Abstract
Continuous glucose monitoring (CGM) is commonly used in the outpatient setting to improve diabetes management. CGM can provide real-time glucose trends, detecting hyperglycemia and hypoglycemia before the onset of clinical symptoms. In 2011, at the time the Endocrine Society CGM guidelines were published, the society did not recommend inpatient CGM as its efficacy and safety were unknown. While many studies have subsequently evaluated inpatient CGM accuracy and reliability, glycemic outcome studies have not been widely published. In the non-ICU setting, investigational CGM studies have commonly blinded providers and patients to glucose data. Retrospective review of the glucose data reflects increased hypoglycemia detection with CGM. In the ICU setting, data are inconsistent whether CGM can improve glycemic outcomes. Studies have not focused on hospitalized patients with type 1 diabetes mellitus, the population most likely to benefit from inpatient CGM. This article reviews inpatient CGM glycemic outcomes in the non-ICU and ICU setting.
Up to 30% of all hospitalized patients have Diabetes Mellitus (DM).1-3 In 2007, an estimated 40.7 million hospital days were incurred by DM patients in the United States. 4 As of 2012, this estimate increased to 43.1 million hospital days, 5 a significant proportion of which without goal glycemic control, with hyperglycemia detected in up to 38% of all patients associated with increased morbidity and mortality.6-13 Moreover, hypoglycemia is detected in up to 30% of Type 2 Diabetes Mellitus (T2DM) patients treated with subcutaneous insulin.8,14-16 In the ICU, hypoglycemia occurs more often with intensive insulin therapy and is associated with increased mortality.17,18 Glycemic variability is associated with increased hospital length of stay and 90-day mortality. 6 Glycemic excursions occur in the setting of poor provider understanding of DM therapy, medications promoting insulin resistance, sudden nutritional changes, and acute illness.3,19-21
In the ICU and non-ICU setting, continuous glucose monitoring (CGM) has the potential to limit glycemic excursions, thereby limiting morbidity and mortality. As of 2011, the Endocrine Society recommended against inpatient CGM due to lack of published safety and efficacy data. 22 Since that time, numerous publications have evaluated inpatient CGM accuracy, reliability, and safety;23-31 however, few studies have evaluated the glycemic outcomes of subcutaneous CGM, which are limited by the lack of Type 1 Diabetes Mellitus (T1DM) subjects and the use of different CGM systems. This article will review subcutaneous CGM glycemic outcomes in the adult ICU and non-ICU setting.
Search Methods
To obtain studies for this review article, two independent reviewers (DLL and EKS) conducted an electronic PubMed search. We used various search queries to identify appropriate studies, including: “inpatient continuous glucose monitoring,” “inpatient CGM,” “intensive care unit continuous glucose monitoring,” “intensive care unit CGM,” “ICU continuous glucose monitoring,” “ICU CGM,” “non-critical care continuous glucose monitoring,” “non-critical care CGM,” “non-ICU CGM,” and “hospital CGM.” This review excluded studies evaluating CGM accuracy and reliability, which have been reviewed previously.23-31 While we reference isolated inpatient non-ICU CGM cases, we primarily included observational and randomized CGM glycemic outcome studies in this review (six ICU studies, three non-ICU studies), reflecting the scarcity of evidence on this topic, and the need for further studies.
ICU CGM Studies
ICU CGM has been studied with the ultimate goal of improving the outcomes of the critically ill, albeit primarily evaluating CGM accuracy and reliability. Table 1 describes the six studies evaluating the glycemic outcomes of ICU subcutaneous CGM. Notably, studies differ significantly, including subjects with different types of diabetes or even subjects without diabetes, using different types of CGM systems. More well-designed studies are needed to further evaluate the use of CGM among patients with diabetes in the critical care setting.
ICU Continuous Glucose Monitoring (CGM) Studies.
