Abstract
Objective:
To evaluate the association of sociodemographic, clinical, and pathological factors with response and survival in triple negative breast cancer (TNBC) undergoing neoadjuvant chemotherapy (NACT).
Methods:
Clinical-pathological and sociodemographic data were obtained from medical records of 235 eligible women with TNBC diagnosed between 2010 and 2014 undergoing NACT and surgery at the Brazilian National Cancer Institute. They have been assessed for pathological complete response (pCR), event-free survival (EFS), and overall survival (OS). Both univariate and multivariate Cox regression analyses were performed.
Results:
The median follow-up was 64.3 months. Most patients had advanced clinical stage (III: 85.1%; cT3/T4: 86.4%; cN1-3: 74.4%) and high-grade tumors (72.1%). Clinical staging (III vs II, adjusted hazard ratio [HR] = 2.95, P = .012) significantly influenced the pCR rate. Alcohol intake negatively influenced EFS (adjusted HR = 1.67, P = .006) and OS (adjusted HR = 1.89, P = .005). Women with pCR showed better EFS (crude HR = 0.15, P < .001) and OS (crude HR = 0.12, P < .001) compared with non-pCR. The ypT (<0.001) and ypN (<0.001) gradually influenced survival outcomes.
Conclusion:
Clinical stage III were associated with lower response rate and worse survival. Alcohol intake, pCR, and burden of post-NACT residual disease have shown considerable influence on survival outcomes.
Keywords
Introduction
According to GLOBOCAN 2018 cancer statistics, breast cancer is the most common cancer in women in the vast majority of countries around the world, as well as the leading cause of cancer death in more than 100 countries. 1 Triple negative breast cancer (TNBC) accounts for approximately 12% to 17% of primary breast tumors. 2 Histologically, it is defined as tumors lacking estrogen receptor (ER), progesterone receptor (PR) expression, and human epidermal growth factor receptor 2 (HER2). 3 Generally, TNBCs are mostly high-grade tumors with higher incidence in younger women, associated with poorer overall prognosis, increased risk of early distant relapse, and higher risk of premature death. Metastases tend to be visceral, mostly occurring at the pulmonary, pleural, hepatic, and central nervous system sites. These clinical features represent a major medical concern in the management of these patients.4,5
Unlike luminal or HER2-positive types, which are known to be sensitive to hormone therapy and anti-HER2 agents, respectively, the treatment of TNBC is based on cytotoxic chemotherapy. In this context, neoadjuvant chemotherapy (NACT) has become the standard of care for most locally advanced TNBCs. This is due to the possibility of greater chances of breast-conserving surgery and consistent evidence of pathologic complete response (pCR) as a strong predictor, or even surrogate, of long-term survival outcomes. 6 Anthracycline and taxane-based chemotherapy regimens are the current standard therapy in most cases, showing pCR rates of around 17% to 40% in some studies. 7 However, recent clinical trials have proposed a refinement of these therapeutic schemes with new drugs such as platinum-based agents, immunotherapy and Poly [ADP-ribose] polymerase 1 (PARP) inhibitors, or even dose-dense regimens.8,9
Some sociodemographic, clinical, and pathological factors may influence the outcomes of NACT in patients with locally advanced TNBC. Nevertheless, there is scarce data in the literature about the role some of these variables have in this specific setting. This study aims to evaluate the association of these factors with tumor response and survival outcomes, as well as present the institutional profile of women with TNBC undergoing NACT at the Brazilian National Cancer Institute (INCA).
Materials and Methods
Study design and ethical considerations
This retrospective cohort was designed to assess the influence of sociodemographic, clinical, and pathological factors on the prediction of clinical response to NACT and on survival outcomes. The study was approved by the Ethics in Human Research Committee of INCA, Rio de Janeiro, Brazil, under number CAAE 61675516.9.0000.5274, and conducted in accordance with Good Clinical Practice guidelines.
Patient selection
Patients newly diagnosed with breast cancer at INCA between January 2010 and December 2014 were included if all the following criteria were met: (a) women more than 18 years old; (b) diagnosis of TNBC (tumors with ER and PR score < 1%, as well as HER-2 score 0/1+ or 2+ with negative FISH) by the INCA Pathology Department (DIPAT/INCA) following the American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) guidelines10,11; (c) stage IIb-IIIc by the 7th AJCC (T3-4NanyM0; TanyN1-3M0); and (d) undergoing NACT and curative surgery at INCA. In turn, patients with synchronic or anachronistic tumors, previously exposed to antineoplastic agents were excluded, as well as patients who remained with unresectable tumors, even after standard NACT and complementary treatment with chemotherapy and/or radiotherapy.
