Abstract
Nowadays, hepatocellular carcinoma (HCC) is the second leading cause of cancer deaths, and identifying the effective factors in causing this disease can play an important role in its prevention and treatment. Tumors provide effective agents for invasion and metastasis to other organs by establishing appropriate communication between cancer cells and the microenvironment. Epithelial-to-mesenchymal transition (EMT) can be mentioned as one of the effective phenomena in tumor invasion and metastasis. Several factors are involved in inducing this phenomenon in the tumor microenvironment, which helps the tumor survive and migrate to other places. It can be effective to identify these factors in the use of appropriate treatment strategies and greater patient survival. This study investigated the molecular differences between tumor border cells and tumor core cells or internal tumor cells in HCC for specific EMT genes. Expression of NOTCH1, ID1, and LST1 genes showed a significant increase at the HCC tumor border. Targeting these genes can be considered as a useful therapeutic strategy to prevent distant metastasis in HCC patients.
Keywords
Introduction
Nowadays, hepatocellular carcinoma (HCC) is the second leading cause of cancer deaths. Viral hepatitis caused by hepatitis A virus (HAV) and hepatitis C virus (HCV) is among the causes of this type of cancer in developing countries. 1 Alcoholic liver cirrhosis and nonalcoholic steatohepatitis are the most common causes of HCC in developed countries. Distant metastatic disease is found in 13% to 36% of patients at the time of initial diagnosis. Distant metastasis is considered as prognosis in the advanced stages of the disease. The results of many studies showed that there is a significant relationship between distant metastasis and reduced patient survival.2,3 Metastasis is defined as the spread of cancer cells from the place where they first formed to another part of the body, which can be portrayed as a 2-phase process: The first phase involves the physical translocation of a cancer cell to a distant organ, whereas the second encompasses the ability of the cancer cell to develop into a metastatic lesion at that distant site, which is a complex process involving a variety of genes and pathways, and is considered an important factor in tumor progression and 90% of cancer deaths, as well as a major challenge in cancer treatment. It is impossible to remove metastatic tumors through surgery, radiotherapy, or chemotherapy as opposed to tumors without metastasis. Acquisition of special properties by cancer cells gives them the ability to move and invade other organs and tissues, as well as therapeutic resistance, and reduces life expectancy in patients despite the use of a variety of therapies. 4 To improve patient survival, it is necessary to understand the underlying mechanisms of metastasis to allow for targeted intervention. 5 According to most studies, there is a simple linear relationship between tumor size and the frequency of metastases. 6 The most common sites of extrahepatic metastasis are lung, regional lymph nodes, bone, adrenal glands, and peritoneum/omentum.2,3,7 Hepatocellular carcinoma patients with extrahepatic metastasis do not live longer than 4 to 6 months.4,5,8 In different types of cancers, there is a significant relationship between metastasis with increasing mortality rate in cancer patients and controlling metastasis significantly increases the patient’s life expectancy. 9 Several studies have demonstrated a link between epithelial-to-mesenchymal transition (EMT) and the metastatic initiating potential of tumor cells. According to studies, the tumor cells lose their epithelial properties at the tumor border and healthy tissue, and acquire mesenchymal characteristics called EMT. An EMT is a biologic process in embryogenesis and is described as a differentiation process. 10 Furthermore, EMT is an important event for tumor cells to invade lymph tissue and vessels. 11 Epithelial-to-mesenchymal transition first involves dedifferentiation and the acquisition of self-renewal and stemness, followed by differentiation into intracellular cells. The most important feature of EMT is the decreased expression of epithelial markers E-cadherin and ZO1, and the increased expression of mesenchymal markers including N-cadherin, TWIST, and SNAIL. 12 Loss of E-cadherin may lead to disconnection of cell-cell adhesion junctions, which increases the invasion and migration characteristics of cancer cells. 13
Tumor microenvironment is a heterogeneous environment that includes different cells and immune tissue with dynamic transaction that can have a potential clinical effect on metastasis by proliferation and differentiation. 14 At the border between tumor tissue and its surroundings, there is a significant relationship between fibroblasts, inflammatory cells, and immune cells with tumor cells. 15 Epithelial-to-mesenchymal transition and metastasis are most likely to occur at the tumor tissue border. According to the results of studies on different types of cancer, there is a significant relationship between the expression of EMT markers such as Snail and TWIST in this area with an increased risk of metastasis. 16 Macrophage filtering to the tumor border has been reported to be found in patients with metastatic HCC. Macrophages induce EMT and increase invasion and migration of tumor cells by secreting cytokine interleukin 8 (IL-8) by activating the JAK2 (Janus kinase 2)/STAT3 (Signal transducer and activator of transcription 3)/SNAIL signaling pathway. 12 According to the results of studies, differentiation occurs following the expression of EMT factors. Increased expression of TWIST and SNAIL factors in epithelial cells turns them into mesenchymal cells with high differentiation capacity. 10 Complex communication in the microenvironment of the tumor with the surrounding cells and other cells in distant areas is possible through various mechanisms that regulate these mechanisms by various factors such as molecules, genes, cytokines, micro RNAs (miRNAs). This study was carried out aimed to investigate the factors affecting tumor progression through EMT induction that the identification of these factors can be useful in applying appropriate treatment methods.