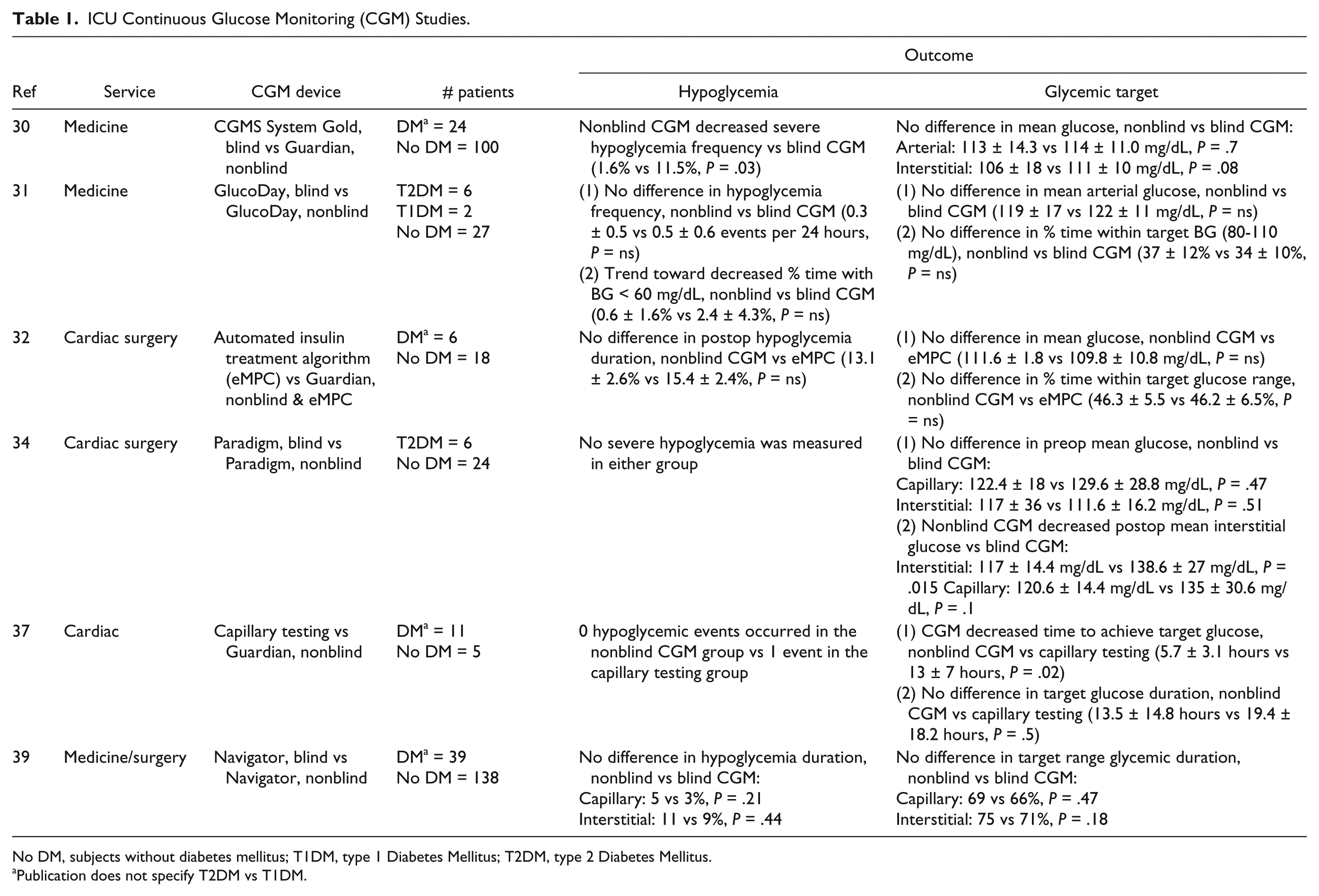
No DM, subjects without diabetes mellitus; T1DM, type 1 Diabetes Mellitus; T2DM, type 2 Diabetes Mellitus.
Publication does not specify T2DM vs T1DM.
To determine if CGM could be an effective tool to titrate intravenous insulin infusion, Holzinger et al enrolled 124 medical ICU patients (24 DM, 100 non-DM) to undergo intravenous insulin titration based on nonblinded Guardian CGM versus arterial blood glucose (BG) with blinded CGM (CGMS System Gold) (both manufactured by Medtronic MiniMed, Northridge, CA).
32
In the intervention group, nurses were instructed to take real-time glucose readings at least every 2 hours, at nursing discretion. Arterial BG values were checked every 1-2 hours in the control group. Study design aimed for target BG between 80 and 110 mg/dL. The primary end point was percentage of time at a glucose level <110 mg/dl. No difference was detected in percentage of time at a glucose level < 110 mg/dL (59.0 ± 20 vs 55.0 ± 18%,
Appraising the use of CGM in patients receiving intravenous insulin, De Block et al randomized 35 cardiosurgical ICU patients to nonblinded or blinded GlucoDay CGM for 96 hours.