Variables and outcomes
Patients were identified through internal database. Data were collected from electronic hospital records and medical charts. The following sociodemographic and treatment variables were evaluated: age at diagnosis, ethnicity (Caucasian or others according to national institutional statistical classifications, IBGE 12 ), schooling (<8 or ⩾8 years), smoking and alcohol consumption (previous or current habit), body mass index (BMI), distance from home to hospital (set by Google Maps), type of standard NACT (detailed in Box 1: FAC, FAC-T, AC-T, or CT), time from diagnosis to NACT onset, time from the end of standard NACT to surgery, compliance to standard NACT (median cycles; complete vs incomplete treatment), and site of progression. The clinical and pathological variables evaluated were clinical stage (II-III), clinical T stage (cT), clinical nodal stage (cN), pathological T stage (ypT), pathological nodal stage (ypN), lymphovascular invasion (LVI), perineural infiltration (PI), Elston histological grade (1-2: low grade; 3: high grade), and type of surgery (radical or conservative, axillary approach type).
Neoadjuvant chemotherapy regimens.

Abbreviation: NACT, neoadjuvant chemotherapy; FAC, fluorouracil, doxorubicin and cyclophosphamide; FAC-T, fluorouracil, doxorubicin and cyclophosphamide followed by docetaxel; AC-T, doxorubicin and cyclophosphamide followed by docetaxel or followed by weekly paclitaxel; CT, cyclophosphamide and docetaxel.
Non-anthracycline option defined by the institutional tumor board for selected cases.
Following the routine of the oncology team, patients with tumors considered unresectable soon after NACT were exposed to complementary chemotherapy and/or salvage radiotherapy to achieve clinical response to enable the surgical approach.
The pCR was defined as no viable tumor in the breast or axilla (ypT0N0). 6 Event-free survival (EFS) was calculated from the date of diagnosis to the earliest date of disease progression, death from any cause, or discontinuation of treatment for initiation of complementary treatment due to poor response to standard NACT. Overall survival (OS) was calculated from the date of diagnosis to the date of death or censored if the patient was known to be alive on the last day of data collection.
Statistical analysis
Statistical analyses were conducted using R environment. 13 All continuous variables were evaluated by the Shapiro-Wilk test of normality. For the pCR outcome, logistic regression was used for each variable assessed to calculate the odds ratio (OR). Survival rates were calculated by Kaplan-Meier curves for each factor and were compared by log-rank test. The crude hazard ratio (HR) for each factor was calculated by the Cox proportional hazards model. All variables associated with survival outcomes at P < .20 on univariate analysis were included in multivariate models. The Akaike criteria was used to pick the most suitable model for multiple Cox analysis. A P-value of .05 or less was considered to indicate statistical significance. The missing data were excluded from the analysis.
Results
Patients’ characteristics
A total of 235 cases of TNBC were eligible for the study (Figure 1). The main characteristics of the patients are summarized in Table 1. The mean age was 50.1 years (range, 23.5-75.8), most women were Caucasian (47.6%) and had completed 8 or more years of education (55.2%). The median BMI was 28.1 kg/m2 (interquartile range [IQR], 24.4; 32.5). Smoking and alcohol consumption were reported by 24.2% and 22.9% of the patients, respectively. The median home distance to INCA was 28 km (IQR, 17; 40). At diagnosis, most patients had advanced clinical stage tumors (⩾IIIa: 85.1%; cT3/T4: 86.4%; cN1-3: 74.4%) and the predominant histological subtype was high-grade invasive ductal carcinoma (72.1%). Metaplastic carcinoma accounted for only 4.7% of cases, with the other patients (95.3%) having non-special-type invasive carcinoma. At surgery, LVI and PI were present in 23.4% and 11% of cases, respectively, and the pathological nodal stage was predominantly ypN0-1 (76.6%).

Study profile. HER-2 indicates human epidermal growth factor receptor 2; INCA, Brazilian National Cancer Institute; NACT, neoadjuvant chemotherapy; TNBC, triple negative breast cancer.
Baseline sociodemographic characteristics of eligible patients.

Abbreviations: BMI, body mass index; IQR, interquartile range; LVI, lymphovascular invasion; PI, perivascular infiltration; SD, standard deviation.
