Abstract
Metaplastic breast carcinoma is an invasive carcinoma with a high differentiation rate of the neoplastic epithelium toward mesenchymal-like epithelium. It comprises of only less than 1% of all breast cancers. Although 80% to 90% of metaplastic breast carcinomas are triple-negative cancers, they usually have worse outcomes than other triple-negative breast cancers (TNBCs). Metaplastic carcinoma is also often refractory to cytotoxic chemotherapy. Here, we reported a case of a 61-year-old female patient, presenting with a solitary and pedunculated mass in the right axillary tail breast tissue, whose biopsy revealed metaplastic breast carcinoma with chondroid differentiation. She had failed neoadjuvant chemotherapy and immunotherapy. Although she received debulking surgery, the tumor regrew even faster before surgery. Despite receiving palliative chemotherapy, the patient died 11 weeks after surgery. This case draws attention to physicians that early recognition and surgery may be more beneficial than chemotherapy in combating metaplastic breast carcinoma.
Keywords
Introduction
Metaplastic breast carcinoma is invasive and comprises less than 1% of all breast cancers. 1 The neoplasm is characterized by cells differentiating into heterologous elements, such as squamous, spindle, and chondroid, without a transition zone between them. Approximately 80%-90% of metaplastic breast carcinoma are negative for estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor 2 (HER2), but they usually have worse outcomes than other triple-negative breast cancers (TNBCs). 2 Treatment of metaplastic breast carcinoma usually relies on radiotherapy and chemotherapy; however, overall survival is usually much lower than other TNBCs. 3 In addition, metaplastic breast cancer is often refractory to cytotoxic chemotherapy. 4
To date, only limited cases of metaplastic carcinoma have been reported, but none have been reported regarding metaplastic carcinoma in the axillary tail breast tissue. Ultimately, little is known regarding the treatment of this cancer. Here, we presented a case of a 61-year-old woman with metaplastic breast carcinoma in the axillary region of the breast who was initially misdiagnosed to have TNBC. The tumor showed a high malignancy stage, rapid progression, and poor prognosis.
Case Report
Here, we report a case of a 61-year-old woman referred to our clinic to evaluate a solitary and pedunculated mass in the upper external quadrant of the right breast. The patient was first diagnosed in another hospital with a multicentric breast tumor in August 2022, showing a 0.6 cm tumor at the right lower out quadrant of the breast (Figure 1A) and a large mass of 4.5 × 3.0 × 3.5 cm in her right axilla (Figure 1B and C). Her first core needle biopsy showed TNBC. The tumor-node-metastasis (TNM) stage was cT1bN2aM0 at the time of the first diagnosis. Therefore, the patient received 2 courses of neoadjuvant chemotherapy with lipo-doxorubicin-cyclophosphamide (adriamycin-cyclophosphamide; AC). However, no apparent therapeutic response was seen on the right axilla tumor. The patient continuously received another 2 courses of AC and 3 courses of docetaxel-carboplatin (taxotere-cyclophosphamide; TC) plus pembrolizumab from October 2022 to January 2023. While the right lower outer quadrant tumor was responsive and showed complete disappearance, the axillary tumor was still progressing, with a TNM stage of ycT3N2M0 at the end of the treatment.

(A-C) Representative CT images scanned at first diagnosis (August 2022). (A) Axial view showing a 0.6-cm tumor (yellow arrow) at the right lower out quadrant of the breast. (B) Coronal and (C) axial views showing right-sided axillary breast cancer (yellow arrow). (D) Clinical photograph taken at the time when the patient was referred to our clinic (February 2023). A solitary and pedunculated mass was seen in the upper external quadrant of the right breast. (E and F) Representative CT images of the (E) coronal and (F) axial views showing an enlarged breast cancer, unresponsive to chemotherapy and immunotherapy (February 2023). CT indicates computed tomography.
In February 2023, the patient was transferred to our clinic. On physical examination, a pedunculated mass occupied her right axilla, which was tender and fixed to the chest wall (Figure 1D). There was also a satellite nodule measuring 3.0 cm in size, which invaded the skin with ulceration and bleeding. The axillary tumor measured 7.0 × 8.0 × 7.5 cm on a computed tomography (CT) scan, whereas the small tumor initially located at the right lower outer quadrant of the breast was not seen. The patient denies any relevant family history of cancer.