33
The primary outcome, percentage time within target BG (80-110 mg/dL), did not improve with nonblinded CGM (37 ± 12% vs 34 ± 10% with blinded CGM,
Kopecky et al randomized 12 post–cardiac surgery patients to use nonblinded Guardian CGM vs arterial BG monitoring to titrate intravenous insulin therapy using a proprietary automated treatment algorithm.
34
Guardian CGM interstitial glucose values were input into the insulin infusion protocol every 15 minutes. While this study demonstrated ICU CGM accuracy and reliability, it did not demonstrate improved mean glucose (111.6 ± 1.8 vs 109.8 ± 10.8 mg/dL,
Logtenberg et al also studied CGM in the cardiac surgery population. 31 cardiac surgery subjects were randomized to blinded versus nonblinded Paradigm CGM (Medtronic MiniMed, Northridge, CA) one day prior to surgical intervention.
35
Outcome measures included mean glucose and percentage time spent in different glucose ranges in the preoperative, postoperative ICU, and postoperative ward setting. No significant difference in preoperative mean interstitial glucose was found, 117 ± 36 vs 111.6 ± 16.2 mg/dL with blinded CGM (
In the setting of acute coronary syndrome, CGM was assessed by measuring time-to-target glucose in 16 subjects who were administered intravenous insulin for 48 hours.
38
Eight subjects used nonblinded Guardian CGM, checked every hour, to titrate insulin infusion each hour. Eight control subjects had insulin infusion titrated by capillary BG, which was checked every four hours. Target glucose was defined as ≤ 140 mg/dl. One hypoglycemic episode (glucose ≤ 70 mg/dL) occurred in the control group. The CGM group achieved target glucose levels within 5.7 ± 3.1 vs 13 ± 7 hours in the control group (
ICU CGM utilization is hypothesized to improve nursing efficiency and hypoglycemia detection. Boom et al recruited from a mixed medical-surgical ICU, randomizing 177 subjects to either blinded or nonblinded FreeStyle Navigator CGM (Abbott Diabetes Care, Alameda, CA) to guide insulin treatment.
40
Among the group, 39 subjects had DM (18 in the nonblinded CGM group, 21 in the blinded CGM group). Target glucose was defined as 90-162 mg/dL, with glycemic threshold alarms triggered in the nonblinded CGM group when glucose levels were outside of this range. Arterial BG checks were performed six times daily. Outcome measures included mean glucose, length of stay, mortality, incidence of glycemic excursions and percentage time within target range. Severe hypoglycemia (<40 mg/dL) incidence was similar in both groups (
Non-ICU CGM Studies
Commonly, patients continue to use their personal CGM device when admitted to the hospital. 42 While a limited number of observational studies have retrospectively evaluated glucose patterns in the non-ICU setting with blinded CGM, randomized nonblinded CGM studies have not been conducted to date (Table 2).
Non-ICU Continuous Glucose Monitoring (CGM) Studies.
