Abstract
Basal-like breast cancer has been reported to be the most aggressive and deadly carcinoma sub-type. Patients diagnosed with this subtype have a less than 50% five-year survival. In addition, many studies have reported that this sub-type is more prevalent in specific ethnic groups and is believed to be a key factor that drives certain ethnic disparities in mortality. In order to effectively study this sub-type and determine unique gene expression and biochemical pathways which sustain this cancer's growth, we sought to identify human breast cancer cell lines that represent a model for the basal-like subtype. Here, we report our findings which indicate the African American cell line CRL-2335 is a true representative of basal-like breast carcinoma.
Introduction
Breast cancer has a high level of heterogeneity throughout the global population. 1 While the disease as a whole is often reported to the public through national surveillance databases as a single disease, oncologists and researchers agree that there are several clinical types of breast cancer, with distinct etiologies and distribution of both incidence and survival among women.1–6 Based on histological analyses which measure presence of Estrogen Receptor (ER), Progesterone Receptor (PR), human epidermal growth factor receptor 2 (HER2/Neu) gene amplification, Epidermal Growth Factor Receptor (EGFR) and cytokeratin 5/6 (ck 5/6), breast cancer is clinically grouped into five pathologically distinct subtypes: Luminal A, Luminal B, HER2 amplified, basal-like, and unclassified.7–9 These subtypes illustrate the etiology of distinct cancer diseases that respond to specific treatment regimens.
There is an observed bias of incidence distributions among ethnic populations of women for each of these subtypes. While African American women have a lower incidence of breast cancer than Caucasian women, they are more likely to present with tumors that are early onset, higher grade, and ER negative and therefore have a lower survival rate.10–12 Premenopausal African American women have a higher prevalence of basal-like tumors than Caucasian women of the same age,12–16 while Caucasian women more often present with Luminal subtypes. This same trend has been seen in Britain, 17 and a recent study determined that triple negative tumors represent the majority of breast cancer tumors in the West African population. 12 In addition, a number of biological differences have been found in breast cancers in African American women, including BRCA mutations, higher expression of cell cycle regulators, and polymorphisms in cytochrome p450 and nucleotide excision repair genes. 15
While there is recent controversy as to the disparities of these subtypes among different ancestral groups, all clinical data agree that the most aggressive subtype, which associates with the highest mortality rate, is the basal-like subtype.5,18,19 This particular subtype is a derivative of the Estrogen Receptor (ER) negative category of breast cancers, but it is further characterized by having no amplification of the HER2 gene, and by expression of EGFR as well as the basal epithelial marker, cytokeratin 5/6. 2 Due to this lack of ER expression and HER2 overexpression there are currently no targeted therapies for basal-like tumors. This lack of therapy, in addition to the subtypes high proliferative capacity8,9 leads to poor clinical outcomes. 13
As we investigate the genetic and epigenetic mechanisms that drive this basal-like cancer, we sought to identify cell lines that may act as an in vitro model for experimental screen and potential tumorigenesis work. A recent publication has outlined approximately 17 cell lines and characterized them into the Luminal and basal subtypes. 20 However, we report an additional basal-like cell line which is derived from a African American female, which will be an essential tool in unraveling not only basal-like carcinoma specific mechanisms, but additionally genetic ancestral associations among these mechanisms related to basal-like breast cancer disparities.
Results
CRL-2335 is confirmed ER negative
The cell line annotation report provided by ATCC indicates this cell line was derived from a primary, stage IIB, grade 2, ancantholytic squamous cell carcinoma with no lymph node metastasis. The line was designated as negative for Her2-neu amplification as well as negative for the expression of ER and PR.
To confirm its retained ER negative status, we conducted immunofluorescence to measure the presence or absence of ER in this cell line (Fig. 1A). As a positive control we determined that CRL-2315, which was designated to be positive for the expression of ER, 21 has high expression of ER as indicated in our findings (Fig. 1A). These data indicate that CRL-2335 is in fact an ER negative cell line. Xu et al also recently confirmed that CRL-2335 is negative for the expression of ER, PR, and Her2 by western blot analysis. 22