Materials and Methods
Microarray analysis
Gene expression profile of GSE84598 that was maintained on Illumina HumanHT-12 V4.0 expression beadchip (GPL10558) was extracted from Gene Expression Omnibus (GEO) database (https://www.ncbi.nlm.nih.gov/geo/) and then subsequently analyzed using GEO2R, a tool which is based on the R language. At first, samples were categorized into “tumor border” and “tumor core” groups based on supplementary information of GSE84598. Then, mRNA expression profile of 22 samples of invasive border of the liver tumor was selected to compare with 22 samples of internal tumor cells with normal distribution to obtain dysregulated genes (Figure 1A). Differentially expressed genes (DEGs) were obtained by considering P < .05 and |log2FC| ⩾ 0.5 as the cutoffs. Since EMT is considered a cell differentiation pathway, and cell differentiation is a biological process (BP) that can be analyzed in Panther (http://pantherdb.org/), a source of gene ontologies (GOs), all of the DEGs were traced in this database to first identify those altered genes that are involved in cell differentiation (GO:0030154) that latterly determined as differentiation differentially expressed genes (dDEGs). These dDEGs were further analyzed to garner EMT-related DEGs in tumor border cells.

Discovering DEGs and dDEGs and associated GOs: (A) value distribution of selected samples in both groups; (B) volcano plot showing significant downregulated (purple dots) and upregulated (green dots) genes considering P < .05 and |log2FC| ⩾ 0.5; and (C) GO analysis results from DAVID.
Gene ontology and pathway analysis
To discover the cellular and molecular consequences of dDEGs, GO analysis including BP, molecular function (MF), and cellular component (CC) analysis was performed using Database for Annotation, Visualization, and Integrated Discovery (DAVID) (https://david.ncifcrf.gov/). Significant GOs were obtained by P < .05 filtration. Next, with the purpose to identify the association of dDEGs and cellular pathways and their effect on EMT induction and metastasis, pathway analysis was established by using Kyoto Encyclopedia of Genes and Genomes (KEGG) (https://www.genome.jp/kegg/pathway.html) and WikiPathways (https://wikipathways.org/). Remarkable pathways were detected with the lowest P value.
Protein network analysis
To reveal key proteins with regulatory roles that are involved in cell differentiation and possibly EMT, a protein-protein interaction (PPI) network was at first constructed by the Search Tool for Retrieval of Interacting Genes/Proteins (STRING) (https://string-db.org/) and then subsequently further analyzed by Cytoscape (version 3.8.0) with CentiScape and CytoHubba plugin. Hub nodes were determined regarding degree (the greatest number of the closest nodes to a node) and radiality (the centrality of a node in a network) as the criteria.
Stage-dependent expression analysis
The final liver hepatocellular carcinoma (LIHC) progression to stage IVb is related to distant metastasis which is the outcome of EMT phenomenon. By screening gene expression alterations through LIHC progression to stage IVb, final candidate genes contributing to EMT could be acquired. To reach this goal, a differential expression stage plot analysis in gene expression profiling interactive analysis (GEPIA) database (http://gepia.cancer-pku.cn/) was recruited to monitor the expression alterations of the microarray candidate genes in The Cancer Genome Atlas (TCGA) and The Genotype-Tissue Expression (GTEx) RNA-sequencing data. Previous candidate genes that were confirmed in this step were represented as the final molecular candidates.
Survival rate analysis
To figure out the correlation of overall survival in LIHC patients and final candidate gene expression, Kaplan-Meier plot analysis was carried out using RNA-seq data of TCGA in GEPIA. Median was considered as the group cutoff. 95% confidence interval and hazard ratio based on Cox proportional-hazards model (Cox PH) model were constituted to draw Kaplan-Meier plots.