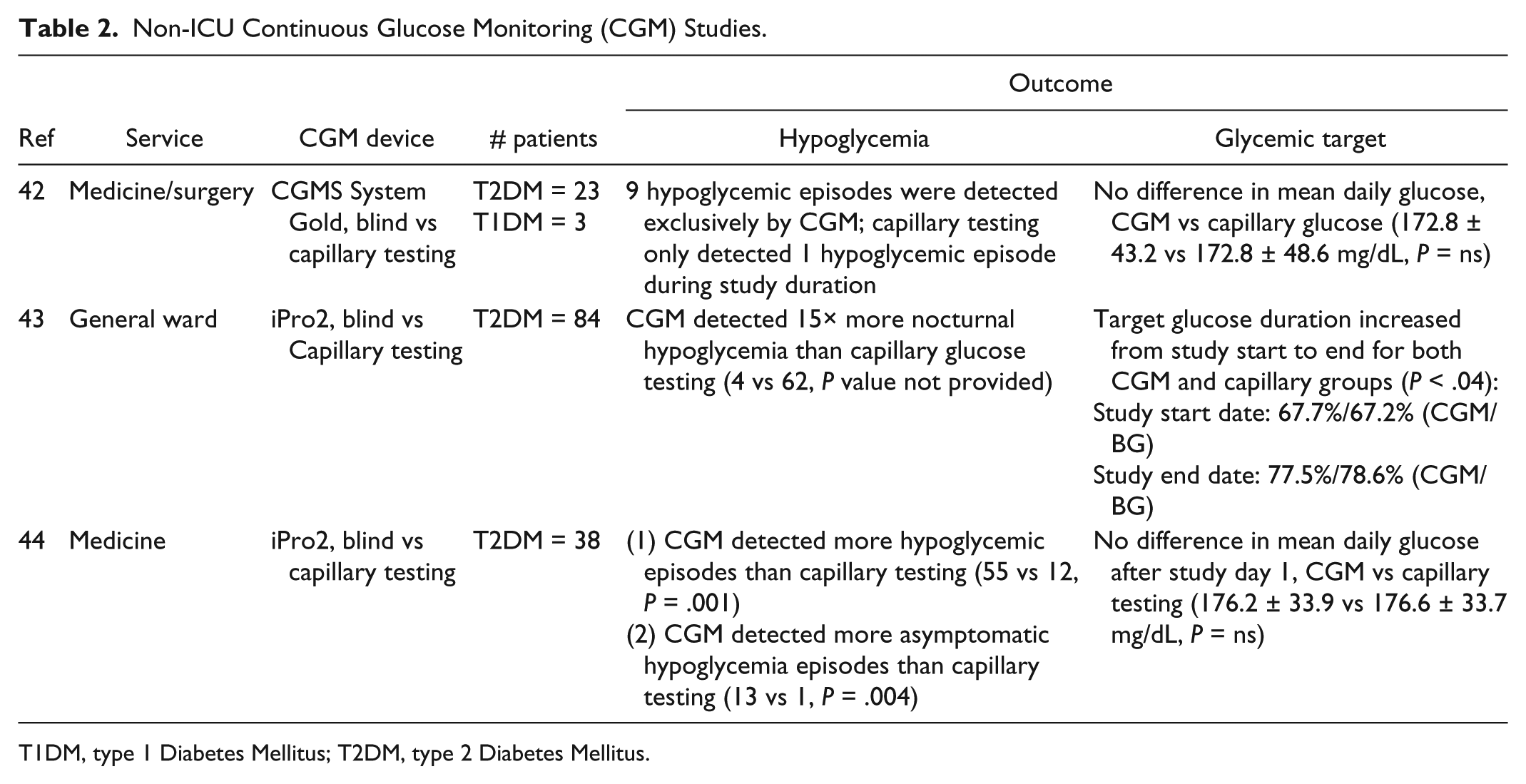
T1DM, type 1 Diabetes Mellitus; T2DM, type 2 Diabetes Mellitus.
Past studies have examined CGM primarily in type 2 diabetes mellitus (T2DM) patients, not T1DM. An Australian observational study of 26 adult DM patients (23 T2DM, 3 T1DM, 69% male, BMI 33 ± 12 kg/m2), treated with basal-bolus insulin during hospitalization, used blinded CGMS System Gold in medical and surgical general wards.
43
The authors hypothesized that fingerstick BG would underestimate mean glucose concentration. However, mean glucose was similar between interstitial and capillary monitoring (172.8 ± 43.2 mg/dL vs 172.8 ± 48.6 mg/dL with capillary testing,
CGM can be an effective tool for detecting occult nocturnal hypoglycemia in the non-ICU setting. Schaupp et al recruited 84 T2DM patients admitted to the general ward and applied a blinded iPro2 CGM device until hospital discharge, transfer to a different ward, or after 21 days.
44
CGM (n = 140 424) and capillary glucose (n = 2066) data from the first and last days of study duration were reviewed retrospectively. The percentage of glucose values within target range, 70 to 180 mg/dL, was higher by the end of study duration: 67.7%/67.2% (CGM/capillary) glucose values were reported on day 1 of the study, compared to 77.5%/78.6% (CGM/capillary) on the last day (
A similar observational study recruited 38 adult T2DM subjects to use blinded iPro2 CGM, with retrospective review of interstitial data.
