months. Although up-to-date HbA1c readings are captured in the NDR as part of clinical reviews and transferred from electronic health records, patients may not attend as frequently as requested, thus some people with T2DM will register fewer readings within the 12-month study timeframe. Non-attendance for someone with T2DM can occur for many reasons, including poor diabetes control or the concurrent management of comorbidities, so it is not possible to infer a selection bias in the reduced size of the 12-months cohort. A longer observational period could help better understand the longitudinal trend in HbA1c. Further, our analysis does not include a control group from within the NDR, matched for baseline characteristics but who were not incident users of the FreeStyle Libre system between June 2016 and June 2019. This is a limitation since it would have highlighted the impact of other factors on glycemic control over the study period, independent of FreeStyle Libre. Since use of the FreeStyle Libre system is only one of a number of variables in our retrospective cohort study, it is possible that other factors may have had an impact on the observed outcomes. For example, registration of the FreeStyle Libre system within NDR may indicate that the incident user has also received device training or specific diabetes education from their healthcare professionals during the initiation process. This can result in improved diabetes self-care behaviors that cannot be controlled for in this retrospective cohort analysis. In this context, our analysis does not include a control group from within the NDR, matched for baseline characteristics but who were not incident users of the FreeStyle Libre system between June 2016 and June 2019. Because the incident users with T2DM in our study may have initiated their use of the FreeStyle Libre system after measuring extreme higher glucose values, both the overall effect of FreeStyle Libre use on the change in HbA1c, and specifically the association between the effect and the baseline HbA1c, could be impacted by the phenomenon of regression to the mean. The possibility that reductions in HbA1c are simply due to improved self-care related to training and diabetes education at the point of starting the FreeStyle Libre system is not inconsistent with the lack of a significant reduction in HbA1c amongst incident users of the FreeStyle Libre system with confirmed prior experience of CGM, although the small number of incident users in this group precludes clear interpretation in this context. It must also be acknowledged that, as with investigation of all registries, the validity of the outcomes relies on the quality of interpretation and application of the variables within the registry. However, the completeness of our data in regard of the variables under consideration and the 100% intra-patient coherence for diabetes type across the incident users of FreeStyle Libre (Supplementary Figure S1) give us confidence that this is not a factor for our interpretation of the NDR data. However, we acknowledge that an updated study, including an appropriate control group, would validate our current findings.
Strengths of our study include its large population of people with T2DM and the fact that the NDR has almost 90% coverage of all adults with diabetes in Sweden. Since clinical type of diabetes is stored at the person level in the NDR and is updated to be the last recorded observation, there should be no error in the variable containing clinical diabetes type. Thus, interpretation of our study outcomes in T2DM is not confounded by errors in classification of diabetes type. This means that selection bias should not be a factor.
Conclusions/summary
This real-world observational study on a large cohort of individuals with diabetes seen in clinical practice, has shown that people with T2DM in Sweden can significantly reduce their observed HbA1c for at least 12 months following initiation of the FreeStyle Libre system. These reductions are evident by 6 months after starting with flash glucose monitoring and the degree of change in HbA1c is correlated with the baseline HbA1c of users prior to initiation. Across the age distribution of users, significant reductions in HbA1c for people with T2DM were evident for all adults 25–74 years old. This analysis has significant implications for person-centered clinical care in diabetes and also for long-term health economic outcomes in the treatment of diabetes at a national level.
Supplemental Material
sj-pdf-1-dvr-10.1177_14791641211067418 – Supplemental Material for Real-world study of flash glucose monitoring among adults with type 2 diabetes within the Swedish National Diabetes Register
Supplemental Material, sj-pdf-1-dvr-10.1177_14791641211067418 for Real-world study of flash glucose monitoring among adults with type 2 diabetes within the Swedish National Diabetes Register by Katarina Eeg-Olofsson, Ann-Marie Svensson, Stefan Franzén, Hodan Ahmed Ismail, Michael Törnblom, and Fleur Levrat-Guillen in Diabetes and Vascular Disease Research
Footnotes
Acknowledgments
We would like to dedicate this work to our colleague Ann-Marie Svensson, who passed away during the manuscript peer review process.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: F.L.G. is employed by Abbott Diabetes Care. H.A.I and M.T are employed by IQVIA who received reimbursement from Abbott to perform the analysis. K.E-O. reports personal lecturing fees from Abbott, Lilly, Novo Nordisk, Sanofi and Bayer.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge funding support from Abbott Diabetes Care. The funding did not affect the collection, analysis, or presentation of the data. KEO acknowledges funding support from the Swedish state under the agreement between the Swedish government and the county councils, the ALF agreement (ALFGBG-698991).
Medical writing and editorial assistance
Medical writing and editorial assistance in the preparation of this article was provided by Dr Robert Brines of Bite Medical Consulting. Support for this assistance was funded by Abbott Diabetes Care
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