Results
LST1 and ID1 as the most altered genes with effects on cell differentiation
At first, the “tumor border” and “tumor core” groups have been compared. Then, a total of 13405 including 1294 significant DEGs that contain 159 downregulated and 662 upregulated genes were collected with respect to P < .05 and |log2FC| ⩾ 0.5 (Figure 1B). Hepcidin antimicrobial peptide (HAMP) and actin-related protein 5 (ACTR5) had the biggest change in upregulated and downregulated genes with log2FC = 3.75 and −1.31, respectively. To investigate EMT and metastasis of the tumor border cells, cell differentiation was counted in Panther to define dDEGs. Monitoring the expression changes in the dDEGs showed LST1, ID1, PCK1, CD79A, and HCLS1 in upregulated and MAFF (MAF BZIP Transcription Factor F) in downregulated dDEGs with the highest expression alteration level (Figure 1C). LST1 and ID1 were the genes with the greatest expression changes among dDEGs by log2FC = 1.93 and 1.87 which contribute to differentiation and perhaps EMT and metastasis.17,18
Discovering associated gene ontologies and pathways
Gene ontology analysis in DAVID indicated enrichment of negative regulation of osteoblast differentiation, negative regulation of transcription from RNA polymerase II, regulation of cell shape, peptidyl tyrosine phosphorylation and cell differentiation for BP, protein tyrosine kinase activity, transcription factor activity, sequence-specific DNA binding, semaphorin receptor binding, and chemorepellent activity for MF and B-cell receptor complex, plasma membrane, integral component of plasma membrane, extrinsic component of cytoplasmic side of plasma membrane, and nucleoplasm for CC as critical GOs in tumor border cells in accordance with gene count and FDR (Figure 1C). Besides, pathway analysis demonstrated axon guidance, B-cell receptor signaling pathway, Ras signaling pathway, PI3K-Akt signaling pathway and transforming growth factor-beta (TGF-β) signaling pathway in KEGG (Figure 2A), and B-cell receptor signaling pathway, ID signaling pathway, TYROBP Causal Network, miR-517 relationship with Archain 1 (ARCN1) and Ubiquitin Specific Peptidase 1 (USP1) and Ras signaling in WikiPathways (Figure 2B), was the associated signature pathways that were disrupted through EMT and metastasis greatly (Supplemental File 2). Recognizing of positive regulation of cell shape migration, mesenchymal cell development (GO:0030335), mesenchymal cell development (GO:0014031), cardiac EMT (GO:0060317), EMT involved in endocardial cushion formation (GO:0003198) in DAVID in tumor border cells, and EMT in colorectal cancer (WP4239) in WikiPathways, in which HEYL, SEMA4A, NOTCH1, FGR, SEMA4D, CSF1R, ID1, and ID2 were detected to be engaged, underpin the contribution of EMT as a possible positive control of promoted of this pathway (Table 1).
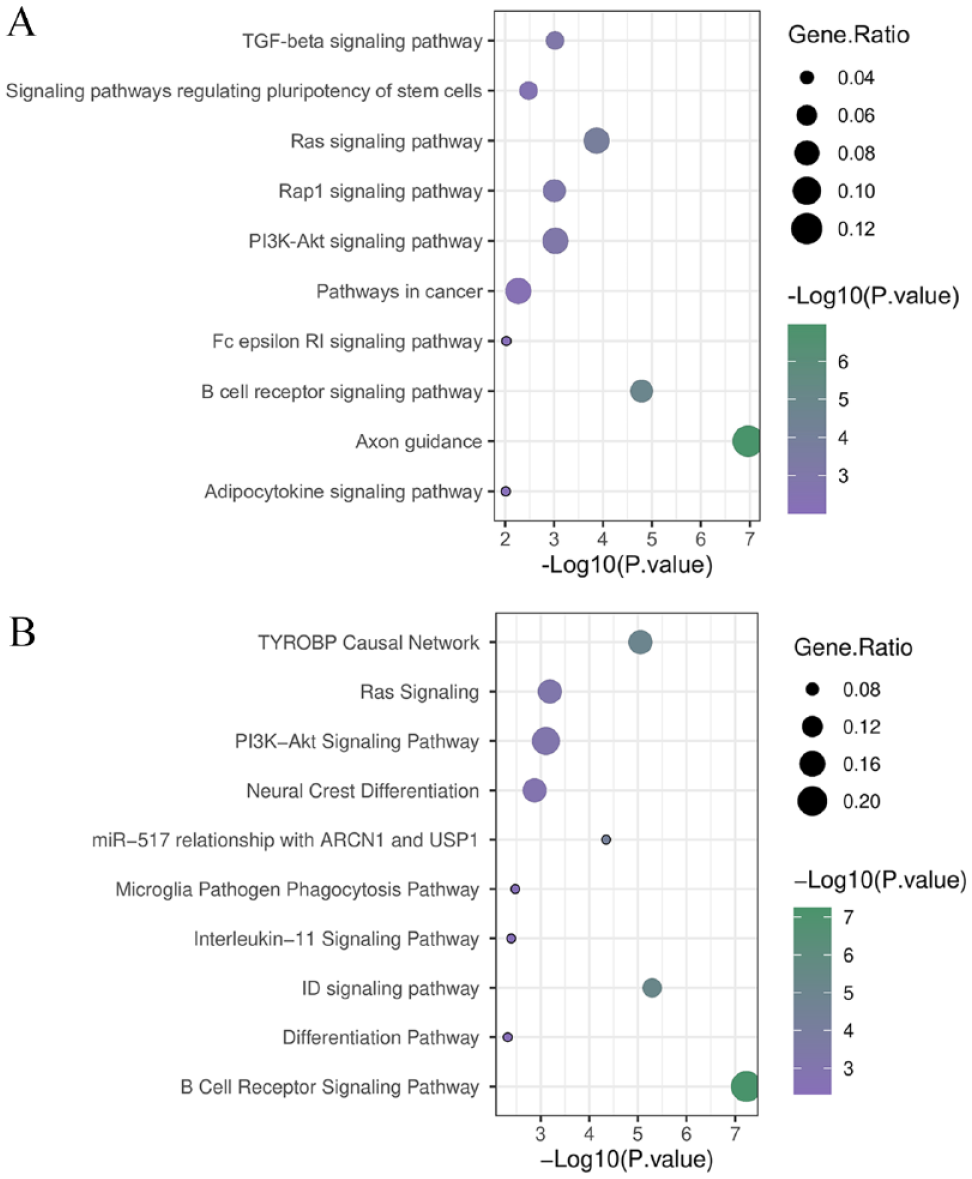
Pathway analysis results using KEGG and WikiPathways. (A) KEGG and (B) WikiPathways significant pathways considering P < .05. Gene ratio refers to the relative number of the involved genes in a pathway and all genes that were identified in significant pathways.