45
All subjects were administered basal-bolus insulin during admission. Capillary testing was performed preprandial, 2-hour postprandial, at bedtime, and 3 AM. As in the prior study, CGM detected more hypoglycemia than capillary testing (55 vs 12 hypoglycemic episodes detected by capillary BG,
Discussion
Many advocate to further study inpatient CGM, in both the ICU and non-ICU settings.42,46-55 There are few randomized trials on this topic. A 2017 ICU CGM review was published in
Non-ICU patients, especially T1DM patients with a high likelihood of labile inpatient glycemic control 58 and hypoglycemic unawareness, may benefit most from CGM. In general, the non-ICU studies reflect increased hypoglycemia detection with CGM (Table 2). This effect would likely be amplified if studied in a larger non-ICU T1DM population. More effort should be made to recruit T1DM subjects for future inpatient CGM studies.
The T2DM non-ICU studies all report increased hypoglycemia detection with CGM, reflecting the potential benefits to be gained from widespread inpatient CGM adoption. The expectation is that results from T2DM studies should extrapolate to T1DM. In 2016, we demonstrated successful initiation and prevention of hypoglycemia with nonblinded CGM for a non-ICU inpatient with T1DM. 56 Only one other case report documents T1DM CGM initiation in the non-ICU setting, albeit blinded. 57
The majority of ICU CGM studies use established protocols for intravenous insulin infusion,32-34,38 in which capillary BG is usually monitored hourly, which may obviate the need for subcutaneous CGM. Hyperglycemic ICU patients not receiving continuous insulin infusion may benefit from CGM, as they are less likely to receive frequent glycemic monitoring. In these patients, CGM could be used as an additional vital sign. 48 While CGM could be limited by the lag time between interstitial and capillary glucose before detecting glycemic excursions, 59 Schaupp et al have demonstrated the ability of CGM to predict glycemia 20-30 minutes in the future in a cardiothoracic surgery population via simple linear extrapolation. 60 In addition, we hold out hope that CGM could be beneficial in the ICU, as these patients are likely at higher risk for glucose variability, in the setting of significant stress and numerous medications. The studies discussed in this review do not evaluate glycemic variability in the context of inpatient CGM. If evaluated in future inpatient CGM studies, it’s possible that there could be decreased glycemic variability. Increased mortality has been associated with increased ICU glycemic variability previously.61-63
CGM devices specifically developed for the hospital setting such as the Medtronic Hospital Glucose Management System (renamed Sentrino CGM), which attaches to a bedside monitor displaying glucose values every minute, may overcome inpatient barriers, although not in widespread use. Currently, the Sentrino CGM device is one of the few systems approved in Europe, not the United States, for inpatient use.53,64
In a well-designed clinical trial, accounting for CGM inaccuracy, glycemic excursion false positives and negatives, sample size, and study duration, CGM glycemic data can be used as a meaningful outcome measure. 65 Comparison of the glycemic outcomes of the aforementioned ICU studies may be difficult, as different variables were evaluated, including different CGM manufacturers, blind and nonblind CGM use, in addition to different patient populations (DM and non-DM subjects).
There are functional limitations when using CGM for hospitalized patients. The CGM device is supposed to be discontinued during MRI or CT scanning, nor should it be used when administering acetaminophen due to chemical interference. 66 The effect of ketoacidosis and lactic acidosis on CGM has not been studied. 22 It may be difficult to calibrate the CGM device during an admission, in the setting of multiple comorbidities. It should be noted that a capillary blood sample might cause inaccurate CGM calibration if taken from a site of tissue edema or low perfusion. Moreover, CGM calibration should only occur during a period of glycemic stability.48,67
Summary
A limited number of studies have evaluated glycemic outcomes in the inpatient setting. In the non-ICU setting, CGM detects hypoglycemia at a greater frequency than capillary BG testing. In the ICU, CGM may not improve glycemic outcomes when patients are receiving intravenous insulin administration, which already requires frequent glucose monitoring. There are significant design differences in the ICU CGM studies, which may hinder the results. Additional studies are needed using nonblinded CGM and in patients with T1DM.
Footnotes
Abbreviations
BG, blood glucose; CGM, continuous glucose monitoring; DM, diabetes mellitus; ICU, intensive care unit; T1DM, type 1 diabetes mellitus; T2DM, type 2 diabetes mellitus.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EKS has received research support (supplies) from DEXCOM for conducting inpatient studies.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