Missing values: race/ethnicity (2; 0.8%), schooling (5; 2.1%), smoking (1; 4.7%), alcohol consumption (13; 5.5%), home distance (5; 2.1%), LVI (35; 14.9%), and PI (64; 27.2%).
Treatment data
Mastectomy was the treatment of choice in 97.4% of cases and axillary dissection was performed in 86.8% of the patients, as shown in Table 2. Regarding systemic treatment, 94.1% underwent chemotherapy with anthracycline and taxane-based regimens and 83.4% completed all the cycles of NACT as scheduled. Complementary chemotherapy was performed in only 6.8% of cases and neoadjuvant radiotherapy in 4.7%. The median time from diagnosis to initiation of treatment was 90.0 days (IQR, 58; 126.5) and slightly more than a quarter of patients (27.2%) started treatment in less than 60 days.
Data from neoadjuvant chemotherapy and surgical treatment.
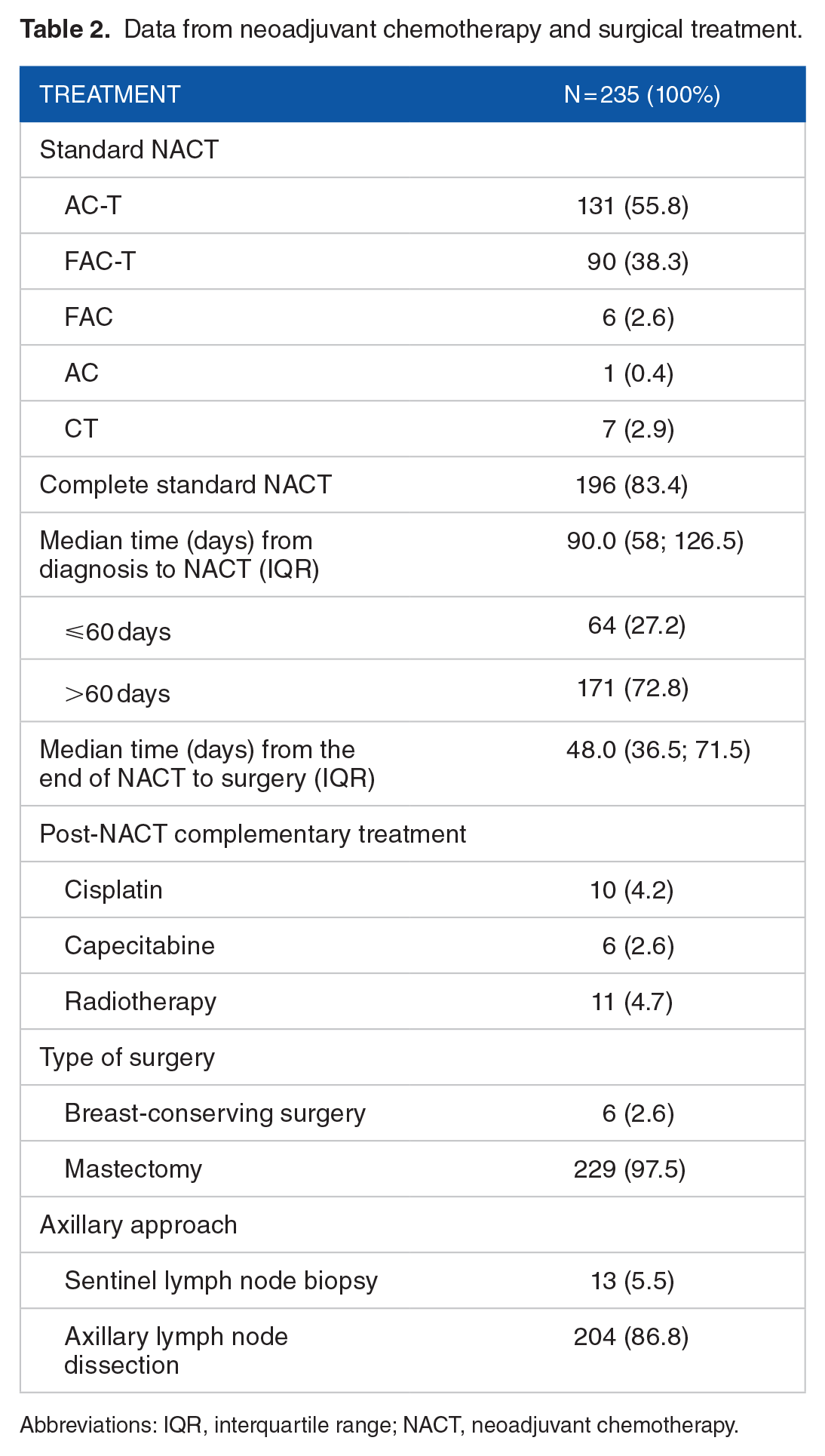
Abbreviations: IQR, interquartile range; NACT, neoadjuvant chemotherapy.