Genes that were identified to be involved directly in cell migration or EMT.

Abbreviations: BPs, biological processes; EMT, epithelial-to-mesenchymal transition; GO, gene ontology;
NOTCH1 as the key protein in cancer cell differentiation
To discern hub nodes among dDEGs, a PPI network was constructed by STRING and ensuing further analyzed by Cytoscape 19 with CentiScape 20 and CytoHubba 21 plugin. At first, within 43 proteins, 30 proteins via 63 edges were interacted (Figure 3A). According to degree, NOTCH1, SYK, FGR, ETS1, and RAC2 were the important proteins. However, because of radiality to distinguish central proteins, NOTCH1, RAC2, EST1, EST2, and CSF1R were the critical nodes. Analyzing the network with CytoHubba represented NOTCH1 as the crucial protein in both radiality and degree network (Figure 3B). Further analysis of NOTCH1 by CentiScape 2.2 manifested the same results with CytoHubba and illustrated NOTCH1 as the most important protein among dDEGs (Figure 3C).
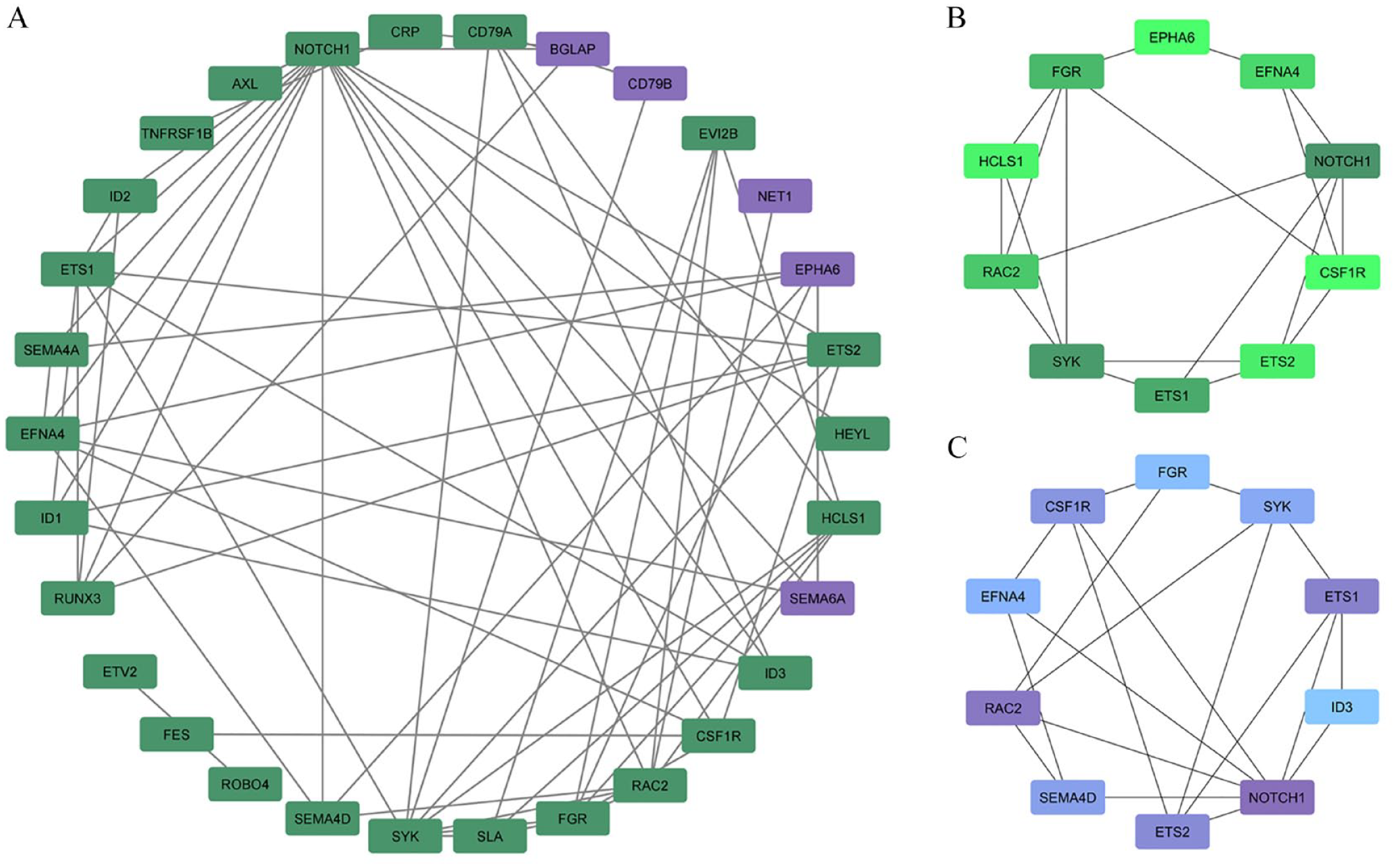
Protein network construction and analysis in Cytoscape. (A) PPI network that was constructed by STRING and visualized by Cytoscape. Purple and green nodes represented downregulated and upregulated dDEGs, respectively. (B) Degree and (C) radiality models constructed by CytoHubba indicated NOTCH1 as the critical protein. The darker the color, the higher the value.