Immunostaining images.
It should be noted that in the past few months ATCC has changed the molecular status of CRL-2315 from ER positive to ER negative. Our data, however, indicates that our line is still positive for the expression of ER, which demonstrates the importance of the periodic validation of cell lines for molecular status in the laboratory setting.
CRL-2335 Expresses Basal-Like Markers, EGFR and ck 5/6
In order to determine if this cell line qualifies as a basal-like tumor model, we conducted additional immunofluorescence to measure the presence of the basal-like tumor markers, EGFR and ck 5/6. We found that CRL-2335 was positive for both markers, while the normal cell line CRL-1947 did not exhibit staining with either of the markers (Fig. 1B and C). The functional relevance of these differences is currently under investigation.
To validate the positive immunostaining results for EGFR and ck 5/6 we conducted qPCR with CRL-2335 and CRL-2315. We normalized average Ct values to the endogenous control, 18s, to get average dCt values, which are shown in Figure 2. Cell lines that showed negative protein staining also showed an increase in the average dCt, which correlates with lower expression and the lack of staining (Fig. 2). qPCR results suggest that EGFR is transcriptionally active in CRL-2315 with an average dCt of 12.04. However, the lack of protein staining suggests that the transcript is degraded and not translated into a functional protein. This demonstrates why in a clinical setting we rely on protein staining for diagnostics and not transcriptional analysis alone.
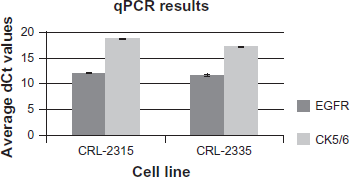
qPCR data. Data is represented as Average dCt values, normalized to the endogenous control 18s. A high average dCt value indicates a low expression value.
Discussion
Molecular classification has important clinical implications for breast cancer patients, as it is used to select treatment options and for prognostic predictions. Breast cancer cell lines are readily available and are commonly used in breast cancer research. Since these cell lines are representative of a heterogeneous group of diseases, it is necessary to determine their sub-classifications in order to correlate molecular data with tumor subtypes. Often we find that cell line studies have contradicting results from what is observed in clinical tissue studies. Part of this dilemma could be due to lack of subtype characterization of the cell lines. Even if derived from a certain tumor type, it is important that we characterize the molecular status of the in vitro lines.
Based on transcriptional and immunofluorescent analysis, we have characterized CRL-2335 as a basal-like tumor cell line. Given its derivation from an African American female, this line will be useful not only in breast cancer subtype studies, but also in genetic ancestry relevant analyses.
Experimental Procedures
Cell culture
CRL-2335, CRL-2315, and CRL-1947 breast carcinoma cell lines were obtained from the American Type Culture Collection (ATCC). Cells were maintained at 37 °C with 5% CO2 and were cultured in appropriate medium (see Table 1). For qPCR, cells were harvested after reaching confluence with 0.25% Trypsin-EDTA (Invitrogen) and washed once with 1X PBS before RNA isolation. For immunofluorescence, cells were grown on alcohol and flame sterilized selected micro glass coverslips (VWR) in 6 well plates (Corning) for 48 hours prior to immunofluorescence.
Cell culture conditions for breast cancer cell lines.

RNA isolation, cDNA synthesis, RT-PCR and qPCR
Cell pellets were resuspended in 1 mL TRIzol Reagent (Invitrogen) and RNA isolation was carried out according to the manufacturer's instructions. cDNA was then synthesized using 300 ng RNA and qScript cDNA SuperMix (Quanta Biosciences) according to the manufacturer's instructions. RT-PCR was performed using expression primers for 18s rRNA, ck 5/6, and EGFR (Table 2) with GoTaqGreen Mastermix (Promega) according to the manufacturer's instructions using 1 ug of cDNA. Amplification was resolved on a 1% agarose gel containing 0.5 ug/mL EtBr run at 120V for 45 minutes and single bands were visualized under an ultraviolet light (Supplemental Fig. 1). qPCR was performed using the same expression primers (Table 2) with RT 2 SYBR Green/ROX qPCR MasterMix (SABiosciences) according to the manufacturer's instructions using 1 ug cDNA.
Expression primers.

Immunofluorescence
EGFR, ck 5/6, and ER immunohistochemical analysis was performed using purified mouse monoclonal antibodies (Santa Cruz Biotechnology, CA). Coverslips were rinsed twice with 1X PBS and fixed in 4% PFA/PBS for 15 minutes. After fixation, coverslips were washed with 1X PBS and then blocked with PBI (1X PBS, 0.5% BSA (Fisher), 0.5% Igepal (Sigma)) for 1 hour. The coverslips were then incubated overnight in corresponding primary antibodies (1:500) with PBI, washed with PBI, then incubated with Alexaflour TRITC goat anti-mouse secondary antibody (2:5000, Invitrogen) in PBI overnight. The coverslips were mounted in 30% glycerol after a 15-minute incubation with DAPI (1:10,000, Sigma) in PBI and two 30-minute washes with PBI. All washes and incubations were performed on a rotator at 4 °C. Confocal images were taken with a Ziess Apotome using Axiovision software.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowlegements
We would like to thank Christina Swoope, Megan Grant and Christopher Walker for assistance with experimental procedures. This work was funded by the Medical College of Georgia Research Foundation.