Pathological response and survival outcomes
The overall pCR rate was 21.2%. By univariate analysis, patients with clinical stage II (crude OR = 0.99, P = .005) and not exposed to alcohol intake (crude OR = 0.38, P = .036) had better pCR rate, as shown in Table 3. In the final model selected for multivariate analysis, only clinical stage II (adjusted OR = 2.95, P = .012) was associated with higher pCR rate.
Univariate and multivariate analysis according to pathological complete response.

Abbreviations: BMI, body mass index; CI, confidence interval; NACT, neoadjuvant chemotherapy; FAC-T, fluorouracil, doxorubicin and cyclophosphamide followed by docetaxel; AC-T, doxorubicin and cyclophosphamide followed by docetaxel or followed by weekly paclitaxel; OR, odds ratio; pCR, pathological complete response. The variables of the final model selected for analysis by the Cox multiple model were highlighted in bold.
Regarding the chemotherapy regimen, only the AC-T vs FAC-T regimens were compared.
Reference.
The median follow-up was 64.3 months (95% confidence interval [CI]: 60.3-68.2). Locoregional recurrence occurred in 51 patients (21.7%) and distant recurrence was observed in 95 (40.4%). The most common distant sites were pleuropulmonary (23%), nodal (14%), hepatic (10.6%), bone (10.6%), and central nervous system (6.4%) (data not shown). For the general population of the study, with 114 events, the probability of 3-year EFS and 5-year EFS was, respectively, 59.4% (95% CI: 53.4-66.2) and 53.3% (95% CI: 47.0-60.5). The median EFS was 76.5 months (95% CI: 44.76-not reached [NR]). For patients with pCR vs non-pCR, the probability of 3-year EFS and 5-year EFS was, respectively, 98.0% (95% CI: 94.2-100) vs 48.9% (95% CI: 42.2-56.9) and 93.3% (95% CI: 86.3-100) vs 42.5 (95% CI: 35.6-50.7) (data not shown). As shown in Table 4, patients with pCR had an 85% reduction in risk of events as compared with non-pCR (crude HR = 0.15, 95% CI: 0.06-0.34, P < .001). Herein, the gradient of post-NACT residual disease burden, represented by ypT0-4 (P < .001) and ypN0-3 (P < .001), also showed a gradual effect on EFS. Still in the univariate analysis, alcohol intake increased by 74% (P = .02) the risk of presenting an event.
Univariate and multivariate analysis according to event-free survival.

Abbreviations: BMI, body mass index; CI, confidence interval; EFS, event-free survival; HR, hazard ratio; NACT, neoadjuvant chemotherapy; pCR, pathological complete response; FAC-T, fluorouracil, doxorubicin and cyclophosphamide followed by docetaxel; AC-T, doxorubicin and cyclophosphamide followed by docetaxel or followed by weekly paclitaxel.
The variables of the final model selected for analysis by the Cox multiple model were highlighted in bold.
Regarding the chemotherapy regimen, only the AC-T vs FAC-T regimens were compared.
Reference.
Regarding OS, with 101 deaths, the estimated probability of patients being alive at 3 and 5 years was, respectively, 68.2% (95% CI: 62.3-74.6) and 59.6% (95% CI: 53.0-66.5). The median OS was 83.36 months (95% CI: 65.66-NR). For patients with pCR vs non-pCR, the probability of 3-year EFS and 5-year EFS was, respectively, 98% (95% CI: 94.2-100) vs 60.2% (95% CI: 53.3-67.9) and 98% (95% CI: 94.2-100) vs 49.9% (95% CI: 42.8-58.2) (data not shown). As shown in Table 5, patients with pCR had an 89% reduction in risk of death as compared with non-pCR (HR = 0.11, 95% CI: 0.04-0.31, P < .001). The residual disease burden gradient, composed of ypT0-4 (P < .001) and ypN0-3 (P < .01), showed a gradual association with OS. In the univariate analysis, alcohol intake increased by 97% (P = .002) the risk of death. Kaplan-Meier survival curves for EFS and OS are, respectively, shown in Figures 2 and 3.
Univariate and multivariate analysis according to overall survival.