NOTCH1, LST1, and ID1 as the final candidate genes
To screen whether the expression changes of NOTCH1, LST1, and ID1 as the microarray candidates are related to distant metastasis, GEPIA stage plot analysis was used. LST1 and ID1 were detected to be upregulated during tumor progression to stage IVb which is consistent with the GSE84598 microarray expression profile. Although the NOTCH1 expression change in the final stage in tumor border cells was hardly detectable, approximately 0.1 log2 (TPM + 1) difference, the same pattern in which NOTCH1 spotted to be overexpressed through LIHC distant metastasis was observed confirming GEO results (Figure 4A). Taken together, GEPIA RNA-seq results validate the previous microarray data for NOTCH1, LST1, and ID1 that these genes are involved in distant metastasis through differentiation processes by their overexpression.

GEPIA analysis results: (A) stage-dependent expression analysis correlated LST1, ID1, and NOTCH1 higher expression and distant metastasis in LIHC, and (B) Kaplan-Meier plot was drawn and demonstrated the correlation of higher expression of correlated LST1, ID1, and NOTCH1 and lower chance of survival in LIHC patients in late stages.
NOTCH1, LST1, and ID1 high expression level causes death in late stages
The higher expression level of LST1, NOTCH1, and ID1 after the 80th month was clearly correlated to a lower survival chance in LIHC patients. Also, high transcript level of NOTCH1 and ID1 before the 80th month was not associated with lift in mortality rate. Besides, although upregulation of LST1 leads to death during the 40th to 80th month, lower transcription of this gene was concerned with increased mortality. These data suggest that NOTCH1, LST1, and ID1 are attributed to final tumor progression and feasibly EMT and distant metastasis consisting with previous results.
Discussion
Tumors and their tumor microenvironment have an organized “ecosystem” in which tumor cells and immune system cells are in a dynamic state. 22 Tumor microenvironment can play a key role in tumor progression and metastasis. Some of the stromal cells of this environment are as follows: cancer-associated fibroblast (CAF), tumor endothelial cells (TESs), tumor-associated macrophage (TAM), tumor-associated adipocyte (TAA), T lymphocyte, and B lymphocyte, which each of these cells exhibits a specific ability to promote tumor growth. 23 Evidence suggests that the tumor microenvironment inhibits drug delivery to the tumor and may lead to chemical and drug resistance. 24 Hepatocellular carcinoma is now the fifth most common cause of cancer worldwide and the second leading cause of cancer deaths. 25 Various molecular compounds of tumor microenvironment (HCC) play an important role in tumor growth and metastasis. Also, these factors have a significant effect on the effectiveness of treatment methods. 26 Liver progenitor cells (LPCs) and circulating mesenchymal stem cells are among the stem cells that have the ability to produce mesenchyme and liver cell lines. Furthermore, these cells have the ability to inhibit the immune system. Myofibroblasts and hematopoietic stem cells activated in the tumor environment are the main source of collagen secretion, which can act as a basis for cell movement and migration. This study was carried out aimed to find the variables in the tumor and its environment and the border of the tumor and healthy tissue. These factors can be altered signaling molecules or pathways that in turn can generate new perspectives on therapies. 27
The EMT plays a key role in the proliferation and metastasis of malignant hepatocytes during the progression of HCC disease. According to the results of previous studies, although several factors trigger the EMT in HCC, but this event is mainly regulated by the 3 main groups of transcription factors SNAIL, TWIST, and ZEB (EMT_AFTs). EMT_AFTs play an important role in oncogenic deformation, regulating the stemness of cancer cells, inhibiting immune activity against cancer, including apoptosis and cell aging, and increasing angiogenesis. 12 Increasing or decreasing the expression of some genes also plays an important role in tumor invasion and metastasis. Increased expression of ID1, NOTCH1, and LST1 genes in tumor border cells compared with internal tumor cells has been observed in this study by enrichment analysis. These genes play a key role in differentiation processes and EMT. More and more evidences revealed that increased expression of ID1, NOTCH1, and LST1 genes is involved in EMT progression.28-30 According to the performed analyses, genes were selected whose expression change was incremental and their log fold change (FC) was more than 1. These genes include ID1 and LST1 genes. ID1 (inhibitor of DNA) is a helix_loop_helix protein and is one of the important regulators of cell cycle and cell differentiation. High expression of this gene is often associated with tumorigenicity, cell aging, cell proliferation, and survival. There is a significant relationship between overexpression of ID1 and increased metastasis in tumors.31-36 Results of studies show relationship between increased ID1 expression and increased EMT and cancer progression in more than 20 types of cancer, including HCC.37-39 ID1 expression is increased not only in HCC but also in hepatocytes infected with hepatitis B virus and viral infections 40 and is considered as an important prognosis in HCC caused by hepatitis B virus. 