Microscopical examination of the axillary tumor re-confirmed a metaplastic-type TNBC with heterologous mesenchymal (chondroid) differentiation. The tumor was assigned an American Joint Committee on Cancer (AJCC) histopathologic grade of 3 (scores 8-9) and a Nottingham histopathologic score of 8. A positron emission tomography (PET)/CT scan suggested no definite abnormal 18F-fluoro-2-deoxy-d-glucose (FDG)-6P accumulation in other lymph nodes or areas of the body.
The patient was given a single regimen of carboplatin (area under the curve [AUC] = 4; 500 mg), paclitaxel (80 mg/m2), and pembrolizumab (100 mg) 1 day after the diagnosis of metaplastic breast carcinoma, but the tumor was still gradually enlarged, as a CT scan performed 1 week after the treatment showed that the tumor had grown to 9.4 cm (Figure 1E and F). Given the poor effect of tumor treatment, a right total mastectomy and axillary lymph node dissection (ALND) were performed. However, because the tumor had already adhered to the patient’s brachial plexus and axillary vessels, part of the tumor tissue remained on the nerves, as the patient refused forequarter amputation (Figure 2A). A breast with a nipple was resected along with 5 axillary lymph nodes (Figure 2B). After sectioning the specimen, a tumor with a firm consistency and irregular surface, measuring 10.0 × 10.0 × 7.5 cm, was observed. The 5 axillary lymph nodes were involved by the tumor, as observed macroscopically. Histopathological analysis suggested a pathological stage of ypT4bN2a, with lymphovascular and perineural invasion.
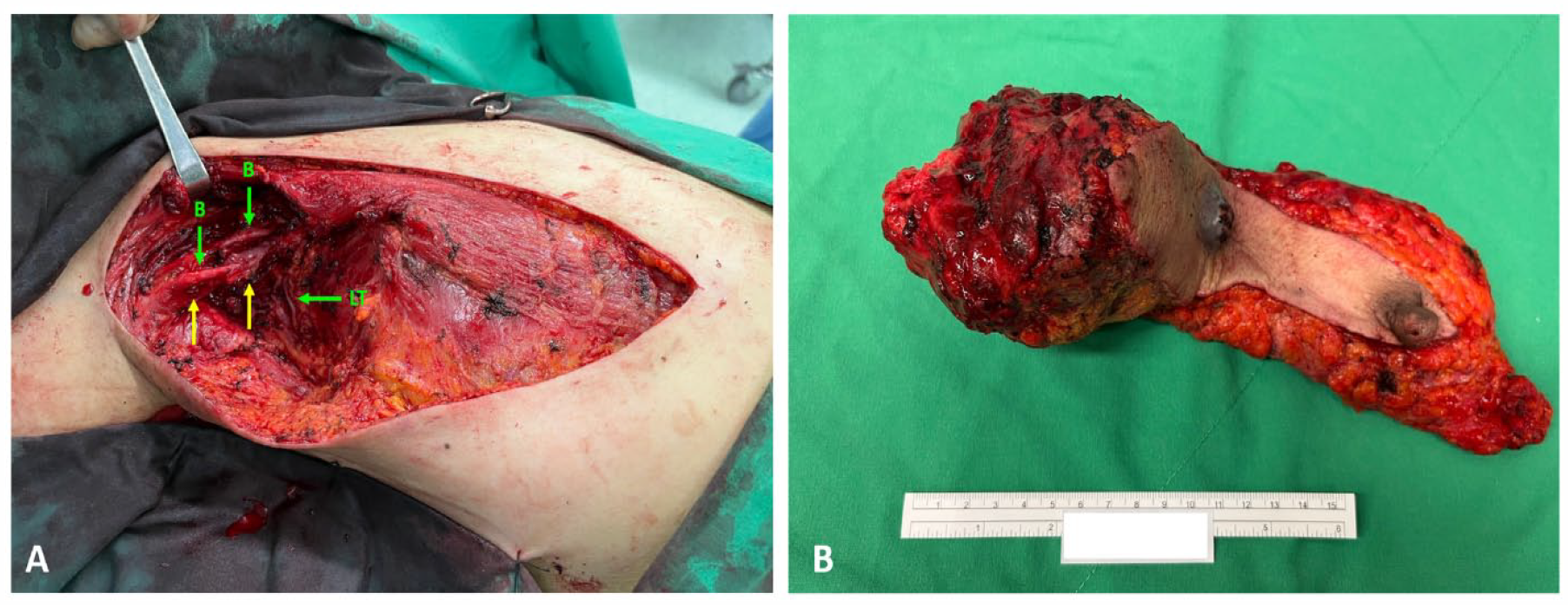
(A-B) Clinical photograph taken at the surgery (March 2023). (A) Right total mastectomy and axillary lymph node dissection were performed, but part of the tumor tissue (yellow arrow) was left behind on the major nerves and axillary vessels. (B) Breast with a nipple and areola-containing overlying skin was resected with 5 axillary lymph nodes. B indicates brachial plexus; LT indicates long thoracic nerve.