Abbreviations: BMI, body mass index; CI, confidence interval; HR, hazard ratio; NACT, neoadjuvant chemotherapy; OS, overall survival; pCR, pathological complete response; FAC-T, fluorouracil, doxorubicin and cyclophosphamide followed by docetaxel; AC-T, doxorubicin and cyclophosphamide followed by docetaxel or followed by weekly paclitaxel.
The variables of the final model selected for analysis by the Cox multiple model were highlighted in bold.
Regarding the chemotherapy regimen, only the AC-T vs FAC-T regimens were compared.
Reference.

Kaplan-Meier event-free survival estimates according to (A) alcohol consumption, (B) pathological tumor stage, (C) pathological nodal stage, and (D) pathological complete response. pCR indicates pathological complete response.

Kaplan-Meier overall survival estimates according to (A) alcohol consumption, (B) pathological tumor stage, (C) pathological nodal stage, and (D) pathological complete response. pCR indicates pathological complete response.
As shown in Table 4, following the Akaike criteria, a model with 3 variables were selected for the EFS multivariate analysis. Clinical stage III increased the risk of event vs stage II by 2.57-fold (P = .016), alcohol intake increased the risk of recurrence or death by 67% (P = .010), and treatment compliance showed no association with EFS (P = .08).
The final model for OS consisted of 4 variables (Table 5). Patients with clinical stage III had a risk of death 2.21 times higher than stage II (P = .046) and alcohol intake increased the risk of death by 89% (P = .005). Smoking (P = .903) and compliance to NACT (P = .248) did not influence OS.
Discussion
This study evaluated the association of sociodemographic, clinical, and pathological variables with response and survival outcomes in women with TNBC undergoing NACT at INCA. To the best of our knowledge, with 235 women included, these were one of the largest cohorts in this subset. Overall, the patients showed a trend toward early recurrence, mostly as distant metastases. The results showed that clinical stage III and alcohol consumption were associated with lower pCR rate and shorter survival. However, patients who achieved pCR had considerably longer survival.
Early age at diagnosis and overweight were mostly found in the patients of this cohort, corresponding to the data presented in previous series.14,15 Likewise, other ominous features of TNBC also prevailed such as high-grade tumors and locally advanced disease at diagnosis with axillary nodal involvement, which in some extent explains the fact that almost all patients underwent radical surgery. 16
The alcohol consumption and smoking rate were quite similar to other series that included women with TNBC.17-20 Further information on the dose, duration, and type of exposure to alcohol and tobacco unfortunately were not available in the records of the patients in this study. A meta-analysis published in 2013 pointed out that only a more regular and heavy alcohol intake, greater than 20 g/day, would be consistently associated with increased breast cancer mortality and earlier recurrence. 21 Apparently, there may be some interference of alcohol on pharmacokinetics of chemotherapy as well as social implications that lead to lower adherence to treatment. As for smoking, it could not be confirmed as a predictive factor for NACT or as a prognostic factor. The data in the literature is quite controversial, with some negative results contrasting with others where smoking had a negative impact on survival. Interestingly, smoking cessation after the diagnosis of breast cancer is likely to reduce the risk of breast mortality.20,22,23
Schooling data were similar to those of another Brazilian cohort 23 and did not show association with response or survival outcomes. Similarly, a Norwegian cohort 24 also showed no influence of schooling on survival or response, whereas other results suggested that higher level of education may be associated with better survival and quality of life.25,26 In the same way, there is not much data in the literature about distance from home to the treatment center. The great variability of this social factor among the cases of this cohort may explain the negative results.