41 Furthermore, there is a significant relationship between increasing the expression of this gene and reducing the life expectancy of patients. 42 Recently, more studies indicate that EMT is a crucial mechanism taking place during tumor invasion and metastasis. As Notch signaling pathway is desperately involved in many cellular processes such as cell proliferation, survival, apoptosis, migration, invasion, angiogenesis, and apparent evidence suggests that Notch regulates EMT, result in tumor invasion and metastasis. In addition, some researches revealed that Notch signaling pathway could be downregulated by its inhibitors. 28 ID1 has been shown to be increased in a number of advanced cancers and is associated with EMT. A study was investigated the expression of ID1 in bladder cancer and its association with EMT. It demonstrated that overexpression of ID1 could lead to fibroblastic spindle cell phenotype along with loss of cell-to-cell contacts. Western blotting and immunofluorescent staining of bladder cancer cell lines showed that the expression level of ID1 is correlated with the expression of mesenchymal markers and is inversely associated with the expression of epithelial markers. Moreover, results of collagen invasion and wound closure assays indicate that ectopic ID1 expression leads to increased ability of invasion and migration 30 A study confirmed through RNA-sequencing analysis that 7 genes (NME1-NME2, LST1, KAT7, BMX, CLIC1, KANSL2, and UFL1) involved in the non-small-cell lung cancer (NSCLC) metastasis were related to EMT function. 29
During EMT, cells acquire the ability to metastasize and invade by acquiring a mesenchymal property called tumor-initiating cells. These cells eventually lead to the formation of metastatic clones.43-46 Aberrant activation of Notch signaling has linked to the metastasis of HCC. Notch signaling, an essential pathway for tissue development, also contributes to tumorigenesis in many cancers includes HCC. A study evaluated NOTCH1 expression and its clinicopathological parameters in 82 human HCC patients. In metastatic HCC cells, abnormal expression of NOTCH1 was associated with increased expression of Snail1 and repressed expression of E-cadherin; the NOTCH1-Snail1-E-cadherin association can also be found in HCC patient tumors. Inhibition of NOTCH1 by shRNA reversed Snail1 expression, which enhances re-establishment of repressed E-cadherin in metastatic HCC cells. Thus, abnormal NOTCH1 expression is strongly associated with HCC metastatic disease, which could be mediated via the NOTCH1-Snail1-E-cadherin pathway. Knock-down of NOTCH1 drive back HCC tumor metastasis in a mouse model. These data indicate that targeting of Notch signaling inhibits tumor metastasis. Also, RING finger protein 187 (RNF187) is a necessary factor for NOTCH1 to further invasion and metastasis of HCC. Clinical relevance shows that activation of NOTCH1-RNF187 associate with a worse prognosis of HCC patients. These findings help for developing novel strategies to tackle HCC metastasis.47,48
According to the results of studies, increasing the expression of ID1 plays an important role in the formation of these cells, increasing the ability to migrate and establish metastatic sites.43,45,46,49 Increased androgen receptor (AR) expression in HCC cells increases ID1 expression, which leads to invasion and migration of tumor cells in HCC. 50
According to the results of the studies, there is a significant relationship between ID1 expression and MAPK/ERK signaling pathway and with increasing C-MYC level in HCC. 51 Furthermore, ID1 induces EMT and metastasis by increasing the expression of EMT-specific proteins such as vimentin, TGF-β, integrin-β, and Snail. 52
The results of examining the signaling paths in the database show increased KEGG and WikiPathway of some signaling paths, including axon guidance, B-cell receptor signaling pathway Wp23, and ID1signaling pathway Wp53. Furthermore, increased expression has been observed in important tumor pathways such as Ras signaling pathways, PI3K-Akt signaling pathway, and TGF-β pathway. Transforming growth factor_β is a cytokine that regulates cell proliferation, differentiation, and migration. In the advanced stages of tumorigenesis, this molecule induces EMT, induces angiogenesis, stimulates tumor-associated fibroblasts, and inhibits the immune system. 53 Expression of this signaling pathway increases in HCC. Transforming growth factor-beta activates the ID1 promoter. Transforming growth factor-beta regulates Vascular Endothelial Growth Factor (VEGF) expression via ID1. 52 In addition, it induces Snail and EMT expression at the initial site of the tumor.54,55 ID1 increases MYC expression, thereby increasing the proliferation and self-renewal of cancer cells by regulating axon guidance process. 56