As some macroscopic tumor remnants were left on the nerves, radiotherapy was initially planned to be performed 3 weeks after surgery. However, 13 days after surgery, the patient showed severe swelling of the axillary area. Examination revealed a mass-like lesion of 13.0 cm in diameter on her right axilla and a skin nodule in her right upper arm near the axilla (Figure 3). Fine-needle biopsy confirmed the regrowing of metaplastic carcinoma. Computed tomography angiography showed extravasation at the tumor wound, and the tumor encased the axillary artery. Genetic analysis was done, showing an amino acid change (H1047R) of PIK3CA and heterozygous deletion of specific tumor suppressor genes including PTEN, FLCN, TP53, and CDKN2A. However, because of the rapid growth of the tumor, the patient was given a 3-day combination of MAID regimen: mesna (2000 mg/m2/d), doxorubicin (20 mg/m2/d), ifosfamide (2000 mg/m2/d), and dacarbazine (250 mg/m2/d), instead of PIK3CA or mTOR inhibitors to get faster control of the tumor progression. However, the results were disappointing, and adverse effects including hypokalemia, febrile neutropenia, and septic shock developed. Following resuscitation, the patient’s condition turned stable. Eight days later, the patient was transferred to the hospice care unit as the patient wanted to discontinue the treatment.

Representative CT images of the coronal view showing the regrowing of right breast cancer (red arrow). The scan was performed on the 25th day after the surgery (March 2023). CT indicates computed tomography.
On May 2023, the patient experienced severe right arm numbness and pain. Morphine and lidocaine failed to relieve the pain. A few days later, the patient lost consciousness and her blood pressure dropped; the patient did not survive due to the aggressive course of disease.
Discussion
Metaplastic breast carcinoma is a rare yet particularly aggressive form of breast cancer. The prognosis and survival rate are much lower in metaplastic breast carcinoma than in other breast cancers. Because of the rare occurrence of this cancer, experience regarding treating these patients remains underrepresented. Our case here represented multicentric metaplastic breast carcinoma growing in a rare location, the axillary breast tissue. This patient was initially misdiagnosed to have TNBC. As such, she received neoadjuvant chemotherapy; however, the therapeutic response was poor. Although the small tumor in the right lower outer quadrant was responsive, the axillary breast tumor was unresponsive. After debulking surgery, the tumor regrew even faster than that before surgery.
Current knowledge on metaplastic breast carcinoma is that the TNM stage, tumor size, and axillary node metastasis were significant prognostic factors for the patient’s survival.5-8 Patients were more often treated with upfront radiotherapy or chemotherapy rather than mastectomy if the tumor is large in size or in an advanced stage; however, this cancer is less responsive to current available neoadjuvant chemotherapy than other breast cancers.4,9 In our case, while 1 tumor was responsive to chemotherapy, the axillary breast tumor was unresponsive. The poor response to chemotherapy might result from epithelial-mesenchymal transition, 10 which occurred in the axillary tumor but not in the right lower outer quadrant tumor. Considering the poor effect of chemotherapy, the patient underwent a mastectomy when the tumor burden was relatively high. However, such a delayed intervention cannot benefit in relieving the tumor burden but instead makes the burden more severe and progressive. Currently, there is no literature reviewing the fast-growing of metaplastic breast carcinoma after surgery, but our case presented here has expanded on the idea that performing surgery at first diagnosis, where the tumor is small in size, may be helpful to control tumor progression; yet this may require a trial with a larger sample size.