The median time from diagnosis to treatment onset of 90 days was quite long. Although not shown to be associated with survival outcomes in this cohort, it is highly suspected that delays in NACT onset can negatively influence treatment outcomes. To avoid long delays in the initiation of cancer treatment, the Brazilian Federal Government decreed the “Law of 60 days” in 2012 (Federal Law number 12.732/12). This law was nationally established in 2013 and defines the maximum range that a patient with cancer has to wait to initiate the specific treatment. However, due to public health system infrastructure issues, this goal is still far from being achieved. 27
A recent comprehensive patient-level meta-analysis 28 has pointed pCR as a strong surrogate of long-term survival outcomes. In the current cohort, the pCR rate was quite similar to other studies using anthracycline and taxane-based NACT. However, this rate was considerably modest when compared to recent clinical studies, in which pCR reached rates over 50%. Some possible reasons for this may be the narrow definition of pCR (ypT0ypN0) and the proportional greater number of women with larger tumors in this study, as well as the use of dense-dose regimens, and the addition of new drugs to NACT in the other studies, such as PARP inhibitors, immunotherapy, and antiangiogenic agents. 29
The use of carboplatin and the PARP inhibitor (veliparib) in the Brightness trial has prompted a substantial increase in pCR rate by more than 20%. 9 Preliminary results from KEYNOTE-173 30 and I-SPY 2 trial 31 have shown a pCR rate of over 50% with the association of pembrolizumab. Other agents such as bevacizumab, nab-paclitaxel, capecitabine, and eribulin showed less significant results. 32 A recent meta-analysis suggested that dose-dense chemotherapy with anthracycline and taxane-based regimen may reduce the risk of death for patients with hormone receptor-negative breast cancer by up to 20%. 33
The ypT and ypN staging were pointed out as reliable prognostic factors. These findings are consistent with the results of a Brazilian cohort that evaluated patients undergoing axillary lymph node dissection, which suggested that the greater the number of positive axillary lymph nodes, the lower the median disease-free survival and OS. 23 Other systems that measure the degree of response to chemotherapy were reported since 2013 and validated in some studies.34-36 The Residual Cancer Burden score may be more reliable than the TNM system for post-NACT staging and evaluation for prognosis, providing an index with good reproducibility in terms of predicting long-term survival. 37 In this cohort, pCR and residual burden disease (ypT and ypN) showed a considerable association with survival outcomes in all models tested for Cox multiple analysis. However, following Akaike criteria, perhaps because they may strongly influence other variables, the final model selected did not include these variables.
Some strengths of this study must be mentioned. The patient inclusion criteria allowed a more uniform sample for evaluation of a broad panel of factors, some of them with interesting and unpublished data. Besides that, using real-world data, the current cohort has drawn a detailed portrait of the harsh sociodemographic reality of women with TNBC treated at a Brazilian public health institution.
On the contrary, some important limitations must be highlighted. As a single-institutional retrospective cohort with lack of standardization of medical records, there was considerable missing data for some variables and some patients were censored for short-term follow-up. Furthermore, the lack of a specific questionnaire to measure the exposure gradient (dose, duration, and type) to alcohol, considering parameters determined by the case-control study conducted by White et al, 38 and tobacco, considering the cutoff of more than 20 pack-years of smoking suggested by Saquib et al, 39 hindered a more detailed analysis of the influence of these factors both in the outcomes. New treatment regimens such as dose-dense schedules, addition of platinum agents to NACT, and maintenance adjuvant capecitabine for patients with residual disease after NACT are not yet available at the institution. Finally, tumor-infiltrating lymphocytes have not been evaluated and there was no molecular analysis of the sample by gene expression profile or BRCA mutation testing. Some molecular subclassifications have predicted different patterns of response to NACT and survival outcomes. 40
Conclusions
In summary, a timely and thorough evaluation of predictive and prognostic factors was carried out. Alcohol consumption and clinical stage III were determinants of lower response rate and worse survival. However, studies with better characterization of type, time, and dose of alcohol consumption are entirely necessary. The burden of post-NACT residual disease, represented by ypT and ypN, is likely to be usable as prognostic factors. Herein, pCR showed a strong association with better survival outcomes, being a potential surrogate for long-term outcomes.
Footnotes
Acknowledgements
We are greatly indebted to the DIPAT/INCA for providing all information regarding the pathological data of the patients included in the study.
Author Contributions
JLdS and ACdM contributed to the concept and design of the study. LCST and ACdM contributed to supervision of the study. JLdS and BHRdP contributed to data collection and/or processing. JLdS, IAS, LCST, and ACdM contributed to analysis and/or interpretation of data. JLdS, BHRdP, IAS, LCST, and ACdM contributed to literature search. JLdS, BHRdP, IAS, LCST, and ACdM contributed to writing the manuscript. LCST and ACdM contributed to critical review of the manuscript.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data used to support the findings of this study are available from the corresponding author. Upon request, may be released after review of the Institutional Review Board.
Ethical Approval
All procedures were in accordance with the ethical standards of the institutional and national research committee. The study was approved by the Ethics in Human Research Committee of INCA, Rio de Janeiro, Brazil, under number CAAE 61675516.9.0000.5274, and conducted in accordance with Good Clinical Practice guidelines.
Informed Consent
As this study has a retrospective observational design, the absence of the informed consent form was approved by the Institutional Review Boards.