The results of GO and signaling pathways in the BP database show that the MF of genes involved in cell shape regulation, cell differentiation, and transcription factor activity in the process EMT has a significant increase. The ID1 gene plays a key role in regulating differentiation and EMT processes in HCC by influencing the expression of some transcription factors. E47 is one of the transcription factors belonging to class 1 bHLH transcription factor, which plays an important role in the EMT process in HCC cells. E47 expression is increased in HCC. 57 E47 induces EMT by suppressing E-able expression. 58 The interaction of factor E47 and ID1 prevents the activation of the E cadre promoter, and continuous expression of factor E47 is necessary to maintain the phenotype and mesenchymal shape of cells after EMT. 59 Also, there is a significant relationship between ID1 and E47 with the basal cancerous cells phenotype (basal-like cancerous cells and EMT active in tumors). Therefore, the collaboration between E47 and ID1 is necessary to maintain the EMT phenomenon. 58 Kruppel-like factor (KLF) is one of the transcription factors that contains the second zinc finger, which plays an important role in cellular processes such as proliferation, differentiation, apoptosis, and cell migration.60-64 This molecule inhibits the EMT and metastasis process by modulating ID1 inhibitors, and KLF17 inhibits it by binding directly to the ID1 promoter. 60
Given the essential role of ID1 in the progression of HCC, its inhibition can be an important therapeutic strategy. 65 A study was performed on the use of berberine which led to inhibit the EMT and HCC metastasis to lung tissue and tissues around the liver. According to studies, berberine inhibitory function is exerted by inhibiting ID1 and ID1 expression, leading to decreased EMT, invasion of HCC cells and their growth. 65
This study reported an increase in the expression (leukocyte-specific transcript 1) of LST1 in tumor border cells compared with tumor center cells. LST1 is an major histocompatibility complex III (MHCIII) membrane protein. Several studies have reported that there is a significant association between LST1 gene and EMT event, morphological deformation (formation of amoebic cell dilatations from cancer cells), and metastasis.29,66,67 According to the results of the study of signaling paths in the BP, MF, and CC databases, the paths associated with the plasma membrane and the path of cell shape are increasing. According to the results of previous studies, LST1 plays an important role in changing the morphology of different cell types by being placed in the cell membrane. 68 LST1 as a membrane scaffold forms a multimolecular complex that eventually leads to the polymerization of actin and Tunneling nanotube (TNT). 69 Tunneling nanotubes establish direct cellular connections at long distances independent of environmental soluble factors. 70
Evidence suggests that TNT plays an important role in tumor survival and progression, enabling long-distance signal exchange between cells 71 and acting as a rail for cell movement after karyokinesis, indicating their role in the migration and proliferation of tumor cells. Cancer cells use TNT to communicate with other cancer cells and stromal cells in the tumor microenvironment, such as mesenchymal stem cells, fibroblasts, macrophages, and neurons. 71 Membrane myoferlin, together with LST1, is involved in the induction and deformation of the plasma membrane and its fusion to form TNT.55,69,72 Myoferlin expression is increased in HCC.73-79 Myoferlin as an oncogene protein is involved in various HCC progression processes including proliferation and invasion, metastasis, and EMT. 80 According to the results of studies, there is a significant relationship between the formation of TNT and EMT. 81 Inhibition of LST1 gene expression can reduce the formation of TNTs and EMTs and can be used as an important therapeutic approach for the treatment of HCC. 69 According to the analysis in Cytoscape and Protein Network based on degree and radiality, Notch1 transmembrane protein has the most interactions and is the main regulator of EMT in HCC (Figure 3). NOTCH1 signaling pathway is one of the conserved biological pathways that plays an important role in the development of hepatic vessels and differentiation of LPCs.48,82 Mutations in this signaling pathway not only lead to development defects but also lead to the development and progression of some cancers, including HCC.83-86 The metastatic tissue of HCC greatly expresses Notch1.27,47,48,87-89 Jagged-1 ligand in epithelial cells as an inductor of the Notch pathway plays an important role in the EMT process. 90 Notch1 promotes EMT in HCC by decreasing E-cadherin expression and increasing vimentin expression. 91 NOTCH1 binds directly to the RNF187 promoter, and RNF187 expression leads to increased EMT and metastasis in HCC patients.92,93 There is a significant relationship between increased expression of NOTCH1 and RNF187 with decreased patient life expectancy.94,95
Calcium signaling is a key mechanism for rapid translation of tumor microenvironmental signals into cellular responses associated with EMT, cancer migration, and invasion.5,6,96 Calcium uptake by mitochondria depends on the calcium uptake channel and the mitochondrial calcium uniporter (MCU) and its regulatory subunit mitochondrial calcium uptake1 (M1CU1) and mitochondrial calcium uptake regulatory 1 (MCUR1).97-99 Expression of this complex increases in HCC and leads to the production of reactive oxygen species (ROS), EMT, and metastasis.100,101 Nrf2 and its inhibitor Keap1 protein act as regulators of cell redox levels and increase Notch1.101,102 Following the activation of the NOTCH1 path, the EMT begins in the HCC. This pathway suppresses the expression of ophthalmic markers such as E-cadherin and induces the expression of mesenchymal markers such as vimentin, slug, Snail1, and Snail2.47,96 Increased angiogenesis is a feature of metastatic HCC. NOTCH1 plays a key role in the formation of tumor blood vessels and induction of the EMT.103-107 By activating the EMT, NOTCH1 promotes distant metastasis, which is one of the reasons for reduced survival in HCC patients.103,108,109