Recent studies have suggested that metaplastic breast carcinomas frequently overexpress immune checkpoint markers programmed death ligand 1 (PD-L1).11-13 However, in the phase 3 clinical trial, KEYNOTE-355 (ClinicalTrials.gov Identifier: NCT02819518), therapeutic benefits of pembrolizumab were only seen in patients with a combined positive score (CPS) more than 10, but not in those with a score between 1 and 10. 14 Similar results were seen in the KEYNOTE-119 trial (ClinicalTrials.gov Identifier: NCT02555657), where pembrolizumab showed more prolonged overall survival in patients with CPS of more than 20 but not in those less than 20. 15 Although these clinical trials were not focused on patients with metaplastic carcinoma, targeting PD-L1 may provide us with an alternative way to combat metaplastic breast carcinomas, and routine testing of PD-L1 expression may be helpful.
Other reports have demonstrated the role of mitochondrial metabolism with cancer cell progression, 16 and PD-L1 blockade has been found to be more effective in cancer cells with higher cellular respiration and adenosine triphosphate (ATP) turnover rate. 17 Therefore, the efficacy of anti-PD-L1 agents may be increased with mitochondria-modulating drugs such as bezafibrate, an agonist of peroxisome proliferator-activated receptor-γ (PPAR-γ) coactivator-1α (PGC-1α)/transcription factor complex. 18 Analyzing mitochondrial metabolism may also serve as a predictive biomarker for anti-PD-L1 treatment. 19
Metaplastic breast carcinoma also frequently showed aberrations in the PI3K/AKT/mTOR pathway, as PIK3CA mutations were detected more often than other breast cancers.20,21 In a phase 1 trial, treatment with the combination of liposomal doxorubicin, bevacizumab, and mTOR inhibitors in patients with PIK3CA mutations had a higher objective response rate than those without mutations. 21 In our case, the patient also harbors a mutation of PIK3CA. However, because of the rapid progression of the tumor, the patient was given MAID chemotherapy instead of PIK3CA or mTOR inhibitors to get faster control of the tumor progression. Nevertheless, the outcome of this treatment was unsatisfying, similar to a recent study showing similar disappointing results. 22
On the contrary, our patient also showed loss of PTEN, which is presented in only 5% of metaplastic breast carcinoma and 2% of other breast cancers. 20 PTEN is a negative regulator of the PI3K/AKT/mTOR pathway, 23 and the loss of PTEN may result in lesser tumor-infiltrating T cells and decreased T-cell trafficking into tumors. 24 Furthermore, loss of PTEN has been shown to induce PD-L1 expression on tumor cells, leading to worsening outcomes. 25 In addition, low density of tumor-infiltrating lymphocytes (TILs) or low level of PD-1 expression on TILs may affect the therapeutic response of PD-1 inhibitors. 26 Consequently, this may be associated with our patient’s poor response to pembrolizumab.
Metaplastic breast carcinoma processes a higher number of forkhead box P3 (FOXP3)-expressing regulatory T (Treg) cells. 11 An increase in FOXP3-positive Treg cells is usually associated with improved survival in TNBC27,28; despite that, they negatively correlated to the survival of patients with metaplastic breast carcinoma. 11 It has been suggested that PD-L1 and FOXP3-positive Treg cells may act synergistically to promote tumor immune evasion in breast cancer,29,30 as PD-L1 can modulate the differentiation of naive T cells to Treg cells. 31 In this regard, targeting Treg cells may enhance the therapeutic effect of PD-L1 blockade therapy in metaplastic breast carcinoma. Currently, multiple Treg depletion therapies, such as antibody against CD25 (camidanlumab tesirine; ClinicalTrials.gov Identifier: NCT03621982), CTLA4 (ipilimumab; ClinicalTrials.gov Identifier: NCT03409198), or CD73 (uliledlimab; ClinicalTrials.gov Identifier: NCT05001347), have been used in combination with PD-1 or PD-L1 inhibitors. However, none of these trials were done on metaplastic breast carcinoma. In addition, it would be valuable to conduct future trials on this rare breast cancer subtype.
Conclusions
Clinical trials for metaplastic breast carcinomas are difficult to achieve because of their rarity and unique clinical features. Although personalized medicine on selected patients has yielded promising results, more large-scale studies are warranted to meet the clinical need. In addition, routine testing of immuno-oncology markers such as PD-L1 or FOXP3 may be helpful in selecting suitable therapeutic methods for these patients.
Footnotes
Acknowledgements
Not applicable.