The results of signaling paths in the KEG database show that axon guidance and PI3K-Akt increase. Axon guidance through the Notch messaging path is one of the important regulators of the EMT process during development. 110 According to the results of previous studies, increasing NOTCH1 expression along with increasing PI3K-Akt pathway activity increases EMT and metastasis in HCC.111,112
The results of GO and signaling pathways in the BP database show that the MF of genes involved in the negative regulation of osteoblast differentiation is increased. Several studies have shown that the NOTCH1 gene plays a key role in inhibiting osteoblast differentiation. 113 Furthermore, based on the results of GO and signaling pathways in the MF database, the MF of the genes involved in semaphorin receptor binding is increased. NOTCH1 increases semaphorin receptor expression, PLXND1, resulting in increased EMT and migration into cancer cells. 114
Given the importance of the NOTCH1 signaling pathway in increasing EMT and metastasis, therapies that target this pathway can be considered as a new treatment for HCC patients. Inhibition of NOTCH1 by DAPT decreases EMT and metastasis in HCC, and activation by Jagged-1 increases EMT.115-117 Also, inactivating calcium channel by PV_mito significantly inhibits EMT and HCC invasion. 90 By binding to mRNA3’UTR NOTCH1 mRNA, miR449a inhibits its expression and thus inhibits the EMT event in HCC tumor cells. Decreased expression of miR449a in HCC patients leads to increased tumor invasion. Therefore, the use of this miRNA can be a therapeutic strategy to inhibit EMT and metastasis in these patients.118-120
The results of this article show that increased expression of ID1, LST1, and NOTCH1 genes in the past months of patients with HCC reduces survival (Figure 4). Therefore, new therapies can help increase the survival and longevity of patients with HCC by reducing the expression of these genes and reducing EMT.
Conclusions
In conclusion, this study elucidates the intricate dynamics between tumor cells and the microenvironment in HCC, highlighting the pivotal role of the microenvironment in disease progression and metastasis. Various stromal cells and their contributions to tumor growth and drug resistance are explored, with a central emphasis on EMT and its regulators, including Snail, Twist, and Zeb. The gene ID1 is identified as a key facilitator of HCC invasion, influencing tumorigenicity, cell aging, and survival. The Notch signaling pathway emerges as a significant player in EMT regulation and metastasis. Genes such as ID1, LST1, and NOTCH1, exhibiting increased expression, correlate with reduced patient survival. Signaling pathways such as Ras, PI3K-Akt, and TGF-β are implicated in cell processes, while therapeutic targets such as ID1, LST1, and NOTCH1 offer potential avenues to inhibit EMT and improve HCC prognosis. The study also delves into the role of TNTs and the calcium signaling pathway, suggesting novel strategies, including berberine, DAPT, and miRNA449a, to hinder EMT and metastasis in HCC. In conclusion, this comprehensive analysis provides valuable insights for developing innovative therapeutic approaches to enhance patient outcomes in HCC.
Supplemental Material
sj-docx-1-bbi-10.1177_11779322241227722 – Supplemental material for Functional Enrichment Analysis of Tumor Microenvironment–Driven Molecular Alterations That Facilitate Epithelial-to-Mesenchymal Transition and Distant Metastasis
Supplemental material, sj-docx-1-bbi-10.1177_11779322241227722 for Functional Enrichment Analysis of Tumor Microenvironment–Driven Molecular Alterations That Facilitate Epithelial-to-Mesenchymal Transition and Distant Metastasis by Mahnaz Abdolahi, Parnian Ghaedi Talkhounche, Mohammad Hossein Derakhshan Nazari, Haniyeh Sadat Hosseininia, Niloofar Khoshdel-Rad and Amin Ebrahimi Sadrabadi in Bioinformatics and Biology Insights
Supplemental Material
sj-xlsx-2-bbi-10.1177_11779322241227722 – Supplemental material for Functional Enrichment Analysis of Tumor Microenvironment–Driven Molecular Alterations That Facilitate Epithelial-to-Mesenchymal Transition and Distant Metastasis
Supplemental material, sj-xlsx-2-bbi-10.1177_11779322241227722 for Functional Enrichment Analysis of Tumor Microenvironment–Driven Molecular Alterations That Facilitate Epithelial-to-Mesenchymal Transition and Distant Metastasis by Mahnaz Abdolahi, Parnian Ghaedi Talkhounche, Mohammad Hossein Derakhshan Nazari, Haniyeh Sadat Hosseininia, Niloofar Khoshdel-Rad and Amin Ebrahimi Sadrabadi in Bioinformatics and Biology Insights
Footnotes
Acknowledgements
The authors acknowledge the support of the CytoTech research team.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MA and HSH analyzed the data and wrote the first draft of the manuscript. PGT and MHDN performed data collection, data interpretation, and bioinformatics analysis. NK-R reviewed the analysis and contributed to the preparation of the manuscript. AES conceived and provided a conceptual framework of the study, made critical revisions, and reviewed the bioinformatics analysis. All authors reviewed and approved the final manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
